Abstract
The Symptom Checklist: Frequency and Severity Scale (SCL) is a symptom-specific questionnaire that measures the patient’s perception of the frequency and severity of arrhythmia-related symptoms. Worldwide, the SCL is one of the most used symptom-specific questionnaires for this purpose. Until now, there has not been a valid Swedish translation of the SCL. This study aimed to translate the SCL into Swedish and test the face, content and construct validity of the translated version. The questionnaire was translated using the WHO’s recommendations for translation. Using the ‘think aloud method’, we gathered data from 12 patients regarding the intelligibility of the questions. Using exploratory factor analysis (EFA), we evaluated the face validity, construct validity and internal consistency properties of the questionnaire. The study included 646 patients with different types of supraventricular tachycardia, atrial fibrillation (AF) atrial flutter (AFL), atrioventricular reentrant tachycardia (AVRT), atrioventricular nodal reentrant tachycardia (AVNRT), and ectopic atrial tachycardia (EAT). Translation and face validity were found to be acceptable. Construct validity evaluated using EFA and principal component analysis with varimax rotation identified four factors with eigenvalues greater than 1 and factor loading equal to or greater than 0.364, accounting for 58% and 53% of the variance in the frequency scale and the severity scale, respectively. Reliability evaluation revealed high internal consistency. For both the frequency and severity scale, the Cronbach’s alpha coefficient was 0.88. Our results show evidence that the Swedish version of the SCL has good psychometric properties according to face and construct validity and internal consistency for patients with AF, AFL, AVRT, AVNRT and EAT, and is reproducible and valid.
Introduction
Atrial fibrillation (AF), atrial flutter (AFL), and paroxysmal supraventricular tachycardia (PSVT) cause palpitations, fatigue, light-headedness, dyspnoea, and more rarely angina and syncope, all conditions that decrease health related quality of life (HRQOL) and are associated with functional impairment.1–7 Arrhythmias occur in all ages and can be paroxysmal as well as permanent. Treatment strategies include drug therapy, cardioversion, or catheter ablation with or without pacemaker implantation. 6 Since these arrhythmias are very seldom life threatening, the purpose of treatment is to improve the patient’s HRQOL and decrease arrhythmia-related symptoms.8,9
The Symptom Checklist: Frequency and Severity Scale (SCL) version 3, developed by Bubien et al., measures the perception of the frequency and severity of arrhythmia-related symptoms. 10 According to personal communication with the SCL authors (personal mail contact with Louse Jenkins and Rose-Mary Bubien, 14 April 2010), they began developing the SCL (initially Distress) in 1987 in patients scheduled to undergo atrioventricular nodal junction (AVJ) ablation for recurrent or permanent AF or AFL who reported symptoms during trans-telephonic event recordings. Patients were asked to indicate which symptoms they experienced and the degree or severity of distress the symptoms produced. Symptoms were catalogued and the cardiology faculty at the University of Alabama at Birmingham were asked to evaluate the patient-reported list of symptoms for completeness of known symptoms based on their clinical experience. Pilot testing of version 1 was conducted on patients undergoing AVJ ablation and implantation of a pacing system. Patients were assessed prior to, at one month, and at six months following AVJ ablation and pacemaker implantation. In 1993, patients undergoing radiofrequency ablation for atrioventricular reentrant tachycardia (AVRT), atrioventricular nodal reentrant tachycardia (AVNRT), AFL, AF, and ventricular tachycardia were examined. Pilot testing of version 2 demonstrated measurable changes in HRQOL from pre ablation to one-month post ablation. Subsequently, a prospective study was initiated in January 1994 of all patients undergoing ablative therapy over a six-month period. A total of 212 patients were enrolled and the formatting changes resulted in SCL version 3, consisting of 16 items (i.e. symptoms) with one frequency and one severity scale.
In the frequency scale, the respondents mark how frequently they have experienced each symptom (e.g. tiredness or lack of energy, heart fluttering, dizziness, and shortness of breath): never = 0, rarely = 1, sometimes = 2, often = 3, or always = 4. In the symptom severity scale, the respondents rate their symptoms as mild (1), moderate (2), or extreme (3). The symptom severity scale depends on the frequency scale. If a patient answers ‘never’ on the frequency scale, the severity scale is left empty. The raw scores from the SCL range from 0 to 64 for the symptom frequency scale and from 0 to 48 for the symptom severity scale. Permission to translate and revise the SCL (version 3) to Swedish was granted by Louise Jenkins, one of the authors, who also copyrighted the final questionnaire.
