Abstract
Background:
The objectives of this study were to investigate the changes in routine laboratory tests and rotational thromboelastometry variables after cardiopulmonary bypass and to investigate the association of these variables with postoperative blood loss.
Methods:
Blood was obtained at baseline, at admission to the intensive care unit, and at the first postoperative day. Rotational thromboelastometry tests and measurements of haemoglobin, haematocrit, platelet count, and plasma fibrinogen level (Clauss method) were performed. Excessive bleeding was defined as >1000 mL of chest tube drainage after surgery within 24 h.
Results:
After cardiopulmonary bypass, haemotocrit was decreased by 21%. Platelet count and fibrinogen level decreased more (35% and 42%, respectively). The largest change was found for clot formation time of INTEM (intrinsically activated coagulation) (+73%). Platelet count was not reflected by maximal clot firmness of PLATEM (platelet component specific coagulation). The decreases in FIBTEM (fibrinogen specific coagulation) maximum clot firmness and fibrinogen at the intensive care unit admission were comparable. Test results outside the reference range were associated with higher median blood loss when compared to values within the reference range, with most significant association for fibrinogen (p < 0.001) and FIBTEM maximal clot firmness (p = 0.016). At baseline, the ability of rotational thromboelastometry parameters to predict postoperative blood loss >1000 mL is higher than the predictive value of fibrinogen (area under the curve = 0.642; 95% confidence interval: 0.487–0.796), but only areas under the curve of APTEM (extrinsically activated coagulation with exclusion of fibrinolysis component) were significantly higher (p⩽0.05). At intensive care unit admission, the highest area under the curve was 0.689 (95% confidence interval: 0.549–0.828) for FIBTEM maximum clot firmness.
Conclusion:
Rotational thromboelastometry could be a useful point-of-care method to monitor haemostasis because it is a faster and more informative method, and it has a slightly higher predictive ability for blood loss compared to fibrinogen.
Introduction
Cardiac surgery is associated with intra- and postoperative bleeding1,2 due to the haemostatic disturbances related to cardiopulmonary bypass (CPB). Platelet dysfunction is often considered to be the most important acquired defect in haemostasis in the early postoperative period. 3 Besides, excessive postoperative bleeding has been attributed to increased fibrinolysis. 4 In addition, the perioperative plasma fibrinogen concentration is associated with postoperative bleeding and transfusion requirements after CPB. Early characterisation of the haemostatic defects responsible for bleeding is crucial for optimising management of the patient. However, routine laboratory parameters provide inadequate guidance for timely and goal-directed haemostatic intervention. 4 Whole blood viscoelastic tests like thromboelastography (TEG) and rotational thromboelastometry (ROTEM) are more suitable for timely decision-making (<15–20 min) in haemostatic interventions than tests performed in plasma. These whole blood tests, providing information on the contribution of both fibrinogen and platelets to clot formation, produce clinically relevant correlations with platelet count and plasma fibrinogen levels5,6 and exhibit a high sensitivity and specificity for detecting subnormal fibrinogen levels. 5 In addition, it is postulated that whole blood tests like ROTEM better reflect disturbances occurring during CPB 7 and may therefore be helpful as a guide for transfusion therapy.8–10 However, the precise relationship between ROTEM values and postoperative bleeding remains unclear.10–14
The aim of this study was to investigate the changes in routine laboratory tests and ROTEM variables, with subsequent association with blood loss after cardiac surgery with CPB.
Methods
Patients
After approval by the local ethical committee (reference number: Nl.34179.068.10/MEC 10-2-095) and written informed consent, 100 patients undergoing elective cardiac surgery with CPB were included in this prospective observational study. Written informed consents are stored in a folder. The patients were scheduled for coronary artery bypass grafting (CABG), aortic valve replacement (AVR) or a combination of both (CABG/AVR). Patients with preoperative coagulation disorders; who used oral anticoagulants, clopidogrel, or thrombolytics within 5 days before surgery; renal insufficiency; hepatic disorders or active sepsis/endocarditis; and patients with active malignancy were excluded from study participation.
Clinical management
General anaesthesia was induced using weight-related dosing of sufentanil and etomidate, and muscle relaxation was achieved with pancuronium bromide. General anaesthesia was maintained during surgery using propofol. An initial dose of 300 IU/kg of body weight of heparin (Heparin Leo, Leo Pharmaceutical Products BV, Weesp, The Netherlands) was injected into a central venous line before the initiation of CPB. After this loading dose, the activated clotting time (ACT) was measured, and if the value was >400 s, CPB was initiated.If necessary, additional heparin was added to achieve an ACT of at least 400 s. At the end of CPB, heparin was reversed by protamine chloride (Valeant Pharmaceuticals, Eschborn, Germany) at a 1:1 ratio of the loading dose, regardless of the total heparin dosage.
