Abstract
Background:
Infective endocarditis is a microbial infection of the endothelial surface of heart and great vessels frequently involving Mitral and Aortic valve. Pulmonary valve endocarditis leading to Severe Pulmonary Regurgitation is extremely rare and is often diagnosed late due to non specificity of symptoms. We intend to report such a case to bring the attention of everyone the long term consequences of Pulmonary Regurgitation. Here we also describe method of replacing Pulmonary valve with Bioprosthetic Aortic valve.
Methods:
We describe the technique of replacement of pulmonary valve with Bioprosthetic Aortic valve in case of severe Pulmonary regurgitation.
Result:
The patient successfully underwent replacement of both Pulmonary and Tricuspid valve with bioprosthetic valve and was discharged from the hospital on seventh post operative day. During successive follow up there was considerable improvement in exercise tolerance and she being presently in NYHA class II.
Conclusion:
Pulmonary valve replacement with bioprosthetic tissue valve should be considered for patients with Pulmonary regurgitation and volume overload of Right ventricle. Emphasis should be on Right ventricle dilatation before setting of RV dysfunction rather then severity of Pulmonary regurgitation alone.
Keywords
Introduction
Infective endocarditis (IE) involving pulmonary valve is extremely rare affecting less than 1.5%–2% of patients suffering from IE. The majority of IE on the right side of heart involves tricuspid valve, usually common in IV drug abusers. We report a case of pulmonary valve regurgitation and secondary tricuspid regurgitation (TR) postsurgical closure of patent ductus arteriosus (PDA) in childhood now presenting with symptoms of right heart failure in the adulthood.
Case report
A 37-year-old female patient presented with dyspnoea on exertion and palpitation and occasional oedema of lower limbs. She was in New York Heart Association (NYHA) class III. She underwent surgical closure of PDA at 7 years of age. The history suggestive of previous IE could not be elicited as the patient herself was a child at the time of PDA closure.
Routine haematological and biochemical investigations were within normal limit. Her electrocardiogram (ECG) shows patient to be in sinus rhythm.
Chest X-ray (Figure 1) revealed cardiomegaly with gross enlargement of pulmonary artery, right atrium (RA) and right ventricle (RV).
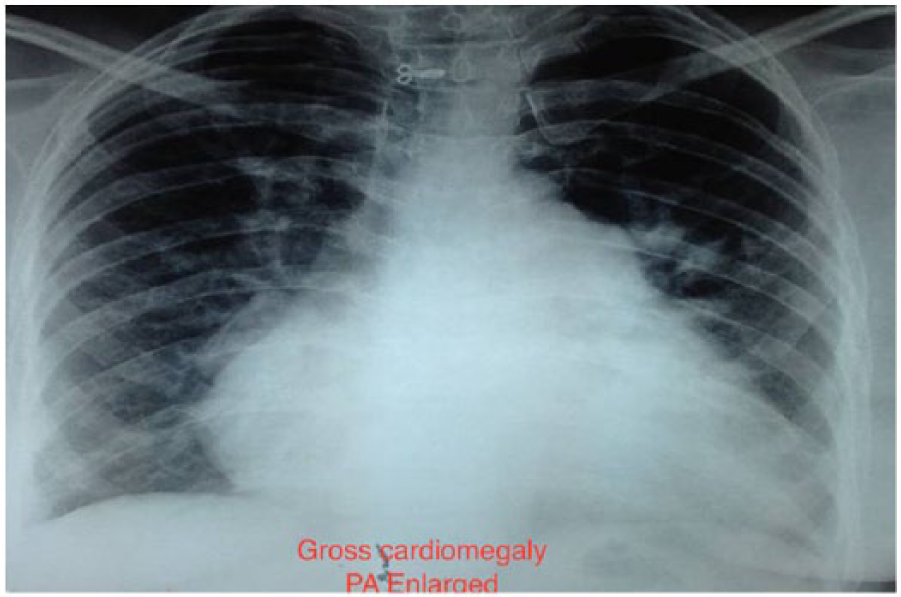
Chest X ray showing Gross cardiomegaly with enlarged RA/RV/PA.
Two-dimensional echocardiography with colour Doppler revealed severely dilated RA/RV and tricuspid valve annulus (Figure 2). Pulmonary valve leaflet shows an old healed vegetation (Figure 3). Pulmonary valve regurgitation with severe tricuspid valve regurgitation is shown in Figure 4. RA and RV were dilated with paradoxical motion of interventricular septum (IVS) with biventricular dysfunction. The main pulmonary artery (MPA) was dilated 3.3 cm, right pulmonary artery (RPA) 1.9 cm and left pulmonary artery (LPA) 1.4 cm, with pulmonary annular dilatation 3.4 cm and tricuspid annulus size 6 cm. There was no mitral regurgitation (MR) or aortic regurgitation (AR). These findings were further confirmed by Transoesophageal Echocardiography (TEE) which revealed old healed vegetations on pulmonary valve with pulmonary annulus size of 2.8 cm, tricuspid valve annulus size of 6 cm with severe TR (right ventricular systolic pressure (RVSP) 51 mmHg) and severe pulmonary regurgitation (PR), markedly dilated RA/RV cavity with features of RV volume overload. PR was so severe that it extends well beyond the right ventricular outflow tract (RVOT) (Figure 5). Her coronary angiogram revealed normal status of coronary arteries.
