Abstract
The COVID-19 pandemic has shown how equitable vaccine distribution still remains unaccomplished. While access to COVID-19 vaccines has been efficient in the EU, the Latin American situation is very different. For example, the Andean Community reports that, as of December 2022, 83% of the population had received at least the first dose of the vaccine while only 43% had received the third dose (http://www.orasconhu.org). Motivated by the challenges and successes endured by countries in the Andean Community vis-à-vis the EU during COVID-19 vaccine roll-outs, we analyse key legal, policy and management factors affecting vaccine equity. We are especially critical of the strategy followed in the Andean Community, namely, of increasing dependency on imports and donations instead of harnessing other more effective policies. We highlight the specific role of the factors identified in achieving coherent policy strategies and propose recommendations to promote equitable vaccine distribution in the Andean Community, and beyond.
Introduction: highlighting factors affecting vaccine equity
During the COVID-19 pandemic, countries from the global South (including albeit not limited to the geographical areas this paper focuses on, namely the Andean Community), struggled to access vaccines despite efforts through organisations such as the Pan American Health Organization (PAHO) or COVAX. Needless to say, these countries were at considerable economic disadvantage in comparison to those from the global North that secured access to top-of-the-line vaccines by investing aggressively in vaccine R&D (White House Fact Sheet 2020) or procurement practices (EU strategy for COVID-19 vaccines, COM(2020)245). Most developing countries lacked the financial, logistic and other necessary capabilities to secure access to COVID-19 vaccines. However, it is certainly questionable whether economic power and wealth should be the driving factors for access to drugs in cases of global health pandemics. Indeed, the topic of access to medicine has been widely discussed in the literature, both in general (Cadillo-Chandler 2014) and in the specific context of other global health crises (e.g. HIV, Ebola and H1N1). Here, issues related to public health policies and use of intellectual property rights (IPR) exceptions have been addressed, highlighting, for instance, the need for continuity in health policy decision making (e.g. Brazil and HIV/AIDS) and implementation of bulk procurement practices (e.g. Chile). These aspects denote the political and ethical connotations in addressing past and recent pandemics. Solutions were proposed and tested before the COVID-19 pandemic; for example, in the South American context, distribution hubs had been proposed as a way to circumvent challenges related to supply chains and distribution, even though it still posed a series of challenges, such as capacity-building. In fact, COVID-19 reminded us how, when faced with global health challenges, equitable access to medicine is often just an idealistic concept.
Several factors have influenced equitable access to vaccines. In the following, this paper is motivated by the challenges endured and successes enjoyed by countries in the Andean Community vis-à-vis the EU (in general) during vaccine roll-outs. Even though a micro-comparative exercise is carried out, the purpose of the paper is not an in-depth comparison of the functioning of the Andean Pact with the EU framework. By referring to areas where similarities exist, we intend to shed light on the topic at hand, that is, access to medicines and vaccine equity. We look specifically at the key factors that affect the governance models of vaccine equity, namely the legal, policy and management factors. This multidisciplinary perspective calls for a multi-method approach that builds on existing literature from law, policy and management angles. Thus, we combine methodologies traditionally used in legal analysis, namely normative approaches, legal dogmatics and comparative law, with policy evaluation methodologies. The legal dogmatic method is normally used to identify legal rules and to solve their indeterminacies (Tuori 2002), while the comparative law method is used to compare laws and legal systems of different countries for various purposes, such as to improve or construct domestic law, as a teaching instrument, as a tool of harmonisation or, in the EU, as a tool to achieve a common European law (Husa 2023). In this article, the dogmatics method is used to build a normative framework for understanding and systematising the existing legal framework in the EU, on the one hand, and in the Andean Community, on the other, in terms of health policy related to vaccines’ equity. Subsequently, the comparative method is used to compare these legal frameworks (i.e. the EU's and the Andean Community's) in order to improve domestic law (in our case Andean Community law) and as a teaching tool to achieve a better global legal and policy framework for vaccine equity. Both legal dogmatics and comparative law are crucial methods to provide with de lege ferenda recommendations for future legislative and policy developments in this context. Moreover, the article deploys policy evaluation methodologies (Hanberger 2001) to problematise the widely accepted idea that one of the main obstacles to vaccine equity relates to the IPR protecting innovations of the vaccine in question. This multi-method approach enables us to address our main research question, namely, to identify and evaluate key aspects affecting vaccine equity in the Andean Community trading bloc. Overall, this paper aims to fill the gap in the literature by (a) providing a concrete suggestion on how the trading bloc could develop health governance cohesively and (b) publishing an international outlet in the English language will raise further awareness on the lessons and developments taking place in the Global South.
The paper is structured in three parts. Part 1 highlights key health governance aspects in the EU, on the one hand, and the Andean Community, on the other. Next, Part 2 elaborates on the factors affecting vaccine equity in general, with particular reflections on the Andean Community case. Part 3 opens the discussion by pondering how we could better achieve global equity in vaccine production and distribution. Following the analysis, the paper concludes that achieving vaccine equity requires effective global coordination and collaboration that cannot be limited to sharing IPR, but instead extends to actual transfer of technology and know-how, capacity building, and supply chain arrangements. We propose how this could be achieved.
EU and Andean Community health governance: some starting points
The existing literature on vaccine equity, particularly in the English language, that compares the EU and the Andean Community approaches is very limited. However, to understand the key factors affecting vaccine equity in these regions the research conducted on health governance in the EU, on the one hand, and in the Andean Community, on the other, provides a good starting point.
First, it should be noticed that, while definitions of governance are many, in this article our starting point is an understanding of governance as a network of practices formed essentially by exercise of power. Indeed, as Grazia Borrini-Feyerabend and Rosemary Hill (2015, 171) point out, whereas management refers to what ‘is done in pursuit of given objectives and the means and actions to achieve such objectives’, governance is about ‘who decides what the objectives are, what to do to pursue them and with what means as well as how those decisions are taken, who holds power, authority and responsibility and who is (or should be) held accountable.’ Specifically, in (health) procurement, governance refers to the overall procedural arrangements and systems established to ensure that the procurement processes adopted allow appropriate levels of control (see, e.g. https://single-market-economy.ec.europa.eu/single-market/public-procurement_en). Thus, governance regimes include legal instruments, such as IPR legislation, as well as procurement policies behind legislation, process management procedures, practices in implementing decisions taken, practices that define who was involved in decision-making in the first place, resource allocations, as well as controls and review processes and mechanisms of dispute resolution (Borrini-Feyerabend and Hill 2015).
