Abstract
Giant pulmonary artery aneurysms are rare with a poorly understood natural history. They can be idiopathic but often associated with congenital cardiac deformities, pulmonary hypertension or vasculitides. Indications for surgery and clinical course are still not well defined. We present a case of a giant pulmonary artery aneurysm corrected surgically using a Dacron bifurcation graft with concomitant single concomitant coronary artery bypass graft and 4-year radiology follow-up.
Introduction
Aneurysms of the pulmonary artery are rare, with an estimated incidence of approximately 1 in 14,000. Almost half of all pulmonary artery aneurysms (PAAs) occur in the setting of congenital heart disease with pulmonary hypertension. Other aetiologies include vasculitis, syphilis, tuberculosis, cystic medial degeneration, connective tissue disorders and idiopathic causes. 1 This rapidly expanding giant PAA was managed surgically with concomitant coronary artery bypass graft on a background of Wegener’s granulomatosis and complication-free after 3 years.
Case report
A 79-year-old man had a 7.6-cm giant PAA found incidentally on a computed tomography (CT) of chest (Figure 1 – sagittal; Figure 2 – axial; Figure 3 – three-dimensional (3D) reconstruction of aneurysm) as investigation for a 4-year history of progressive breathlessness and 2-year history of central chest pain and orthopnoea. He was referred to us for further management. Due to the size of the aneurysm and progressive symptoms, we decided the best option was for surgery and pulmonary artery replacement. Intraoperative transoesophageal echocardiogram confirmed a PAA with a maximal diameter measuring more than 8 cm. The pulmonary valve was normal in structure with only trivial regurgitation. Figure 3 is a 3D reconstruction from the CT scan. The aneurysm involved the main pulmonary trunk and extends to the left and right main pulmonary arteries.
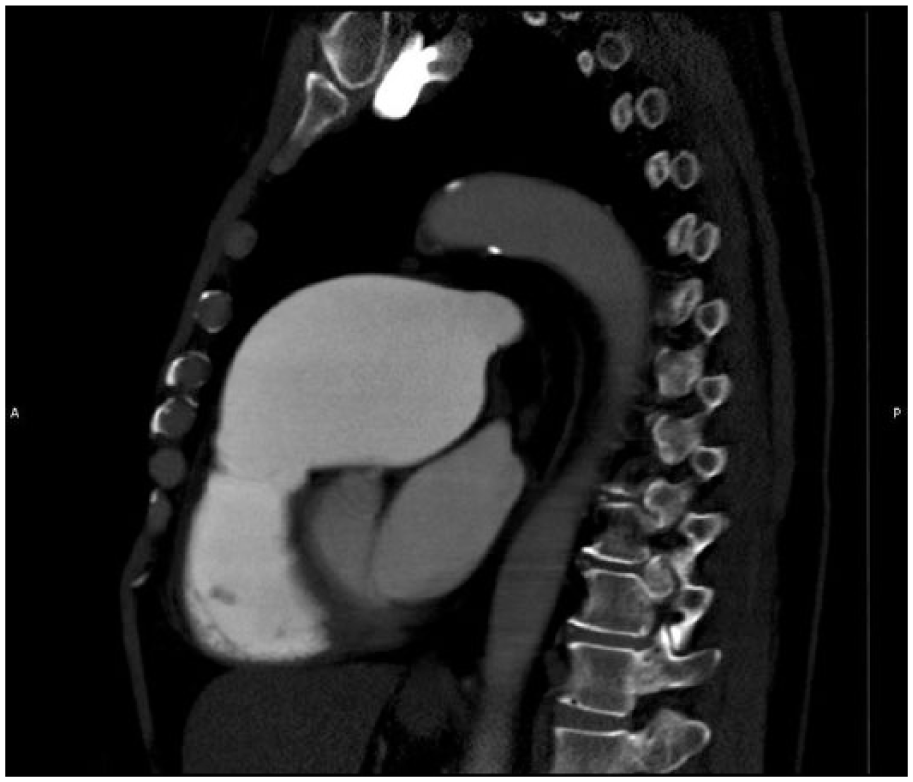
Sagittal view of PAA.
