Abstract
Introduction
Evaluation of manual skills training for spinal rehabilitation has been stagnant for 40 years. Observation and mimicry are coupled with feedback from “expert” observers to train and evaluate learners, relying on rater experience to discern speed and force. Spinal manipulation is a controlled act under the Regulated Health Professions Act (1991) as it is not without risk. The discordance between current methods for evaluating proficiency and the potential risks of inadequate application is a critical gap.
Methods
This work reports a novel wearable device that measures finger forces via microstrain bending of the physiotherapist’s nail. The device leaves the tactile finger pad unobstructed and does not interfere with treatment application. Five expert-level physiotherapists performed a standard postero-anteriorly directed spinal segmental mobilization treatment for 1 min at 1–1.5 Hz rhythm.
Results
The device successfully measured all treatment forces (0.2–27.3 N). Physiotherapists applied a maximum force rate of 0.03 ± 0.01 N/s with a rhythm of 1.76 ± 0.38 Hz. In 15 trials, there were no device failures. The device was easily applied and removed, and physiotherapists were able to walk about and interact normally with patients.
Conclusions
These results indicate that this technology can be integrated into a skills training program to provide quantitative feedback for objective assessments.
Keywords
Introduction
Manual and manipulative therapy (MMT) are cornerstones of physical therapy and chiropractic education and treatment. Despite a universal desire for excellence in MMT training, the field as a whole suffers from near stagnation in training and evaluation strategies since its introduction into Canada in the mid-1970s. Slight differences in context or delivery aside, the overarching strategy has remained steadfast: learners observe an expert perform a technique and then attempt to mimic and master that technique on a colleague. At best, learners receive direct observation and verbal feedback from an instructor or from their (live) healthy colleague. After some period of practice and attempted mastery, learners perform their techniques on “mock patients” (other healthy volunteers) while being observed and rated by expert raters. If the rater perceives the performance to be satisfactory, then the learner is granted a credential indicating adequate competence has been attained. Some exceptions aside, the “patients” in these simulated clinical encounters being healthy volunteers, the evaluation cannot be outcomes based (since there are in fact no symptoms to speak of), rather competence is most commonly based on observed performance using indicators such as speed, amplitude, and force under the untested assumption that these parameters are important for improving clinical outcomes.
The discordance between teaching or evaluation method and real-world application is realized when considering that MMT is not without risk. Arguably the most recognizable risk is that of stroke or death 1 often cited by the lay press 2 most commonly associated with “adjustments” (manipulations) of the upper cervical spine thought to result in cervical arterial dissection (CAD). This has led to repeated calls to ban the practice.3,4 However, it should be noted that the true incidence of serious adverse events has been difficult to ascertain owing to factors such as unclear definitions5,6 and expected underreporting of events. 7 A recent population-based case–control study found that the incidence of CAD following chiropractic visits was no greater than that following primary care physician visits. 8 Regardless of the true incidence, all are in agreement that upper cervical manipulation holds the potential for serious adverse events especially in the absence of proper training. Other more common complications are less serious but not completely benign and include dizziness, increased pain, headaches, or radiating numbness and tingling. 5 It is not surprising that adverse events are most likely to occur when these techniques are applied by novices rather than by experts, 9 yet the only way to learn the technique is to apply and practice it on colleagues or fellow students. While never empirically quantified, our own anecdotal experiences are of learners inadvertently harming their practice partners thereby forcing many in MMT programs to sit out a session as a result of ongoing symptoms from a technique performed on them a day prior. It is clear that a more quantifiable approach to developing spinal therapy competencies is required.
The only published attempt at quantifying and evaluating performance of people learning spinal manipulation of which we are aware was that of Cuesta-Vargas and Williams, 10 who used a single, triaxial accelerometer at 100 Hz sampling frequency to evaluate the effect of real-time acceleration feedback on learning outcomes. The only significant effect found was that of increased peak angular velocity posttraining. Those authors concluded that the use of the sensors improved learning of the skill, but this is neither borne out by the results nor is it clear that peak angular velocity is in fact an important part of the performance of manipulation. We believe that the first step toward moving this field forward starts with understanding what makes a “good” manipulation and to develop a portable method to capture those important parameters. This would facilitate the development of systems for immediate feedback during learning and practice, filling the need for knowledge of performance (KP) described by Triano et al. 11 as a key component of biomechanical skill acquisition.
