Abstract
Introduction
Electrostimulation devices stimulate the common peroneal nerve, producing a calf muscle-pump action to promote venous circulation. Whether knee joint angle influences calf neuromuscular activity remains unclear. Our aim was to determine the effects of knee joint angle on lower limb neuromuscular activity during electrostimulation.
Methods
Fifteen healthy, older adults underwent 60 min of electrostimulation, with the knee joint at three different angles (0°, 45° or 90° flexion; random order; 20 min each). Outcome variables included electromyography of the peroneus longus, tibialis anterior and gastrocnemius medialis and lateralis and discomfort.
Results
Knee angle did not influence tibialis anterior and peroneus longus neuromuscular activity during electrostimulation. Neuromuscular activity was greater in the gastrocnemius medialis (p = 0.002) and lateralis (p = 0.002) at 90°, than 0° knee angle. Electrostimulation intensity was positively related to neuromuscular activity for each muscle, with a knee angle effect for the gastrocnemius medialis (p = 0.05).
Conclusion
Results suggest that during electrostimulation, knee joint angle influenced gastrocnemii neuromuscular activity; increased gastrocnemius medialis activity across all intensities (at 90°), when compared to 0° and 45° flexion; and did not influence peroneus longus and tibialis anterior activity. Greater electrostimulation-evoked gastrocnemii activity has implications for producing a more forceful calf muscle-pump action, potentially further improving venous flow.
Keywords
Introduction
Reduced mobility following surgery, such as hip or knee arthroplasty, presents a risk of deep vein thrombosis (DVT) in patients. 1 Clot formation arising from venous stasis 2 and lower limb muscle inactivity 3 can be prevented by mechanical counter-measures (i.e. compression stockings/devices). Although commonly used, the bulk and discomfort of mechanical devices can result in poor compliance. 4 In contrast, neuromuscular electrostimulation devices offer a non-invasive, practical and economical alternative to reduce the risk of venous thromboembolism.5,6
Electrostimulation devices stimulate the common peroneal nerve to induce an involuntary, isometric muscle contraction of calf extensor muscles (i.e. tibialis anterior and peroneus longus) and an additional stretch of the flexor gastrocnemii muscles. The passive stretch compresses the antagonist gastrocnemii, as the muscle is pulled in a distal direction during dorsiflexion. 7 The passive motion of the flexor gastrocnemii acts as the calf muscle pump to promote venous circulation by raising intramuscular pressure. In healthy adults, 5 min periods of lower leg electrostimulation has been shown to enhance venous volume (flow up to 100%) and velocity, with minimal discomfort at maximum stimulation intensity. 8 Recently, Zhang et al. 7 trialled an electrostimulation device by modelling venous stasis in healthy adults using an automated tourniquet. Short periods (10 min) of electrostimulation were shown to (i) augment calf muscle-pump action and (ii) reduce DVT-associated rises in blood volume and tissue deoxygenation. Alongside reduced limb volume, others have shown reduced venous transit-time and venous ambulatory pressure in the young. 9 Clinically, stimulating lower limb venous circulation with electrostimulation can also reduce limb volume oedema in orthopaedic, 10 diabetic and cardiovascular disease patients. 11 During electrostimulation, the activated tibialis anterior becomes an agonist, and the medial gastrocnemius an antagonist. Force and EMG recordings indicate that electrostimulation intensity relates directly to ankle dorsiflexion (and muscle-pump) force. 7 This involuntarily stretches the gastrocnemii, reducing the muscle anatomical cross-sectional area and subsequently venous diameter to eject blood to a greater extent than voluntary contraction alone. 12
Interestingly, Khanbhai et al. 9 reported greater change in limb volume and venous function with electrostimulation applied in a lying position, when compared to sitting and standing. Standing elevates lower limb volume 13 and venous pressure, 14 in comparison to lying and sitting. In these positions, knee joint angle (and therefore muscle length) may influence muscle tension of the bi-articular gastrocnemii prior to innervation. 15 Furthermore, altering muscle length (via joint angle) during electrostimulation is recommended to promote spatial motor unit recruitment. 16 Clinical observations from our group support a visible twitch response during electrostimulation when seated (∼90° knee joint angle), but little visible twitch with the knee extended (∼0° knee joint angle) in orthopaedic patients. Receiving electrostimulation whilst lying may be preferable to standing, in terms of gravitational pressure influencing peripheral haemodynamics. However, the common peroneal nerve becomes displaced from the fibular head by approximately 17 mm when standing or sitting with 0° knee flexion, when compared to sitting with 90° knee flexion. 17 It is reasonable to assume that if an individual is upright and unable to sit whilst receiving electrostimulation, they will experience less calf muscle activation (and potentially muscle-pump action). This proof-of-concept study will assess the impact of knee joint position on the neuromuscular responses of calf muscles during electrostimulation. A subsequent study will incorporate haemodynamic, alongside neuromuscular assessments, with post-operative orthopaedic patients.