The CPB system consisted of a standard closed bypass circuit with a hollow-fibre membrane oxygenator (Capiox SX18R, Terumo, Tokyo, Japan), a cardiotomy reservoir (Capiox, CXCRXA, Terumo), and an arterial line filter (Pall, LeukoGuard LCG, Pall, Portsmouth, UK). All circuit components were poly-2-methoxyethylacrylate coated (Terumo).
The standard priming of the CPB circuit included 1300 mL of 4% Gelofusine (B. Braun AG, Melsungen, Germany), 200 mL of 20% mannitol, 100 mL of 20% human albumin, and 50 mL of 8.4% NaHCO3. The total volume of the CPB priming amounted to 1650 mL clear fluid, containing 6500 IU/L heparin (Heparin Leo, Leo Pharmaceutical Products BV). In most of the cases, a technique to reduce the large priming volume by 200–500 mL was used, the so-called retrograde autologous priming (RAP), which resulted in less haemodilution.
Myocardial preservation was provided by infusing St Thomas’ No. 1 solution at 4°C via the aortic root. Target flow rates of ⩾2.4 L/min/m2 were maintained throughout the CPB period. Normothermic (⩾36°C) pulsatile perfusion was used during the period of cross-clamping. Mean arterial blood pressure was maintained above 70 mmHg, and if needed, phenylephrine (0.4 mg/mL) was titrated via an infusion pump. Pericardial and pleural blood was drained and washed with a cell saver device. After termination of CPB and decannulation, the residual volume of the heart-lung machine was displaced into the cell saver device (C.A.T.S., Fresenius Kabi, Germany) and transfused to the patient at the end of surgery. As antifibrinolytic therapy, all patients receive 2 g of tranexamic acid pre-CPB and 1 g during CPB. Transfusions of red blood cells, platelets, fresh frozen plasma or fibrinogen were according to standard procedures guided on routine laboratory results or clinical observation of bleeding.
Measurements
Blood samples were taken, after discarding the first 10 mL, at three time points: (T1) directly before the induction of general anaesthesia, (T2) at admission to the intensive care unit (ICU), and (T3) at the first postoperative morning. Blood was collected in 4.0 mL tubes with 7.2 mg spray-dried K2EDTA for analysis of haemoglobin, haematocrit, and platelet count and in 4.5 mL, 3.2% citrate tubes (BD Vacutainer, Becton Dickinson, NJ, USA) for analysis of fibrinogen and ROTEM. Complete blood count has been carried out on a Sysmex XN-9000 analyser (Sysmex Corporation, Kobe, Japan), an automated analyser. Fibrinogen levels were determined using the Clauss coagulation method on a Sysmex CA-7000 analyser (Sysmex Corporation) (Dade Thrombin Reagent, Siemens Healthcare Diagnostics Products, Marburg, Germany).
Thromboelastometric measurements were performed with a four-channel ROTEM device (ROTEM®, Pentapharm, Munich, Germany). ROTEM was performed according to the manufacturer’s instruction at 37°C. The whole blood samples were recalcified with calcium chloride, and coagulation was activated with specific reagents (TEM Innovations GmbH, Munich, Germany) using a semi-automated electronic pipette system. Analysed ROTEM parameters were as follows: EXTEM clotting time (CT), clot formation time (CFT), alpha angle, maximal clot firmness (MCF) (addition of tissue factor); INTEM CT, CFT, alpha angle, MCF (addition of ellagic acid); FIBTEM CT and MCF (addition of tissue factor/cytochalastin D); and APTEM CT, CFT, alpha angle and MCF (addition of tissue factor/aprotinin). As a measure for the contribution of platelets, the ROTEM variable PLATEM was calculated by subtracting FIBTEM from EXTEM (eliminating the contribution of fibrinogen). PLATEM was also used in previous studies.4,6,15 Only MCF was determined for PLATEM.
Blood loss was measured postoperatively and was determined by volume of chest tube drainage after closing the chest until 24 h after surgery. Transfusion requirements were also recorded in the whole perioperative period until 24 h after surgery.
Statistical analysis
A sample of 100 patients undergoing complex surgery was studied to explore the association between coagulation variables and blood loss. No formal sample size analysis was performed.
Continuous variables are described as means with standard deviation (for normally distributed data, tested by Kolmogorov–Smirnov) or as median with interquartile range (for data without normal distribution). Changes in ROTEM and routine laboratory parameters were expressed as percentage from the baseline and were calculated as: (end value − baseline value/baseline value) × 100%. Values before and after surgery were compared with a t-test or the non-parametric Wilcoxon test for paired samples where appropriate.