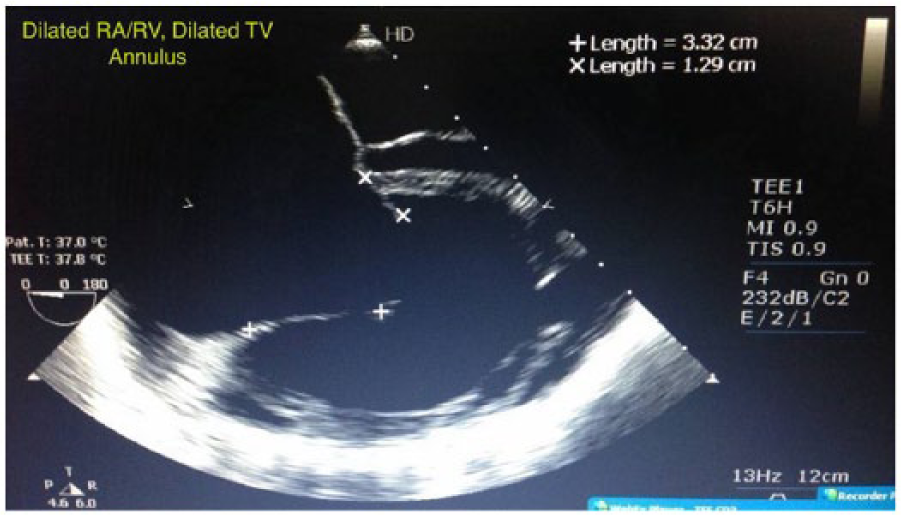
Echocardiogram showing dilated Tricuspid valve annulus.
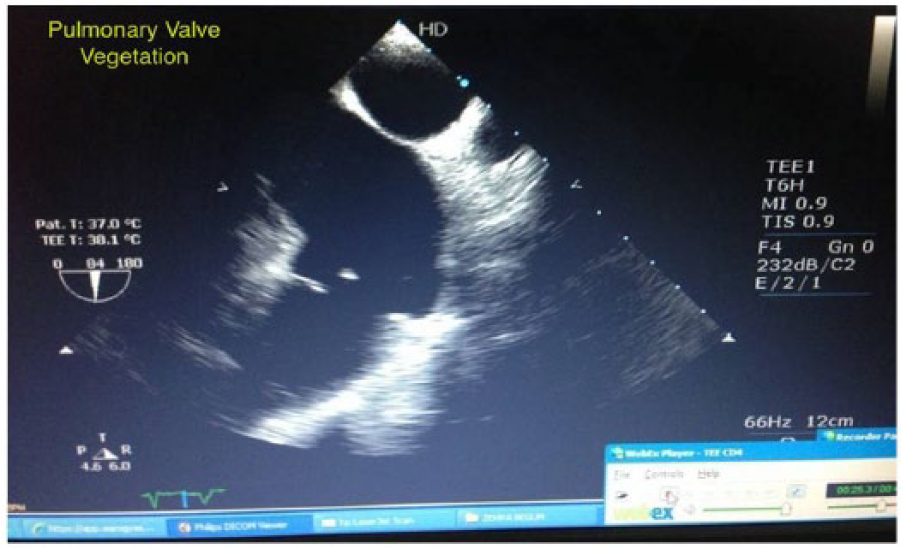
Echocardiogram showing Old healed vegetation on pulmonary valve.
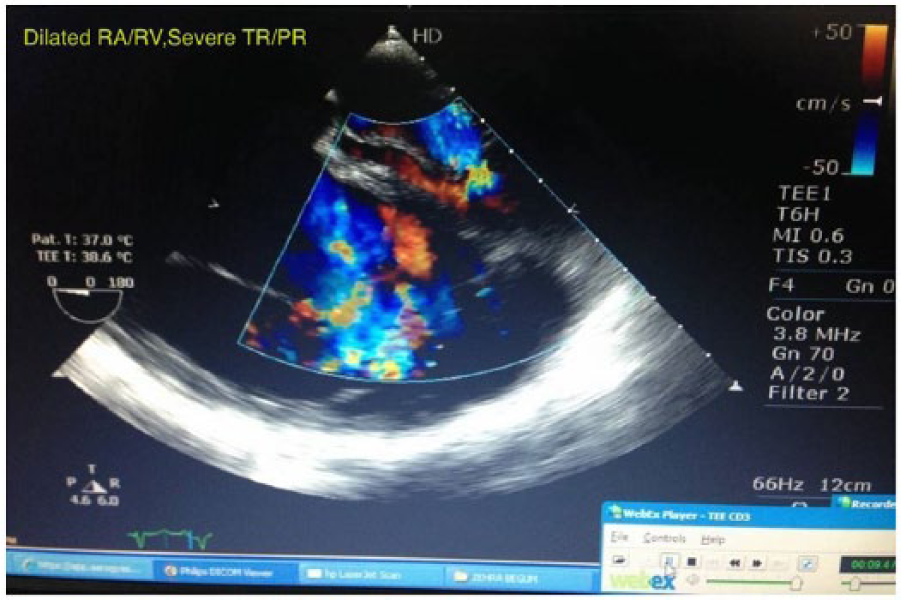
Echocardiogram showing Tricuspid and Pulmonary regurgitation.