What is not clearly understood is whether knee joint angle influences the neuromuscular activity of the lower leg muscles, particularly the gastrocnemii co-contraction (and therefore the effectiveness muscle-pump action) during electrostimulation. This pilot study aimed to assess the effect of seated, knee joint angle on the neuromuscular activity of the (i) gastrocnemii (co-contractor muscle pump) and (ii) peroneus longus and tibialis anterior (innervated) muscles during electrostimulation in healthy, older adults.
Methods
Participants
Demographic characteristics of recruited older adults.

Note: Values are mean ± SD. BMI: body mass index; PASE: the Physical Activity Scale for the Elderly; SD: standard deviation.
Inclusion and exclusion criteria for participation.

DVT: deep-vein thrombosis; BMI: body mass index; PASE: the Physical Activity Scale for the Elderly.
Experimental protocol
Participants visited the laboratory once to undergo 60 min of lower leg transcutaneous electrostimulation, with knee joint at three different angles (20 min administrations each (Figure 1)). Online software (sealedenvelope.com) was used to randomly allocate electrostimulation joint angle order (0° first, n = 4; 45° first, n = 5; 90° first, n = 6); no order effect was found for knee joint angle on neuromuscular activity for each muscle (p > 0.1). Each 20 min bout was separated by 60 s rests. Pilot testing (n = 3 (males), age 56 ± 2 years) confirmed no fatigue effect from monitoring EMG signals during three, 25 min electrostimulation bouts. Instruction was given to arrive hydrated, having maintained habitual physical activity levels in the preceding 48 h (Appendix 1). Upon arrival at the laboratory, the experimental protocol was re-explained, body mass was then recorded using digital scales (Seca Ltd, Birmingham, UK) and height with a stadiometer (Holtain Ltd, Crymych, UK). All subsequent measures and electrostimulation treatments refer to the non-dominant limb.
Schematic of the experimental protocol to examine the effect of leg position on electrostimulation. Leg position order was randomised. Black arrows indicate beginning (0–1 min), middle (9–10 min) and end (19–20 min) time-points for electromyography (EMG) root mean square (RMS) analysis; discomfort was assessed in the end time-point, only grey arrows indicate the mid time-point (9–10 min).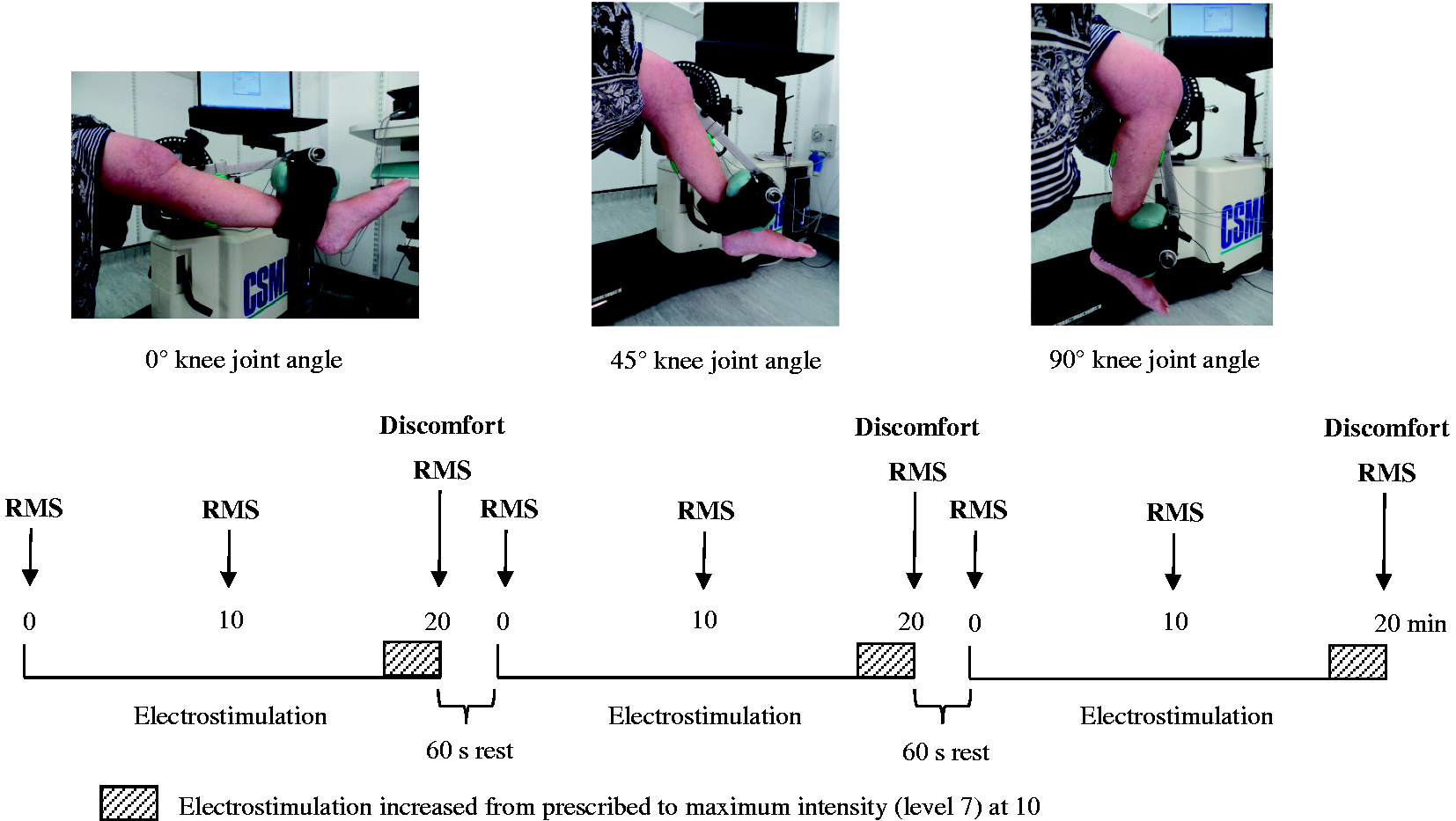