In order to evaluate the association of ROTEM and routine laboratory parameters with blood loss, patients were categorised into subgroups: (1) with values within the reference range 16 of the Maastricht University Medical Centre (MUMC+) (G1) and (2) with values outside the reference range of the MUMC+, indicating a lower haemostatic potential (G2). Median blood loss between the two subgroups was compared with a t-test for independent samples or with the non-parametric Mann–Whitney U test where appropriate. The ability of ROTEM and routine laboratory parameters before and after surgery to predict excessive blood loss in individual patients was investigated by receiver operating characteristic (ROC) analysis and was expressed as area under the curve (AUC) with 95% confidence intervals (CI). Excess bleeding was defined as >1000 mL within the initial 24 h for two reasons. This volume of blood loss has been previously used as a definition of excessive bleeding after CPB,17,18 and additionally 1000 mL represents the 75th percentile of the patient population in this study. AUCs of ROTEM parameters were compared with the highest AUC of the routine laboratory parameters, to evaluate whether the difference in AUC was statistically significant. Values of p ⩽ 0.05 were considered to indicate statistical significance. All analyses were performed using SPSS version 20 (SPSS Inc., Chicago, IL, USA), excepting comparison of AUCs, which was performed with STATA version 13. All patient-related data are saved in an SPSS database.
Results
A total of 100 patients, scheduled for CABG, AVR, or a combination of CABG/AVR, participated in this study. Three patients were excluded, one because of complicated postoperative course with three times a re-exploration with exceptional blood loss and one patient because of the necessity to use an extracorporeal life support system for cardiac support postoperatively. Due to logistic reasons it was impossible to perform ROTEM measurements at the first postoperative day (T3) for the third patient. This made us decide to exclude this case. Demographics, clinical data, and information about blood loss and transfusion requirements are shown in Table 1.
Demographic and clinical variables, transfusion requirements and blood loss of all patients.
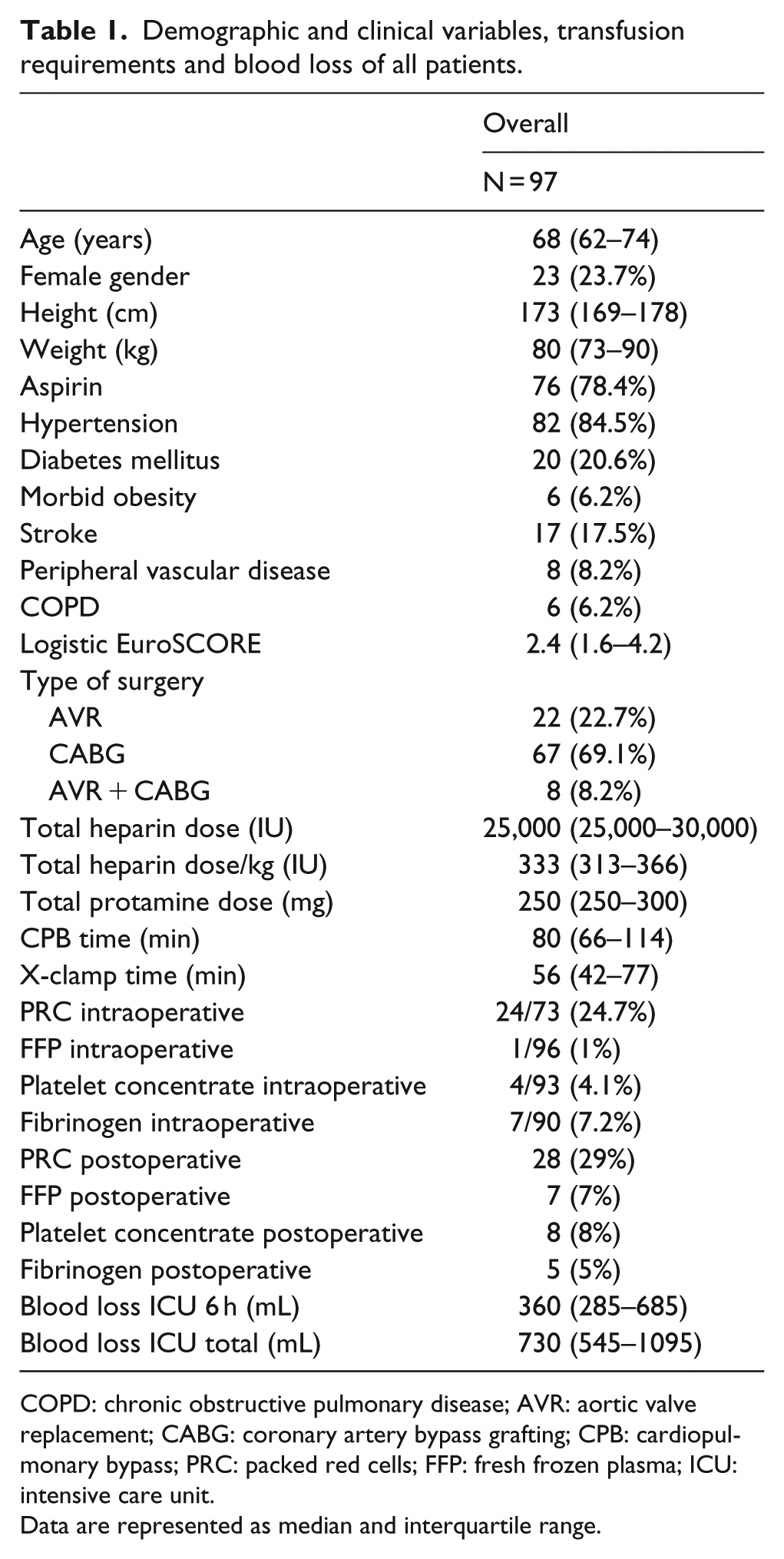
COPD: chronic obstructive pulmonary disease; AVR: aortic valve replacement; CABG: coronary artery bypass grafting; CPB: cardiopulmonary bypass; PRC: packed red cells; FFP: fresh frozen plasma; ICU: intensive care unit.