The EU vaccines Strategy states that ‘Safe and effective vaccines against COVID-19 are the best way out of the pandemic’ (EU Vaccines Strategy, 2020). Along these lines, the EU early on followed a common approach to fighting the pandemic. Thus, the EU Commission Decision approving the agreement with the Member States on procuring COVID-19 vaccines on behalf of the Member States and related procedures was key in establishing joint procurement mechanisms through advanced purchase agreements to acquire COVID-19 vaccines (EUC(2020) 4192 final). Overall, this strategy focused on setting up the foundations for individual Member States to follow when designing their own national vaccine strategy. Specifically, the EU sought to ensure quality, safety, and efficacy of vaccines, together with timely, equitable and affordable access thereto. The strategy also seeks to address supply chain challenges, such as transportation and deployment needs (EU Vaccines Strategy 2020). The EU press release of 17 June 2020 highlights financial and regulatory aspects as key elements in achieving the goals established by the strategy. In this regard, € 2.7 billion funding up-front vaccine development, plus a commitment to engage with the regulatory flexibility required, are considered the two main pillars of this strategy (Press release, Coronavirus: Commission unveils EU vaccines strategy 2020).
On the other hand, the approach followed by the Andean Community was more based on individual-state needs (Cervantes and Sherwood 2021) rather than Community-bloc coordination. The Andean Community, or Andean Pact, is a regional integration organisation between four countries, namely, Bolivia, Colombia, Ecuador and Peru. The organisational structure includes institutions of supranational character and those without. Health governance occurs within the Andean Health Organization (ORAS-CONHU) as derived from the Hipólito Unanue Agreement. This body has no supranational character, hence encouraging cooperation amongst Andean countries’ health authorities with the aim of integrating health systems in the region (Noticias ORAS-CONHU 2022). Notwithstanding, the supranational nature of key institutions such as the Andean Tribunal of Justice (ATJ), the Andean Commission of National Executives, and the General Secretariat, allowed the region to harmonise intellectual property rights (IPR) through Decision 486. Within the Andean Pact context, decisions have direct implementation effect and, once adopted, are also to be applied by national courts, the ATJ and other administrative agencies (Helfer, Alter, and Guerzovich 2009). Close interaction between the ATJ and the administrative agencies has contributed to a balanced approach in IPR protection in the region. From an IPR perspective, especially pipeline patents, a data exclusivity 1 regime, and patents for secondary uses were initially resisted in the Andean Community. This is particularly important in the case of pharmaceuticals and access to medicines.
Data exclusivity is a challenge for vaccine distribution within both the Andean Pact and the EU. We should remember that the Agreement on Trade-Related aspects of Intellectual Property Rights (TRIPs) harmonises IPRs at the international level. As part of the mandatory package of ratification for countries within the World Trade Organization (WTO), this needs to be implemented at the national or regional level. Thus, under this framework, undisclosed information or information required by health authorities for the purpose of granting marketing authorisation needs to be protected against unfair commercial use. However, each WTO member state decides at the individual level on how best to protect such information. In the Andean Community, in order to advance IPR protection in the region, clarification was required by its member states. Thus, Decision 632 follows TRIPs and also leaves it as a prerogative of the state. 2 In general, Andean Community Decision 486 harmonises IPRs in the region, and although it protects undisclosed information it does so similarly to TRIPs, so that no harmonised data exclusivity system exists in the region, either.
This differs from the EU framework, where the system is to an extent harmonised in terms of medicines approved through a centralised procedure (Cadillo Chandler 2020). In this regard, Colombian (t’ Hoen 2022) and Peruvian legislation (Raquel Artecona and Rosine Plank-Brumback 2016) contain data exclusivity clauses within their pharmaceutical regimes to ensure that commercialisation by originators enjoys a market exclusivity period of five years from the date marketing authorisation was granted. Even though disputes related to data exclusivity are not new in the region, these do not seem to have played a role in the context of vaccine distribution in general. Admittedly, the structure of the Andean Community's health governance does not enjoy direct implementation effect, so efforts to fight COVID-19 in the region required close cooperation between the countries concerned. A series of activities took place during the pandemic to address its progress, to share statistics, and to design measures for each Andean Community country to fight the spread of the virus (Mariano, Bressan, and Ferreira 2022). Of all these efforts, one key achievement is the proposal to harmonise the Community Directive ‘Andean Strategy on Medical Devices’ of the Andean Parliament (Noticias ORAS-CONHU, 2022). Health governance has remained at the individual member-state level, which means that harmonising the strategy signifies further cohesion and collaboration within the trading bloc, making the Andean Community more closely resemble its inspirational foundation, the EU.
While in the Andean Community the response to the pandemic was largely dependent on the protocols designed and implemented by each country, at the EU level the strategy was more uniform and coordinated. Our hypothesis is that this divergence is entrenched in the structural difference between the two community blocs in themselves. The following section seeks to understand further some of the factors impacting vaccine equity in the Andean Community.
Vaccine equity governance: key factors
Vaccine equity has been defined by the UN Development Programme, as the ‘allocation of vaccines across all countries based on needs and regardless of their economic status’ (UNDP, 2022). Although the term became a mantra during the pandemic, the elements for achieving it have not received enough attention. Therefore, this section seeks to understand the tools embedded within initiatives such as COVAX and other vaccine strategies to actually fulfil the goal of vaccine equity, that is, the building blocks of health governance. In the first place, this naturally draws attention to the concept of health governance itself, as presented above.
Procurement measures
COVAX is a pooled procurement initiative belonging to the Access to COVID-19 Tools (ACT) Accelerator (see: https://www.who.int/initiatives/act-accelerator). Its purpose is to foster ‘global collaboration to accelerate the development, production, and equitable access to COVID-19 tests, treatments, and vaccines’ (GAVI 2021). Pooled procurement measures like COVAX have also been used in the past, for instance, to address the purchase of HIV medicines (t’ Hoen, Kujinga, and Boulet 2018). Generally, this mechanism allows for several buyers to be combined and operate under a single entity that purchases, for example, vaccines on behalf of those buyers (Immunization Financing Toolkit The World Bank and GAVI Alliance 2010).