The SCL is currently one of the most used symptom-specific questionnaires for arrhythmia patients.3,11–17 As there has been no validated Swedish translation for the SCL, the English version has been used for comparison when developing new symptom-specific arrhythmia questionnaires in Swedish.18–20 It is likely that the SCL was used for this purpose because there were no other suitable translated and validated questionnaires available. To address this shortcoming, this study aimed to translate the SCL into Swedish and test the face, content and construct validity of the translated version.
Methods
Design
The study has a prospective exploratory design.
Sample
Patients referred to Karolinska University Hospital in Stockholm, Sweden, for ablation of AF, AFL, or PSVT between January 2011 and August 2014 and included in clinical studies were consecutively asked to participate in the study. Out of 975, 646 agreed to take part and completed the Swedish version of the SCL before cardiac ablation. There were no differences in terms of arrhythmia, age or gender between patients who accepted to participate and those who declined to participate.
Data collection
The SCL, study information, and informed consent were mailed with a postage-paid reply envelope to the participants two to four weeks before the scheduled ablation. Exclusion criteria were age < 18 years, inability to read or understand Swedish, cognitive impairment, and unwillingness to participate.
Instrument development
The study was performed in several steps. First, the English version of the SCL was translated into Swedish using the recommendations from the World Health Organization (WHO). 21 Next, the translated Swedish SCL was validated using exploratory factor analysis (EFA).
Face validity assessment by expert panel
Per WHO’s recommendations, the first step was a forward translation of the SCL from English to Swedish performed by one of the authors, who was familiar with the English-speaking culture and the terminology of the symptoms for arrhythmias. 21 The second step consisted of a bilingual expert panel of three clinical specialists – one in heart failure, one in electrophysiology, and one in device therapy. The third step was a back-translation to English performed by an independent bilingual translator whose first language is English. Both the expert panel and the independent bilingual translator were tasked with identifying and resolving inadequate expressions/concepts of the translation as well as solving any discrepancies between the forward translation and the existing or comparable previous versions of the questions. The forward-backward translations using the WHO’s recommendations yielded no differences in the descriptions of the items in the SCL.
Content validity
The fourth step was a pre-test in 12 patients (the pre-test group) – six referred for cardioversion and six referred for PSVT ablation. They were interviewed using the ‘think aloud method’ and asked to evaluate the intelligibility of the items in the SCL. 22
The pre-test group found the questionnaire easy to use and to understand, and the items relevant for patients with arrhythmias. Only one minor clarification was made: the heading of the scale was clarified with the word OR between the scales.
Data analysis
To examine the construct validity, exploratory factor analysis (EFA) was performed for the underlying constructs of the questionnaire frequency and severity scales. As such, principal component analysis (PCA) with varimax rotation was applied.23,24 Prior to the this analysis, the Kaiser-Meyer-Olkin (KMO) measure of sampling adequacy and Bartlett’s test of sphericity were applied to analyse whether the data fulfilled the assumptions for carrying out a factor analysis. According to Kaiser’s recommendations for interpreting statistics, KMO values between 0.8 and 1.0 indicate the sampling is adequate, values less than 0.6 indicate the sampling is not adequate. 25
In a factor analysis, eigenvalue can be thought of as index of variance. Each factor yields an eigenvalue, which is the amount of the total variance explained by the factor. 26 Eigenvalues greater than 1 and factor loading equal to or greater than 0.364 were used as acceptable values, which Hair et al deem acceptable in a sample size greater than 300. 27 Internal consistency of the questionnaire was assessed using Cronbach’s alpha coefficient. According to George and Mallery, the general rule is a Cronbach’s alpha coefficient of <0.7 is unacceptable, <0.70–<0.80 is acceptable, <0.80–<0.90 is good, and >0.90 is excellent.28,29 An index of 0.70 or higher was used in this study as a minimum acceptable value. 30
Ethics
The investigation conforms to the principles outlined in the Declaration of Helsinki 31 and the research protocol was approved by the Regional Institutional Ethics Committee. The study was also performed in accordance with the International Conference on Harmonization in Good Clinical Practice Guidelines to protect the rights, integrity, confidentiality, and well-being of the trial subjects. 32 All participants gave their written consent.
Results
Patient population
Demographic characteristics of the study sample (n = 646).
