Abstract
Sleep in hospital is considered a challenge from both a patient and a nursing perspective. To enable nurses to promote patients’ sleep during hospital care it is essential to have valid, reliable and useful sleep assessment tools at hand. The aim of the present study was to evaluate and compare the outcomes of objectively estimated sleep and self-reported sleep quality. Data were collected through continuous wrist-worn actigraphy and completion of the Richards–Campbell Sleep Questionnaire (RCSQ) in a pilot study on 25 patients during hospital care. A correlation between mean values for the two assessments was shown for the second night but not for the third night of assessment, and it was concluded that the two assessments might capture different dimensions of sleep. Furthermore, based on a high response rate and an evident feasibility in a nursing context the RCSQ may facilitate person-centered care.
Keywords
Background
Sleep is important for maintaining good health and it is a basic human need.1–3 Many patients find sleeping in hospital a challenge and describe impaired sleep as a stressor during hospitalization.4–7 Patients’ reported total sleep time in hospital was 83 minutes shorter and awakening time was 43 minutes earlier than when patients slept at home. 8
Nurses are strategically well placed to assess and promote patients’ sleep, and promoting patients’ sleep in hospital ought to be an important goal for the nursing profession.9,10 Despite this, several studies report that nurses identify and observe sleep as insufficient among patients but they do not know how to actively work to promote patients’ sleep and sleep-promoting interventions are rarely used.11–13 The non-use of standardized assessment tools has been described as a barrier for effective sleep management14,15 and there is a call for validated and brief sleep assessment tools that are easy to understand, quick to use, and have positive psychometric properties indicating reliability. 9
The Richards–Campbell Sleep Questionnaire (RCSQ) is a brief questionnaire that evaluates the previous night’s sleep and sleep quality. 16 The RCSQ, amongst other self-administered questionnaires, could be useful in an acute care hospital. It only consists of five questions and is therefore not time consuming or stressful to complete 9 compared to, for example, the Pittsburgh Sleep Quality Index (PSQI) that consists of 24 questions and assesses sleep quality and disturbances over a one-month time period. 17 Alternatively, an objective measure of patients’ sleep may be achieved with an actigraph; a wrist-worn device that can sense and store information about a person’s small movements. Registered data are transformed by computer software into periods of rest and activity, and thus into sleep/wake periods. 18 Studies comparing actigraphy and polysomnography (the gold standard for objective measurement of sleep) have shown a high correlation for assessing total sleep time and awakenings.19–21
Relatively few studies have addressed sleep in hospitalized patients outside the intensive care unit (ICU) and the RCSQ needs to be further tested at regular wards in an acute hospital context. 9 The aim of the present study was to evaluate and compare the outcomes of objectively estimated sleep and self-reported sleep quality. For this purpose, a wrist-worn actigraph and the RCSQ were the assessment tools chosen for the evaluation. To the best of our knowledge no such studies have previously been performed in a Swedish acute hospital.
Methods
This study was a non-experimental prospective pilot study. The research methodology and reporting followed the Standards for Quality Improvement Reporting Excellence (SQUIRE 2.0). 22
Setting and participants
Patients scheduled to be admitted for at least three days on a ward at a hospital in an urban area in Sweden were eligible for inclusion. Two wards at two different hospitals were selected for the data collection period, June 2016 to March 2017. Patients under the age of 18 years or who were unable to understand the purpose of the study due to inadequate language skills or cognitive deficit were excluded. Participants were included consecutively based on the departmental occupancy lists, and all were informed by the same member of the research group. Altogether, 25 patients aged 58 ± 2.2 years (mean ± SEM [standard error of mean]; 17 men and eight women) were enrolled into the study. The recruited patients were instructed to wear the actigraph wristwatch 21 at all times, except in the shower, and to fill out the RCSQ first thing every morning for three days. The first night and morning were considered as a run-in period; only data from Day 2 and Day 3 were used in the analyses.
Measurements
Actigraphy
Actigraph recordings were obtained using Vivago software version PWMI 3400 set to a medium threshold of sensitivity. The wristwatch was placed on the patients’ non-dominant arm to register periods of sleep and wakefulness (sleep/wake patterns) by monitoring body movements, rest, and activity.18,20 Data on the numbers of sleep periods and minutes of sleep were obtained together with graphic presentations of the activity levels. The default settings for sleep time were from 8 pm to noon the following day and for the full 24 hours. In the present study we chose to manually calculate the numbers of sleep minutes from 11 pm to 6 am, which was the time patients could be expected to have a continuous sleep period. The results are presented as sleep minutes (11 pm–6 am) and as percentage of the full seven-hour (420-minute) period. The numbers of sleep periods were obtained from the graphs, and the average numbers of sleep minutes per period (total number of sleep minutes divided by numbers of sleep periods) were calculated.