COVAX was founded in April 2020 as the multistakeholder group and vaccine distribution arm of ACT. Its principal function is to handle the financing of the purchase of the COVID-19 vaccine (Cadillo Chandler, Ballardini, and Nuottila 2022). COVAX is also part of GAVI, the Vaccine Alliance, the Coalition for Epidemic Preparedness Innovations (CEPI), and the World Health Organization (WHO). Thus, COVAX also maintains strong relationships with the World Economic Forum, the Bill and Melinda Gates Foundation and Big Pharma. COVAX is not a partnership for developing vaccines. On the contrary, it functions as a financing body for the purchase of vaccines, or as an intermediary between manufacturers and consumers (these being understood as governments). Countries participating in COVAX are divided between ‘self-financing’ and ‘funded’ ones, which translates into countries having to pay for vaccines or not. Currently, fourteen countries in Latin America are self-financing, and five belong to the funded group (see: https://www.as-coa.org/articles/what-covax-and-what-does-it-mean-latin-america). From the moment of its launch, COVAX promised to immunise 20% of the population in countries funding the programme, hence providing access to vaccines in low- and middle-income countries. By March 2021, for instance, 12 million doses had been shipped to at least 29 countries (Usher 2021). Notwithstanding the positive promises, several challenges remain.
First of all, despite efforts to fulfil its goal – namely, providing access to the vaccine for all countries participating in the financing model – COVAX has also spotlighted how wealthier nations have used the system without actually needing to do so (Sariola 2021). Clearly, ‘vaccine nationalism’, strict IPR protection strategies and wealthier countries’ purchasing power have threatened the success of COVAX, making distribution of vaccines far from equitable. For example, while some countries hoarded vaccines, South America had to wait for vaccines way longer than desired (https://www.vox.com/future-perfect/22440986/covax-challenges-covid-19-vaccines-global-inequity). South American countries only began receiving COVID-19 vaccine doses as early or as late as 21 March 2021 (see: https://www.paho.org/es/covax-americas), by which time the EU had already immunised a large percentage of its population.
Second, in terms of vaccine distribution, IPR may still pose a challenge, for instance the prevalent and different data-exclusivity regimes in the region may affect distribution. While Colombia and Perú have adopted a model that grants protection for five years from the time the marketing authorisation was granted, Bolivia and Ecuador 3 are not providing such protection (Uscátegui 2014).
Third, even though COVAX as a platform was mainly meant for the purchase of vaccines, it also supports R&D and manufacturing of vaccine candidates to later negotiate pricing once these are suitable for use (Gleckman 2021). Considering COVAX's support for the development of vaccine candidates, IPR issues – as well as technology and know-how transfer to guarantee both protection and access to innovations – cannot be ignored. Admittedly, the complexities in manufacturing the vaccine are vast. However, improving or allowing production hubs for the vaccine, for instance in Argentina and Brazil, will in principle lead both to further access and also technology transfer between the originator and authorised third parties (PAHO 2021a).
Fourth, despite efforts towards enhancing collaboration and IP sharing, concerns still exist about widely sharing IPR and know-how. All this raises questions as to the need to subvert IPR, since COVAX seems to be a smokescreen for strong IP protection, hence accentuating vaccine inequality. Indeed, these efforts remain very limited (Silverman 2020) even if some manufacturers have engaged in a certain level of cooperation – mostly through voluntary licences such as the AstraZeneca-CEPI partnership and the GAVI-Serum Institute of India agreement – contracted production (e.g. the agreement between Novartis and BioNtech) and even engaged in technology transfer (e.g. the case of NOVARAX and the Canada Deal). In the case of Bolivia, a national strategic plan was needed before vaccines could be delivered through COVAX. This plan allowed for bilateral cooperation between the country and other institutions, and COVAX itself (PAHO 2021b).
Fifth, to an extent, COVAX provided a certain level of ‘global’ coordination in terms of vaccine distribution. Nevertheless, vaccine distribution in both the EU and the Andean Community was not solely dependent on the scheme. Indeed, these two blocs followed completely different strategies. On the one hand, as previously mentioned, even when countries in South America belong to the Andean Community, coordination strategy as a bloc or coordinated regional response to access COVID-19 vaccines was non-existent. In contrast, the EU, as a bloc, did negotiate access to vaccines through advance purchase agreements and also designed a coordinated strategy for the entire bloc. As we have seen, this different approach in handling the health emergency may be a result of structural differences between the EU and the Andean Community as such. Nonetheless, in terms of IP governance both blocs have harmonised IPR accordingly, which begs the question: why did countries in the Andean Community not seek a coordinated response? To answer this question, it is first necessary to open up how the Andean Community differs from the EU in terms of health strategy design.
While the EU provides common guidelines at Union level for the individual Member States to follow in the health context, the Andean Community leaves each member state to design and implement its own strategy. Although the Andean Community leaders expressed their commitment to fight the effects of the pandemic, this commitment focused on intra community trade by prompting process digitisation, preventing the spread of the disease, and coordinating safety and health measures at work (https://www.comunidadandina.org/notas-de-prensa/estrategia-de-la-comunidad-andina-frente-al-covid19-fue-presentada-por-el-secretario-general-de-la-can-a-embajadores-en-paises-andinos/, June 2020). In terms of a coordinated response, during April 2020 the Andean Community did foresee the possibility to evaluate joint measures for the purchase of medical supplies aiming to effectively address emerging health needs throughout the pandemic (Declaración de los Ministros de Relaciones Exteriores y de Comercio Exterior de la Comunidad Andina respecto a la propagación del coronavirus (COVID-19), June 2020). However, as such, this declaration from the Andean Community leaders did not commit to a strategy to engage in vaccine equity. On the contrary, it appears that all efforts were made in order to strengthen trade, leaving each Andean Community member to define the strategy to follow in regard to accessing COVID-19 vaccines.
Finally – and perhaps most importantly, as pointed out by the country study published by CEPAL in 2022 analysing vaccine inequity within the south-American region – throughout the pandemic the region has been heavily dependent on donations through COVAX (de Castillo and Castillo 2022.) Figure 1 reflects the number of doses per capita purchased by country. This is juxtaposed by the number of doses distributed by COVAX in the region. This demonstrates that South America has been lagging severely behind in regard to vaccine roll-outs and that the doses delivered are still not sufficient to immunise even half of the population in each of the countries participating in COVAX. The strategy of relying on vaccine donations instead of strengthening other legal, policy and management strategies such as those used in the EU (e.g. via the EU Joint Procurement Initiative and European Health Emergency preparedness and Response Authority (HERA)) is instead a major cause of continuous vaccine inequity in the Andean Community.