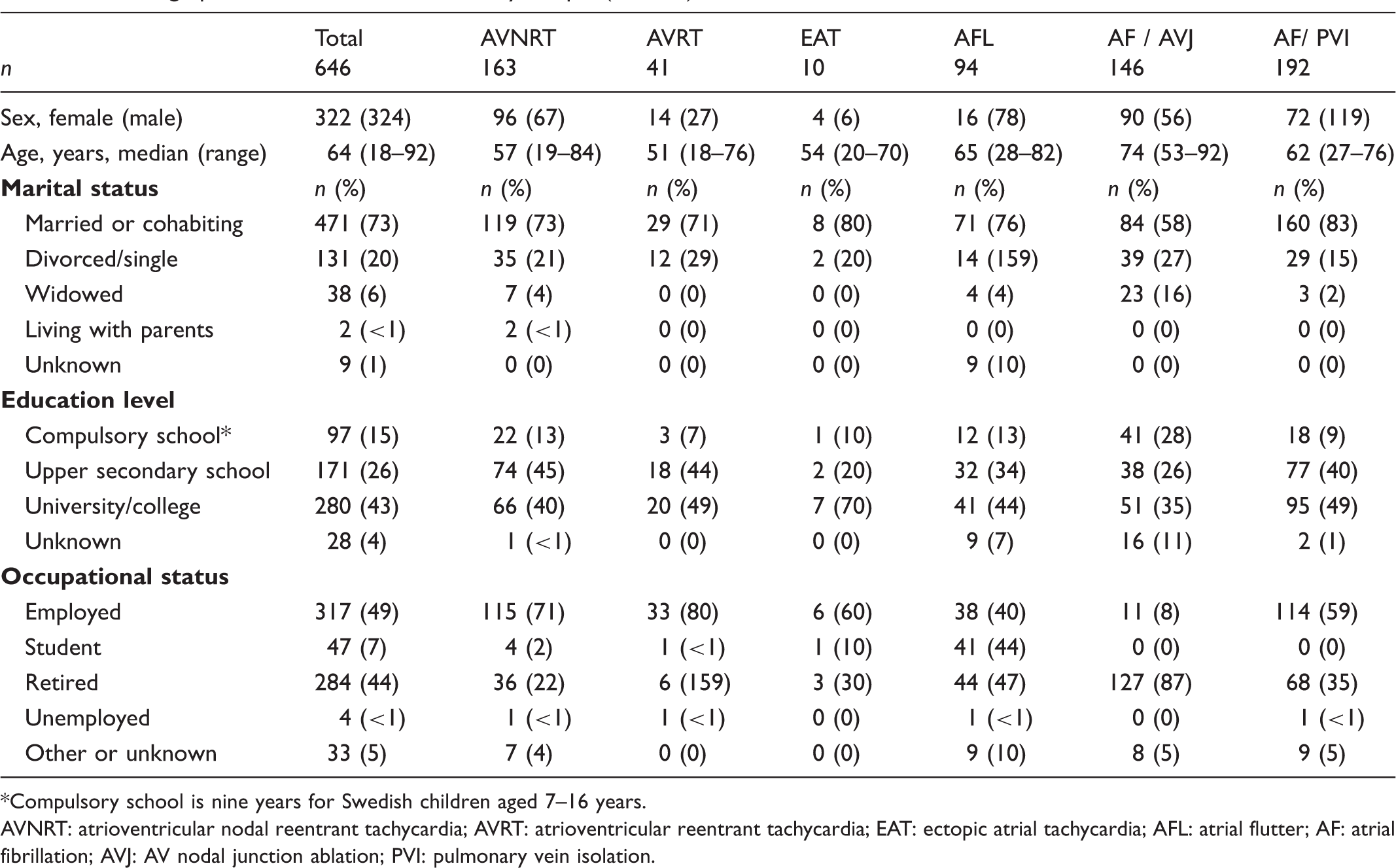
Compulsory school is nine years for Swedish children aged 7–16 years.
AVNRT: atrioventricular nodal reentrant tachycardia; AVRT: atrioventricular reentrant tachycardia; EAT: ectopic atrial tachycardia; AFL: atrial flutter; AF: atrial fibrillation; AVJ: AV nodal junction ablation; PVI: pulmonary vein isolation.
Response rate
Descriptive statistic of the answers on the SCL scales.