Once the electrostimulation device was fitted according to manufacturer instructions (full knee extension (0°); Firstkind Limited, Bucks, UK) and stimulation intensity determined, four EMG sensors were placed on the lower limb with the participant lying prone, and for the tibialis anterior lying supine. Electrostimulation was administered with participants seated upright (hip joint at ∼90°) in an adjustable isokinetic dynamometer chair (Humac Norm, Cybex International Inc., NY, USA) to replicate clinical administration. The lateral femoral epicondyle of the tested limb was aligned to the rotational axis of the dynamometer and the ankle joint was secured to the lever-arm. Participants were guided to extension and flexion limits by the investigator to determine knee joint range of motion (0° = full extension); the lever-arm was then mechanically set to the first knee joint angle (0°, 45° or 90°). Lower limb neuromuscular activity was recorded for 20 min throughout electrostimulation. Participant discomfort was self-reported in the final 60 s only, so as not to interfere with EMG sensor recordings and summatively assess perceived discomfort for each 20 min bout. The procedure above was then repeated for 20 min in the second knee angle and 20 min in the third knee angle. Instruction was given to relax both lower limbs throughout the entire electrostimulation period.
Electrical stimulator
A small (186 mm × 31 mm), non-invasive electrostimulation device (geko™ T2, Firstkind Limited, Bucks, UK) was attached horizontally below the fibula head on the lateral–posterior aspect of the knee, according to the manufacturer’s instructions for use. The device stimulates the common peroneal nerve, which leads to isometric contraction of the peroneus longus and tibialis anterior muscles of the lower leg. Seven stimulation intensities can be selected (50, 70, 100, 140, 200, 280 and 400 μs), to deliver a 27 mA pulse current (200 Ω–5 kΩ load impedance), at a 1 Hz repetition rate. Hereafter, electrostimulation intensities are referred to as levels 1 to 7. Participant stimulation intensity (or level) was determined based upon (i) maximal stimulation effect (slight visible dorsiflexion/eversion movement) and (ii) patient comfort. To investigate a potential staircase effect 20 for knee joint angle on electrostimulation neuromuscular activity, stimulation intensity was increased from the participant’s prescribed level up to maximum (level 7) at 10 s intervals at the end of each 20 min period (Figure 1).
