Abstract
Introduction
Coronary artery disease (CAD) is the leading cause of death worldwide. Lifestyle change is a crucial part of secondary prevention. Only 30% of CAD patients follow the corresponding guideline recommendations. The widespread adoption of smartphones offers the opportunity to integrate secondary prevention into the daily routine of CAD patients.
Methods
We developed an app to integrate secondary prevention into CAD patients’ everyday life (smartphone-guided secondary prevention, SGSP). The app provided a daily 15-minute program that included video-guided exercises, video sessions with background information about CAD, and a tool to record blood pressure and heart rate once a day. The SGSP app was tested with the primary outcome of 28-day adherence. The secondary outcome was a composite of (1) self-reported behavioral changes, (2) gain of knowledge about cardiovascular risk factors, and (3) an increase in quality of life.
Results
Of the 66 patients screened, 43 (65%) were included into the study and, of those, 17 (40%) used the app continuously for 28 days. From this group, 14 (82%) were physically more active and ten (59%) improved their dietary habits. Usage of the SGSP app was also associated with a gain of knowledge about cardiovascular risk factors (70% physical activity, 59% healthy diet).
Conclusion
The regular use of a SGSP app appears to support lifestyle changes in patients with CAD.
Keywords
Introduction
Across the globe, coronary artery disease (CAD) is the number one cause of death. 1 A reduction of cardiovascular risk factors (CVRF) has been shown to significantly lower the occurrence of cardiovascular events in CAD patients.2,3
In addition to the administration of several effective drugs and coronary revascularization, a healthy lifestyle that includes regular physical activity, following a healthy diet, and avoiding tobacco consumption can have a major impact on preventing secondary cardiovascular events.2,3
Previous studies have shown that just 15 minutes of moderate physical activity per day are sufficient to significantly reduce the risk of cardiovascular-related death.4–6
However, the compliance among the patients to change their lifestyle in accordance with these recommendations is low.2,7–9 Recent data indicate that only 30% of CAD patients follow the recommendations of the current guidelines. 2 This means that 70% of acute coronary syndrome patients fail to make the minimal behavioral adjustments needed to significantly reduce their risk of a heart attack. 2 Only 59% of CAD patients exercise in a sufficient manner and only 22% take part in cardiac rehabilitation programs.7–9 When CAD patients were asked make lifestyle changes after a cardiovascular event, only 49% stopped smoking, 40% reduced their consumption of saturated fats, and 35% increased their consumption of fruit and vegetables.2,8,9
There are many different causes that may lead to this lack of compliance. First of all, medical doctors often do not have enough time to explain the importance of these lifestyle changes in the hectic environment of their day-to-day clinical practice. Second, patients might find it difficult to implement the required changes into their everyday life. Finding time to fit regular physical activity into a normal day can be a problem for patients. This problem is further aggravated by uncertainty over the safety and correct performance of certain exercises. Furthermore, it has been shown that long distances to cardiac rehabilitation centers discourage patients from going there. 7
To explore a possible solution for this problem, we tested an app-based approach allowing participants to integrate secondary prevention into their everyday life in patients with CAD. In this study, we show that a smartphone app, which was specifically designed based on the patients’ needs, appears to support lifestyle changes for patients with CAD.
Methods
App development
To develop a mobile app-based tutorial, especially for patients with CAD or patients at high cardiovascular risk, we created a questionnaire about the app features that patients find important to implement secondary prevention into their everyday life. Both out-patients and in-patients with CAD were asked to express their interest (yes or no) regarding the following opportunities: (1) educational sessions, covering basic facts about cardiovascular risk factors, (2) video-guided physical exercises, (3) monitoring function for blood pressure and heart rate, (4) a chat-based health coach, and (5) medication tracking. Prior to the development of our app, some of our patients tested the video-guided physical exercises and gave their feedback.
App functions
The digital program (app) was created based on the responses from the patient’s questionnaire and is intended to be used daily for 10–15 minutes.