Spinal adjustments and similar therapy maneuvers are rapid (in the order of 100–200 ms), 12 which makes it challenging to evaluate a therapist in training and to provide immediate KP-based feedback. Part of the limitation is that it is currently prohibitive to measure the manual forces that a trainee applies to a subject. A small wearable device to measure real-time finger forces would benefit many MMT training programs by providing quantified metrics of finger force coordination that could be used to establish standardized assessments of competencies prior to independent clinical application. The quantifiable feedback would inform trainees on their skills progression and how they compare to their peers and to experienced therapists.
Previous studies that have quantified the forces of such treatments transmitted to subjects have used measurement devices that are interposed between the subject and therapists hands 13 or have measured the load transmitted through the subject and table via floor-mounted load cells using live patients 14 or mannequins. 15 In the former case, interposed devices obstruct the therapist from the subject, which may confound application of the standard therapy protocols. In the latter cases, a floor-mounted load cell setup would require precise tracking of the therapist’s point of load application. Thus, it may not capture tangential load components, or the net load vector may be attenuated by inconsistent positioning of the subject. The use of mannequins or instrumented dummies has been unable to reproduce the real experience of living humans.
In this study, we implemented a novel device to measure forces applied through the fingers and thumbs using a custom sensor that is worn on the finger nail much like an esthetic false nail. As the user interacts manually with objects and subjects, the forces that are experienced at the finger pad cause small deflections in the therapist’s bone and soft tissues underlying the nail, which result in small deflections of the nail that are measured by the high-sensitivity sensor and converted into real-time force output. The low-profile sensor adds no more than 1.5 mm thickness to the nail, allowing the user to move normally. Moreover, unlike other technologies, the finger pad is not obstructed, which allows for the native tactile sensation to be experienced without interference from the sensor.
Methods
Five expert-level physiotherapists with a minimum 15 years of clinical experience, all of whom were Fellows of the Canadian Academy of Manipulative Physical Therapists and instructors in pre- or postprofessional MMT training programs, performed a standard manual mobilization technique requiring the application of a postero-anteriorly (PA) directed pressure using the pads of the thumbs on the fifth and sixth cervical vertebrae. This type of mobilization can be qualitatively “graded” from grade 1 (contact with the skin) to grade 5 (a manipulation or adjustment, taking the segment beyond its normal physiological range). For the purposes of this study, the therapists were instructed to perform a grade 3 mobilization, which is a larger amplitude, rhythmic, application of PA pressure where each push is applied until resistance to movement signals to the therapist that the end of normal physiological range has been reached, after which the pressure is removed and reapplied. 16 Traditional training for therapists endorses a rate of 1–1.5 Hz for a grade 3 application.17,18 The “patients” in this case were healthy university-aged participants who were instructed to provide no feedback to the therapist.
Each finger sensor was constructed from one uniaxial foil type 350 Ω strain gauge (model CEA-13-062UW-350, Micro-Measurements Inc., Wendell, NC), which was cemented to a standard esthetic acrylic nail substrate (Nailene, Pacific World Corp., Aliso Viejo, CA). This device was mounted on the nail with its measuring axis oriented transversely across the finger. The principle of operation is that when a therapist touches the patient, deformations of the soft tissues and bone in the therapist’s finger translate into microscopic bending of the finger nail. As the finger pad is pressed, the bone and tissues underlying the nail push up and tend to flatten the nail’s radius of curvature. The subsequent strain is measured by the gauge in a quarter bridge completion circuit.
A wireless data acquisition transceiver (V-Link 200, Lord MicroStrain Inc., Williston, VT) provided signal conditioning and transmitted microstrains wirelessly to a base station (WSDA-Base-104-LXRS, Lord MicroStrain Inc., Williston, VT) connected to a laptop via USB. This wireless setup allowed the user to walk around freely and untethered, and to apply the treatment unobstructed by the finger force measuring system (Figure 1).