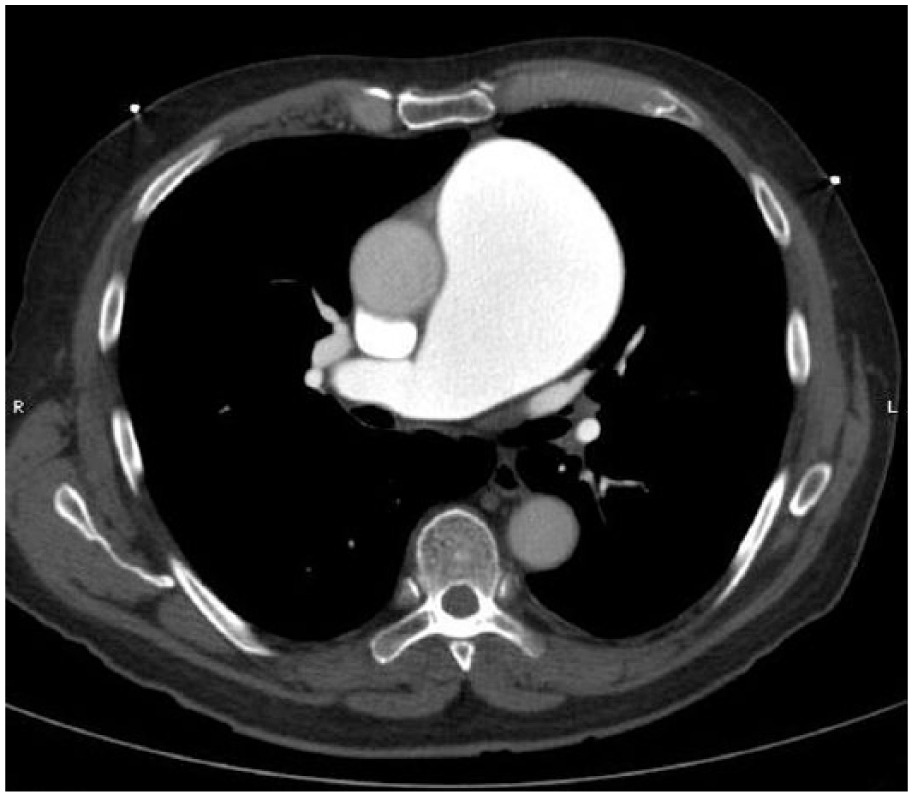
Axial view of PAA.
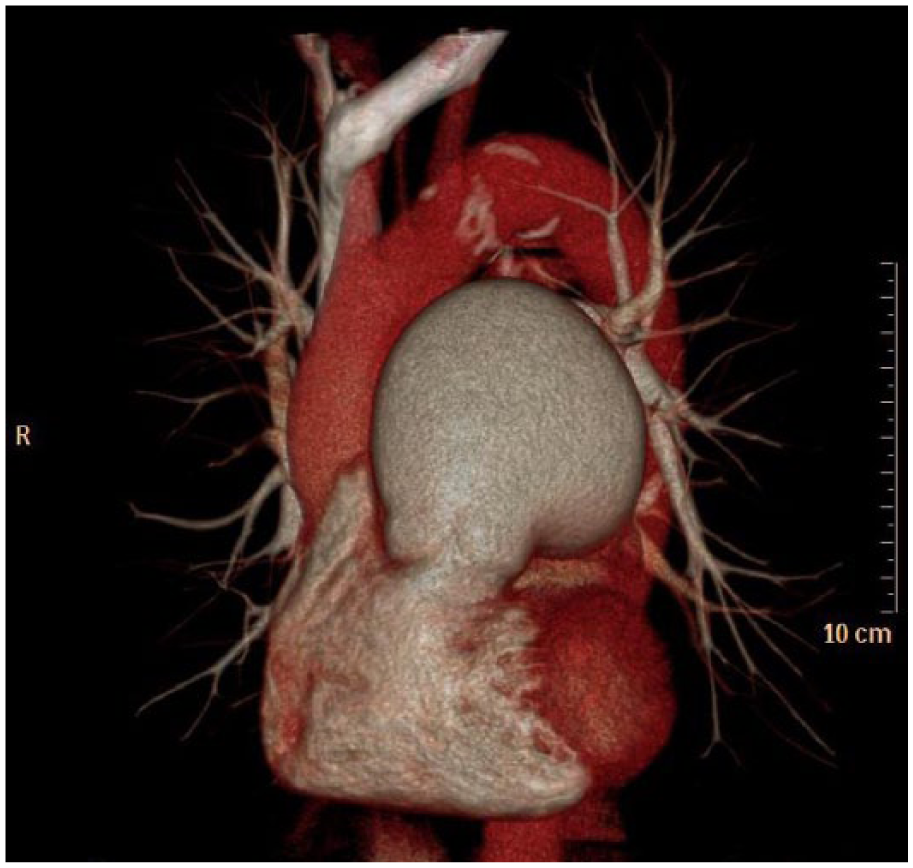
3D reconstruction of PAA (preoperative).
After a median sternotomy was performed, a pedicled left internal thoracic artery was harvested, followed by direct aortic and venous cannulation. Cardiopulmonary bypass was initiated without complication and myocardial arrest achieved with cold blood cardioplegia and systemic hypothermia of 34°C. The PAA was dissected to the left and right main branches and resected. The pulmonary trunk with left and right pulmonary arteries was reconstructed with a bifurcated 24 cm × 12 cm × 12 cm Hemashield® woven Dacron® graft (Boston Scientific, Marlborough, MA, USA). End-to-end anastomosis was performed with continuous 4-0 Prolene® suture (Ethicon, Somerville, NJ, USA) and the suture line sealed with BioGlue® (Advanced Biomedical, Ringwood East, VIC, Australia). The patient was rewarmed as the left internal thoracic artery was anastomosed to the left anterior descending artery and separated from cardiopulmonary bypass with minimal inotropic support.
