Abstract

Background
Regular use of neuromuscular electrical stimulation (NMES) for functional applications can produce changes in stimulated muscles that increase soft tissue health. This has spurred numerous attempts to develop clinical stimulation systems for weight-shifting and pressure injury (PrI) prevention, for example in wheelchair-bound individuals who use their chairs for hours at a time. For these individuals, stimulation of the gluteus muscles and subsequent contraction can cause weight shifts that can substantially reduce PrI development. 1
Surface gluteal electrical stimulation (ES) was first reported and patented over 25 years ago.2,3 Unfortunately, surface NMES has limited efficacy in providing repeated and reliable gluteal contractile responses both clinically and practically. The inferior gluteal nerve lies relatively deep to the buttock surface and close to the sciatic nerve. Reliable surface electrode placement in the upper buttock region for preferential recruitment of the inferior gluteal nerve is difficult to achieve. 4 Repeated application of electrodes to the same area of skin can in itself cause skin damage. 5 In previous work, study participants commented that they were motivated to join a study to evaluate the use of surface ES because they felt there was no effective approach for pressure injury prevention currently available to them. However, they found the surface NMES system cumbersome and reported that daily placement of the electrodes was time-consuming. They stated that they would be unlikely to consider long-term use of a surface NMES system due to these limitations. 6 While use of a custom-fitted garment may alleviate some placement issues; these systems take more than 15 min to don or doff. 7 Weight gain makes any fitted garment challenging to put on and this is exacerbated for individuals with limited mobility. Any increase in subcutaneous fat will also raise the stimulus current required to achieve a contractile response. 8 No surface NMES systems have enabled automated weight-shifting. These interventions have failed to become standard of care and associated patents have lapsed. 9
Implanted stimulation systems for long-term therapeutic use have dual advantages. The stimulating tip of the electrode can be located very close to the motor point of the nerve of interest. This reduces the charge required to elicit a contractile response and ensures that the response is repeatable and predictable. In addition, the user does not have to apply and remove the stimulating electrodes.5,10 Weight gain over time will not alter the responses, since the stimulating tip is adjacent to the nerve. Implanted systems avoid potential repetitive damage to fragile skin and enable use on demand or continuously throughout the day. An implanted system is reliable and simple to use on a day-to-day basis and over the longer term in order to maintain hypertrophy of paralyzed gluteal muscles.
Early implanted stimulation systems included on-board battery power, which necessitated surgical replacement of the entire implanted system when the battery power started to drain. 11 This was recognized as a limitation and mathematical models were developed to reduce the rate of power consumption. 12 Other groups developed wireless, inductively powered stimulators which removed the limitation of loss of power and the associated need for revision surgery.13,14
When considering NMES for intramuscular (IM) stimulation, it is essential that the stimulator have sufficient power to reliably generate selective action potentials and elicit a contractile response. The output must be maintained within safe limits for the duration of the stimulation so that muscle damage does not occur. Compliance voltage (Vcc) is defined as the maximum output voltage of a constant current power supply at which the load regulation is maintained within predefined certain limits. Operationally, the voltage that can be achieved by a stimulator is the supply voltage minus the voltage drop due to the stimulator’s internal resistance. It has routinely been suggested that a 20 V Vcc is required for IM stimulation.
The goal of the current experiment was to evaluate whether the 20 V Vcc routinely used was in fact necessary.
There have been nearly 1000 papers published on NMES for muscle stimulation in humans; however a Pubmed search for “compliance voltage” and “intramuscular stimulation” found only one paper by Vargas-Luna et al. which reported on a systematic evaluation of compliance voltage. 15 The group used surface ES to elicit quadriceps contractions in five healthy young persons. They found that current-controlled surface ES at 150 µs pulse width required an average stimulator Vcc of nearly 40 V to achieve 40% of the maximum force. A further search was carried out on IEEE Explore and found no results for those search terms. A search for “compliance voltage” and “intramuscular stimulation” found one paper by Liu and Yang which presented the use of biomimetic design to provide high compliance voltage in miniaturized neural implants. 16 They did not address intramuscular stimulation.
While surface electrical stimulation systems may easily be designed to apply high Vcc stimulation to electrodes on the skin (e.g. at 30 V compliance or more), implantable systems that are small in size require the use of application-specific integrated circuits (ASICs) to provide the required stimuli. The Vcc achievable for stimulation with implantable ASICs in turn depends upon the fabrication process used at the chip foundry, and often requires mixed-signal analog and digital process capability. Current mixed-signal processes are available that can support up to 20 V compliance, but the transistors used in such circuits can be relatively large, consuming extra silicon area and driving up chip costs. If lower compliance voltages can be employed to achieve effective muscle contractile responses, it becomes feasible to use ASIC fabrication processes with smaller feature sizes but lower Vcc capability, such as ± 5 V or less. This will enable die size and cost to be reduced and digital switching speed improved.