Treatment force measurements system worn by a physiotherapist. The thumb-mounted gauge is wired to the transceiver which transmits nails strains wirelessly to a base station laptop (left). A uniaxial strain gauge on acrylic substrate adhered to thumb nail converts measures treatment force by way of nail strains (right).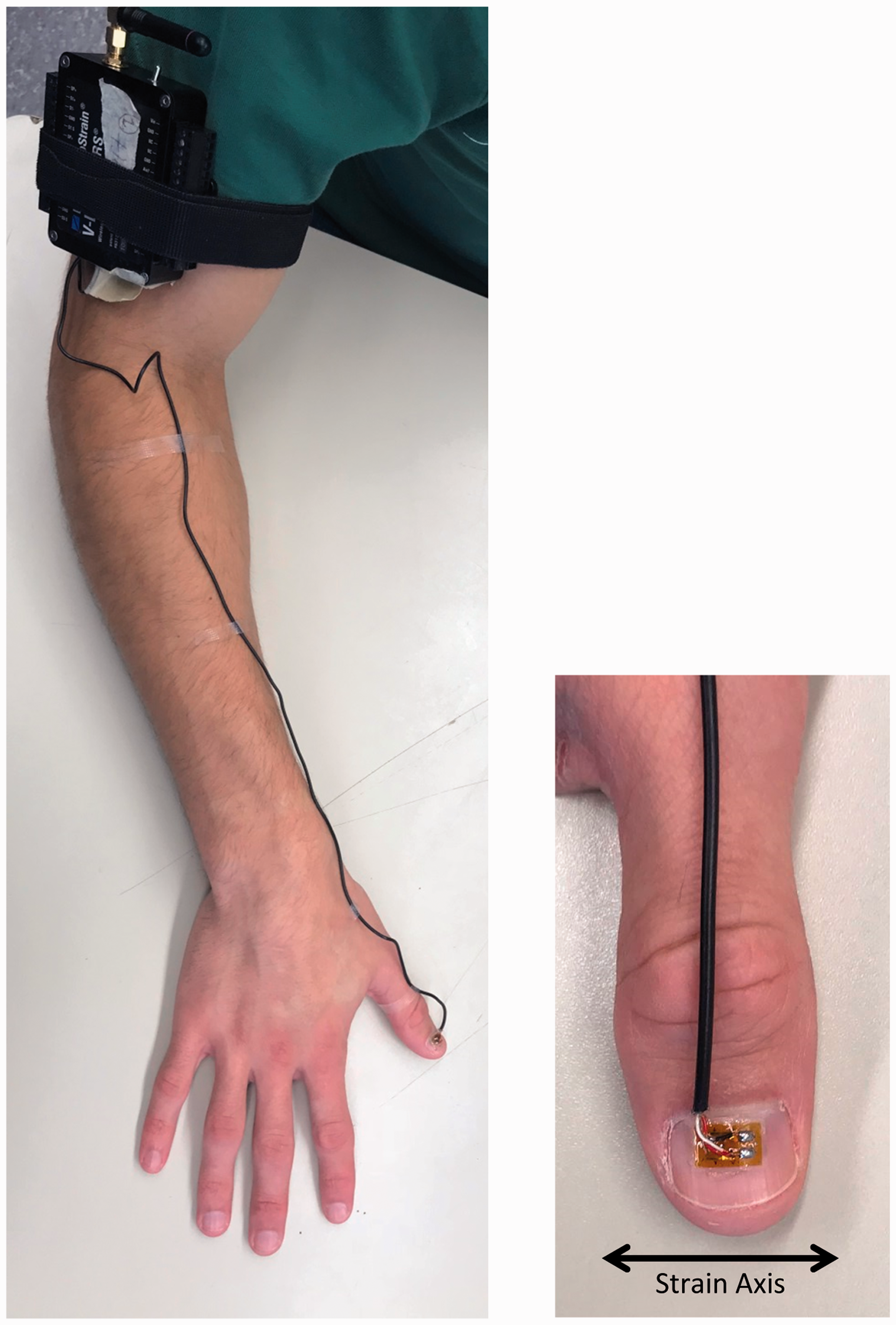
Acrylic substrates were selected according to the size and curvature of each subject’s nails which were determined prior to testing. Nail curvature was important to avoid a high prestrain in the gauge, which could damage the gauge or saturate the measurement system. Prior prototype feasibility tests had revealed that sensor contact with the cuticle or skin adjacent to the nail produced unanticipated tissue recruitment, which caused a severely nonlinear response that was difficult to address with calibration. Thus, each custom-made sensor was trimmed to fit well within the nail boundaries in order to avoid contact with soft tissues.
Each assembled sensor construct was affixed to the nail of the thumb of both hands using the double-sided adhesive provided by the acrylic nail manufacturer. Prior to adhesion, the finger nail was cleaned with 70% isopropyl alcohol to remove natural oils in order to provide reliable adhesion. Then the nail was lightly sprayed with Dehesive® spray (Cramer Products Inc., Gardner, KS) and excess spray was patted off with a paper towel. The Dehesive® step decreased adhesive strength to ensure damage-free removal of the sensors with no discomfort to the user. This protocol ensured consistent and reliable adhesion throughout a 1 h test period. The wireless transceiver was Velcro strapped to the subject’s forearm or upper arm and thin wires, connecting the strain gauges to the wireless transceiver, were secured to the forearm using Micropore Standard Hypoallergenic Paper Surgical Tape (3 M Healthcare).