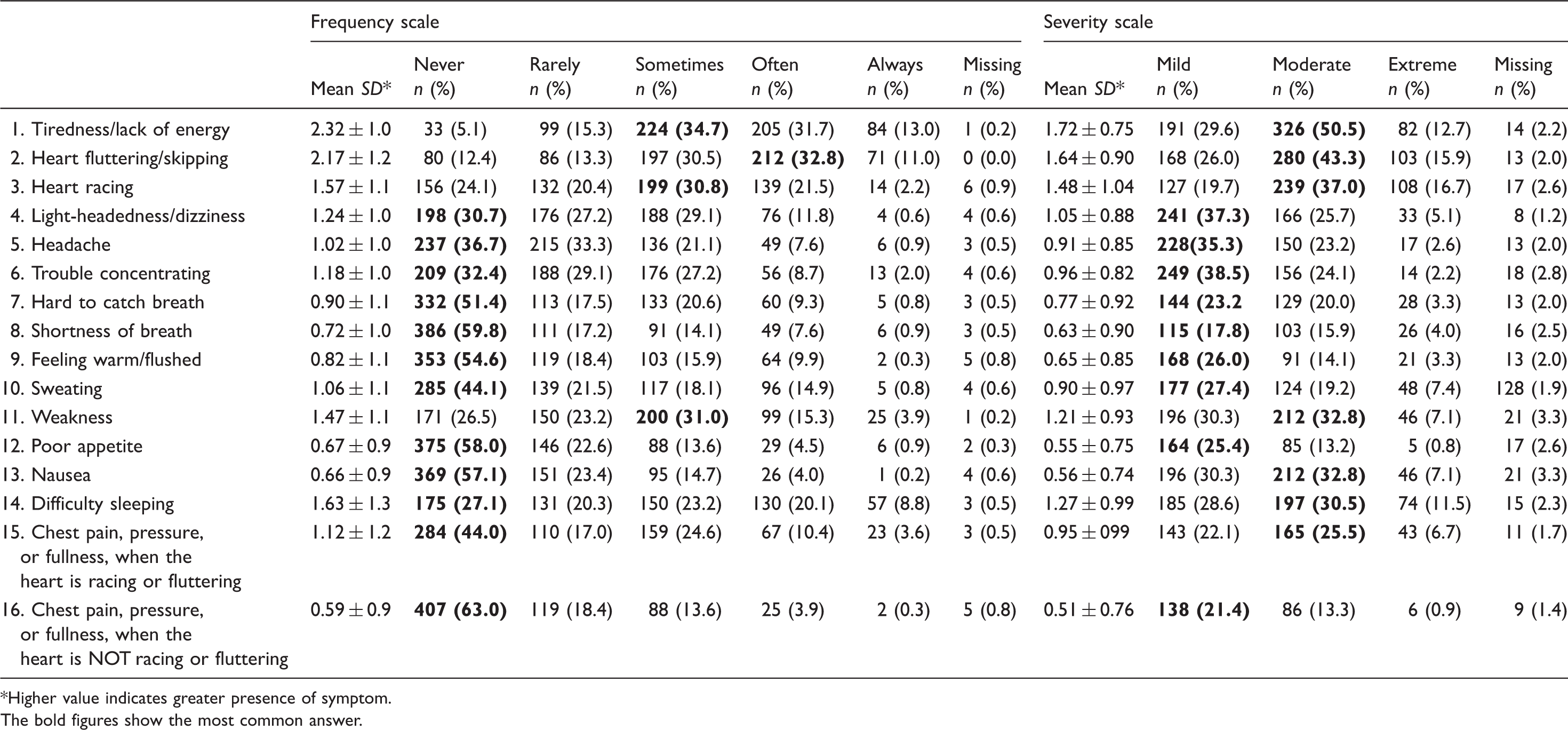
Higher value indicates greater presence of symptom.
The bold figures show the most common answer.
Factor analysis of the symptom severity checklist: Frequency and severity scale
The KMO-index (0.843) and Bartlett’s test of sphericity (<0.001) indicated that the data fulfilled the assumptions for carrying out a factor analysis. Principal component analysis with varimax rotation identified four factors with eigenvalues greater than 1 and factor loading equal to or greater than 0.364, accounting for 58% of the variance observed in the frequency scale:
Factor 1 (Lethargy cluster – representing tiredness, lack of energy, and weakness) includes eight items (1, 4, 5, 6, 11, 12, 13, 14). Factor 2 (Heart failure cluster – representing breathing difficulties) includes two items (7, 8). Factor 3 (Heart cluster – representing tachycardia-related symptoms) includes four items (2, 3, 15, 16). Factor 4 (Feeling of warmth cluster – representing the autonomy turnout) includes two items (9, 10) (Table 3a).