Follow-up CT at 3 months post-operation showed an intact graft with no abnormal dilatation or stenosis. The pulmonary artery anatomy remains stable on CT imaging for over 4 years (Figure 4 – 3D reconstruction).
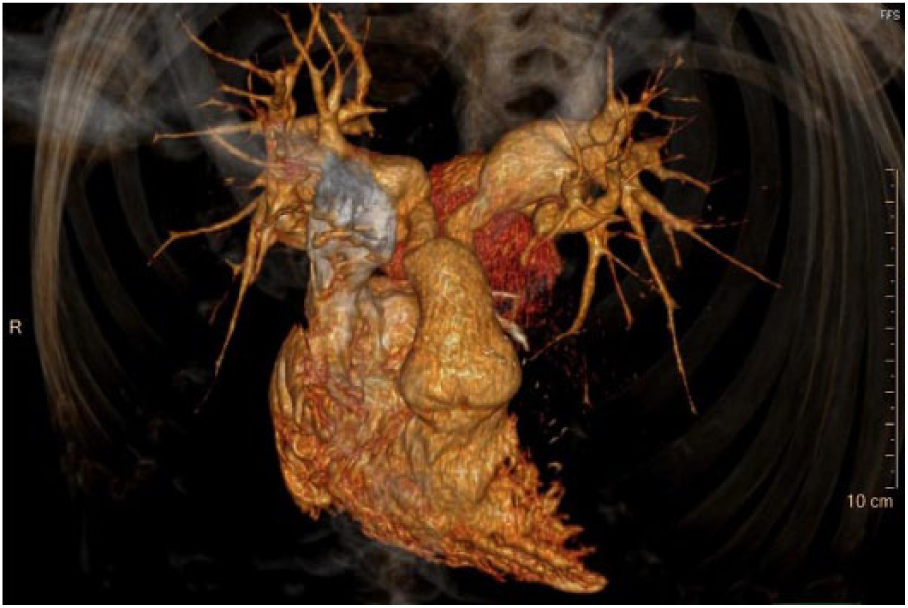
3D reconstruction at 4-year follow-up with bifurcation graft in situ. Dilatation of left and right pulmonary arteries distal to graft remains stable.
Histology
Histological assessment of the resected aneurysm demonstrated stromal degeneration with non-specific, patchy, non-necrotic granulomatous inflammatory changes scattered throughout the media. Given a negative anti-neutrophil cytoplasmic antibody (ANCA) titre, this histological picture is not characteristic of Wegener’s granulomatosis or Churg–Strauss disease. 2 The histology is, however, consistent with that found in aortic aneurysms.
Discussion
Idiopathic PAAs are very rare, and as such, the natural history of untreated disease is unknown. Evidence regarding management of PAA is limited. Conservative management can be successful in the setting of low pulmonary pressures, 3 or where the aetiology is reversible and completely treated, 4 however, surgery remains the preferred treatment option for patients with symptoms, pulmonary hypertension, right heart ventricular dysfunction or valvulopathy. 5 Rupture of a PAA is a very rare but serious complication. 6 However, as it is still unclear whether PAAs have a hinge point in size like that of aortic aneurysms to guide intervention, patients are treated on an individual basis.
We recommend that any patients with symptomatic PAA should undergo surgery. The indications for surgery have been extrapolated from our understanding of aortic aneurysms. They include PAA size greater than 5 cm or progressive enlargement of PAA of more than 0.5 cm per 6 months, presence of dissection 7 or symptoms of dyspnoea, chest pain and haemoptysis. 1 In our patient, the decision to operate was based on size and rapid growth.
PAA is reportedly associated with large-vessel vasculitis such as Behçet’s disease, 4 but its role in small-to-medium vessel vasculitides (such as Wegener’s granulomatosis or Churg–Strauss disease) has not been reported in the literature. Therefore, it is unknown whether Wegener’s granulomatosis is implicated in the development of the patient’s PAA or rather his asthma and associated eosinophilia.
Various surgical methods have been successfully used to treat PAA including Dacron graft repair, aneurysmorrhaphy and autologous pericardial replacement. 3 Concomitant procedures have been performed and reported successful. 8 Without further evidence, we can only base our treatment decision on our current knowledge. However, concomitant PAA replacement and coronary artery bypass graft can be performed safely with good medium term results.
Footnotes
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Patient consent
The authors declare that they have obtained written informed consent for the publication of details in relation to the patient in this report. All possible steps have been taken to safeguard the identity of the patient. This submission is compliant with the requirements of the Barwon Health’s Research and Ethics Committee.