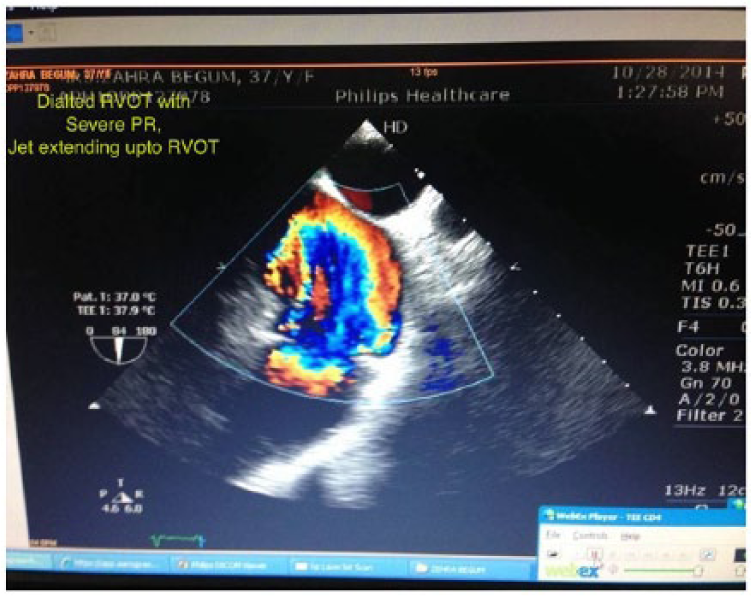
Echocardiogram showing severe Pulmonary regurgitation with jet extending upto RVOT.
She was scheduled for double valve replacement (Pulmonary valve replacement and Tricuspid valve replacement (PVR + TVR)) after completion of preoperative assessment. Narcotic-based induction of anaesthesia was done using graded stimulation. Chest was opened via median sternotomy. After systemic heparinization, aortic and bicaval cannulation and tapes around cava, cardiopulmonary bypass (CPB) was established. Aorta was cross clamped after achieving a core temperature of 30°C; antegrade cold blood cardioplegia administration was done to achieve cardiac stand still. Perfusion pressure during CPB was kept around 60 mmHg. RVOT and MPA junction were opened as shown in Figure 6.
Right ventricular outflow tract and main pulmonary artery opened up.
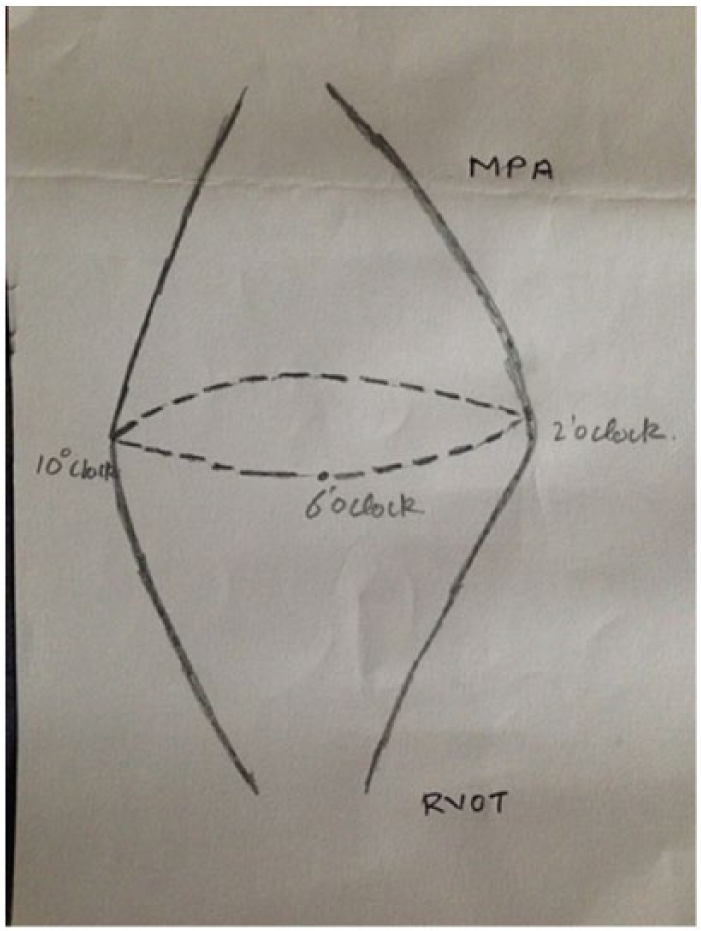
There was a healed vegetation on pulmonary valve leaflet. Pulmonary valve sizing was done using bioprosthetic valve sizer and was decided to implant 23-mm Biocor aortic valve. The principle used is that the prosthetic valve will seat into posterior 2/3 of native pulmonary valve annulus with a slight tilt anteriorly (pt head side) as the pulmonary artery dips down after originating and so if we don’t tilt the valve anteriorly then the struts of prosthetic valve will abut on the pulmonary artery (PA) wall and may injure or perforate it. Using Prolene 3-0 suture (suture A), the prosthetic valve was sutured starting from 6 o’clock position (inferior portion of annulus) and then extending on either side to 2 o’clock position (away from surgeon side) and 10 o’clock position (towards surgeon side) (Figures 7 and 8).