For our study, participants were required to use the app at least five out of seven days per week. Every day, participants were recommended to watch a three- to four-minute-long educational video about cardiovascular risk factors and how participants can actively influence them. These videos included basic facts about cardiovascular disease, information about the importance of medication and fundamental facts about heart-healthy nutrition. The app also presented a daily ten-minute video-based physical exercise program. Here, a professional fitness trainer demonstrated the exercises for participants and further explained what problems to watch out for. After the participants had completed the knowledge-based section and the exercise section, green check marks appeared to indicate that both units had been finished successfully for this day (Figure 1). The program content was not personalized. Furthermore, the participants were required to measure their blood pressure and heart rate with their own devices and to document them once a day in the app (Figure 1).
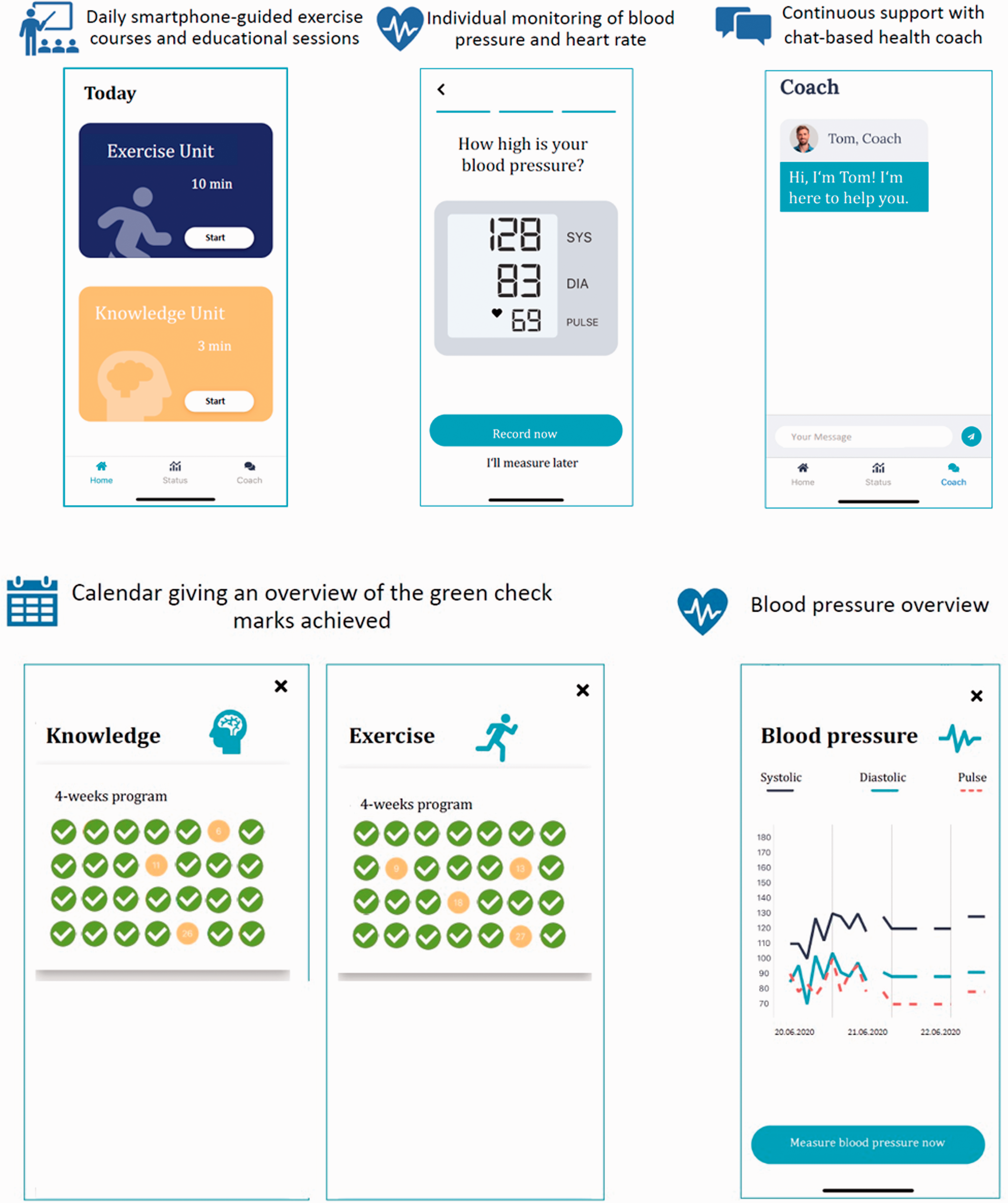
Overview of the different app functions.
