Abstract
Background:
We report three cases undergoing reoperation with previous aortic homograft implantation due to structural valve deterioration.
Case:
For two cases with severe calcified aortic wall of the homograft and dehiscence of the aortic root, aortic root replacement was indicated. For the last case with less calcified aortic wall, aortic valve replacement was indicated via the aortotomy on the homograft, which required patch plasty for the defect of the stiff aortic wall.
Conclusion:
The distribution and extent of calcification of the aortic homograft would be the most important factor to determine operative procedures, contributing to successful outcomes.
Introduction
An aortic homograft is considered as an ideal substitute due to various reasons including excellent hemodynamic performance, the absence of anti-coagulation and low infection rate. 1 However, reoperation due to structural deterioration of the homograft is a great technical challenge. 2 Thus, we retrospectively reviewed patients undergoing aortic homograft implantation to elucidate surgical strategy.
Case 1
A 49-year-old man had undergone aortic valve replacement (AVR) with a 23-mm Carbomedics mechanical valve due to aortitis. After 1 year, aortic homograft implantation was performed due to prosthetic valve endocarditis (PVE). After 14 years, structural valve deterioration (SVD) with severe aortic stenosis was demonstrated by echocardiography. Additionally, computed tomography showed that the sternum involved severe calcified homograft, requiring total exclusion (Figure 1). Then, redo aortic root replacement was scheduled.
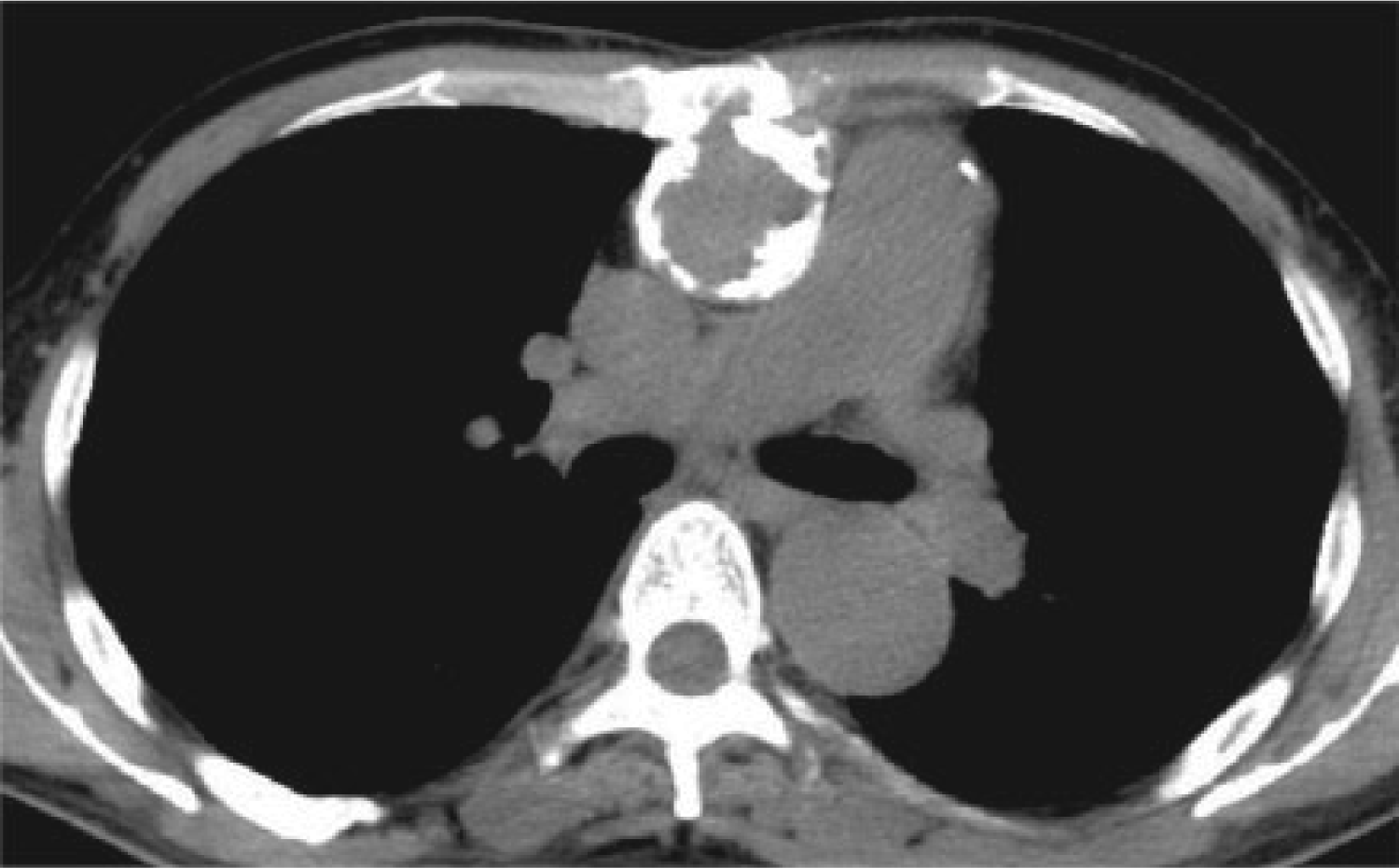
The aortic homograft with severe calcification was involved in the sternum, which required systemic hypothermia to obtain circulatory arrest during sternotomy.