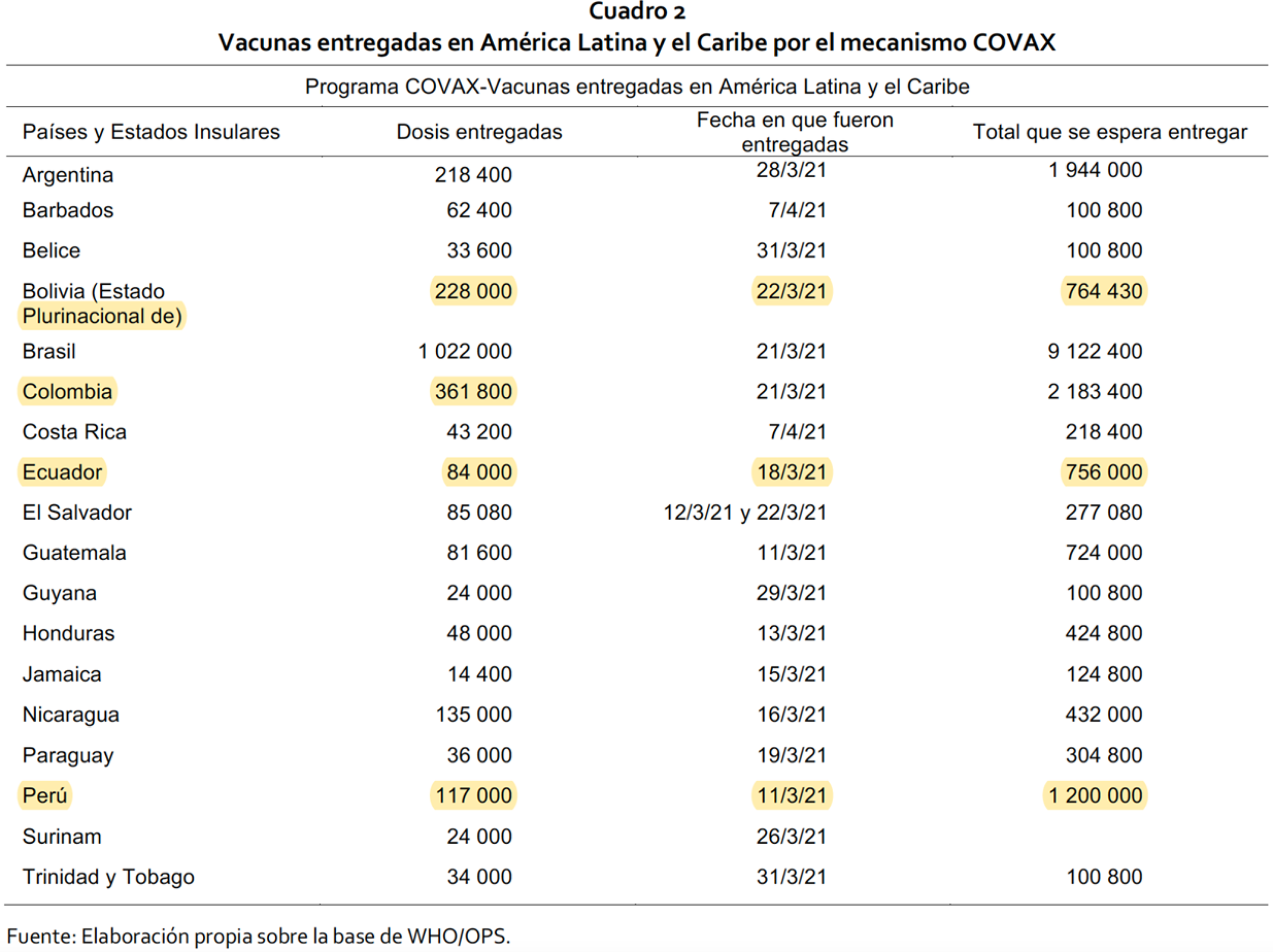
Doses delivered by country, vaccines delivered by COVAX in Latin America and the Caribbean; first column from left to right: countries and island states; doses delivered; delivery date; and total to be delivered. (de Castillo and Castillo 2022).
Figure 1 shows the disparity between vaccine doses needed and the amount actually delivered by COVAX. For instance, while Bolivia expected to receive 764,430 doses, by 22 March 2021 only 228,000 doses had actually been delivered. In proportion, other Andean countries in the list are in an even ‘worse’ position than Bolivia. The situation in the region remains at best challenging.
IPR tools for vaccine equity
As previously mentioned, legal instruments form a key element of most governance models – albeit they are not only about governance models. In the context of governance models of vaccine equity, the most important legal instrument is the IPR system, as already emerges from the sections above. Generally speaking, IPR are both essential and problematic in the context of global vaccine equity. On the one hand, IPR – especially patents and data exclusivities – are essential to enable investors and manufacturers to reap commercial benefits from their innovations, recouping the significant investments needed for the long-term and costly R&D needed for developing new drugs. On the other hand, IPR, and the relative temporary monopoly rights they create, might impose obstacles to accessing, as well as sharing and co-creating, protected innovations (Cadillo-Chandler 2014). Indeed, the TRIPs Agreement requires member states to implement minimum standards of intellectual property protection, including patents for pharmaceutical products, but also contains ‘flexibilities’ – such as compulsory licensing, 4 Bolar/research exemption, 5 parallel importation 6 and an LDC transition period 7 – designed to address barriers to accessing medicines (Tenni et al. 2022). Moreover, national intellectual property laws can also include so-called TRIPs-plus rules (El Said 2022; t’Hoen et al. 2018) that go beyond what is required by the TRIPs Agreement in terms of ‘flexibilities’, for instance in terms of data exclusivities.