Perceived discomfort
Participants self-reported lower limb discomfort during electrostimulation for each knee joint angle. The same investigator presented a 10 cm Visual Analogue Scale (VAS), ranging from 0 (no discomfort/pain) to 10 (extreme discomfort/pain); participants marked perceived discomfort on the 0 to 10 cm scale. A Verbal Rating Score (VRS) was also used, ranging from 1 (no sensation) to 7 (very severe discomfort) that aligned to the stimulation levels. Participants circled perceived sensation.
Electromyography (EMG) recording, normalisation and processing
Peroneus longus, tibialis anterior, gastrocnemius medialis and gastrocnemius lateralis EMG were recorded via SX230-1000 bipolar sensors from a portable Biometrics PS850 system (DataLOG, Biometrics Ltd, Newport, UK) during electrostimulation (Figure 2). The skin was shaved, cleansed and gently abraded to reduce sensor-to-skin impedance. Sensors were placed over the respective muscle bellies according to surface electromyography for the non-invasive assessment of muscles (SENIAM) recommendations,
21
and the reference electrode was strapped over the lateral malleolus of the tested limb. To limit electrostimulation artefacts in the raw electromyogram (EMG) signal, recording sensors were positioned orthogonal to the stimulation electrode and at an inter-electrode distance of ≥2.5 cm. Raw signals were sampled at 1000 Hz by each amplifier-embedded sensor (10 mm diameter, 20 mm inter-electrode distance; bandwidth = 20–460 Hz; common mode rejection ratio = >96 dB (typically 110 Db); input impedance = >10,000,000 MΩ), and processed with a second-order Butterworth filter (bandwidth = 10–350 Hz) to remove DC offset. The root mean square (RMS) was then calculated using a 0.25 s moving window (overlap of 50% window length). EMG data were manually checked for stimulation artefacts by overlaying the RMS envelope on to the raw EMG signal (DataLOG software v. 7.5, Biometrics Ltd, Newport, UK). For RMS analysis, at each knee joint angle, 5 s capture periods were used at the end of the following time-points: 0–1 min, 9–10 min and 19–20 min (nine capture periods in total).
Left leg showing the EMG sensor placements for the tibialis anterior (TA), peroneus longus (PL) (left figure), and the gastrocnemius lateralis (GL), gastrocnemius medialis (GM) and reference electrode (REF) affixed to the lateral malleolus (right figure).
Prior to each 20 min period, the investigator increased the electrical stimulation intensity in a sequential, step-wise manner every 15 s from the lowest (1 (50 μs intensity)) to the highest (7 (400 μs intensity)) setting, whilst measuring muscle activity at respective knee angles. This was used to assess the relationship between stimulation intensity and muscle activity for each participant, at each knee angle. Maximum RMS was determined for a 1 s interval around the peak torque evoked from the participant’s maximum voluntary contraction for each muscle. Joint torque was measured for each muscle using the same isokinetic dynamometer used to secure knee joint angle. In a prone position, participants produced three, 3–5 s maximal voluntary isometric contractions (60 s rests), with verbal encouragement provided by the investigator. Subsequent RMS data were normalised by dividing by the maximum RMS value and then multiplying by 100 to provide percentage of RMS maximum.7,22
Statistical analysis
Shapiro–Wilk tests confirmed neuromuscular activity data were non-normally distributed; non-parametric tests were used to analyse RMS for each muscle. One-way, repeated measures Friedman’s analyses of variance (ANOVAs) were used to compare (i) RMS activity and (ii) discomfort (VAS and VRS) between knee joint angle (0°, 45° and 90°) for each muscle. Paired Wilcoxon Signed-Rank tests identified angle-specific differences. Mixed-design ANOVAs (within-group, repeated measures on levels (7) and degrees (3)) tested whether there was an electrostimulation intensity effect on RMS activity, dependent upon knee joint angle. Relationship between stimulation intensity and neuromuscular activity (normalised RMS) at each knee angle was determined by Spearman’s correlation (based upon group mean (n = 15) for each stimulation intensity).
Data were expressed as mean and standard deviation. Non-normal data were expressed as mean, with 95% confidence intervals (CIs), and the Friedman’s ANOVA test statistic represented as Chi-squared (χ2). Effect sizes (Cohen’s d) were calculated to determine meaningful differences (small = 0.2, moderate = 0.5, large, 0.8) and statistical significance set as p < 0.05.