Data are represented as median and interquartile range.
At the ICU (T2), both routine laboratory tests and all ROTEM variables were changed as a consequence of the CPB procedure. The relative changes from baseline for all parameters were statistically significant (p < 0.001) with the exception of changes for PLATEM MCF (p = 0.922). At the first postoperative day (T3), all variables tended to normalise; however, the values were still significantly different from baseline (T1) (p < 0.001), with the exception of fibrinogen and PLATEM (Table 2) (p = 0.156 and p = 0.137, respectively).
Results of routine coagulation tests and ROTEM parameters at different time points and relative changes at T2 and T3 as percentage, of baseline values, measured in patients with and without haemostatic transfusion requirements.
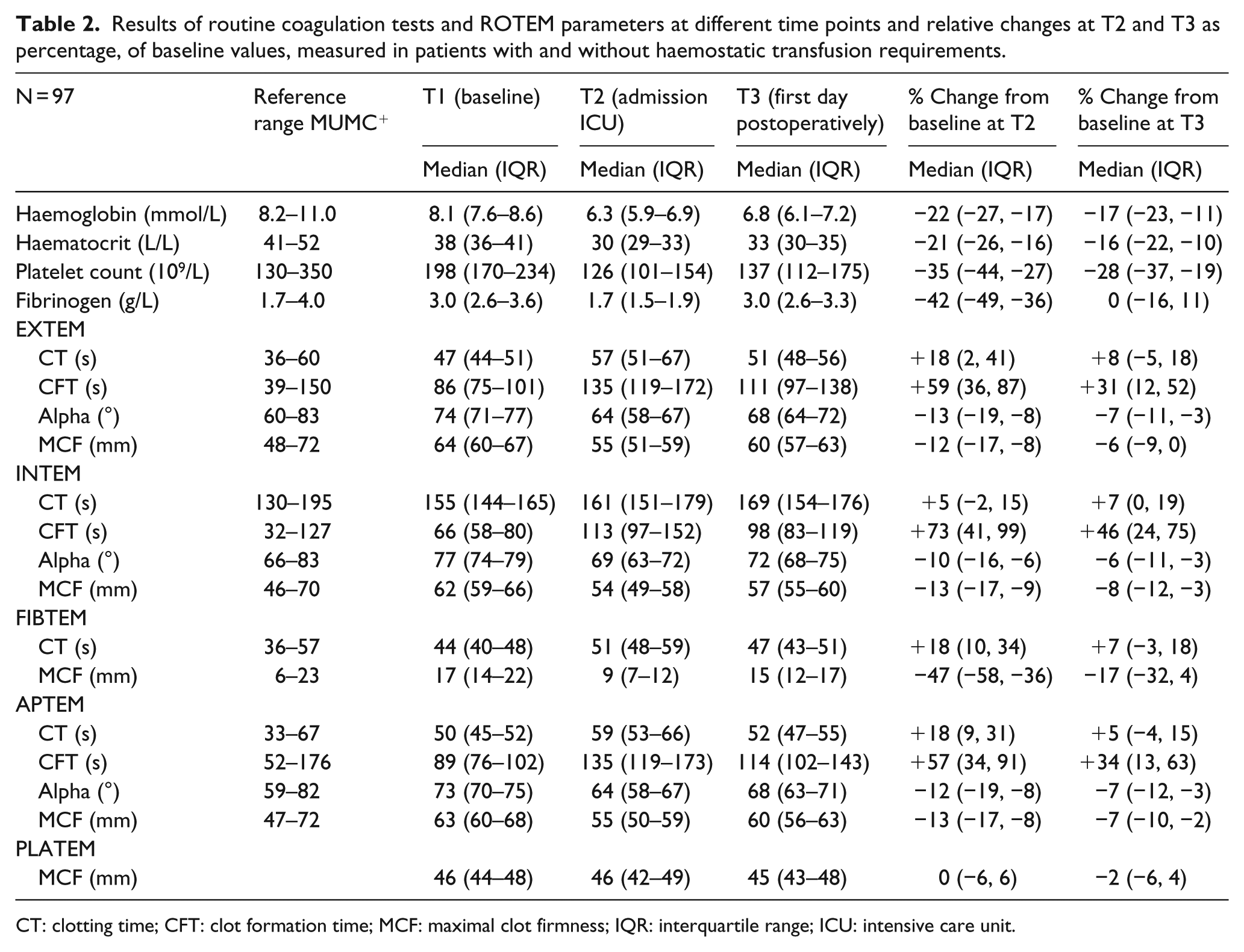
CT: clotting time; CFT: clot formation time; MCF: maximal clot firmness; IQR: interquartile range; ICU: intensive care unit.