Each finger sensor was calibrated for force output by having the physiotherapist press against a clinical finger press load cell (model PF002, NK Upper Extremity Assessment System, NK Biomechanical Corp., Minneapolis, MN) that was placed on a table. Custom written code (LabVIEW, National Instruments Inc., Austin, TX) recorded strain gauge and load cell outputs as the therapist pressed with increasing force to a comfortable maximum. A finger-specific calibration function was determined from the synchronized nail strains and load cell forces. Therapists 1 and 2 preferred a thumb-over-thumb technique. For these two therapists, calibration was also performed in their preferred configuration. Pilot testing revealed that the top thumb’s sensor was sufficient to measure the overall applied force. The specific calibration accounted for any effect that the bottom thumb’s curvature may have had on the top thumb. Thus, the bottom thumb’s sensor output was not included in the final analysis of therapists 1 and 2.
The therapists were then instructed to apply treatment at their preferred force levels for 1 min at a 1–1.5 Hz rhythm (Figure 2). The start/stop times were announced by the coordinator, but the frequency was not controlled. Thumb placement was not specified, and as such, therapists were free to use either a thumbs side-by-side method or a thumb-over-thumb technique as they would normally do in routine practice.
Therapist wearing two thumb force measurement systems (left). Thumbs side-by-side technique (top right). Thumb-over-thumb technique (bottom right).
During each treatment period, voltage data was collected at 256 Hz. Signal conditioned voltages were converted to microstrain in the laptop and calibration functions were applied post hoc to convert the finger nail strains to forces in Newtons. Each therapist performed the treatment three times (3 × 1 min) on the same mock patient. Outcome measures included the peak force, delta time (i.e. time between force peaks), average frequency, and maximum rate of force application (N/s). This latter metric was calculated as the maximum positive slope within each push motion (using at least 10 data points to avoid artificially high values), averaged over a single treatment trial.
Pearson correlations were performed using SPSS (V22.0, IBM Corp., Armonk, NY). Metrics of peak force, force rate, and delta time were available for each push, providing approximately 80–140 measurements per simulated treatment (i.e. trial), depending on the therapist’s speed. All three trials from every therapist were used in these analyses.
Results
In comparison to the load cell, postcalibrated testing demonstrated that sensor accuracy was 0.17 ± 0.02 N, with a minimum sensitivity of 0.02 N. Additionally, pressing to maximum safe levels with minor discomfort to the wearer did not damage the sensors or saturate the data acquisition system, and thus, the sensors recorded the full range of treatment forces.
A sample of sensor output from the first trial of Therapist 1 is illustrated in Figure 3. Table 1 summarizes the metrics over three trials. Average peak force applied by all therapists ranged from 5.6 to 21.1 N. The average frequency was 1.76 ± 0.38 Hz, and three of five therapists were within the target frequency range of 1–1.5 Hz. All five therapists were remarkably consistent in their timing, which is evidenced by the low variability of the delta time metric. The variability of force application was greater, with one standard deviation as a percentage of mean peak force ranging from 9 to 31%. While greater peak force did generally correspond with greater force variability, this was not necessarily true as a percentage, as evidenced by Therapist 3, who applied the highest peak forces, but whose variability was relatively low.