The Richards–Campbell Sleep Questionnaire
The self-administered RCSQ consisting of six questions is designed to measure the previous night’s sleep through patients’ subjective perception of sleep and sleep quality. Answers to the sixth question about noise during the previous night were not included in the analysis in this study. The five items pertaining to sleep are rated on a visual analogue scale ranging from 0 to 100 mm (0 indicating the worst possible sleep and 100 indicating the best sleep) and the questionnaire takes approximately two to five minutes to complete. 16 The five RCSQ items pertaining to sleep were sleep depth, falling asleep, wakefulness, going back to sleep, and overall sleep quality. A total score for the RCSQ was derived for each patient by calculating the sum of the scores for the five items. A mean score was then obtained by dividing the sum of the scores by five. A total RCSQ mean score was obtained for all patients by summarizing the individual mean scores and dividing by the number of patients. The RCSQ’s internal consistency (Cronbach’s alpha coefficient) has been reported as 0.90 23 and for the Swedish version of the RCSQ, a Cronbach’s alpha of 0.82 was reported. 24
Statistical analysis
Data were assessed for normality of distribution, and a Student’s t-test was used to compare differences between groups and days for each of the three days, for each total mean RCSQ score, and for actigraph data. Both RCSQ scores and sleep minutes obtained from actigraph records were shown to be normally distributed, so comparisons were estimated using parametric analysis, Student’s t-test and Pearson’s correlation. 25 Collected data met the criteria to perform linear regression 25 with normally distributed residuals, a mean of zero, and constant spread. Pearson’s correlations were performed to assess the potential extent of association between sleep minutes and the RCSQ score. We performed a within-group comparison. Intra-class correlation was calculated between sleep minutes and the RCSQ total mean score to assess variance of the data. 26 All statistical analyses were conducted using IBM SPSS statistics version 24. A significance value of P < 0.05 was set for all statistical tests. Only data from Day 2 and Day 3 were used for the statistical analyses. Internal consistency was evaluated by using Cronbach’s alpha with a range of values between 0.00 and 1.00 and the general rule is a Cronbach’s alpha of <0.70 is unacceptable, 0.70–0.79 is acceptable, 0.80–0.89 is good, and >0.90 is excellent. 27
Ethical considerations
The study was approved by the Regional Ethical Review Board in Stockholm, Sweden, Dnr: 2016/768-31/4. Patients were enrolled into the study after providing written informed consent in accordance with the Declaration of Helsinki. 28 Written permission to use the RCSQ and the translated version of the RCSQ were obtained from the copyright holders.16,24
Results
A total of 25 patients were enrolled into the study: 17 (11M/6F) were hospitalized on an orthopedic ward at one hospital, and 8 (6M/2F) at a gastroenterology ward at another hospital. Of 150 possible assessment points over three consecutive days with two recordings (RCSQ and actigraph) per day (25 × 3 × 2), a total of 146 data points were obtained for a response rate as high as 96%. The Cronbach’s alpha was shown to be 0.93 for Day 2 and 0.95 for Day 3. A total of 50 and 46 data points were obtained for Day 2 and Day 3, respectively. Four data points on Day 3 were missed due to early discharge. For comparison, 46 paired data points were used.
No significant differences were revealed between the two days of measurements for the RCSQ total mean scores (Table 1). Furthermore, there were no significant differences between Day 2 and Day 3 for any of the five items as indicated by the data in Table 1.
Self-assessed sleep with Richards–Campbell Sleep Questionnaire (RCSQ) on two consecutive days.