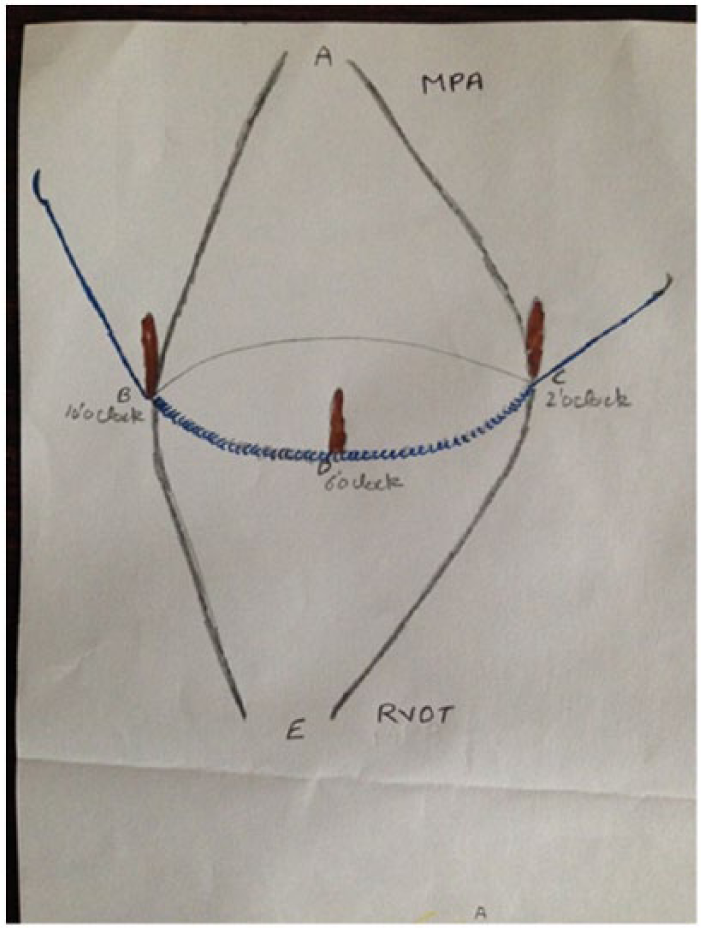
Prosthetic valve sutured to native annulus starting from 6’0 and extending on either side to 10’0, and 2’0 clock position.
Valve sutured onto Posterior 2/3 of annulus.
As we will be approaching 10 o’clock and 2 o’clock positions, our suture line will deviate gradually onto the MPA wall, this will keep the valve slightly anteriorly directed.
Thereafter, gluteraldehyde-treated pericardial patch of appropriate size was used to augment the RVOT junction.
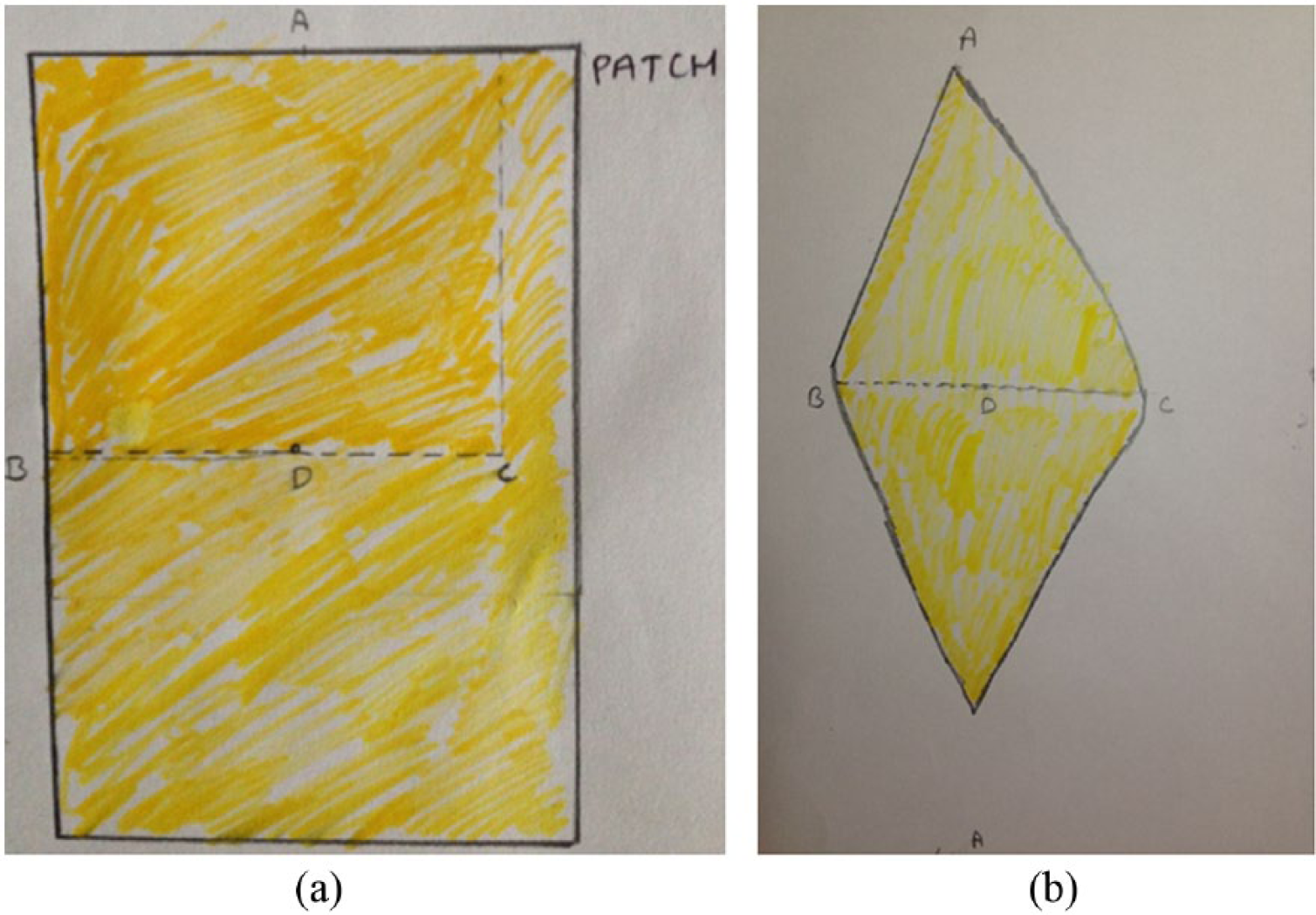