Results
Anthropometry and discomfort
Perceived discomfort VAS and VRS during electrostimulation at each leg position.

Values are mean ± SD. VAS: Visual Analogue Scale; VRS: Verbal Rating Score; SD: standard deviation.
Neuromuscular activity and knee joint angle
During electrostimulation, knee joint angle did not affect RMS activity of the tibialis anterior (χ2(2) = 1.857, p = 0.4, d = 0.07; Figure 3(a)) and peroneus longus (χ2(2) = 3.0, p = 0.2, d = 0.08; Figure 3(b)). However, knee angle did influence gastrocnemius medialis RMS activity (χ2(2) = 12.0, p = 0.002, d = 0.54), with greater RMS activity at 90° knee joint angle, when compared to 0° (p = 0.003, d = 1.07) and 45° (p = 0.003, d = 1.06; Figure 3(c)) angles. Knee joint angle influenced gastrocnemius lateralis RMS activity (χ2(2) = 16.714, p = 0.0001, d = 0.49), with greater RMS activity at 90° knee joint angle, when compared to 0° (p = 0.002, d = 0.99) and 45° (p = 0.002, d = 1.31; Figure 3(d)) angles. Gastrocnemius lateralis RMS activity was greater at 45° knee joint angle, when compared to 0° (p = 0.02, d = 0.27) angle.
Normalised EMG activity of the (a) tibialis anterior, (b) peroneus longus, (c) gastrocnemius medialis and (d) gastrocnemius lateralis during 20 min of electrostimulation, at different knee joint angles. Time-points refer to beginning (0–1 min), mid (9–10 min) and end (19–20 min). *Significant difference at 90°, #significant difference at 45°, p < 0.05. RMS: root mean square.
Joint angle-dependent effect on electrostimulation intensity
When increasing the electrostimulation intensity from minimum (level 1) to maximum (level 7), knee joint angle did not affect tibialis anterior RMS activity (p = 0.27, d = 0.09), although there was a linear trend (p = 0.004, d = 0.48; Figure 4(a)). Peroneus longus RMS activity was influenced by electrostimulation intensity and knee joint angle (p = 0.02, d = 0.21; quadratic trend: p = 0.01, d = 0.41; Figure 4(b)), with greater effect at 90° knee joint angle than at 45° (p = 0.05). Gastrocnemius medialis RMS activity showed an interaction effect (intensity × knee joint angle) (p = 0.01, d = 0.15; quadratic trend: p = 0.05, d = 0.26), with greater effect at 90° knee joint angle than at 0° and 45° angles (see Figure 4(c)). The gastrocnemius lateralis was influenced by intensity only (p = 0.001, d = 0.52; quadratic trend: p = 0.03, d = 0.32; Figure 4d). There was a positive relationship between electrostimulation intensity and RMS activity for each muscle (n = 15; tibialis anterior: 0°, r = 0.96; 45°, r = 0.97; 90°, r = 0.94; peroneus longus: 0°, r = 0.89; 45°, r = 0.81; 90°, r = 0.90; gastrocnemius medialis: 0°, r = 0.76; 45°, r = 0.78; 90°, r = 0.91; gastrocnemius lateralis: 0°, r = 0.87; 45°, r = 0.85; 90°, r = 0.94; p < 0.001).