The COVID-19 pandemic with its global reach and spread has clearly revealed in multiple ways both elements of this long-known contrast as well as the challenges and limitations of IPR flexibilities in truly enabling ‘medicines equity’ (Ballardini et al. 2022). The EU-Andean Community comparison offers fruitful ground for this discussion, as limitations became evident in terms of relying on the use of compulsory licensing only. Compulsory licensing is one of the regulatory mechanisms typically mentioned as offering ways to circumvent the exclusive effects of IPR: while ordinary licensing involves a contract between the rightholder and the party seeking to use the subject matter covered by the IP right, under compulsory licensing this authorisation is replaced by the decision of a competent authority or court. The mechanism of compulsory licensing is clearly regarded as a last resort, that is, when licensing negotiations between the parties are futile or if other factors stipulate non-consensual solutions. Although in the Andean Community no compulsory licensing was issued in the context of COVID-19 vaccines, the Community countries are not strangers to relying on this instrument. For instance, in the case ‘Proceso 144-IP-2019’, the Andean Court of Justice (ACJ) in 2021 was asked to interpret the legitimacy of a compulsory licence granted by the Ecuadorian Intellectual Property Office on the basis of public interest. Even though neither this compulsory licence, nor the case itself, concern COVID-19 vaccines, this ruling considers the contours of public interest as a rationale for a compulsory licence due to health emergencies and is therefore relevant for the case here under discussion. In fact, the ACJ stated that when a health emergency is caused by a pandemic, authorities granting a compulsory licence are required not only to declare the existence of a pandemic, but also need to identify its negative effects on society. Furthermore, authorities must identify the virus, pathogen, vulnerable population amongst other details that amount not only to a necessity test, but that also seem to limit Andean Countries’ use of compulsory licences in accordance with minimum standards of protection as foreseen in TRIPS (Tribunal de Justicia de la Comunidad Andina-Proceso 144-IP-2019). Another case concerns Perú, which in May 2017 conducted a market analysis to demonstrate abusive practices by the rightholder in relation to sale of the drug Atazanavir in the country. In doing so, Perú sought to enact a compulsory licence on the basis of public interest, as well as an action for abuse of a dominant position on the grounds of violation of competition law rules. Although the compulsory licence was never granted, the pharmaceutical was in fact declared of public interest by the government of Perú. Following the expiration of the patent, the government proceeded to purchase it through the Pan American Health Organization (Castillo 2019). In summary, although compulsory licences are considered to be a powerful bargaining tool to negotiate lower medicine prices (Urias and Ramani 2020), use of this mechanism has not been widespread in the region – nor indeed worldwide – to fulfil access to medicine policy goals and even less in times of global pandemic.
Alternative methods for sharing and promoting co-creation of vaccines and COVID-19 related innovations and IPR were also tested during the pandemic to try to navigate the challenges. For instance, voluntary licensing structures such as IP pools (i.e. agreements between two or more parties to cross-license parts of their current or future IP portfolios) and IP pledges (i.e. publicly announced interventions by IP owners to out-license active IPR for a certain period of time, either ‘free from or bound to certain conditions for a reasonable or no monetary compensation’ (Ehrnsperger and Tietze 2019)) have been used by various companies and organisations to secure access to essential medical equipment during COVID-19, for instance the Open COVID Pledge: https://opencovidpledge.org/. Although these types of voluntary licensing carry advantages, the challenge is that these tools only share IPR, not the trade secrets or know-how often needed, for example, to reproduce the innovation concerned (e.g. related to COVID-19 vaccine production), making use of these tools difficult. Moreover, without solid capacity building in place, these instruments are not so helpful. Thus, it is doubtful whether these instruments have been useful at all in the case of developing countries – or regions such as the Andean Community – to enhance COVID-19 vaccine (or medicine) production and distribution. In addition, the reality is that voluntary licensing tends to be limited both to a particular territory and in time – and to our knowledge there is no evidence that the Andean Community was a target for any of these voluntary licensing strategies.
Finally, it is important to mention that, at the end of 2022, there is no internationally recognised general exception to IPR enforcement for health emergencies. Potential TRIPs ‘waivers’ have been considered to facilitate access to and distribution of innovations in critical situations, as with the COVID-19 emergency (Matthews and Minssen 2021a and 2021b). The Andean Community reportedly acknowledged and supported the proposal for an IPR waiver in times of pandemic. One the one hand, Bolivia in the forefront of the COVID-19 fight and in order to access vaccines not only signed an agreement with Johnson and Johnson to manufacture the vaccine (Martell 2021), but also emphasised the need for the WTO to waive patent rights over any vaccine to ensure equal distribution (Martell 2021). On the other hand, the Andean Community members acknowledged that vaccines, pharmaceuticals and medical devices are a common good, and as such exclusive rights such as IPR should not become a barrier to accessing vaccines or any other health/medicine-related device to control or fight the pandemic (ORAS-CONHU in Comunicado N° 5 from 11 May 2021). However, all these discussions have led to not much more than lengthy and highly political negotiations on how to resolve the ‘IP problem’ in emergency cases, without bringing about any concrete solution.
Know-how and technology transfer
In the case of global pandemics like COVID-19, demand for vaccine and medicine doses explodes and is counted in billions. Inevitably, this leads to major challenges for key manufacturers with supply chain management, availability of raw materials, production capacity, and logistics. In the harshest stages of the COVID-19 pandemic, these factors were slowing down production scale-up for several COVID-19 vaccine manufacturers. When considering vaccine equity, these problems raise concerns far beyond the discussion on access to IPR on vaccine-related innovations. Indeed, even assuming a solution to get wider access to vaccine-related IPR utilising TRIPs flexibilities, IP pools, or IP pledges, the valid question remains: how to incentivise and foster local capacity for vaccine production? For this to happen, both technology transfer and access to the know-how and trade secrets needed to produce the vaccines are crucial – yet not achievable by simply opening up or sharing IPR on the innovations concerned (Krishtel and Hassan 2021; Nicholson Price, Rai, and Minssen 2020).
The technology transfer challenge is nothing new. In the latter half of the previous century, pharmaceutical production, including vaccines, was consolidated into large, global pharmaceutical corporations, often referred to as ‘Big Pharma’. This centralisation model led to the clear consequence that these companies were amongst the only ones to be able to invest in developing new vaccines and modernising production facilities (Hendriks 2017; Homma and Knouss 1994). Another consequence was a radical decrease in local production of vaccines. This was also because many countries did not have national public policies to favour local vaccine production, nor did they prioritise development and maintenance of in-country production capacity (Homma and Knouss 1994; Milstien, Gaulé, and Kaddar 2007). Moreover, vaccine development and production, being highly investment-intensive, are profitable only in high volumes. As an example of market dynamics, when PAHO established a revolving fund in 1977 for the purchase of vaccines by Latin American and Caribbean countries, it managed to obtain good-quality vaccines at low prices from the global market; however, at the same time this was not encouraging for local producers to invest in modernised local production (Homma and Knouss 1994). Overall, this development led to a problematic situation. For example, many diseases basically ceased to exist in developed countries (Manner, Graef, and Dent 2019). At the same time, however, limited commercial incentives to invest in vaccines for these same diseases, particularly compared to pharmaceutical products for diseases common in developed countries, have made it difficult to get affordable access to these vaccines (LaForce et al. 2007).