Sizing of the patch is very important to prevent the paravalvular leak. The B-C on patch should correspond to the 10 o’clock to 2 o’clock position and A-D on the patch should correspond to the vertical distance between Point A and 6 o’clock position on the previous Figure 7 (Figure 9(a)). Patch was cut out in diamond shape (Figure 9(b)) and was sutured (suture B) beginning from the point A and extending to point B (10 o’clock) and point C (2 o’clock) on either side (Figure 10). Also tie suture A and suture B at point B and point C to prevent any paravalvular leak.
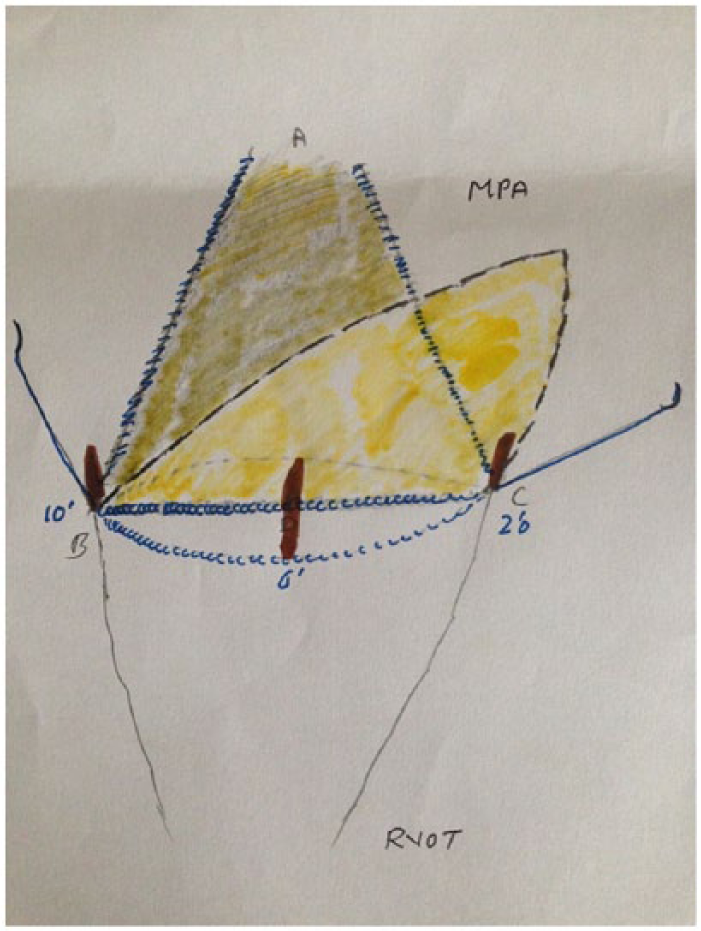
(a) Gluteraldehyde Treated pericardial patch is sized (b) Cut in a spindle shape (see text for description).