Relationship between the electrostimulation intensity and the normalised EMG activity (% of maximum stimulation intensity level 7) of the (a) tibialis anterior, (b) peroneus longus, (c) gastrocnemius medialis and (d) gastrocnemius lateralis, at 0°, 45° and 90° knee joint angles. *Significant difference at 90°, p < 0.05. RMS: root mean square.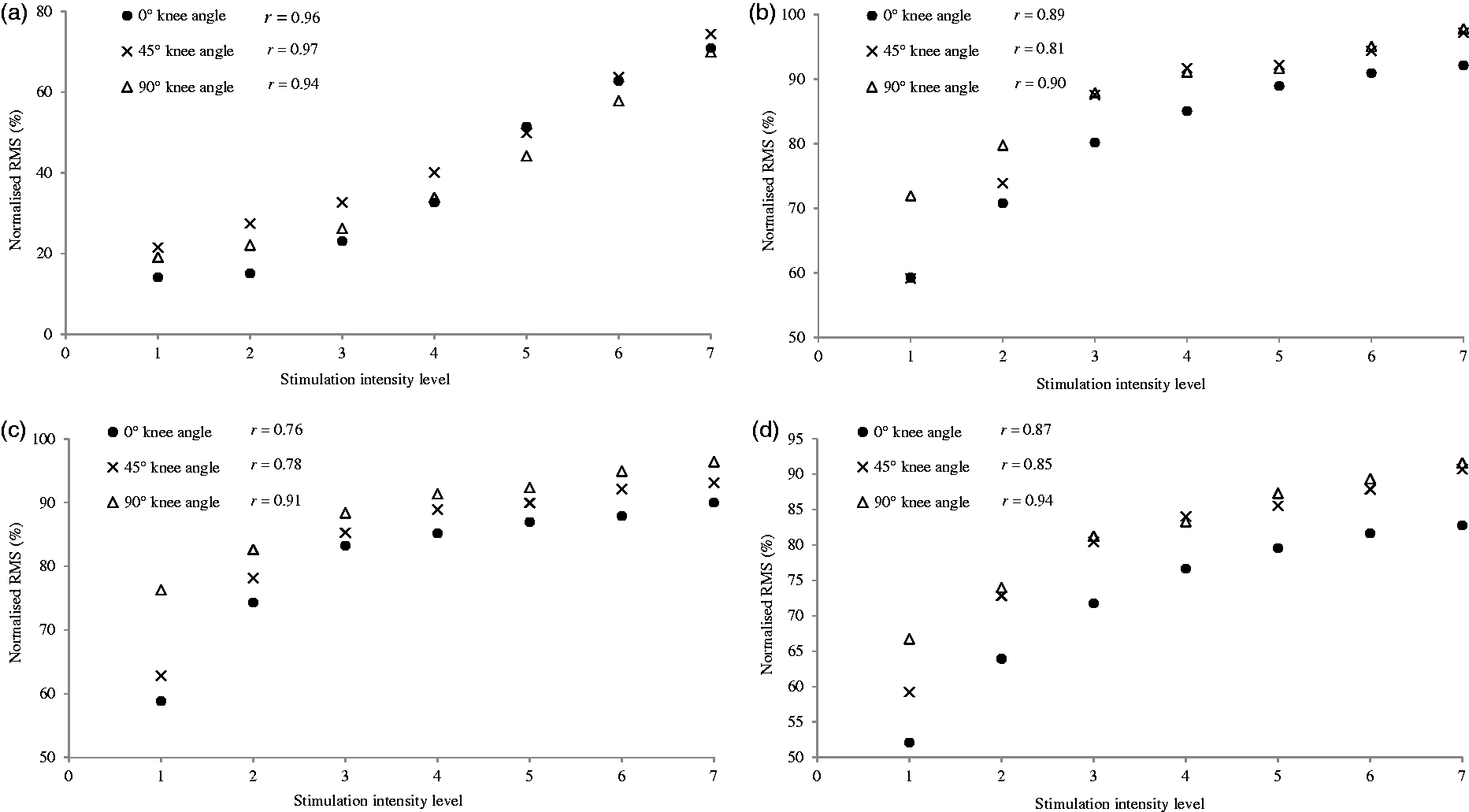
Discussion
This proof-of-concept pilot study investigated whether knee joint angle influenced lower limb neuromuscular activity during electrostimulation in healthy, older adults. It is recommended that the joint angle (and therefore muscle length) remains the same during electrostimulation, 15 as the device stimulates the common peroneal nerve to activate the calf muscle pump to subsequently promote venous circulation in the lower limb. We examined the muscles responsible for the muscle-pump action: the peroneus longus, tibialis anterior and the co-contractor gastrocnemii (lateral and medial heads) at three different knee joint angles (i.e. 0° (full extension), 45° and 90° knee flexion). We found that during electrostimulation positioning the knee joint at 90° flexion (i) influenced gastrocnemii (ankle plantarflexor) muscle activation; (ii) increased gastrocnemius medialis activation at each stimulation intensity (from minimum (level 1) to maximum (level 7)), when compared to 0° and 45° knee flexion and (iii) did not affect activation of the peroneus longus (ankle plantarflexor and evertor) and the tibialis anterior (ankle dorsiflexor) muscles.