Before sternotomy, cardiopulmonary bypass was installed with femoral cannulation to obtain systemic hypothermia (25°C). Although eventual left ventricular distention due to ventricular fibrillation is one of the serious concerns for systemic hypothermia, venting form the left atrium through the thoracotomy was not performed to prevent subsequent hemorrhage. Under hypothermic cardiac arrest, the chest was re-entered via median sternotomy, and the calcified homograft was sharply and bluntly dissected to exclude from the sternum. After removal of the homograft, native ascending aorta was cross-clamped to discontinue circulatory arrest. The aortic root was replaced with a composite graft created by a 21-mm Carbomedics mechanical valve and a 24-mm J graft. Low output syndrome did not occur after withdrawn from cardiopulmonary bypass. Postoperative course was uneventful without neurological complication.
Case 2
A 55-year-old man had undergone AVR with a 25-mm St Jude Medical mechanical valve due to aortic regurgitation. After 1 year, aortic homograft implantation was performed due to active infective endocarditis (IE). After 6 years, echocardiography demonstrated severe aortic regurgitation, severe mitral regurgitation and a dilated left ventricle. Computed tomography showed pseudoaneurysm arising from the distal anastomosis of the homograft penetrated into the left atrium (Figure 2(a) and (b)). Thus, redo aortic root replacement plus mitral valve surgery was indicated.
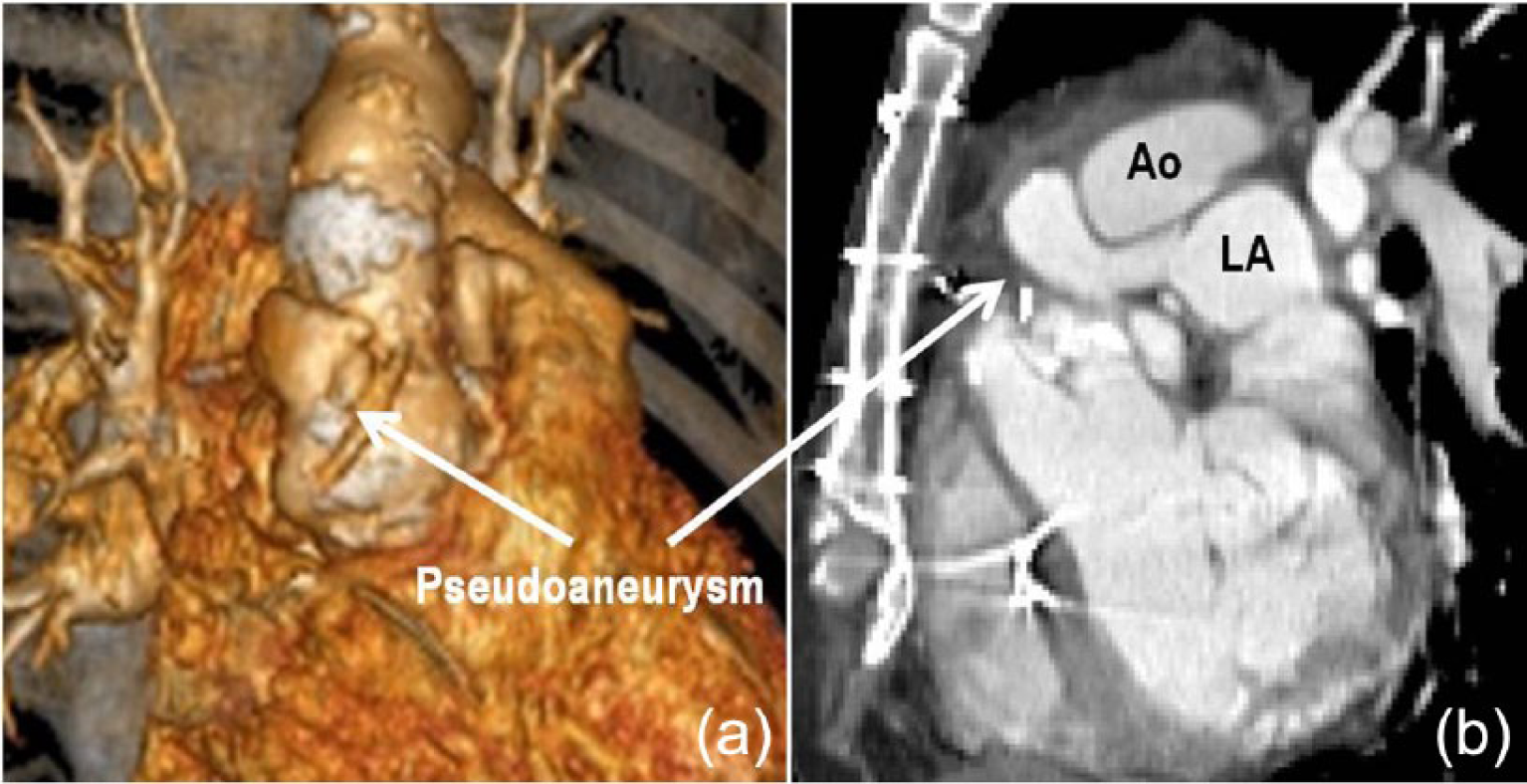
Pseudoaneurysm (arrow) arising from the proximal anastomosis for the homograft and the ascending aorta (Ao) penetrated into the left atrium (LA).