To give the participants an overview of what they had achieved so far, the app contained a status section. In a monthly calendar view, participants could monitor how many green check marks they had collected for the physical activity and knowledge units. Based on the daily values entered for blood pressure, a blood pressure curve was shown as an overview in the status section (Figure 1). Another important feature of the app was the option to contact a health coach to solve technical issues for the participants if needed. The smartphone app was jointly developed by the Heart Center Bonn and Vantis GmbH.
Trial population
Patients with known CAD or at least three cardiovascular risk factors were enrolled between November 1 and December 13, 2019. The inclusion criteria were as follows: (1) age between 18 and 75 years, (2) presence of acute or chronic coronary syndrome or at least three cardiovascular risk factors, (3) access to a smartphone and the ability to use it, (4) being physically active (Frailty Score ≤3), and (6) sufficient language proficiency. Participants with dyspnea (New York Heart Association classification, NYHA >III) or a malignant primary disease were excluded. The mobile operating system of their smartphone was not relevant. The app software was available on the Apple App Store (iOS) and the Google Play Store (Android).
Out of the 66 participants who were assessed for eligibility, a total of 43 (65%) were enrolled in the study (Figure 2). From those participants, 27 (63%) had been hospitalized and 16 (37%) were enrolled from the out-patient clinic. From the 23 participants that were not enrolled in the study, 11 were excluded because they did not own a smartphone and 12 declined to participate, mainly because they were aware of or following the guidelines already (Figure 2).

Flow chart of the study, from the patients’ questionnaire prior to development of the app to the screening, inclusion, and follow-up of the participants.
Follow-up and outcomes
Included participants were followed up with 28 days after the start of the app usage. The primary outcome was 28-day app adherence (i.e. usage of the app for at least 20 days within the 28-day study). The secondary outcome was a composite of (i) behavioral changes (increased physical activity and implementation of a healthier diet), (ii) a gain of knowledge about cardiovascular risk factors, such as lack of physical activity, unhealthy diet, stress, and smoking, and (iii) a change in the patient’s quality of life.
Prior to the start of the trial, candidates were asked to assess their knowledge of the above-mentioned cardiovascular risk factors on a scale from 1 (very good knowledge) to 5 (no knowledge at all). Additionally, they estimated their level of physical activity in minutes per week and rated their implementation of a healthy diet on a scale from 1 (always) to 5 (never).
At the end of the study, the adherence and the secondary outcomes were assessed through a telephone interview. The participants were asked whether they had used the app regularly (five out of seven days per week). Only participates that reported regular usage were asked for a second time to rate their knowledge of cardiovascular risk factors, their level of physical activity, and their implementation of a healthy diet. Then, the pre-study to the post-study results of the adherent patients were compared. An increase of one point or more was defined as an “increase in physical activity”, “implementation of a healthy diet”, or “relevant gain of knowledge”. To measure the quality of life, we have chosen two questions from the 12-Item Short-Form Health Survey (Did you have more energy while using the app? Did you experience less limitation in physical activity while using the app?) and added another question that we found important (Did you experience an improved feeling of being able to positively influence the disease by using the app?). These questions were yes/no questions. Moreover, the reasons for non-adherence were obtained by open questions (Figure 2). Written informed consent was obtained from all participants.
Ethics approval for the study was granted by the ethics committee of the Medical Faculty of the University of Bonn (Nr: 234/19).
Statistical analyses
Data are presented as the mean ± SD or as the median and the interquartile range. Categorical variables are given as percentages. For continuous variables, a Student t-test was used for comparison between two groups. For categorical variables, Fisher's exact test was used. All tests were two-sided and a p value <0.05 was considered statistically significant. Statistical analyses were conducted by using GraphPad Prism version 8.0.0 for Windows, GraphPad Software, San Diego, California USA, www.graphpad.com.
Results
App development for secondary prevention based on a patient questionnaire
We questioned 20 outpatients and hospitalized patients at the Heart Center Bonn about the features that they were interested in for an app to help prevent heart disease. Each of the included patients suffered from CAD. Among them, 13 (65%) were male and seven (35%) were female. The mean age of the group was 62 years and the mean BMI 30 kg/m2. Five patients (25%) were current smokers. In total 17 patients (85%) rated their implementation of a healthy diet as good or very good. The education level of the participants was not obtained.