There was a significant correlation between stimulation intensity and muscle activation for each calf muscle. The strongest correlation was observed at 90° knee flexion for the peroneus longus, gastrocnemius medialis and gastrocnemius lateralis (Figure 4(b) to (d)).
When receiving calf electrostimulation seated, our cohort showed greater gastrocnemius medialis (co-contractor) activity with the knee at 90°, when compared to partial knee flexion (45°) and knee extension (0°). A similar joint angle-dependent effect was shown for the gastrocnemius lateralis, which, in addition, displayed greater activity at 45° than at 0° knee flexion (full extension). The gastrocnemius medialis and lateralis are similar in fibre-type composition 23 but controlled by different afferent pathways from the same neural origin. 24 We did not examine neural pathways, but differences in gastrocnemius medialis and lateralis neuromuscular activity at 45° flexion are likely to derive from a wider 95% CI for the gastrocnemius lateralis and therefore a small-to-moderate effect size. Activation increased for the gastrocnemius medialis by 31.3% and lateralis by 32.4% during 20 min of electrostimulation with the knee at 90° flexion, when compared to 0° flexion. Varying the knee and ankle joint angles influences the gastrocnemii muscle length 25 and force-producing capacity, 26 as well as the passive knee flexion moment. 27 As the human gastrocnemii operates on the ascending limb of the force–length relationship, passive tension begins to develop at short muscle lengths (i.e. in 90° knee flexion), before approaching near-maximum at longer muscle lengths (i.e. 0° knee extension). 28 As a consequence, at longer muscle lengths, the contribution of the active, contractile component becomes near-maximum 11 with greater passive force exerted. 2 Therefore, electrostimulation may be less effective at activating the calf muscle pump with the knee extended with the gastrocnemii muscle-tendon unit at a longer muscle length.
In our study, to ensure potential changes in gastrocnemii neuromuscular activity were attributable to knee joint angle, and not ankle joint angle, the participant’s ankle was held in a neutral position (∼0°) throughout electrostimulation. During electrostimulation, we found greater gastrocnemii neuromuscular activity with the knee flexed (90°) and the muscle in a shortened position, when compared to the knee extended (0°) and the muscle in a lengthened position. This is chiefly attributable to displacement of the common peroneal nerve from the fibular head (by ∼17 mm) with the knee in 0° flexion, 17 which would result in sub-optimal peroneus longus and tibialis anterior activation. In addition, stimulating lengthened gastrocnemii, with longer contractile and/or elastic component would likely affect muscle activation. For example, as the gastrocnemius is an agonist in knee flexion, stimulation at 90° flexion would innervate an already ‘active’ muscle under tension. As an antagonist in knee extension, gastrocnemii activation increases during voluntary knee flexion 29 but decreases during voluntary knee extension. 30 In a lengthened position (0°), the stimulation would have to overcome a stretched gastrocnemii tendon and greater passive force.27,31 Therefore, a proportion of muscular tension evoked by electrostimulation would be attenuated by the Achilles tendon of the gastrocnemius, which accounts for ∼73% of the total muscle-tendon length change (in contrast, the tibialis anterior tendon accounts for ∼45% length change). 25
Another possible explanation for the increased gastrocnemius medialis activity with electrostimulation at 90° knee flexion arises from neuromuscular propagation. Decreases in contraction time and half-relaxation time during progressive muscle shortening 32 reflect a requirement for higher excitation rates to produce the same evoked torque. Greater activation at 90° knee flexion may indicate a need to increase activation at a shorter muscle length. However, this seems unlikely, given that the gastrocnemii muscle is at a favourable position on the length–tension relationship at 90° knee angle. Others have reported decreased gastrocnemii activation at pronounced knee flexion angles up to 60%,33,34 which disagree with our findings. However, it should be noted that these studies evoked muscle activity by maximal voluntary contraction, whilst manipulating ankle angle.