Under cardioplegic cardiac arrest, mild calcified homograft was opened. The dehiscence of the homograft resulted in aortic regurgitation and pseudoaneurysm was ruptured into the left atrium, which might be caused by previous infection. The resultant shunt flow induced functional mitral regurgitation. Then, the defect of the left atrium was directly closed and mitral ring annuloplasty was performed. The aortic root was replaced with a composite graft created by a 27-mm Carbomedics mechanical valve and a 30-mm Gelweave valsalva graft. Postoperative course was uneventful without infection.
Case 3
A 58-year-old man had a history of three times cardiac surgery, including AVR due to IE, re-AVR due to PVE and aortic homograft implantation due to PVE. After 6 years, SVD with severe aortic stenosis and mild regurgitation was demonstrated by echocardiography. Computed tomography showed diffuse calcification of the ascending aorta, which would prohibit the standard aortotomy for AVR (Figure 3). Then, redo AVR via the aortotomy on the less calcified aortic wall of the homograft was planed.
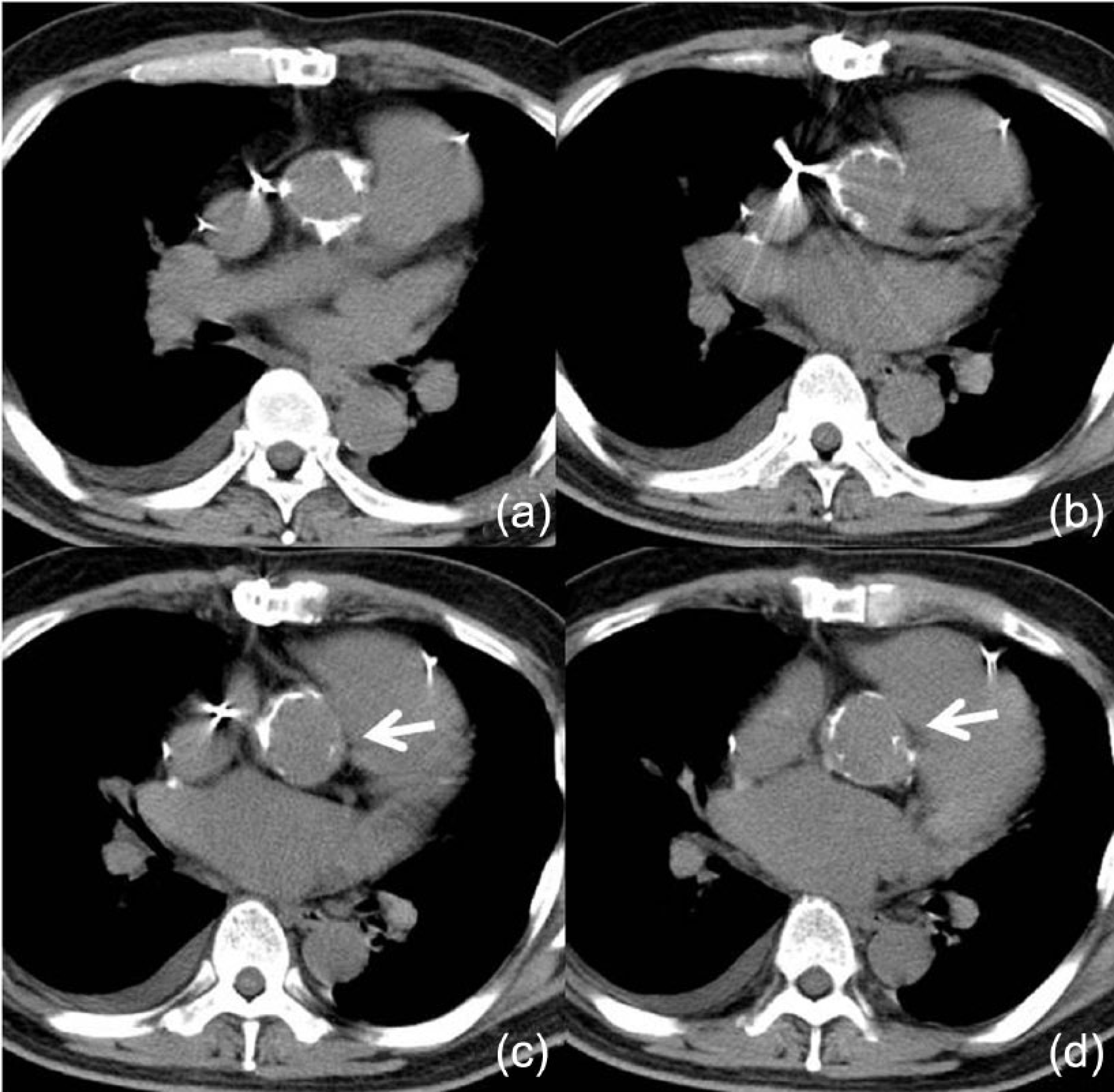
The ascending aorta of the homograft included diffuse calcification. The aortotomy was placed on the left-posterior side of the aortic homograft, which involved less calcification compared with other lesion (arrow).
Under cardioplegic cardiac arrest, the left-to-posterior wall of the homograft with less calcification was opened to complete AVR. After placement of the prosthetic valve, the defect of the stiff aortic wall was closed with a Hemashield patch. Postoperative course was uneventful without infection.
Discussion