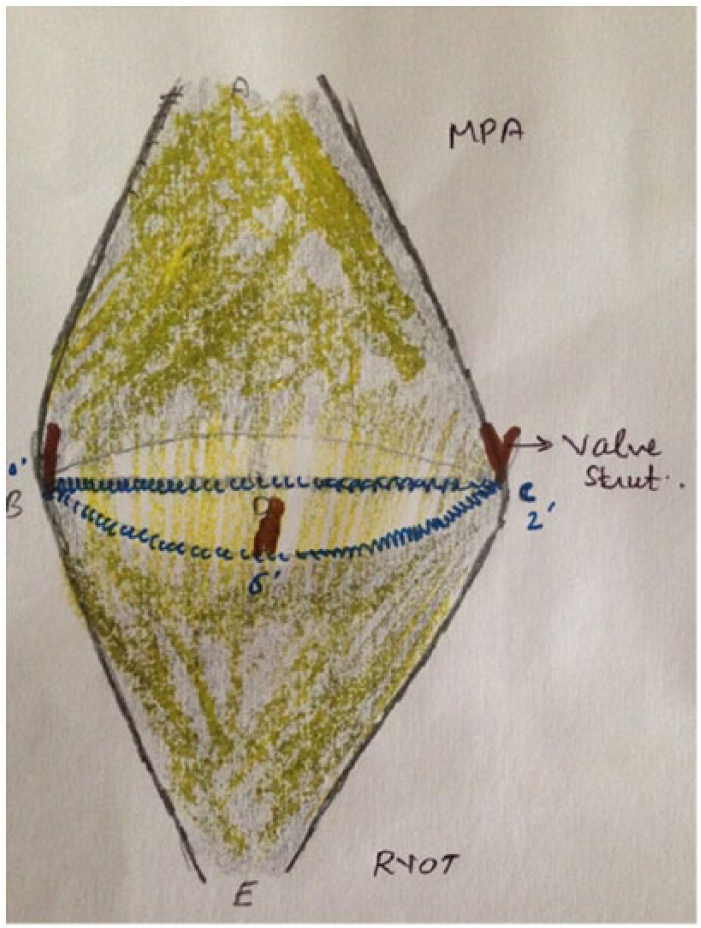
Patch sutured onto Main pulmonary artery and sewing ring of prosthetic vale (see text).
Between B and C, the patch was folded onto itself as shown in Figure 10 and sutured with prosthetic valve with previous Prolene 3-0 suture (suture A) (Figure 11). Thereafter, the folded portion of the patch was unfolded and sutured onto the RVOT, using suture B, thus augmenting the RVOT (Figures 12 and 13).
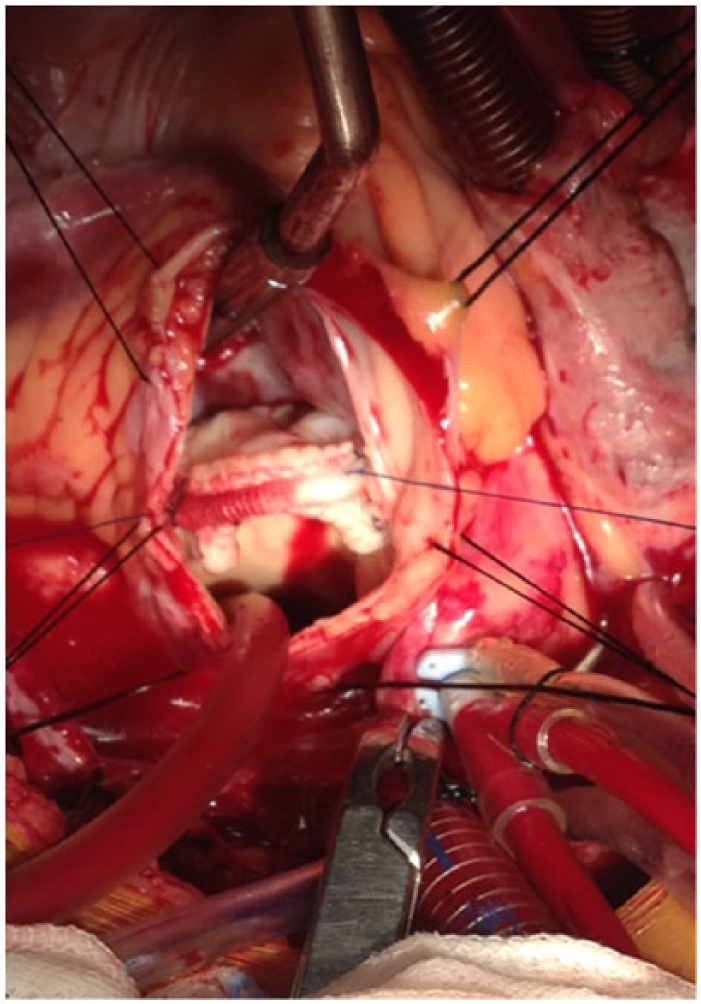
Valve stitched onto the posterior 2/3 of annulus.
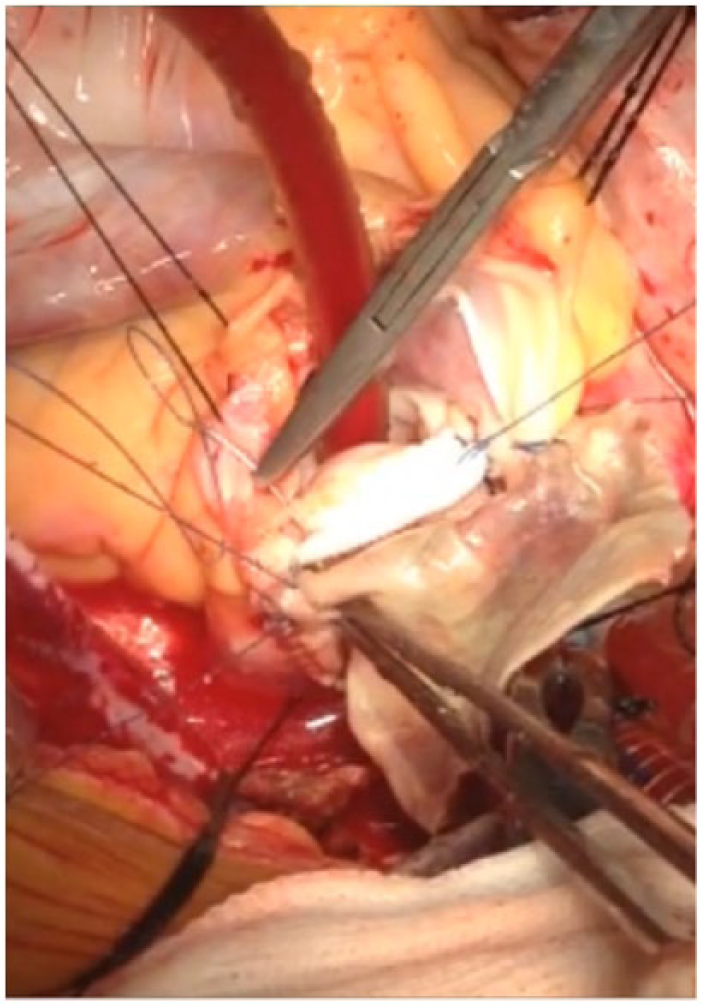
Pericardial patch augmentation of RVOT.