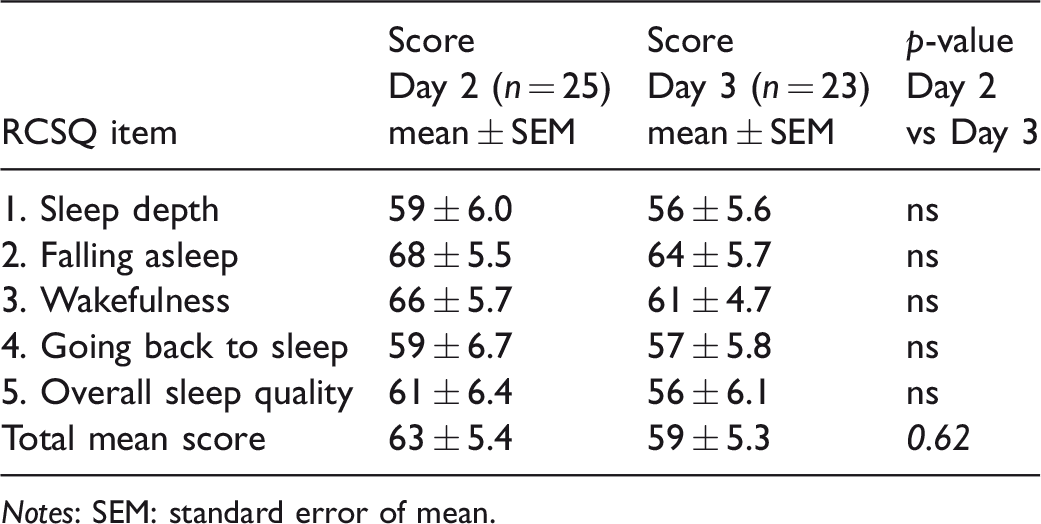
Notes: SEM: standard error of mean.
The total mean scores for the RCSQ varied from 0 to 100 both inter and intra-individually, but no pattern describing better sleep quality on the first or second night of monitoring (Day 2 and Day 3, respectively) was detected. Twelve patients scored higher on Day 3, nine patients scored lower on Day 3 and two patients scored approximately the same low value both days (Figure 1a). Likewise, there was a great variation in the number of minutes of sleep between the two nights inter- as well as intra-individually but again no pattern could be visualized. For 11 patients, a higher number was registered on Day 3, for seven patients a lower number was registered on Day 3, and five patients showed approximately the same number of sleep minutes for both nights (Figure 1b).
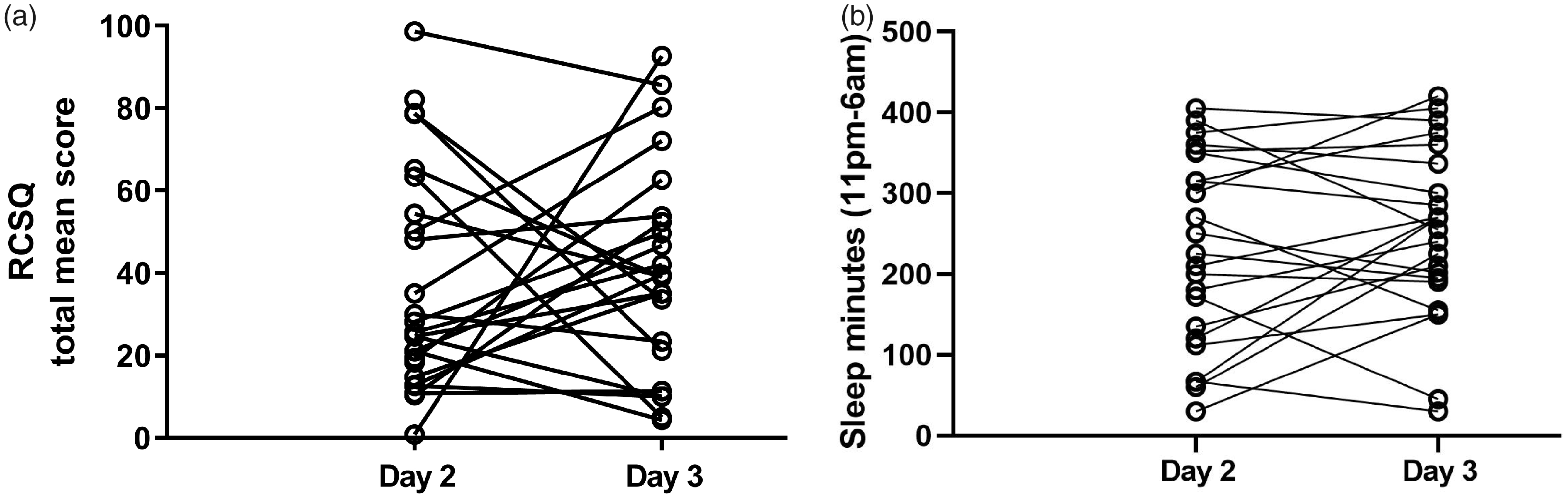
(a) Individual Richards–Campbell Sleep Questionnaire (RCSQ) scores for Day 2 and Day 3 for 23 patients. (b) Individual actigraph scores for Day 2 and Day 3 for 23 patients.