Sample force application from first trial of Therapist 1. One minute of treatment was analyzed beginning at the detection of the first peak, indicating the therapist’s first push. In this example, it is apparent that the therapist applied a higher force to begin with, before quickly settling into a rhythm. Force magnitude was less repeatable than rhythm. Grade 3 mobilization metrics averaged over three trials of simulated treatment.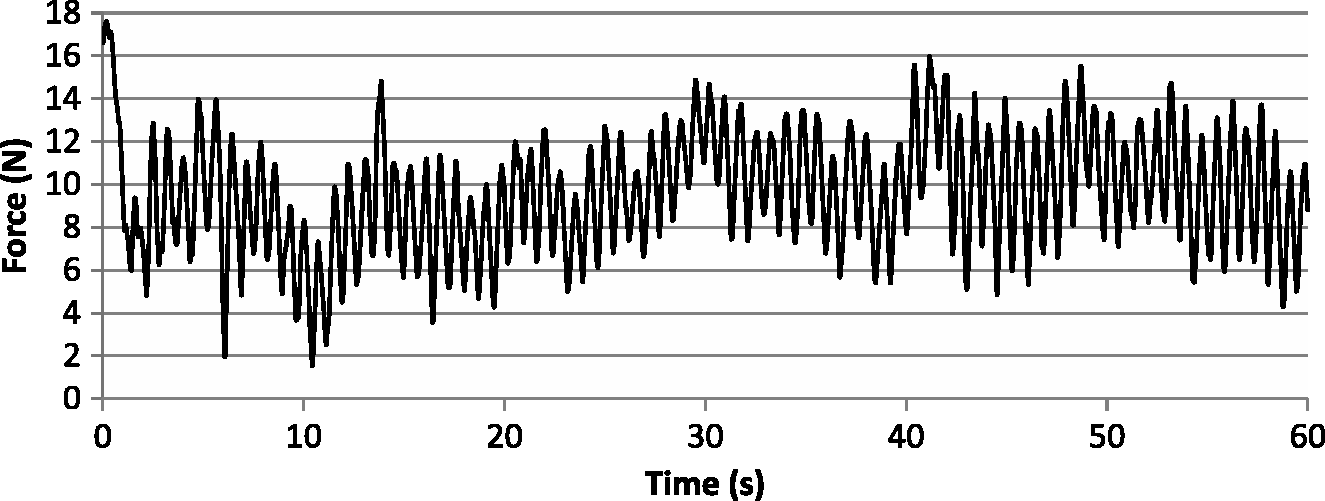

Peak force was positively correlated with the rate of force application (i.e. positive slopes in Figure 3) (p < 0.001), indicating that faster pushes caused higher peak forces. On a peak-for-peak basis, peak force was also positively correlated with delta time (p < 0.001), indicating that reaching higher forces required a greater amount of time.
Discussion
Most devices that quantify hand function are in the form of a glove.19–21 However, glove constructs have found little application in therapy because they occupy the tactile-sensitive finger pads of the therapist. To our knowledge, this is the first study of a low-profile sensor that takes advantage of the natural bending of the human nail to measure force applied by the finger pad. This allowed the therapists to feel their patient normally, as no part of the sensor obscured the therapist’s tactile finger/thumb pads.
Our results show that this simple and cost-effective method can be employed in a real therapeutic environment and that it is sufficiently reliable and sensitive to measure specific performance metrics for therapist skills assessments. Informal feedback from the therapists indicated that after a short period of acclimation to the feeling of the acrylic nail sensors, they were able to perform the clinical maneuver in the lab-based setting in a way similar to that of a real patient treatment.
Two out of five therapists who used a thumb-over-thumb technique were not hindered by the device. Posttesting analysis revealed that the top thumb sensor was sufficient following a specific calibration. Of course, any interthumb difference in force applied during testing would increase error if it was not accounted for during calibration. This may be a limitation, and although the small sample size of this study was not designed to elucidate this, the results from these two therapists were not different from the group. Future work is needed to investigate this reliability factor further.
While peak force is intuitively relevant to potential injury (the higher the force applied, the greater the displacement of underlying tissue, the higher risk of injury), we felt it was equally important to define a maximum rate of force application because the viscoelastic property of tissue means that its strain rate is a function of force application rate. This implies that injury can occur below the static (or quasi-static) yield strength of soft tissue. The maximum rate of force application (N/s) was always greater than peak force values (N), and this was owing to the definition of force rate, which was defined as the steepest linear slope of a segment of data points on the positive increasing slope of the push movement, and as such, it did not include the highest (i.e. peak force) and lowest points in the push cycle. This was done purposely because a sinusoidal-shaped curve demonstrates a rate decreasing to zero approaching each peak and trough, thus the maximum rate lies somewhere in between.
We chose to report the maximum rate of force application without regard to its relative occurrence with respect to the time of peak force; however, it could be argued that a more relevant measure would be the average rate of force immediately leading up to the peak force, which would be related to the speed at which the therapist pushed just before reaching the maximum tissue displacement. As an example, reaching a peak of 20 N may be safe at 30 N/s, but perhaps not at 100 N/s. These metrics can continue to be defined and refined with future testing in both healthy and clinical cohorts.