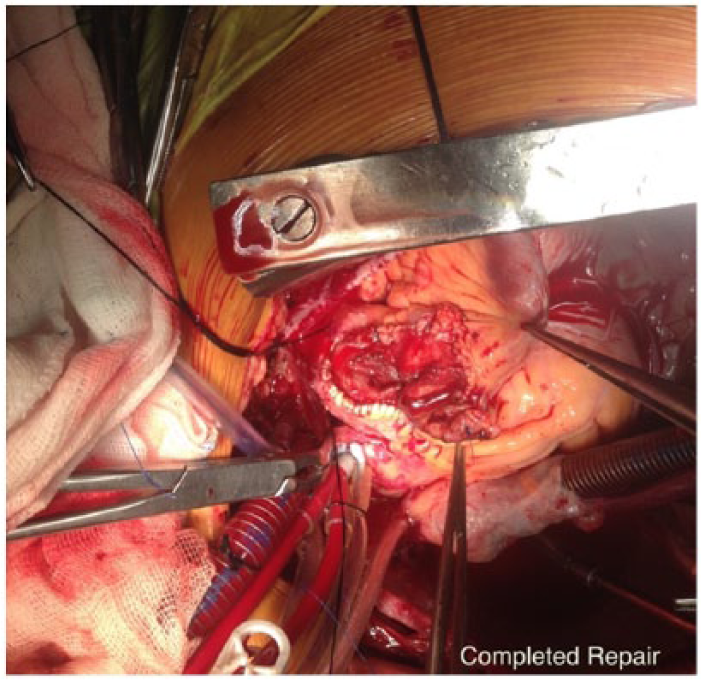
Completed Repair.
Thereafter, RA was opened posterior to anterior atrioventricular groove; tricuspid valve shows gross annular dilatation, thus it was replaced with 27-mm Biocor Mitral valve taking care that suture on the septal leaflet side should pass through base of septal leaflet and not from the tricuspid valve (TV) annulus to prevent the bundle from injury. RA was closed following replacement.
Patient was weaned from CPB after complete rewarming, with ionotropic support of adrenaline 0.04 µg/kg/min, levosimendan 0.1 µg/kg/min and vasopressin at 0.0003 U/kg/min. Total bypass time was 185 min with aortic cross clamp time of 120 min.
On table echocardiography revealed normal prosthetic valve function. Thereafter, reversal of heparin with protamine was done, hemostasis achieved and chest closed in layers.
In the intensive care unit (ICU), haemodynamics was monitored and cardiac output optimized with ionotropes and vasopressors. Broad-spectrum antibiotics were administered for 3 days. Patient was extubated on first postoperative day (POD). After 24 h, levosimendan infusion was tapered off and central venous saturation was monitored daily and was found to be above 60%, reflecting adequate cardiac output.
From first day itself patient was given Heparin 5000 IU subcutaneously and also acitrom was started to maintain international normalized ratio (INR) above 2.5.
Patient was discharged on seventh POD with acitrom and ecosprin. Echocardiogram prior to discharge revealed normal prosthetic valve function and no paravalvular leak.
Discussion
Pulmonary valve disease is very uncommon. Majority of pulmonary valve lesions occur due to congenital heart anomalies, but the pulmonary valve may rarely be damaged as a result of infection (IE) or as a result of disease (e.g. carcinoid disease, Marfan’s syndrome or pulmonary hypertension). The case we are reporting had a surgical closure of PDA in the childhood, the details of the surgery and the history of her past illness could not be elicited as the patient was a child herself.
Possibility of a congenital PR and secondary tricuspid valve involvement cannot be ruled out though isolated stenotic lesions are more common than regurgitant lesions. Here, on table finding of a healed vegetation on the valve leaflet with a near complete destruction of valve is more in favour of the IE in the past leading to severe PR, right ventricular dilatation and TR.