All data obtained by the actigraph and used for evaluation are shown in Table 2. Although the numbers of total sleep minutes were not significantly different between the two days of measurements (230 ± 24 vs 249 ± 22, mean ± SEM, P = 0.55), the intra-individual variation in number and length of sleep periods seemed to be broad, as indicated by the high SEM values (Table 2).
Objective measurements of sleep as assessed with the VIVAGO actigraph.
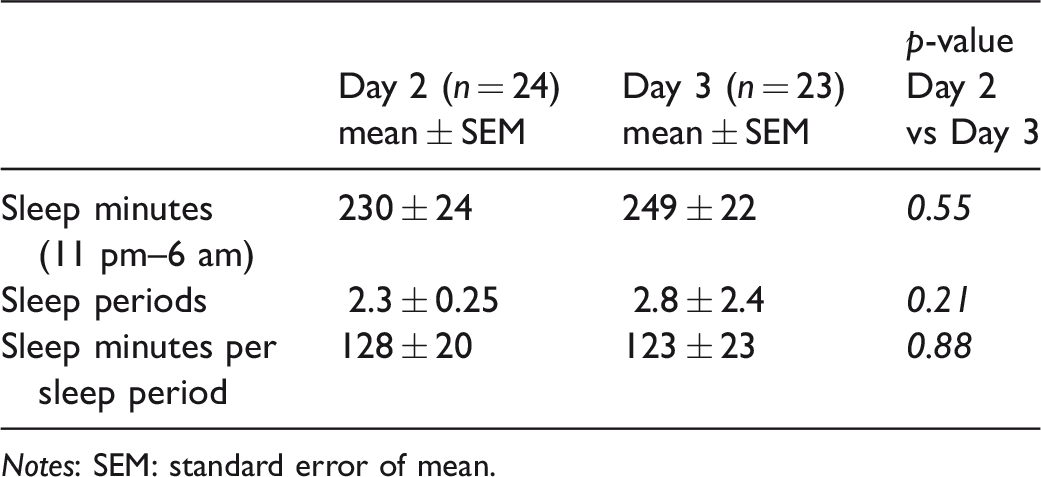
Notes: SEM: standard error of mean.
A significant correlation between RCSQ and actigraph data was obtained for Day 2 (R square = 0.487, P = 0.0002); the degree of explanation for the relationship was about 49% (Figure 2). No significant correlation was obtained for Day 3 (R square = 0.033, P = 0.405).