For a periodic function, frequency is the inverse of delta time. We reported a single frequency value corresponding to the average of a complete 1 min treatment period. However, the term “frequency” is not defined for a single cycle, and while all five physiotherapists were highly repeatable, their sinusoid-like forces did not perfectly return to the same value at regular intervals. Thus, these data do not meet the strict definition of a periodic function, which is why we chose to also report average delta time, with each delta time value being the time between subsequent push cycles. It is an interesting result that peak force was positively correlated with delta time, and thus by extension, inversely correlated with frequency. Peak force must occur when tissue displacement reaches its maximum, and so this result suggests that a therapist using a higher frequency may be applying lower treatment forces. In fact, Therapist 2 was notably the fastest at 2.4 Hz, and this same therapist applied the lowest peak forces of 5.6 ± 0.5 N. Thus, it may be that therapists (perhaps unknowingly) who apply a faster treatment speed leave less time between push cycles, and therefore less time to reach desired spine segment displacement. While our study was not sufficiently powered to elucidate this possible relationship, it is notable that the two fastest therapists produced significantly lower peak forces (p < 0.001). Future work with this device could investigate whether a higher frequency at lower force produces roughly the same “work” as a higher force at lower frequency, how work relates to clinical outcome, and perhaps a new metric could quantify “total work” of treatment as the area under the force–time curve.
It may seem intuitive that faster treatment should be associated with less time between peaks (i.e. lower delta time). We did not observe this relationship; however, we did not necessarily report treatment speed, but rather, the force application rate, which refers solely to the positive slope of the push. It should be noted that there are multiple factors that affect treatment speed. We reported the force application rate, the average peak-for-peak delta time, and the overall frequency of the 1 min treatment. Other factors that we did not report, but also affect the speed of a sinusoidal curve, are the negative slope, the turnaround time at each peak and trough, and any potential pause at each peak and trough. This wearable technology does provide the ability to measure all of these metrics, and doing so may lead to a quantified definition of a standard treatment.
Broadly speaking, the intended functions of this type of clinical maneuver are the attenuation of pain and/or increase of spinal segmental mobility. While evidence is generally supportive of an effect of cervical manual mobilization on pain 22 and mobility, 23 the true magnitude and mechanisms of those effects are largely unknown. Traditional thought in the clinical field is that the biomechanical effects of the displacement of one (or more) spinal segments relative to others are responsible for normalizing tissue length and tension. The new measurement technique described herein opens a whole new field of research where these questions can be directly explored. For example, the consequence of a higher frequency may produce inadequate treatment effect if sufficient amplitude of tissue displacement is not achieved consistently. In contrast, higher frequencies did correspond with lower variability (i.e. more consistency) in peak force application. Force, frequency, acceleration (rate), and repeatability of the maneuver can now be captured to empirically explore relationships between performance parameters and real-world treatment effectiveness.
We have established quantitative levels of performance corresponding to expert therapists, which may be used as targets for objective skills-based assessment programs. A training program based on this technology will give trainees immediate feedback on their performance compared to their peers and to experts, and provide a record of their progression. This technology can also provide real-time feedback to the physiotherapist, to give a visual or auditory alert if treatment force (or its application rate) approaches an unsafe level. This should be particularly valuable to learners and educators during skill acquisition. Whether used clinically or as part of a training program, this quantifiable feedback may reduce the risk of adverse events.
Conclusion
The new method reported in this work represents the potential for a dramatic shift in manual skills training and opens new opportunities for clinical trials of spinal manual mobilization and manipulation. With this wearable device, we can quantify performance metrics during acquisition of competency-based rehabilitation skills training and mastery. Additionally, and for the first time, it appears to be possible to standardize the physical performance components of spinal mobilization and manipulation, and objectively explore important clinical questions that stand to impact the teaching, evaluation, and performance of spinal therapies. For example, common limitations in prior clinical trials of spinal mobilization/manipulation treatment have been an inability to standardize the dosage parameters of the applied intervention beyond coarse measures such as an instrumented treatment bed. This new wearable approach appears to provide greater precision of individual clinician performance and interindividual differences. With additional testing, this will facilitate translation to the clinical arena where rigorous outcomes-based research can be conducted.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial supportfor the research, authorship, and/or publication of this article: This work was supported by the Ontario Ministry of Research, Innovation and Science (Project ER16-12-140).
Guarantor
LF.
Contributorship
RK developed the sensors and cowrote the manuscript. JO collected data and analyzed results. JS and DW were involved in protocol development, gaining ethical approval, and subject recruitment. LF and DW cowrote the manuscript and researched the literature. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Research ethics
This study was approved by the Health Science Research Ethics Board at Western University (protocol ID no. 108044).
Acknowledgments
None