Changes in routine laboratory tests
At the ICU admission, haemoglobin and haematocrit decreased by 22% and 21%, respectively, whereas the decreases in platelet count and fibrinogen were higher (35% and 42%, respectively). Fibrinogen was the only variable that returned to baseline at the first postoperative day (Table 2).
Changes in ROTEM variables
Table 2 shows that the most pronounced change compared with baseline was an increase in CFT in all assays (EXTEM 59%, INTEM 73%, and APTEM 57%) at admission to the ICU, indicating that the most substantial decline in clot formation seems to take place after the initiation phase of clotting (indicated by CT). The increase in CFT was much higher in INTEM than in EXTEM, at both admission to the ICU and the first postoperative day. CTs of EXTEM, FIBTEM and APTEM were also increased. In addition, INTEM CT had the smallest change of all variables at admission (only 5%), but increased further the first postoperative day (7% increase compared with baseline). Changes in alpha angle and MCF were more or less comparable between EXTEM and INTEM. The reduction in MCF of EXTEM and INTEM was not that robust as the reduction in FIBTEM MCF (47%), the latter indicating a considerable reduction in the contribution of fibrinogen to clot strength. The results for APTEM were comparable with EXTEM, with only a slightly smaller change. There were no differences in PLATEM MCF between baseline and the postoperative measurements.
Association of haemostatic variables with postoperative blood loss
For variables measured upon admission to the ICU, patients were categorised into subgroups according to whether the value was within or outside the reference range. With respect to ROTEM parameters, values remained within the reference range for the majority of patients. Median blood loss was higher in the subgroups with values outside the reference range (G2) when compared with the subgroups with values within the reference range (G1), but statistically significant differences were only found for the parameters fibrinogen, EXTEM alpha, INTEM CT, FIBTEM CT/MCF and APTEM CT (Table 3).