We surgically treated patients undergoing reoperation after aortic homograft implantation due to SVD. The main degenerative alteration of a homograft was calcification, which affected the ascending aorta other than aortic leaflets. Although the distribution and extent of calcification of the ascending aorta would be the most important factor to determine operative procedures, structural alterations caused by previous infection should be also considered for these patients. Thus, reoperations with calcified aortic homograft would become complex and atypical due to various structural deteriorations, which would increase technical difficulties.
An aortic homograft is one of the useful substitutes for patients with acute endocarditis, and satisfactory results had been already demonstrated. 3 Although the durability of an aortic homograft was acceptable, reoperation would be necessary during follow-up of 20 years. 4 Kumar et al. 5 demonstrated that operative risk was not increased for patients with previous aortic homograft replacement at the time of redo AVR. However, redo aortic root replacement would be also indicated due to major homograft calcification or endocarditis. Thus, reoperation after aortic homograft implantation presented a relatively high perioperative morbidity because redo aortic root replacement was indicated in about 50% of such cases. 2 However, operative risk would be reduced by new innovative technologies such as trans-catheter approach and sutureless aortic valve prosthesis for these patients.6,7
Conclusion
We reported successful reoperations undergoing previous aortic homograft implantation due to SVD. The distribution and extent of calcification of the aortic homograft would be very important to determine operative procedures.
Footnotes
Acknowledgements
This article was presented at the 20th Annual Meeting of the Asian Society for Cardiovascular and Thoracic Surgery, Bali, Indonesia, 2012.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