Earlier studies in South America identified some key production-related preconditions for successful transfer of vaccine technology (Homma and Knouss 1994). Firstly, local governments need to support the transfer politically and financially. It is also important that local, national, and regional institutions are involved in the process. A scientific and technical infrastructure needs to be in place and capable of absorbing the technology. And finally, raw materials and supplies need to be accessible. Even if the technology transfer is successful, there is a risk that the receiving party is not able to keep up with technological development, for example, they do not receive technology updates from the originator, or cannot develop the technology further by themselves.
When the PAHO revolving fund and UNICEF (later joined by the GAVI alliance) began to buy vaccines on behalf of, and for, developing countries in the 1980s, there was a challenge to set the quality criteria to tender not only on the basis of price (Milstien, Gaulé, and Kaddar 2007). The WHO participated in setting the quality criteria by developing a prequalification procedure for vaccine manufacturers who were qualified to participate in tendering. Vaccine manufacturers from developing countries had difficulty in meeting the prequalification standards and many of them ceased production in the 1980s (Milstien, Gaulé, and Kaddar 2007). The situation began slowly to improve when the Developing Countries Vaccine Manufacturers Network (DCVMN) was founded in 2000, and several UN organisations started to support technology transfer-related initiatives (DVCMN 2022; Jadhav et al. 2008). As a result, vaccine production is emerging in developing countries, mainly to meet local demand (Ca 2007; Milstien, Gaulé, and Kaddar 2007) but usually these are old, bulk vaccines, not new patent-protected vaccines. However, there are no companies from the Andean community in DCVMN. From South America, there is one company from Argentina and two are from Brazil. On the other hand, DVCMN's role is important for vaccine supply in the Andean community. On average, 70% of the vaccine doses procured by PAHO in 2007 were delivered by a DVCMN member (Jadhav et al. 2008).
Technology transfers have not been easy to organise in general. In 2006, the WHO received funding to start local influenza vaccine production in six countries but did not manage to find any established vaccine manufacturer to participate, mainly due to the required long-term commitment (Friede et al. 2009). The WHO also investigated technology transfer opportunities for influenza vaccines and found no major IP barriers for transfer, but recognised the main obstacle as being access to technical know-how and regulatory dossiers (Friede et al. 2011) In the end, a general technology transfer hub was founded in the Netherlands to support new manufacturers to work with the related technologies free of intellectual property barriers (Beurret, Hamidi, and Kreeftenberg 2012; Friede et al. 2009). These efforts were successful, with mass production starting in several new production facilities, for example, in Brazil, India and Indonesia (Dhere et al. 2011; Miyaki et al. 2011; Suhardono et al. 2011). Technology transfers included intensive training of the recipient parties (Suhardono et al. 2011).
Looking at COVID-19, there are several challenging issues related to possibilities for successful technology transfers. First, many of the COVID-19 vaccines utilised recently developed vaccine technologies including mRNA, protein subunit, and viral vector (Asundi, O’Leary, and Bhadelia 2021; Wouters et al. 2021). Clearly, new technologies include barriers to transfer in the form of intellectual property rights or regulatory procedures that in a sense relate to IPR, namely, marketing authorisations/data exclusivities (Gopakumar, Chetali, and Sangeeta 2021). In addition, challenges go beyond IPR. These technologies also require new production and logistics infrastructure, technical know-how, manufacturing expertise, and supply chain capabilities (Wouters et al. 2021).
Capacity building
Capacity-building, as defined by the United Nations, refers to ‘the process of developing and strengthening the skills, instincts, abilities, processes and resources that organisations and communities need to survive, adapt, and thrive in a fast-changing world’ (UN Capacity Development 2022). There are some good examples of long-term capacity building in the past. Funded by institutions from Sweden and Vietnam, the National Laboratory of Bacteriology of Sweden educated young Vietnamese researchers between 1978 and 1994 (Ca 2007). This cooperation initiated vaccine research, development and manufacturing in Vietnam, producing millions of doses annually (in 2007). Another long-term success is in the shape of operations led by the RIVM, the Netherlands’ public health institute together with the Netherlands Vaccine Institute (NVI). Starting in the 1970s, the RIVM has produced dozens of international courses on vaccine development and production in cooperation with the WHO for public and private vaccine producers, and for vaccine authorities and control laboratories in many countries (Hendriks 2012). In addition to supporting development of scientific and technical human capacity in the recipient countries, the RIVM also coordinated several important vaccine technology transfers to initiate vaccine production in developing countries between 1972 and 2021 (Hendriks 2012). From the beginning of 2013, their operations were moved to the Institute for Translational Vaccinology, which provides services for vaccine researchers from discovery towards the start of production (Hendriks et al. 2013).
The importance of national capacity for translational research is recognised as a key factor enabling vaccine production in developing countries (Kumraj et al. 2022). This means national policies and strategies for efficiently translating the findings from basic research into vaccine development by the national pharmaceutical industry (Homma et al. 2013). For example, in Brazil, a strategic effort has been made to support basic research on vaccines and manufacturing technologies. As a result, two Brazilian vaccine producers are now members of DVCMN. By possessing relevant resources and facilities, one of them, the Bio-Manguinhos Institute of Technology on Immunobiologicals at the Oswaldo Cruz Foundation (Fiocruz), acquired a licence for COVID-19 (adenovirus vector) vaccine from AstraZeneca (Medeiros et al. 2022). The commercial arrangement also included technology transfer to enable independent, national production. The licensing process and technology transfer was supported by the Brazilian Federal Government, which opened a large credit to fund the operations initiating national manufacturing and local supply. Fiocruz started production of COVID-19 vaccines with imported active pharmaceutical ingredients in March 2021, and totally independent production in August 2021, only 18 months after the start of the pandemic (Medeiros et al. 2022). This case demonstrates a successful technology transfer with local scientific and technology capacity and a governmentally supported bilateral commercial licensing agreement.
In terms of capacity building and governmental efforts in the fight against COVID-19, Bolivia was praised by the Pan American Health Organization in a report published in 2022. This report highlights how the Bolivian government facilitated investment, granted access and availability of vaccines, moved needed resources and eased regulatory and communication processes (PAHO 2021a). Admittedly, capacity building within the context of IPRs and the COVID-19 vaccine relate more to actual training and cooperation to reproduce the vaccine. However, allocating funding and collecting data though technical cooperation may be a step forward in the capacity building required. Technical cooperation between PAHO and the Bolivian government, the case in point, is claimed to have led to significant improvements in handling the pandemic as well as advancing health systems.