The neuromuscular activity of the tibialis anterior and peroneus longus during electrostimulation were not influenced by knee joint angle. Tibialis anterior activation at 45° (59.3%) and 90° knee flexion (64%) appeared greater than 0° knee flexion (49.9%) after the first minute, yet this did not reach significance. Additional linear trend analyses (p = 0.008) indicated that tibialis anterior neuromuscular activity increased proportionally from minimum (level 1) to maximum (level 7) stimulation intensities similarly across each knee joint angle. However, these findings are unsurprising given that both are mono-articular muscles and span only the ankle joint, whereas the bi-articular gastrocnemii spans the ankle and knee joints. The tibialis anterior is composed predominantly of slow twitch, type I fibres, with slower contraction time, 32 which may also contribute to the electrostimulation-evoked muscle activation being lower at each knee angle, when compared to the other muscles (Figure 4(a) to (d)). Additionally, the common peroneal nerve first passes the peroneus longus, which when activated, will oppose force produced by the tibialis anterior. 32
Knee joint angle did not influence discomfort, with the majority of perceptual ratings showing that stimulation involved minimal discomfort, and only the highest stimulation setting, level 7 (pulse current: 27 mA; intensity: 400 μs; repetition rate: 1 Hz), reached mild discomfort. Similar discomfort values have been reported during percutaneous electrostimulation administered intermittently (5 min stimulation, 10 min rest for 4 h) in healthy adults 8 and in hip arthroplasty patients of similar age. 3 Electrostimulation settings were participant-specific and determined according to the manufacturer’s instructions which recommend that the appropriate stimulation intensity should evoke a visible twitch in the foot. Even at 0° knee flexion, tibialis anterior and gastrocnemius medialis activation increased by a minimum of ∼49% maximum with little discomfort using prescribed settings. As lower limb blood flow can be increased by a muscle producing 30% of maximal contraction, 35 our preliminary results show promise with regard to electrostimulation at 90° knee flexion enhancing neuromuscular activity, and potentially venous blood flow, with minimal discomfort.
From a clinical perspective, these pilot data from healthy, older adults suggest that receiving electrostimulation when seated at 90° knee flexion can enhance gastrocnemii activation, when compared to seated at 45° or 0° knee flexion. The electrostimulation device stimulates the common peroneal nerve to evoke an involuntary, isometric contraction of the peroneus longus and tibialis anterior muscles simultaneously. The gastrocnemius then undergoes as passive stretch as the antagonist flexor muscle. This calf muscle-pump action improves venous blood flow in bed-rest, 36 sitting for prolonged periods 8 and during venous stasis. 7 The gastrocnemii contributes a greater physiological cross-sectional area (96.1 cm2) of the calf muscle pump than the tibialis anterior (18.5 cm2) and peroneus longus 37 and therefore has greater potential for force-producing capacity and venous circulation. However, straightening the leg to 0° knee flexion may displace the common peroneal nerve from the fibular head 17 and reduce the impact of the calf muscle pump. Based on our pilot observations, future work should determine whether receiving electrostimulation seated, with 90° knee flexion can increase gastrocnemius activation and, in turn, produce a more forceful muscle-pump action to enhance venous blood flow in clinical cohorts (i.e. orthopaedic patients undergoing hip/knee arthroplasty).
The main limitations of this proof-of-concept pilot study were that we did not measure electrostimulation-evoked (i) torque-production or (ii) venous blood flow. Ankle torque would have been difficult to assess given that our experimental aim was to study the potential influence of knee joint angle on electrostimulation. Zhanget al. 7 assessed electrostimulation-evoked torque and during isometric ankle dorsiflexion with participants lying prone. They were able to fix a load cell in this position, whereas our dynamometer lever-arm (measuring torque) was used to fix knee joint angle. Our 20 min electrostimulation periods were too brief to accurately apply both EMG and Doppler ultrasound to measure venous blood flow.
Conclusions
This pilot study presents the first observation that knee joint angle can influence gastrocnemii activation during seated electrostimulation in healthy, older adults. The results suggest that receiving electrostimulation when seated, with the knee flexed at 90°, can augment increases in gastrocnemii activity shown with the knee partially flexed (45°) or extended (0°). This could have implications for an electrostimulation device stimulating a more forceful calf muscle-pump action and, in turn, further improving lower limb venous blood flow with little discomfort.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by Bournemouth University Grants Academy and the Orthopaedic Research Institute.
Guarantor
JPG
Contributorship
All authors were involved in the study design and manuscript writing. JPG and MC were responsible for gaining ethical approval, participant recruitment, data collection and data analysis. JPG wrote the manuscript drafts, with guidance on clinical interpretation from TWW. JPG, MC and TWW critically revised, and approved the final manuscript.
Acknowledgements
The authors would like to thank all those who volunteered their time to participate in the study, as well as Debbie Gale whose laboratory support was invaluable. The findings of this paper were presented at the 20th European Congress of Physical Rehabilitation Medicine, Lisbon, Portugal, 2016.