Comparison of median blood loss (IQR) in the total patient population with routine laboratory and ROTEM values outside and within the reference range at T2.
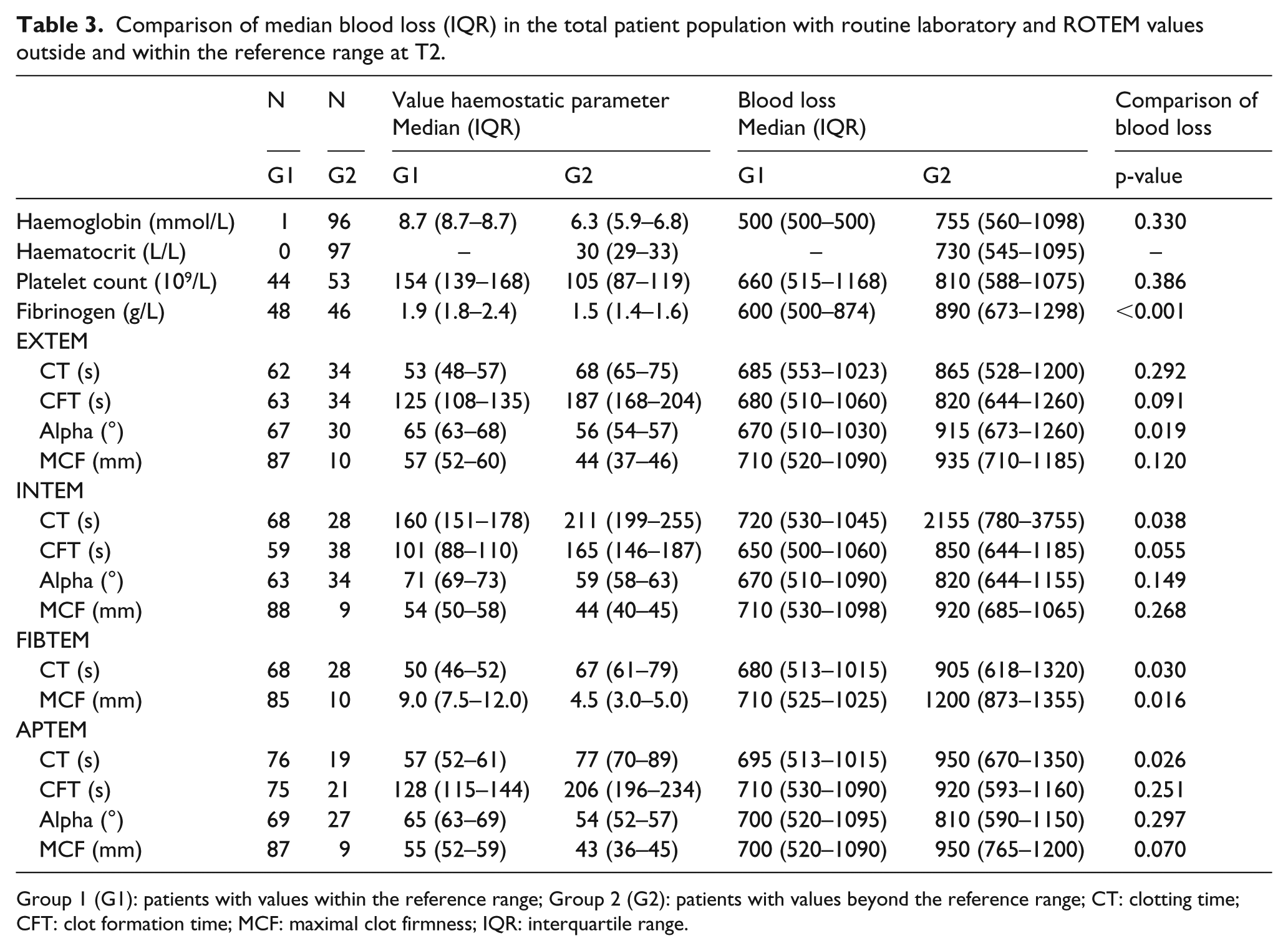
Group 1 (G1): patients with values within the reference range; Group 2 (G2): patients with values beyond the reference range; CT: clotting time; CFT: clot formation time; MCF: maximal clot firmness; IQR: interquartile range.
ROC analysis was performed to assess the capacity of ROTEM and routine laboratory tests in predicting blood loss of more than 1000 mL. We restricted the analysis to patients without treatment with haemostatic transfusion requirements during the whole perioperative period since any haemostatic transfusion could possibly affect chest tube drainage and disturb the independent influence of the routine laboratory/ROTEM parameter on blood loss. A total of 78 patients were included in this analysis (Table 4). AUCs of routine laboratory parameters were highest for plasma fibrinogen both at baseline and ICU admission (AUC = 0.642 and 0.646, respectively). Concerning the ROTEM assays, CFT, alpha, and MCF values at baseline were predictive for blood loss >1000 mL. The alpha angles showed highest AUCs (AUC⩾0.746). At the ICU admission, INTEM CT and FIBTEM MCF were significantly associated with increased bleeding (AUC = 0.669 and 0.689, respectively). Furthermore, AUCs of all ROTEM parameters were compared with the AUC of fibrinogen at baseline and at ICU admission. Only the predictive values of APTEM CFT and APTEM alpha at baseline for postoperative blood loss were significantly higher (p = 0.05 and 0.01, respectively) than fibrinogen (Table 4).
Predictive value for postoperative blood loss (>1000 mL versus ⩽1000 mL) expressed as AUC with 95% CI for routine laboratory variables and ROTEM variables for patients without haemostatic transfusion requirements and comparison of AUCs of ROTEM parameters with the AUC of plasma fibrinogen level.