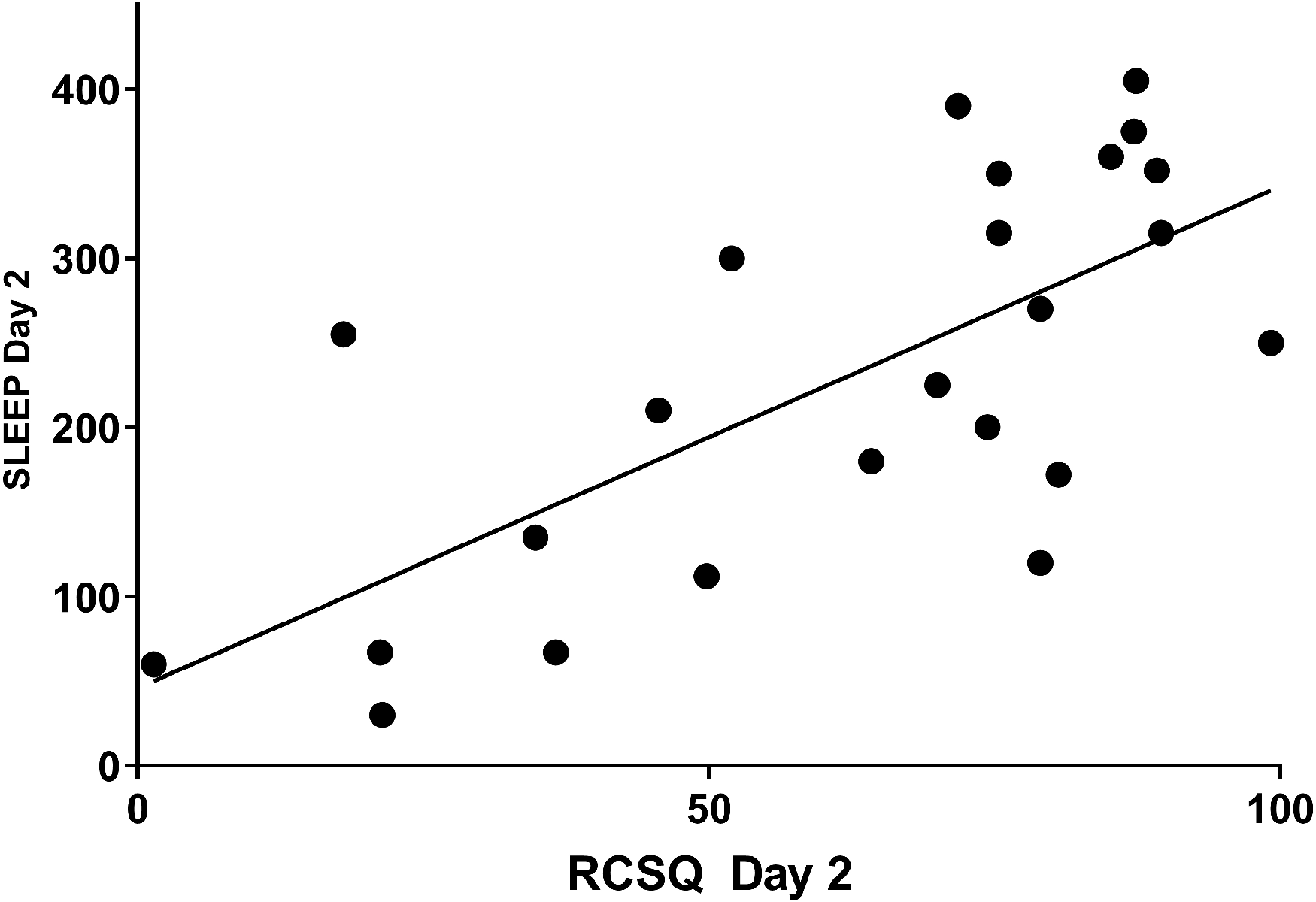
Relationship between Richards–Campbell Sleep Questionnaire (RCSQ) and Vivago® assessments.
A relationship between the two assessments of patients’ sleep was also investigated by relating the patients’ individual self-assessment of sleep during the night, i.e. the RCSQ score, to the activity graph for that particular night. Two typical examples are illustrated in Figure 3: Vivago graphs depicting a ‘bad night’s sleep’ (Subject 18, Day 2, RCSQ mean score = 1.4, sleep = 60 minutes, 14% of the period 11 pm–6 am) and a ‘good night’s sleep’ (Subject 10, Day 2, RCSQ mean score = 89.2, sleep = 352 minutes, 84% of the period 11 pm–6 am), both showing a good agreement between RCSQ score and sleep minutes in individual patients.