Bolivia's access to and distribution of vaccines was dependent both on bilateral governmental efforts and on cooperation with COVAX. In regard to bilateral efforts, direct negotiations with the industry, and governmental willingness to adopt nationwide strategies and allocation of funds seem to have played a significant role: in short, a proactive approach to accessing vaccines, instead of relying solely on donations. On the capacity-building end, diagnosis and development of protocols were amongst the highlights in the report (PAHO 2021b). Thus, capacity-building in the Bolivian case does not seem to be exclusively about having the infrastructure to manufacture the vaccine, but also points to a series of complexities embedded in tackling health emergencies.
As for transfer of technology and capacity building in the region, in general, an initiative from the WHO, the Medicines Patent Pool (MPP) and the PAHO to collaborate in developing the ground-breaking mRNA vaccine technology, has brought together Fiocruz in Brazil, and Sinergium Biotech, a private biopharmaceutical company in Argentina (PAHO 2021c). Nevertheless, strictly speaking, no similar initiative has taken place in the Andean Community country members’ jurisdiction. Admittedly, the facilities in Argentina and Brazil will enable the Latin American region to obtain vaccines, mRNA being the case in point, much faster than if relying exclusively on donations or imports.
How do we achieve global equity in vaccine production and access?
Finding a solution, or even aiming at realising vaccine equity, calls for a closer look into the concept itself. As previously mentioned, the United Nations has stated that ‘vaccine equity, means that vaccines should be allocated across all countries based on needs and regardless of their economic status. Access to and allocation of vaccines should be based on principles grounded in the right of every human to enjoy the highest attainable standard of health without distinction of race, religion, political belief, economic, or any other social condition.’ (UNDP 2022).
Allocation of vaccines, as stated in this quote, would somehow point to the existence of a supranational entity guiding not only the direction of investments, R&D and production, but also distribution of end products. COVAX, as a pooled procurement mechanism, aims to achieve or contribute to vaccine equity. However, it is quite clear that coordination is a major problem in the transition. As an example of this, to date there is no systematic holistic global database containing key information on, for example, the world's R&D needs and related initiatives. In an effort to map out the data on the global roll-out of COVID-19 vaccines with relevant socioeconomic information, the UNDP, together with the WHO and the University of Oxford in cooperation across the UN system, began a joint initiative titled the Global Dashboard for Vaccine Equity. This initiative clearly highlighted the need for further global cooperation that goes beyond the manufacture of vaccines. Yet it only represents a starting point, and much still needs to be done to achieve the goal of reducing fragmentation of knowledge and boosting coordination, collaboration and co-creation of innovations and sharing of crucial information. In line with SDG17 ‘Partnership for the goals’, this transition requires efforts to further develop the policy presented above, legal as well as supply chain management and capacity building action through the lenses of values such as global participation, inclusiveness and diversity. Intelligent cooperation would enable finding win-win types of collaboration, instead of relying on governance models, IPR, skills and capacity all held individually by different partners in an uncoordinated and fragmented manner. Overall, this could trigger an important step towards achieving vaccine equity in general, including in the context of COVID-19 vaccines and in relation to the Andean Community case.
From a policy point of view, for instance, this vision could be concretised via a paradigmatic policy change, meaning by igniting global cooperation and coordination policies driven by ‘life’ as the key value. In essence, paradigmatic policy change means both a new direction in state policy that represents a new way of thinking (Bryant 2002). Traditionally, IPR policy and health policies have been developed in isolation, although in practice we have seen how both fields are intertwined (Cadillo Chandler 2016). Thus, policy developments in these areas have not radically changed, but instead continued accentuating a traditional approach to IPR protection. The global pandemic has demonstrated that a continued fragmented approach to use of traditional IPR flexibilities would not be suitable in times of global health crises. With that in mind, countries like Mexico have made use of a paradigmatic policy approach at the national level by temporarily integrating an IPR exception that allows approval for commercialisation of biotechnological medicines, biosimilars and bioequivalent in the country without having to replicate clinical trials in the country if the product's interchangeability studies or biosimilar products have already been approved for commercialisation abroad. 8 Even though Mexico is not part of this paper's analysis, the above mentioned policy approach highlights a necessary level of complementarity between IPR and public health policymaking (Cadillo Chandler 2014) that to our knowledge has not been done before.
At the international level, a paradigmatic policy change would entail a multi-layered policy approach that takes into account the complexities of addressing a global pandemic. In other words, a policy guideline to follow in times of pandemic that takes into consideration all areas needed to achieve vaccine equity, namely: funding – for example, investment – IPR, supply chain and infrastructure. Admittedly, this proposal may resemble a global agreement. However, by committing to share information about R&D needs, availability and types of resources, providers, and stakeholders, countries would enable setting up a database that maps out the global situation. In this case, technologies such as artificial intelligence (AI) may be useful. Such a commitment should be achieved through alternative tools, such as science diplomacy – a concept that organically emerges, as it entails scientific (i.e. pharma knowledge) collaboration among nations, scientists, public and private parties to address common problems to build constructive international partnerships (Nina V. Fedoroff, Science and Technology Adviser to the Secretary of State and to the Administrator of the U.S. Agency for International Development (USAID) 2009) – or soft law that enables coordination and cooperation, which so far have been lacking at the Andean Community level. True, the situation at the global level shows fragmentation of initiatives, and at the Andean Community level even when no uniform health policy exists, during COVID-19 cooperation and collaboration was strengthened through political will and common regional objectives (ORAS-CONHU Comunicato N°5).
Moreover, cooperation, collaboration and sharing could be concretised in the legal frameworks that the governance model relies on – thus in the IPR system – in various ways in order to optimise mitigation of both monetary and health-related losses. An option could be to develop new legal tools to support co-innovation and open innovation structures as well as licensing models both to promote more vaccine-related co-development between actors operating in developed and developing countries, as well as better diffusion of existing innovations. As above presented, one of the main challenges with most of the currently in-use structures for IP co-creation and sharing (for instance, compulsory licensing, IPR pools and pledges) is that even when IPR is shared, the trade secrets and know-how needed to reproduce innovations or put them into use are not. Indeed, any legal constraints forcing manufacturers to share crucial information on their innovations must be carefully balanced with the fact that substantial investment is needed to develop the required innovations, especially in the case of emergencies when new drugs are needed at an unprecedentedly high speed – as in the case of COVID-19 vaccines. Another point to consider is that often drug innovations are not in silos, while crucial inventions relevant for one drug (e.g. COVID-19 vaccines) can also be crucial for other medicines (not necessarily relevant for the emergency at hand). In this regard, for instance Ballardini et al. (2022) have proposed that similar constructions to those used in the context of the mobile networks industry in relation to standard essential patents could be a tool to be explored. In sum, they suggest that when national governments predict shortages of specific IP-protected equipment that is declared ‘essential’ to fight an emergency (e.g. equipment needed to produce COVID-19 vaccines), ‘those organisations holding IP rights on these products or methods would assign their rights to, for instance, specific collective management organisations under Fair, Reasonable and Non-Discriminatory (FRAND) licensing conditions to enable manufacturing to be ramped up globally’ (Ballardini et al. 2022). This could foster more equitable distribution globally. However, more should be done also to strengthen local capacity building in under-developed countries like the Andean Community. Indeed, sharing knowledge (e.g. IPR and know-how) is notably not sufficient without a local infrastructure in place.