Methods
The testing carried out was approved by the local institutional review board. It is the policy of this board that study approval numbers are not publically available. Neuromuscular stimulation response was tested in 16 intramuscular (IM) electrodes with percutaneous leads that were chronically implanted in an individual with spinal cord injury (SCI). All channels were functional, and had shown stable impedance values for several years. All the IM electrodes produced active muscle contractions when stimulated with a 15 V Vcc at pulse amplitude 8 mA and pulse duration 150 µs using a portable laboratory/intraoperative stimulator (NeuroControl, Cleveland, OH). Figure 1 shows the setup for all testing.
Clinical testing setup.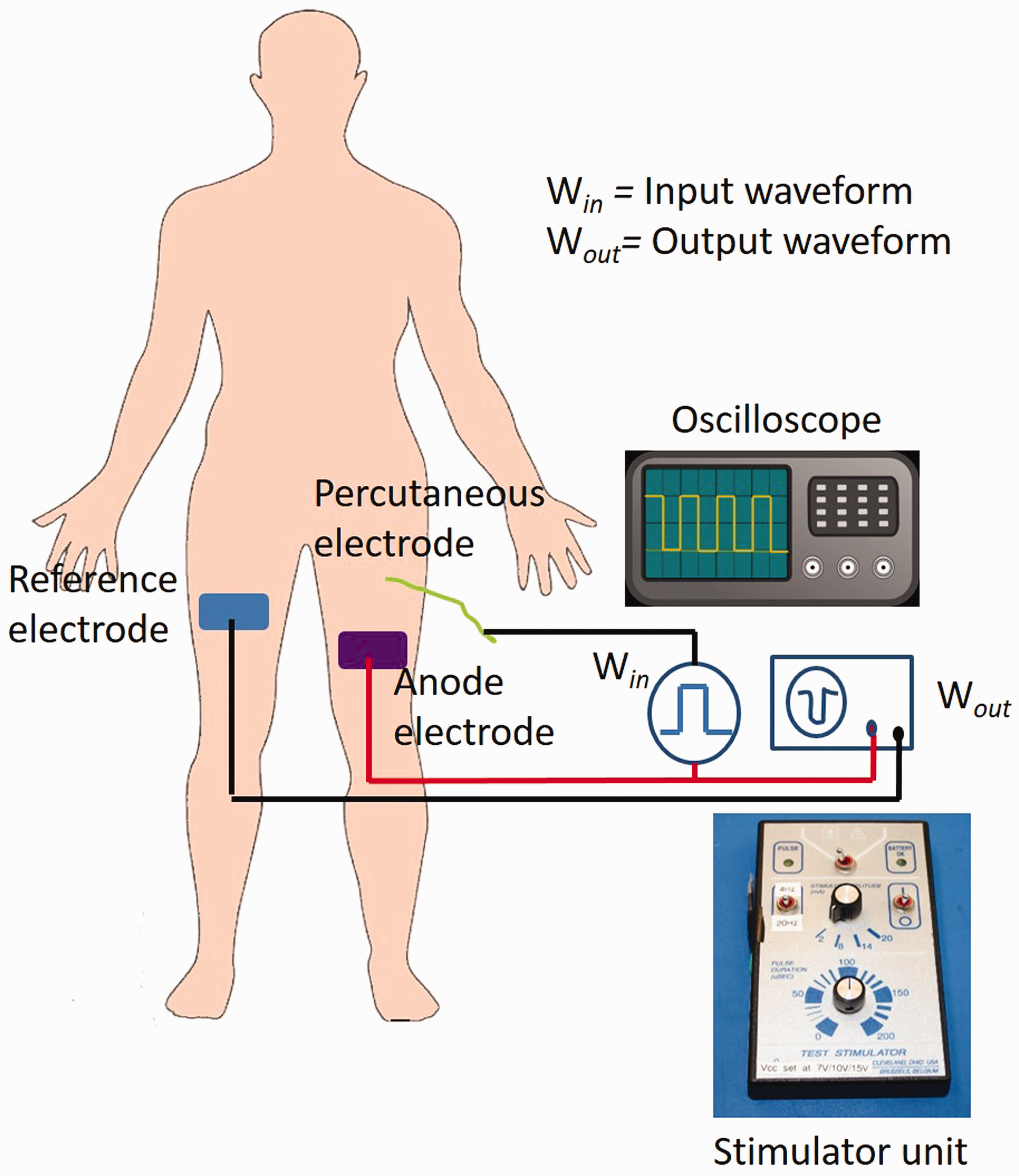
Next, the stimulator was modified to enable selectable Vcc settings. An external 249 Kohm resistor could be switched in or out during the experiments to reduce the baseline Vcc from 15 V. The 249 Kohm resistor is in series with the output of the stimulator, and drops the maximum output compliance voltage from that which was set internally in the handheld stimulator box (in this example, an internal Vcc of 15 V) down to 10 V. Thus, when switched on the Vcc was modified to 10 V. Benchtop testing of the stimulation waveforms was carried out using the modified stimulator to drive stimuli across a 499 ohm resistor which was used as a “dummy load” to simulate the resistive component of a typical intramuscular electrode in a worst-case scenario. The dummy load was placed across the stimulator output, or in this case, from the output side of the 249 Kohm voltage-dropping resistor, to ground. The stimulation frequency was set to 20 Hz, and the pulse duration was set at approximately 200 µs for all tests. Pulse amplitude was varied from 2 to 20 mA to produce charge-balanced, current-controlled outputs as shown in Figures 2 and 3. After the initial phase of the pulse, the electrode is shorted to ground, and the current required to return the electrode to 0 V potential flows to balance the charge.