Overall, 17 patients (85%) were interested in using educational video sessions, 14 (70%) in video-guided physical activity, and 11 (55%) in monitoring. Five (25%) were interested in a chat-based health coach and ten (50%) in medication tracking.
Based on the obtained information, an app was developed to contain daily educational and physical activity sessions, blood pressure and heart-rate monitoring, and optional access to a health coach.
Participants characteristics
The baseline characteristics of the 43 participants enrolled in the app usage part of the study are demonstrated in Table 1. The mean age was 60.5 years. From all participans, 33 (77%) were men, 32 (74%) had hypertension, 26 (61%) had hyperlipidemia, and only 12 (28) had diabetes. Eight participants (19%) were current smokers and 19 (44%) were former smokers. With regard to concomitant diseases, CAD was present in 41 (95%) of the participants, 19 (44%) of the participants had a history of myocardial infarction <6 months prior to the study, and 14 (33%) had a history of myocardial infarction >6 months. One participant had a history of stroke (Table 1). There were no significant demographic differences between the adherent and non-adherent groups.
Baseline characteristics.
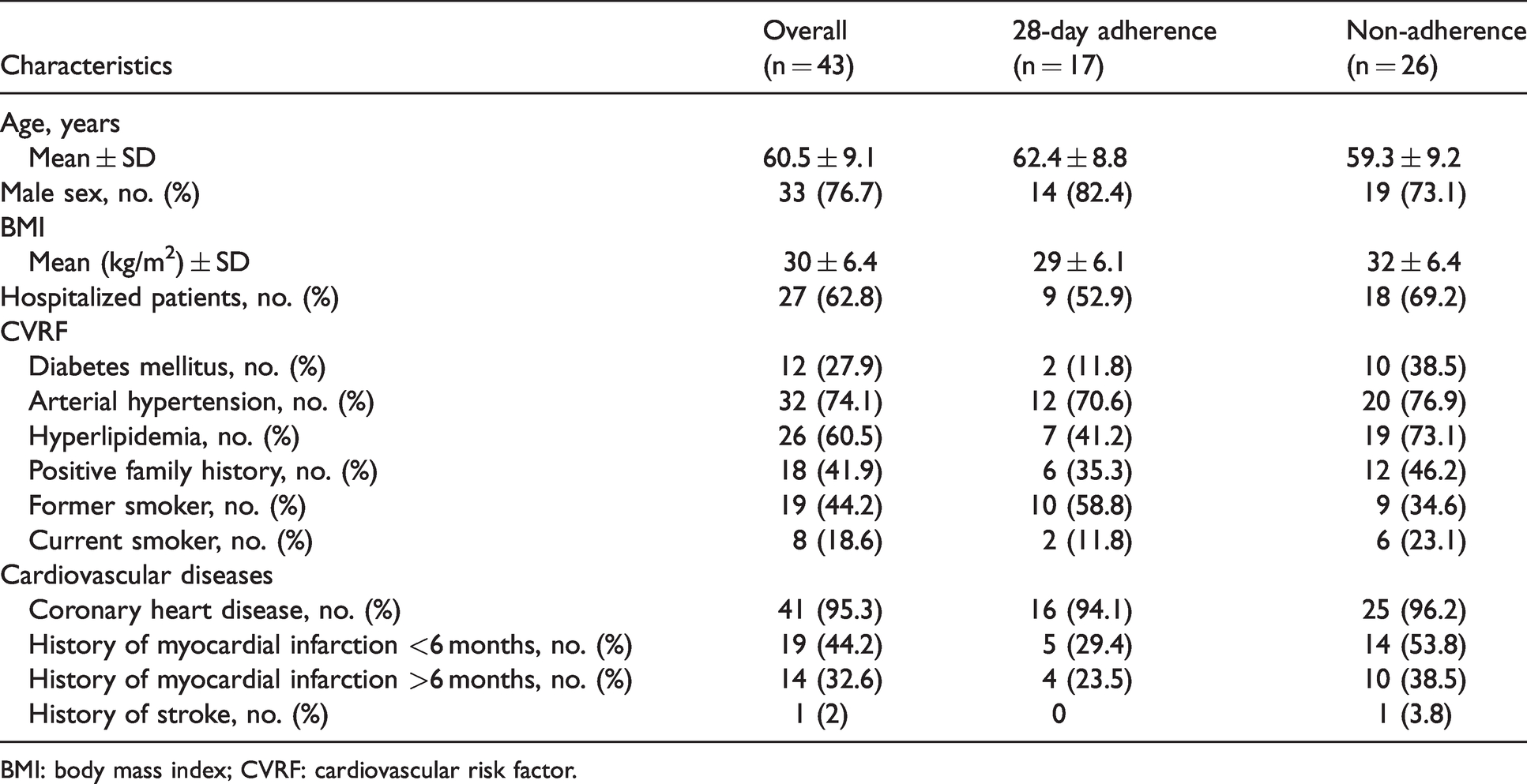
BMI: body mass index; CVRF: cardiovascular risk factor.