For the severity scale, varimax rotation identified three factors with eigenvalues greater than 1 and factor loading equal to or greater than 0.364, accounting for 53% of the variance observed:
Factor 1 (Lethargy cluster, see above) includes eight items (1, 4, 5, 6, 11, 12, 13, 14). Factor 2 (Heart failure cluster, see above) includes two items (item 7, 8). Factor 3 (Heart cluster, see above) includes five items (item 2, 3, 4, 15, 16) (Table 3b).
Reliability
Results obtained from exploratory factory analysis of the SCL frequency scale.
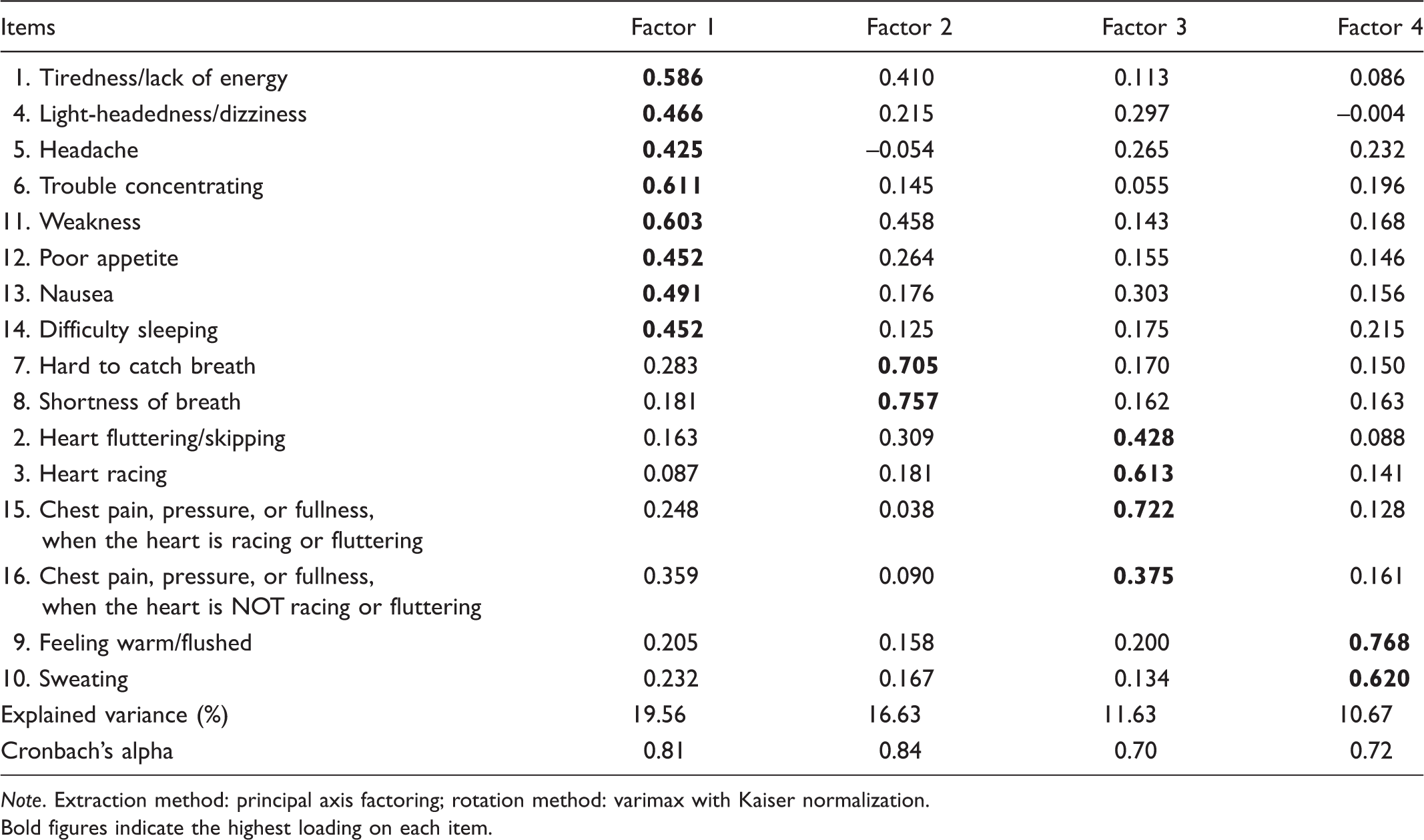
Note. Extraction method: principal axis factoring; rotation method: varimax with Kaiser normalization.