Isolated pulmonary valve endocarditis is rare. 1 The incidence of right-sided IE range from 5% to 10% in different series2,3 and tricuspid valve being most commonly involved. The low incidence of infection on the pulmonic valve as compared with other cardiac valves may be due to low pressure gradients in right heart, low prevalence of congenital malformations, lower oxygen content of venous blood and the difference in covering and vascularization of right heart endothelium. 3 However, most cases of pulmonary valve endocarditis have been reported in children with congenital heart disease as in our case.4,5,6
Isolated PR is well tolerated for prolonged period. 6 Review of literature reveals in a meta-analysis that 29% of patients had developed symptoms within 40 years. 7 Some of these patients who had regurgitation as the dominant lesion eventually needed pulmonary valve replacement. 8 Although tolerated well for prolonged periods, chronic PR has a detrimental effect on right ventricular function and exercise capacity leading to right ventricular dilatation, right ventricular dysfunction, ventricular arrhythmias and sudden cardiac death. 9
The reason chronic PR is well tolerated for prolonged period is because of certain features of pulmonary circulation. Forward pulmonary blood flow can be maintained not only by right ventricular contraction but also by work of left heart via the systemic venous return and by right atrial contraction. This mechanism helps for part of each cycle in patients with severe PR and restrictive right ventricle in whom late diastolic forward flow and flow in the pulmonary trunk due to atrial systole is seen and right ventricle temporarily acts as a conduit (during late diastole). The second feature which helps the patient in tolerating PR is the low resistance of pulmonary vascular bed and its location being close to heart. Thereafter, each RV systolic blood moves forward into pulmonary microvessels whose low resistance acts as a watershed into the pulmonary veins which are in turn maintained at low pressure by action of left heart. Blood which flows forward through alveolar capillaries in systole do not pass back again into heart during diastole. 10
The adaptive right ventricular response to volume overload resulting from PR depends on degree and duration of the regurgitant flow 11 and the properties of the right ventricle and of the pulmonary arteries. PR of moderate severity produce RV volume overload, 4 RV dilatation and deterioration of myocardial function. PR which is seen in postoperative Tetralogy of Fallot repair patients is well tolerated in infancy than in adulthood. Adult patients should be considered for pulmonary valve implantation at the time of repair if native pulmonary valve function cannot be preserved. This is based on the speculation that right ventricle is relatively non-compliant in adulthood. Surveillance should be directed at monitoring right ventricle dilatation and its sequel and not only on PR. Clinically right ventricular dilatation leads to decreased exercise tolerance and other signs and symptoms of heart failure, so evidence of right ventricular dilatation should be monitored by chest X-ray, echocardiography or magnetic resonance imaging (MRI). Thus, evidence of progressive right ventricular dilatation with symptoms of heart failure or documented arrhythmias is a clear indication for pulmonary valve replacement as documented in a review of recent surgical pulmonary valve replacement by Davlouros et al. 12
Pulmonary valve replacement may lead to a reduction in right ventricular dilatation, improved exercise tolerance and decreased arrhythmias. There is difficulty in comparing surgical series as patients may undergo surgery at different stages of disease and there may be variations in underlying anatomy and some patients may have additional lesions that need to be addressed at the time of pulmonary valve replacements as in our patient having severe TR. This is secondary TR due to dilatation of tricuspid annulus which alters the geometry of a tricuspid annulus from a saddle shape to a more planar and circular shape.13,14
The presence of severe PR leading to right ventricular dilatation and tricuspid annular dilatation which may further cause papillary muscle displacement, tethering of tricuspid valve leaflet and thus causing eventually TR. 15 Fukuda et al. 16 showed that the severity of residual TR was associated with patient age, tethering distance and severity of preoperative TR and suggested that preoperative TV tethering is an important criteria for selecting valve replacement than repair for correction of severe functional TR. In the cases where annular dilatation is severe, an isolated annuloplasty is often unable to restore a durable competence of the tricuspid valve and may need additional procedures like augmentation of anterior leaflet with pericardial patch. In order to decrease the incidence of early failure and to reduce the recurrence of regurgitation, decision needs to be taken between tricuspid repair and replacement according to the stage of the disease. The presence of TR after surgery is a cause of concern as residual TR is associated with decreased late survival and increased morbidity. 17 It was shown by previous studies that replacement was more effective than repair in subsiding TR, though replacement may have increased late mortality rates. 18 As seen in this patient, repair of primary valve was not possible; given the dilatation of annulus, a valve replacement was done. The rate of thromboembolism is high with mechanical valves so a bioprosthetic valve is preferred in tricuspid and pulmonary position as was done in our patient, though no survival advantage over a mechanical prosthesis has been proven.19,20
Conclusion
In most of the patients with PR, the volume overload on the right ventricle results in right ventricular dilatation. Over a prolonged period, there will be TR. Therefore, emphasis should be laid on monitoring right ventricular dilatation and its sequelae rather than PR only. If there is objective evidence of progressive right ventricular dilatation with symptoms of heart failure or documented arrhythmias, then we should consider the patient for pulmonary valve replacement.
The dilemma again associated in such patients who have accompanying severe TR necessitating tricuspid valve surgery is first to define the indication for surgery, secondary to choose between tricuspid valve repair or replacement and third to assess the effectiveness of various types of valve repair or the type of prosthesis to be used in valve replacement. If timely intervention or tricuspid valve is not done, then the progressive right ventricular dilatation can lead to irreversible deterioration of right ventricular function which will affect the postoperative outcome of the patient negatively, yet the management of functional TR remains a debatable issue.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