Vcc = 10 V. (a) Current amplitude setting = 20 mA; (b) Current amplitude setting = 14 mA; (c) Current amplitude setting = 8 mA; (d) Current amplitude setting = 2 mA. Vcc = 15 V. (a) Current amplitude setting = 20 mA; (b) Current amplitude setting = 14 mA; (c) Current amplitude setting = 8 mA; (d) Current amplitude setting = 2 mA.

Following benchtop testing, the modified stimulator was used to firstly evaluate a highly responsive gluteal muscle in the study participant. The stimulation frequency was set at 20 Hz, and Vcc of 15 V and 10 V were used while applying a range of pulse amplitudes and pulse widths.
The stimulator was then further modified to reduce Vcc to 7 V (resulting in an effective maximum output voltage of 4 V). Neuromuscular stimulation with this further reduced Vcc still resulted in contractile responses in 15 muscles in the same human subject with SCI. The pulse amplitudes with Vcc = 7 V were varied from 2 to 14 mA and pulse width varied from 50 to 150 µs.
Results
Gluteal muscle: Minimum IM stimulation pulse parameters for strong observable contractile responses at higher levels of Vcc = 10 V and Vcc = 15 V.

Muscle responses: Minimum IM stimulation pulse parameters for observable contractile responses at Vcc = 7 V.


PA: pulse amplitude; PW: pulse width.
Representative charge-balanced, current-controlled outputs at lower Vcc are shown in Figure 4. Variations in responsiveness, due for example to varied proximity between the intramuscular electrodes and the motor points of the corresponding muscles, were noted with the higher Vcc compliance stimuli. The same variations were observed with Vcc = 7 V, indicating that the responsiveness was not due to the Vcc differences.
Stimulation waveforms. (a) Left gluteus maximus; (b) Left tensor fasciae latae.
Discussion
The implanted technology used by the individual tested is well-established and has been in use by the individual for nearly 15 years. The current systemic evaluation indicates that contractile responses may be elicited with a Vcc of 7 V, equivalent to 4 V maximum effective output. Furthermore, it is not necessary to lengthen applied pulses to increase the total charge to obtain equivalent contractile responses to those elicited by higher Vcc stimulation. While MMT scores with stimulation for a paraplegic person can never be normal since NMES of paralyzed muscle can only produce about 30 to 50% of normal muscle strength, a ± 5 V power supply may be adequate for IM stimulation using a well-placed electrode. This implies that a high-voltage (20 V or more) mixed-signal ASIC chip for a chronically implantable muscle stimulator is not necessary and that lower voltage CMOS fabrication processes having smaller transistors would be usable.
Conclusions
The efficacy of NMES over time may be influenced by the initial quality of the muscle being stimulated. 17 Lower stimulator compliance voltages can be used if electrode placement is appropriate and the stimulating tip is located close to the motor point.
Recent advances in electronics packaging technology provide the tools to meet to develop an implantable pulse generator (IPG) based on a flexible, chip-scale encapsulated electronics platform. The findings that effective contractile responses can be elicited at lower Vcc expand the possibilities for development of ASICs for effective clinical neuromuscular electrical stimulation using advanced IPGs with both smaller footprint and cost. There are currently no commercially available IPGs that can be placed without major surgery and or located in body regions with lower soft tissue cushioning than the abdomen. These experiments suggest that miniature ASICs with 5 V power supply compliance or less could be developed to fill this gap.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: KMB, DBS are RK are employees of the Department of Veterans Affairs.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Guarantor
KMB.
Contributorship
KMB and DBS researched literature and conceived the study. RK was involved in protocol development, gaining ethical approval and participant recruitment. DBS was responsible for data analysis. KMB wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Acknowledgements
We would like to thank John Schnellenberger for technical assistance in this research.