Bold figures indicate the highest loading on each item.
Results obtained from exploratory factory analysis of the SCL severity scale.
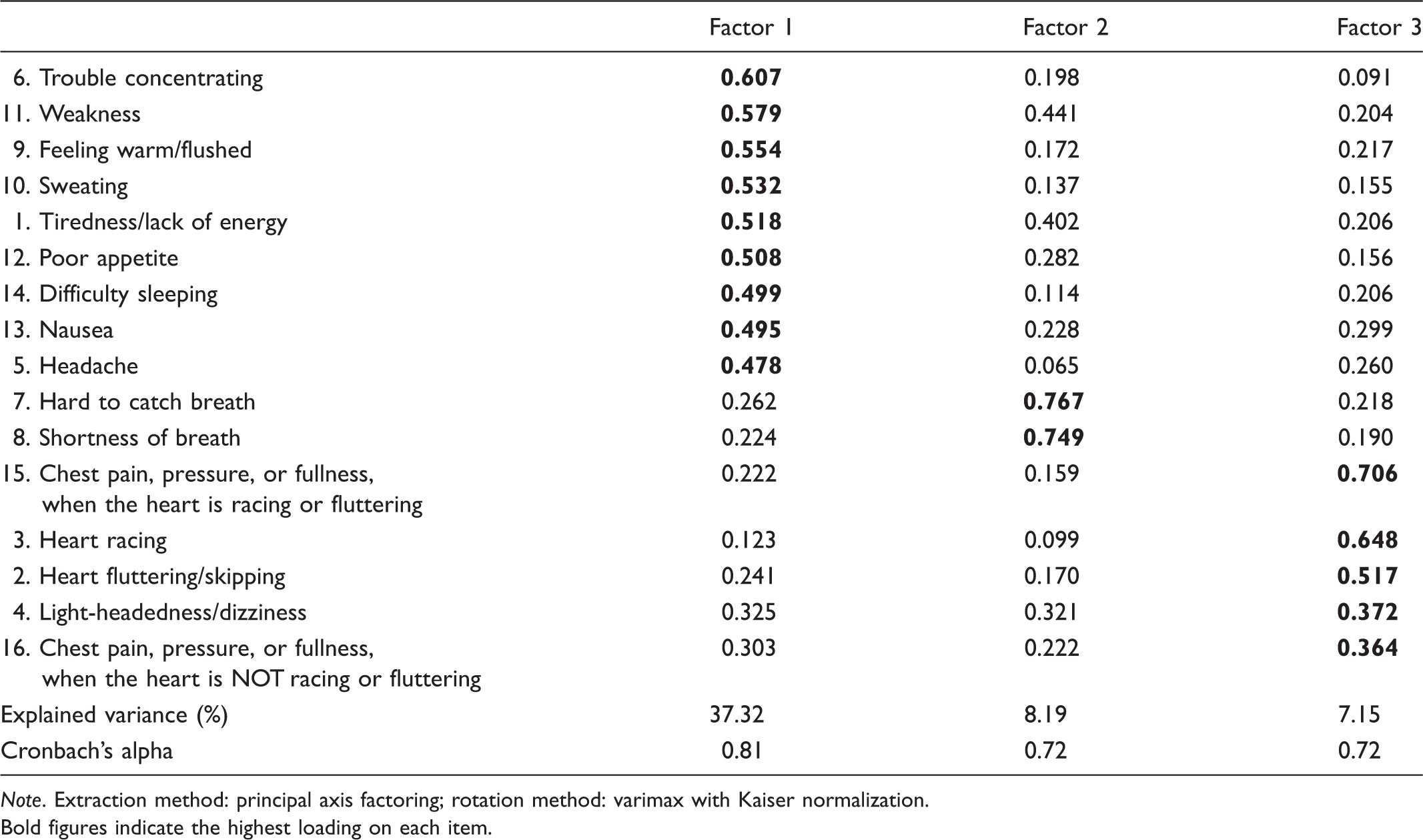
Note. Extraction method: principal axis factoring; rotation method: varimax with Kaiser normalization.