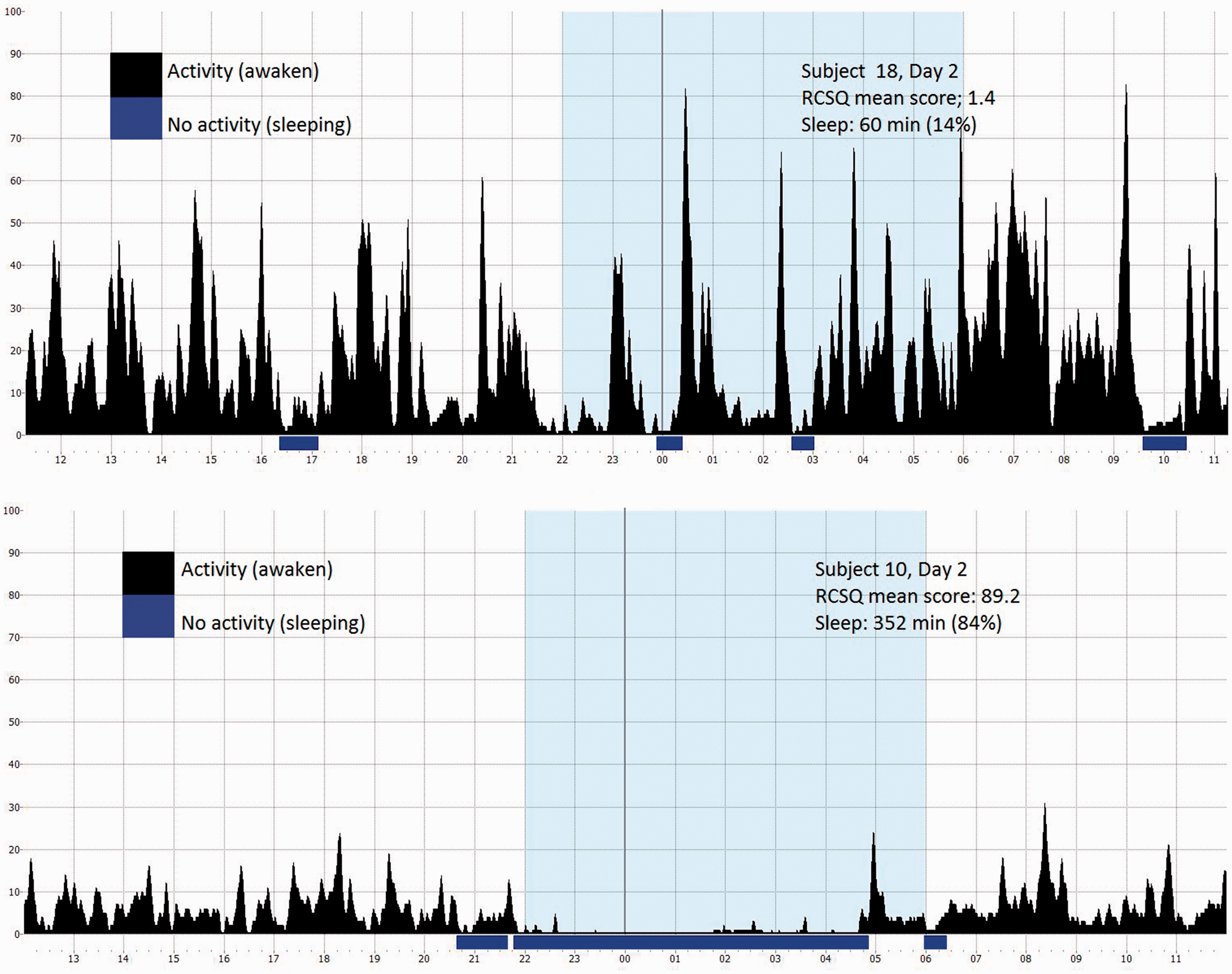
Activity graphs from Vivago® from two patients.
Discussion
The aim of the present study was to evaluate and compare the outcomes of objectively estimated sleep and self-reported sleep quality. The results showed a significant positive correlation between objectively recorded sleep minutes and self-assessed sleep quality (RCSQ) for Day 2, but a similar agreement could not be demonstrated for Day 3.
Our results confirm those of previous studies29,30 showing that actigraphy and subjective measures do capture different dimensions of sleep. Due to this discrepancy it appears that important information obtained by self-assessed sleep may be missed by objective assessments. It is therefore difficult to compare these two approaches clinically. Routinely assessing patients’ subjective experience of sleep with a validated instrument may encourage dialogue, which may in turn indicate the need for sleep-promoting interventions. It may also contribute to person-centered care31–33 in a clinical context where interventions are based on the patient’s perspective. We interpret the results obtained from the self-assessed RCSQ with the understanding that a patient’s subjective sense of sleep quality is affected by several factors, including – but not limited to – the recorded number of sleep minutes. In line with this, the correlation between the RCSQ and number of sleep minutes showed only a 49% degree of explanation, and about 51% was due to other factors. This may be demonstrated by the outliers in Figure 2 with a high RCSQ score but low number of sleep minutes and a low RCSQ score but high number of sleep minutes. A relatively large intra-individual variation was also found in the RCSQ data for Day 2 versus Day 3 (see Figure 1a). Together, these results may indicate that patients do not give their answers without thought, but rather provide a true assessment of their sleep experience the previous night, which adds considerably to the trustworthiness of the questionnaire.