In this regard, improvements could be sought through national, or regional action to support re-establishment of regional capabilities on vaccinology. This includes several activities, in which both public and private actors are necessary. Local vaccine production is not possible without the existing scientific community of vaccine researchers and experts. This requires funding and governmental support for developing universities and research institutes to carry out basic research on vaccines and vaccinology. This is a fundamental enabler for the growth of regional vaccine production. In addition, there is a need for local strategies and national policies to establish the infrastructure supporting vaccine developers and manufacturers. Cooperation between research institutes and pharmaceutical companies should be encouraged and funded. Local production requires large investment by regional companies, so that national and international social investment funds might be required to support local companies. On the other hand, it might be convenient for local governments to act as partial owners of and shareholders in local companies producing vaccines for the region.
It is not realistic to expect that local vaccine production could be started independently. Thus, international cooperation is needed. There are already good examples from the past of long-term scientific and technology capacity building by committed research institutes and centralised vaccine technology and training hubs (examples from Sweden and the Netherlands were mentioned earlier). International collaboration can support educational activities on vaccinology locally, facilitate transfer of advanced vaccine technologies, and accelerate local vaccine production for new and emerging diseases. With these types of activities facilitating long-term capacity building, the international community could support national and regional organisations to prepare for fast-paced technology transfers in case of a new pandemic similar to COVID-19. Local resources with manufacturing capabilities and absorptive capacity would enable a rapid start to production after signing a bilateral commercial licensing agreement with a technology owner, or adopting technology through a TRIPs flexibility arrangement. Surely, this would also improve the capabilities of local research institutes and manufacturers to develop original vaccines against existing and emerging pathogens and decrease the level of dependency on global supply chain in case of an emergency.
The way ahead: a need for global collaboration and coordination
This paper began by drawing a brief comparison between the EU and the Andean Community, with the purpose of understanding the successes and challenges related to vaccine equity during the global COVID-19 pandemic in the Andean Community region. Much has been said in terms of lack of access to COVID-19 vaccines, usually pointing to IPR as the main cause. Nevertheless, while the aforementioned premises were under scrutiny, several challenges emerged that go beyond merely opening up IPR via, for example, TRIPs waivers. Instead, and more generally, governance models stand foremost among the factors affecting vaccine equity.
Furthermore, coherence between policymaking and reality is paramount in achieving sustainable solutions. Shifting accountability from main actors to secondary actors will resolve neither capacity building nor transfer of technology challenges. This implies that fragmented initiatives or donation dependency is not – and cannot be – a sustainable governance model. On the contrary, only by increasing global cooperation and collaboration can vaccine equity be achieved. While initiatives such as COVAX and IPR voluntary licensing mechanisms (such as IP pools and pledges) or IPR flexibilities (such as compulsory licensing) are important steps forward in addressing vaccine equity, these are still – and paradoxically – accentuating inequalities between countries from the Global South and those of the Global North. These initiatives have proven successful when accompanied by local or regional coherent policymaking – cases in point being the EU Vaccine Strategy and the Bolivian example presented in this paper. In both jurisdictions, additional multidisciplinary approaches were followed. The EU sought to ensure quality, safety, and efficacy of vaccines, together with timely, equitable and affordable access thereto. EU strategy also addressed supply chain challenges, such as transportation and deployment needs. Whereas Bolivia worked closely with COVAX to improve its national strategy, it also sought cooperation and collaboration bilaterally with the pharmaceutical industry. Indeed, defining common policy goals and objectives are key to defining global cooperation and collaboration.
In the Andean Community, while the ORAS-CONHU fostered collaboration amongst its members, a common policy in the context of health was missing. Instead, the trading bloc made substantial efforts in relation to strengthening trade. Even when IPRs are trade related, and are the most important legal instrument to both incentivise development and diffusion of key innovations related to vaccines, the global pandemic showed how the current IPR system is not apt to promote and foster local capacity building. Through the brief analysis carried out in the previous sections, it becomes clear that vaccine equity is neither – as often claimed – only about IPR policy development, nor is it about public health alone. Rather, it is a complex phenomenon that demands collaboration and coordination at the global level, particularly when thinking of future and emerging global pandemics. Considering that ORAS – CONHU's objective is to coordinate and support Andean Members’ efforts in improving health (https://www.orasconhu.org/es/presentacion), this sub regional organisation could work as the umbrella organisation to map out both strategic health needs and develop strategic collaboration amongst stakeholders in the region. True, ORAS-CONHU does not have legislative competences, thus, intervention to develop a cohesive policy, needs to come from the Andean Community Commission. Ideally an Andean Strategic Health Common Policy name envisaged by the authors would take into account the Andean Community IP framework in a way that provides for exceptions to, that is, the protection of information in times of global pandemic. Additionally, would grant ORAS-CONHU the competence to manage a strategic fund to develop a regional database mapping out health needs, know-how, innovators, providers in all required fields to address vaccine equity. This fund would also require a joint procurement mechanism – similar to that of the EU – to enhance the Andean bloc's bargaining power. In terms of sharing knowledge, a sound licensing system envisaging fair conditions will enable further collaboration, similarly to FRAND as previously suggested (Ballardini et al. 2022). Indeed, developing a coherent common policy requires collaboration amongst stakeholders (legislators, scientists, members of the public, public-private partnerships, etc), thus, looking at the solutions provided in the previous section already draws the reader's attention to existing means of collaboration and cooperation. Notwithstanding, ours is not another call for solidarity (Van Der Graaf, Browne and Baidjoe 2022) in terms of policymaking; on the contrary, it is a manifesto for consistency and sustainable holistic policy making that brings together needed stakeholders to move forward in the desired direction.