Bold figures indicate the highest loading on each item.
Discussion
The present study evaluates the face and construct validity, and internal consistency of the Swedish translated version of the SCL to assess the patient’s perception of arrhythmia-related symptoms. The results indicate adequate psychometrics properties and provide evidence that the SCL is valid and reliable in a large sample of Swedish patients with arrhythmia-related symptoms.
Response rate
Response rates for the frequency scale and the severity scale were 94% and 84%, respectively. The lower response rate for the severity scale might indicate that it is easy to forget to tick answers in the second table. The reliability measured with Cronbach’s alpha coefficient was found to be acceptable in both the frequency and severity scales according to the recommendations by DeVellis. 29
Exploratory factor analysis
Watson and Thompson have reported that standards for using and reporting factor analysis results are variable. 33 In this study, we chose to use eigenvalues greater than 1 and factor loading equal to or greater than 0.364 as our study size was large (646 participants). Comrey and Lee classify study sample size as follows: 50 = very poor, 100 = poor, 200 = fair, 300 = good, and 1000 = excellent. 34 The exploratory factor analysis indicated a four-factor solution for the Swedish version of the SCL frequency scale namely, ‘lethargy cluster’, ‘heart failure cluster’, ‘heart cluster’, and ‘feeling of warmth cluster’. The severity scale results indicated a three-factor solution: ‘lethargy cluster’, ‘heart failure cluster’, and ‘heart cluster’. Items in the feeling of warmth cluster in the severity scale were included in the lethargy factor according to the loadings. In the severity scale we found factorial complexity with two items: tiredness/lack of energy and weakness. Both these items correlate to the lethargy and heart failure cluster. After discussion, we deemed that these items correlate most to the lethargy factor. Streur et al, looking at symptom clusters in patients with permanent AF, found that the tired cluster was similar to symptoms in patients with heart failure. 35 AF and heart failure are well known to commonly coexist. 36 The most common symptoms in the study were tiredness/lack of energy, weakness, heart fluttering/skipping, and heart racing experienced as mild to moderate. Patients with arrhythmias, particularly AF, often have other cardiovascular illnesses, which can result in similar symptoms such as those. 37 Consequently, in some cases reported symptoms may be due to other factors, e.g. heart failure, and not to the arrhythmia per se.
In EFA, variance should be as high as possible. Stevens recommends a variance of 75% or more; 38 however, Henson and Roberts found that if the extracted factors explain on average 52% of variance 39 this will be acceptable; results that correspond to what we found. The total variance for the frequency scale and the severity scale was 58% and 53%, respectively.
Translation
According to Maneesriwongul and Dixon, information about translation processes is often inadequate and varies widely. 40 Consensus is that a minimum standard for applying an instrument to another language is that it should include back and forward translation. 40 Therefore, we followed the WHO’s recommendation, including a back and forward translation. 21 The pre-test group deemed the translated version of the SCL as user friendly and easy to complete during the think aloud test.
The authors of the SCL recommend that the reliability of the two scales (frequency and severity) of the SCL should be calculated using Cronbach’s alpha to estimate the internal consistency at each data collection point. To avoid missing data, we emphasize the importance of concrete directions about how to complete the questionnaire. We also recommend combining the SCL with a generic questionnaire for comparison and to provide systematic knowledge of the self-reported health in this special patient group.
Limitations and strengths
Some limitations should be considered. The patients were consecutively recruited from four ongoing studies at our institution where the SCL was used to evaluate the arrhythmia symptoms pre and post ablation. Our data were sampled and retrospectively analysed, which made it impossible to have a 100% response rate. On the other hand, the study sample consisted of a large population, a strength of this study despite the different sizes of the arrhythmia groups.
Questionnaires with few response alternatives similar to the SCL scale with the data on an ordinal scale can be handled in different ways. In this study we chose a parametric method instead of a non-parametric method to handle the ordinal data from the questionnaire. According to Polit and Beck, this is possible if the requirement for normal distribution is satisfied by the variables in a questionnaire. The summation of the results from the different questions in the sub-scales to horizontal main scales means that they latter become parametric. 41
Conclusion
Our results show evidence that the Swedish version of the SCL has good psychometric properties according to face and construct validity and internal consistency for patients with different types of supraventricular arrhythmias and is reproducible and valid.