Previous studies of patients’ sleep experiences indicate that psychological factors affect sleep more than environmental factors.34–36 The results of our study indicate through subjective assessments of their sleep quality that patients’ experiences are affected by several factors other than the recorded number of sleep minutes. Although self-assessment by questionnaire is a relatively simple and inexpensive method, self-reporting of sleep may have drawbacks, for example, that daily sleep monitoring may be affected by social desirability bias and misperceptions. 37 Hospitalized patients are very likely to be ill and may have insufficient strength and energy to complete a self-assessment instrument. It is therefore crucial to use an instrument that is easy to understand and appropriate to the context. Several studies with a high response rate have been performed regarding applicability of the RCSQ in ICUs.23,24,34,38 In our study, the external loss justified by early discharge of two patients was almost negligible, with a response rate of 96%. Furthermore, participants in the study who completed the RCSQ left no questions unanswered, thus we had no internal loss. The outcome of our study therefore indicates that the RCSQ may be applicable in a Swedish hospital context. However, it should be considered that the results may be different with a larger study population and with a larger number of measurements.
Data through actigraphy were collected for three consecutive days, but only recordings from Day 2 and Day 3 were used for analysis. In line with this, it is recommended that the actigraph should be worn for a minimum of three days to obtain a comprehensive view of the patient’s sleep/wake pattern. 39 During the course of conducting the current study, another investigation was published 30 that confirmed our design with three days of measurements, suggesting that data from at least two nights should be used when assessing sleep duration.
Considering the broad range of the number of sleep periods and the number of minutes of sleep per period, there appears to be a great variation in sleep quantity and quality, not only amongst patients, but also between days for individual patients, indicating daily sleep assessment as a prerequisite for personalized care. We believe that the RCSQ can provide a good estimate of the overall quality of a patient’s previous night’s sleep and therefore a fair estimation of patients’ sleep during their hospitalization. In agreement with a previous study, 40 we are thus surprised that the RCSQ is not frequently used in a clinical context to assess patients’ sleep. Our results show the importance of further exploring and describing user-friendliness regarding the RCSQ, both from a patient and nurse perspective, in order to implement the RCSQ in a clinical context. Routinely estimating the patient’s sleep is fully in line with person-centered care and may help to highlight this basic need. Furthermore, a routine estimation of sleep increases the opportunities to identify problems within the area, and nursing interventions could be planned and based on an individual level.
Strengths and limitations
One important strength of this study is that the estimation of sleep quality combining subjective assessment and actigraphy was performed in a clinical context. An additional strength was that data were collected for three days in line with recommendations. 30 One limitation of this study could be that the patients were recruited from only two wards which may have affected the heterogeneity of the group. However, it is of great importance to stress that the aim of the study was to compare two means of assessments and not the extent of sleep quality in individual patients. The external validity 41 may be limited due to the limited number of participants in this pilot study, and the result may be different with a larger study population or with a larger number of measurements. Another limitation may be that the comparison between the different outcomes of the two means of assessment are not capturing the same dimension.29,30 The almost negligible occurrence of internal and external loss in the study, together with the evaluated internal consistency by Cronbach’s alpha being 0.93 for Day 2 and 0.95 for Day 3 may be considered as strengths.
Conclusion
The relatively weak relationship between the mean outcomes of the two assessments may indicate that the two measures used in the study capture different dimensions of sleep. RCSQ scoring seems to depend on more variables than number of sleep minutes, indicating a better reflection of the patient’s previous night’s sleep experience. Thus, it may be concluded that the RCSQ in its present form is applicable in a clinical context, in line with person-centered care. The RCSQ, with daily repeated assessments, may help initiate and facilitate nursing actions to promote sleep amongst hospitalized patients. The use of the RCSQ instrument in a clinical context may allow and encourage dialogue about sleep experiences and could enhance the proportion of nursing interventions regarding sleep promotion.
This study illustrates the importance of further investigations that explore and describe the user-friendliness of the RCSQ from a patient perspective. Such knowledge is essential for future implementation.
Footnotes
Author contributions
Gellerstedt, Medin, Kumlin and Rydell Karlsson designed the study. Gellerstedt recruited the participants and collected all data. Gellerstedt, Medin, Rydell Karlsson and Kumlin performed the analysis and drafted the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research was supported by The Sophiahemmet Research Foundations, Capio Research Foundation, Karolinska Institutet, Rut and Arvid Wolff Memorial Foundation.
Conflict of interest
The authors declare that there is no conflict of interest.