Outcomes
Primary endpoint: app usage for 28 days
Out of the 43 participants enrolled in the study, 17 (40%) met the criterion for 28-day adherence by using the app for at least 20 days. The primary cause of non-adherence was a loss of interest (15 patients). Three patients stopped using the app because of health problems, three others because of technical problems, two of the patients were on vacation, and three were hospitalized or were undergoing rehabilitation and could no longer participate (Figure 1).
There was no significant difference between the outpatients and hospitalized patients (50% vs 33%, 95% CI, p = 0.343) regarding adherence to the app (Table 2).
Primary and secondary endpoints.

CVRF: cardiovascular risk factor.
Secondary endpoints: physical activity, lifestyle, and quality of life
Next, we explored whether regular use of the app changed the physical activity, lifestyle, knowledge about cardiovascular disease, and quality of life of the participants. Out of the 17 participants, who used the app regularly, 14 (82%) stated that they were physically more active than before using our program. Ten participants (59%) reported that they followed the nutritional recommendations and changed their dietary habits based on the knowledge provided in the videos (Table 2). Compared to the beginning of the study, 12 participants (71%) stated that their knowledge significantly increased regarding the importance of physical activity to reduce the risk of cardiac events. Ten participants (59%) indicated that their knowledge improved concerning the dietary aspects of heart disease (Table 2).
In addition, participants with 28-day adherence reported a better quality of life while using the app. Nine participants (53%) had more energy and nine (53%) felt less limited in physical activity during and after usage of the app. 15 participants (88%) reported a positive feeling about the opportunity to actively influence the progression of the disease (Table 2).
Finally, we aimed to determine whether participants who completed the 28-day program would be willing to use the program for a longer period of time. Of the participants who successfully finished the 28-day program, 15 (88%) showed a high interest in using the app for a longer period of time (Table 2). This was consistent amongst outpatients and hospitalized participants (Table 2).
Discussion
Coronary artery disease is the leading cause of death worldwide. 1 Previous studies have proven the importance of lifestyle changes as part of secondary prevention measures.2,3 However, 70% of patients with CAD do not follow the current secondary prevention recommendations. 2
A lack of knowledge about the benefits and limited access to conventional rehabilitation programs might be a possible explanation for such incompliance.10,11
Therefore, it appears that the available secondary prevention programs are insufficient for establishing successful secondary prevention measures in a patient’s everyday life.
Increased smartphone adoption amongst elderly patients provides an opportunity to begin to guide lifestyle changes via a smartphone app.12,13
The efficiency of healthcare apps has already been shown in patients with back pain, COPD, and diabetes.14–16 In a study by Toelle et al., the regular use of the multidisciplinary mHealth back pain app (Kaia app) led to lower pain intensity in patients with back pain. 14 Rassouli et al. reported that a 20-day digitalized pulmonary rehabilitation program (via the COPD Kaia app) had a positive effect on health-related quality of life in patients who completed the 20-day program. It is particularly noteworthy that the above-mentioned app provides daily physical exercises, educational videos, and mindfulness techniques for coping with the disease, similar to our Vantis app.15 The potential for app usage among CAD patients has not been systemically studied so far. To address these issues, we tested the adherence to SGSP in CAD patients and the impact it had on lifestyle changes.