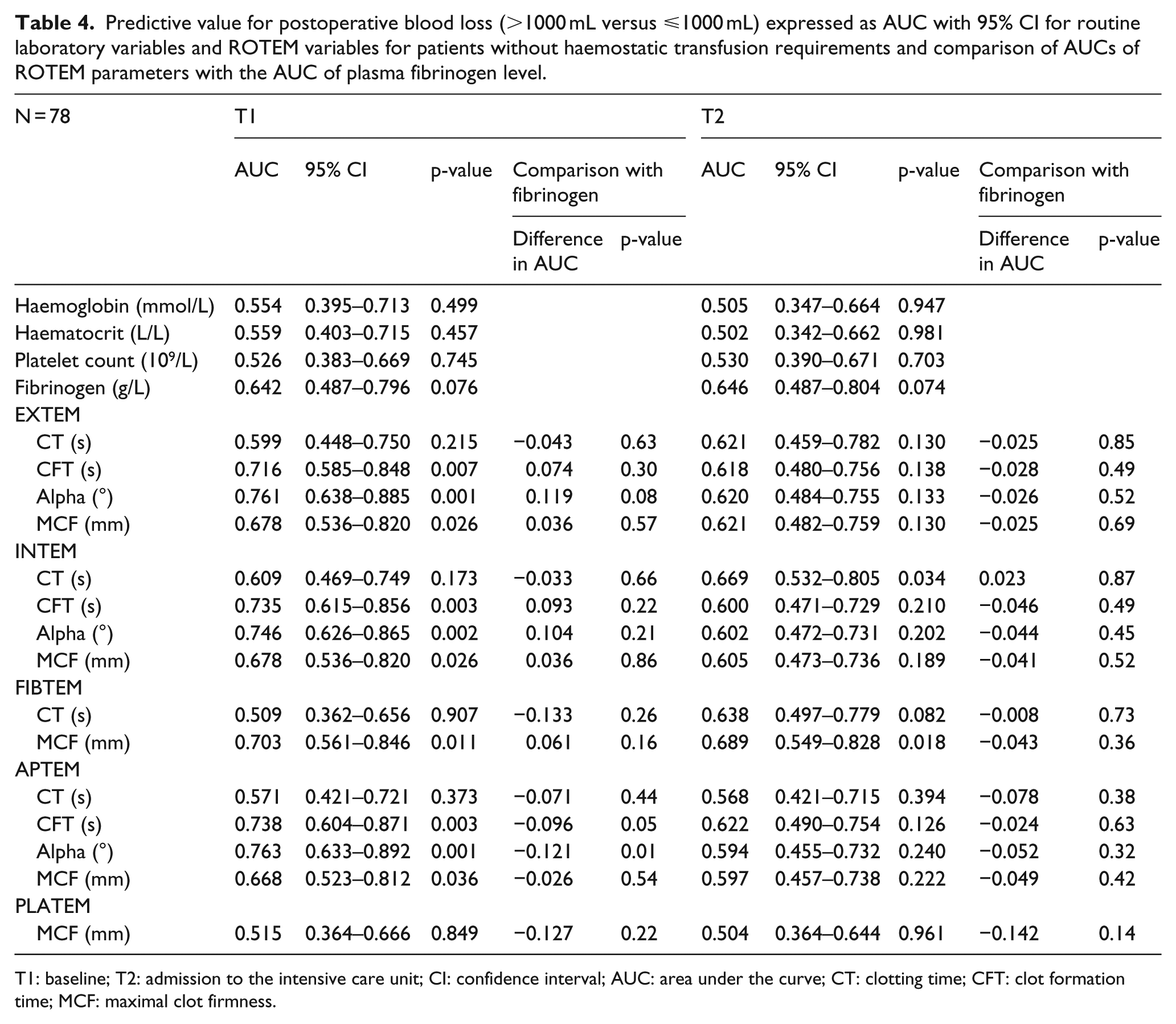
T1: baseline; T2: admission to the intensive care unit; CI: confidence interval; AUC: area under the curve; CT: clotting time; CFT: clot formation time; MCF: maximal clot firmness.
Discussion
In this study, we demonstrated that plasma fibrinogen levels and EXTEM alpha, INTEM CT, FIBTEM CT/MCF, and APTEM CT outside the reference range in the first hours postoperatively were associated with increased bleeding. When analysing the ability to predict blood loss, all ROTEM variables except CT parameters were associated with a moderate predictive value. Only APTEM CFT and alpha had a significantly higher predictive ability than classical fibrinogen determination by the Clauss method.
As expected, all haemostatic variables changed significantly from baseline to directly after surgery. In this study, median platelet count decreased by 35% and fibrinogen levels by 42% in the first hour after surgery. The most important causes of reduction in haemostatic factors are haemodilution, 19 consumptive coagulopathy 20 and excessive fibrinolysis.21,22 Although fibrinolysis was not estimated directly, it likely does not play a significant role in our patients since the results of the APTEM assay were not different from the results of the EXTEM assay at the different time points, indicating no hyperfibrinolysis. This could possibly be explained by the fact that all patients received tranexamic acid during surgery.