Among all 66 patients screened for inclusion, 43 (65%) were willing to take part in the study. Our data revealed that 55 out of 66 screened patients used a smartphone (83%), confirming a high penetration of smartphones in this patient segment. This exceeds previous studies, showing that 71% of cardiac patients use smartphones and 55% use them especially for health problems. 13 The balance of sexes in our study reflects the usual demographic distribution for CAD patients. 1
The primary endpoint of our study was app adherence, which was defined as app usage in 20 out of 28 days. Of the 43 enrolled patients, 17 (40%) reached the primary endpoint of app adherence. The most common reason for non-adherence was a loss of interest (15 patients). Some participants evaluated the video-guided exercises as too exhausting, others as too simple. The same opinion was expressed about the educational videos. Therefore, we estimate that an app that is more personalized and technically more robust will achieve higher adherence rates. One way to achieve this goal would be to evaluate the participants physical and educational status prior to usage of the app and then adjust the exercises to the patient’s fitness and education levels.
Additionally, using gaming elements like goal setting, challenges or recognition of achievements might motivate and engage people to use the app more regularly. People have been shown to be motivated to achieve activity goals through game mechanics, rather than chasing intangible outcomes.17,18 A pilot study has already tested game strategies for CAD patients, which reported high levels of engagement. 19 The display of green check marks in our app does this to some extent. Gaming strategies like progress and health level transparency, tracking improvement of the “cardio age” and risk score, as well as reminders to use the app will be integrated into future versions of the app.
Analysis of the secondary endpoints revealed that after only 28-day usage of the app already an increase in physical activity, a change in nutritional habits, and an increase in knowledge about CVRF could be observed. These results indicate regular usage of the app supports patients with CAD in changing their lifestyle, towards more physical activity and a heart-healthy diet.
Furthermore, using an app as a channel to provide knowledge about CAD leads to better informed patients. The relatively modest increase in knowledge about smoking (6%) as a risk factor may be contributed to the low incidence of current smokers in our study population (two patients) and the widespread existence of smoking cessation programs. However, other areas of cardiovascular knowledge increased greatly.
Interestingly, we detected no significant differences regarding our primary and secondary endpoints between hospitalized patients and outpatients, indicating that the app might be suitable for all patients, including those with a recent coronary intervention as well as outpatients with stable coronary disease.
One major strength of our app-based secondary prevention approach was the convenience it provided for patients. It was easy to integrate the program into their daily routine, including their work schedule and other obligations. The program could be used at any time and any place.
However, it is still unclear how long the app should be used to engender lasting behavioral changes that lead to a reduction of cardiovascular risk. We could also not rule out that the positive effects of SGSP decline after completion of the program.
Previous studies have shown that participation in rehabilitation programs after a cardiac event, even for a short time (28 sessions), decreased morbidity and mortality.20,21 In a study by Chow et al., it was demonstrated that adherence to a healthy diet and exercise for just six months was already associated with a decreased risk for myocardial infarction, when compared to non-adherence. 2 Hence, we hypothesize that even time-constrained usage of an SGSP app, for example for 6 to 12 months, may already improve cardiovascular risk factors and decrease cardiovascular mortality and hospital admissions. This should be explored in future studies with larger patient numbers, a longer follow-up, and a more personalized app.
Limitations
This study has methodological and technical limitations. The main methodological limitations are the limited patient number, a short follow-up period, the assessment of endpoints based on self-reported outcomes, and the lack of a control group. The main technical limitation is the rudimentary nature of the app, which lacked personalization and was developed in a short time on a small budget.
Conclusion
This study indicates that smartphone apps can facilitate secondary prevention for CAD patients. Regular use of the app appears to increase physical activity and improve dietary habits of patients suffering from CAD. Most of the participants also reported a relevant gain of knowledge about cardiovascular risk factors and a better quality of life. However, additional studies using a more personalized app, tested in a larger number of patients, and with a longer follow-up period need to be done to confirm the results of this preliminary study. Long-term smartphone app-guided secondary prevention could be a novel tool to reduce the burden of cardiovascular disease.
Footnotes
Disclosures
F. Jansen is a shareholder of Vantis GmbH.
Acknowledgments
We thank Dr. Meghan Lucas for her critical reading of the article.
Guarantor
Irina Eckardt and Felix Jansen.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: F. Jansen is funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) – JA 2351/2-1 and Project-ID 397484323 – TRR 259 and the Corona Foundation. G. Nickenig is funded by the DFG Project-ID 397484323 – TRR 259.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