It would be expected that changes induced by CPB, like reductions in platelet count and fibrinogen, will be reflected by the ROTEM assay. The decrease in FIBTEM MCF is comparable with the decrease in fibrinogen at the ICU admission. This is in line with the excellent correlations between the two variables.5,6 This suggests that FIBTEM MCF could be an accurate ROTEM parameter for rapid assessment of fibrinogen levels. The reason for a greater change in FIBTEM MCF than the MCFs in other ROTEM assays could be related to compensatory interactions between platelets, red blood cells, and coagulation factors in the other assays. Since fibrinogen is a cofactor that enhances platelet aggregation, 23 MCF values may not decrease extensively if platelet counts are within the normal range.15,24 The median platelet count in our study directly postoperatively was just below the normal range. However, platelet count seems to be high enough to compensate for a decreased fibrinogen level compared to baseline in ROTEM assays other than FIBTEM with regard to the MCF parameter. The decrease in platelet count should be reflected in PLATEM. This was, however, the only variable that did not change after CPB. CPB would also lead to platelet dysfunction, 25 but platelet dysfunction cannot be detected with ROTEM tests because of the strong activation of platelets. 6 According to our results, we conclude that PLATEM MCF does not reflect the contribution of platelets. The role of platelets can probably better be assessed in tests describing the function of platelets, and not by ROTEM.
The specific contribution of fibrinogen and platelets to dilutional coagulopathy remains controversial. While Schols et al. 26 observed that platelets partially compensate to fibrin clot formation measured by TEG in conditions of plasma dilution in vitro, others 27 demonstrated in patients subjected to in vivo dilution that platelets were indeed important in fibrin clot formation, in a fibrinogen independent way. In contrast, Lang et al. 15 concluded in an in vitro study that the clot strength increases in a fibrinogen concentration-dependent manner independent of platelet count, even in case of thrombocytopenia.
According to our predefined analysis, patients having fibrinogen levels below the reference range had a significantly higher median total blood loss than other patients, whereas platelet count was not significantly associated with blood loss. Karlsson et al. 28 also found that low fibrinogen levels, even within the normal range, are an important cause of coagulopathy after cardiac surgery with CPB. Concerning the fibrinogen dependent ROTEM assay, FIBTEM parameters outside the reference range were also related to higher blood loss, suggesting a major role for fibrinogen in clot formation and/or stability. However, the importance of fibrinogen did not clearly emerge when assessing the capacity of the different tests discriminating between patients with blood loss of less or more than 1000 mL with ROC analysis. In general, all ROTEM tests except CT have moderate ability to predict blood loss with higher predictive abilities for postoperative bleeding than the predictive ability of plasma fibrinogen level (showing the highest AUC of the routine laboratory parameters), mainly at baseline. However, only the differences in AUC of APTEM CFT and APTEM alpha angle compared to fibrinogen reached statistical significance. FIBTEM MCF at the ICU admission demonstrated a higher predictive ability for blood loss than the other tests, but this was not significantly different from plasma fibrinogen level. Lee et al. 13 did also investigate ROTEM’s predictive value for chest tube drainage after cardiac surgery with CPB in 321 patients. From this study, it was concluded that ROTEM did not substantially improve prediction of chest tube drainage, beyond routine clinical and laboratory variables, although some ROTEM variables were independently associated with chest tube drainage post CPB. Our results confirm the results of Lee et al. that despite the poor predictive value for blood loss, ROTEM results are not inferior to fibrinogen in predicting relevant blood loss. That ROTEM is only moderately associated with the prediction of blood loss is not surprising, given the numerous factors related to blood loss in cardiac surgical patients, like surgical bleeding, acidosis, CPB time, and body weight.13,29 However, the advantage of ROTEM compared to plasma fibrinogen levels is that the test has shorter turnaround times, it gives information about the complete haemostatic profile of the patient, and it could therefore be a faster method to guide haemostatic therapy.
This study had limited sample size, but was large enough to detect statistically significant AUCs for various ROTEM parameters. However, the power was too low to detect an increase in AUC by at least 0.05 when comparing the discriminative ability (AUC) between ROTEM parameters and fibrinogen. Only AUC differences in the order of 0.1 or larger could be detected with statistical significance. For example, the AUC difference between APTEM CFT versus fibrinogen was 0.096 associated with a p-value of 0.05. Another limitation is that the red blood cell content of the fluid in the mediastinal drains was not measured, which might be different between patients because of progression from blood to serosanguineous drainage.
In summary, results of routine laboratory tests and the ROTEM assay outside the reference range are associated with higher median blood loss when compared with values within the reference range. ROTEM parameters have moderately high predictive abilities for postoperative blood loss and are slightly higher than the predictive ability of plasma fibrinogen level. The results of this study propose a role for ROTEM as a point-of-care method to monitor haemostasis in cardiac surgical patients because of a higher predictive ability for blood loss and a more complete reflection of haemostasis compared to fibrinogen. Besides this, ROTEM is a faster method to guide haemostatic therapy.
Footnotes
Acknowledgements
We acknowledge Rick Wetzels for expert coordination of the ROTEM measurements. This work is registered in the ‘Nederlands Trial Register’: NTR2712.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplementary Materials
All data are saved in folders and in a digital SPSS file to which the corresponding author has access.
