Abstract
Background
Anti-CD20 therapy is a highly effective multiple sclerosis (MS) disease-modifying therapy, but may be complicated by infections, which has led to the utilization of different extended interval dosing (EID) regimens.
Objectives
To compare the efficacy and safety of standard interval dosing (SID) and EID of anti-CD20 therapy in MS.
Methods
We searched PubMed and MS conference abstracts and included all studies comparing distinct cohorts of people with MS (PwMS) treated with SID and EID of anti-CD20 therapy that reported ≥1 efficacy or safety outcome measure. A meta-analysis was performed with effect size calculation (log odds ratio [logOR]) of efficacy and safety outcome measures.
Results
Seventeen studies comparing SID to EID were identified, with 1739 (SID) and 1856 (EID) participants. There was no significant difference in frequencies of PwMS with relapses (logOR = 0.15; p = 0.37), MRI activity (logOR = 0.05; p = 0.7), no evidence of disease activity −3 (logOR = 0.33; p = 0.33), serious infections (logOR = 0.46; p = 0.16), and hypogammaglobulinemia G (logOR = −0.24; p = 0.76).
Conclusions
Data from observational studies suggests that EID of anti-CD20 therapy may have a comparable efficacy and safety profile compared to SID.
Introduction
Anti-CD20 therapy is a highly effective treatment for relapsing-remitting multiple sclerosis (RRMS). 1 The four anti-CD20 monoclonal antibodies currently utilized for treatment of RRMS are ocrelizumab (OCR), rituximab (RTX), ofatumumab (OFA), and ublituximab (UTX). 2 RTX has been used as an off-label MS disease-modifying therapy (DMT) for many years. 3 OCR was approved by the US Food and Drug Administration (FDA) in 2017 to be used as a treatment of RRMS, primary progressive MS, and active secondary progressive multiple sclerosis (SPMS). 4 This was followed by FDA approval for OFA in 2020 for RRMS and active SPMS. 5 Additionally, UTX was approved by the FDA in 2022 for RRMS and active SPMS. 6
RTX, OCR, and UTX have intravenous formulae that can be given by two induction doses two weeks apart with subsequent every-6-month standard interval dosing (SID). OFA is a subcutaneous medication that is given monthly after initial weekly induction doses. 2 Additionally, subcutaneous every-6-month administration of OCR has been recently approved. 7
There is a concern that the usage of B-cell depleting therapy may be associated with an increased risk of life-threatening infection, impaired vaccine response,8,9 and hypogammaglobulinemia.10,11 Additionally, there is a risk of secondary immunoglobulin deficiency involving the immunoglobulin M (IgM) and G (IgG) subtypes, 11 which may be associated with the occurrence of infections, such as urinary tract infection, cellulitis, and pneumonia. 11
A potential solution to mitigate risks of B-cell depleting medications could be extended interval dosing (EID) of anti-CD20 therapy. Individual B-cell repopulation rates are highly variable, and complete depletion may last significantly longer than the dosing interval. 8 Furthermore, the initial B cell subset to repopulate (naïve CD19+/CD20+) is not considered as a driver for disease activity. 8 Consequently, the COVID-19 pandemic and concerns for the potential risks associated with long-term B-cell depleting medication12–14 have led many centers to delay redosing of anti-CD20 agents. Nonetheless, B-cell depletion in the peripheral blood may not reflect deep tissue B-cell depletion, and analysis of the OCR pivotal trial data suggests that lower OCR exposure could be associated with higher rates of disability progression. 15 This could caution the extension of OCR dosing intervals.
To date, a variety of extended dosing approaches have been applied in different centers worldwide, including personalized dosing regimens guided by laboratory evidence of B-cell repopulation. 16 The current study aims to analyze the currently available studies comparing SID to various EID regimes of anti-CD20 therapy.
Methods
This is a meta-analysis of all studies that evaluated standard interval dosing versus extended interval dosing of anti-CD20 therapy in MS. For this, we conducted a PubMed search on 2 April 2025, with the following search term: (“extended interval dosing” OR “EID” OR “alternate dosing”) AND (“ocrelizumab” OR “rituximab” OR “ofatumumab” OR “ublituximab” OR “RTX” OR “OCR” OR “OFA”) AND (“multiple sclerosis” OR “MS”). Additionally, we searched conference abstracts of the European Committee of Research and Treatment in Multiple Sclerosis and the American Committee of Research and Treatment in Multiple Sclerosis from 2020 to 2024 using the search term “extended interval dosing.”
Inclusion criteria included (1) a confirmed diagnosis of MS according to McDonald criteria (2005, 2010, and 2017), (2) comparative studies with two distinct cohorts of people with MS (PwMS) comparing SID to EID of anti-CD20 therapy (RTX, OCR, OFA, and UTX), and (3) ≥1 reported efficacy or safety outcome measure. According to the Preferred Reporting Items for systematic review and meta-analysis (PRISMA) guidelines, as shown in Figure 1, the literature search was conducted by two reviewers (TR and ME). A long discussion was held until final agreement was achieved.

Flow diagram of the identification process for eligible studies per PRISMA guidelines.
For each study, data was extracted, including author names, title, sample size, study design, type of MS, mean duration of dosing interval in weeks, and follow-up duration in weeks, as well as the number of PwMS with relapses, MRI activity, “no evidence of disease activity – 3” [NEDA-3], serious infections, and hypogammaglobulinemia. In case data was not available, the authors were contacted to obtain additional data if available.
A meta-analysis (IBM® SPSS Statistics Version 31.0.1.0) with effect size calculation (effect size measure: log odds ratio [logOR]; random-effects model; estimation method: restricted maximum likelihood) of the following binary efficacy and safety outcome measures was performed: Reported frequencies of participants with (1) clinical relapses, (2) MRI activity (new/enlarging T2 lesions and/or gadolinium-enhancing lesions), (3) NEDA-3, (4) serious infections, and (5) hypogammaglobulinemia G. For outcome measures with ≥10 included studies, random-effects meta-regression was performed with follow-up duration as a continuous covariate to assess whether follow-up duration modifies the intervention effect.
Results
We identified a total of 35 articles, and 18 were excluded after review (review articles: n = 7,17–20 not related to research question: n = 6,21–26 no comparison group n = 2,27,28 comparison of intraindividual changes pre- versus post intervention: n = 329–31; Figure 1). Seventeen studies were included in this meta-analysis with a total of 1739 (SID) and 1856 (EID) participants. The majority of PwMS were diagnosed with RRMS, but there was also a smaller proportion of participants with PPMS (n = 354 [9.85%]) and SPMS (n = 27 [1.31%]). While the studies varied significantly in design, population, and outcome measures, all reported at least one efficacy or safety outcome measure.
All 17 studies had an observational design (prospective: n = 4,32–35 retrospective: n = 13). There were no randomized-controlled clinical trials. The utilized anti-CD20 agent was OCR in 13 studies and RTX in two studies36,37 with the other two studies including both OCR and RTX.35,38 There were no studies available assessing the EID of ofatumumab or ublituximab. The publication date of the included studies ranged from January 2020 to March 2025.
Fourteen studies were conducted in Europe, two in the United States,16,39 and one in Asia. 36 The summary of all included studies is presented in Table 1. The dosing interval in the EID cohorts of the different studies varied substantially from 7.25 to 27 months.
Overview of the 17 included studies including agent used, dosing regimen, MS type and different efficacy and safety outcome measures. PO = prospective observational, RO = retrospective observational, OCR = Ocrelizumab, RTX = Rituximab, SID = standard interval dosing, EID = extended interval dosing, NR = not reported.
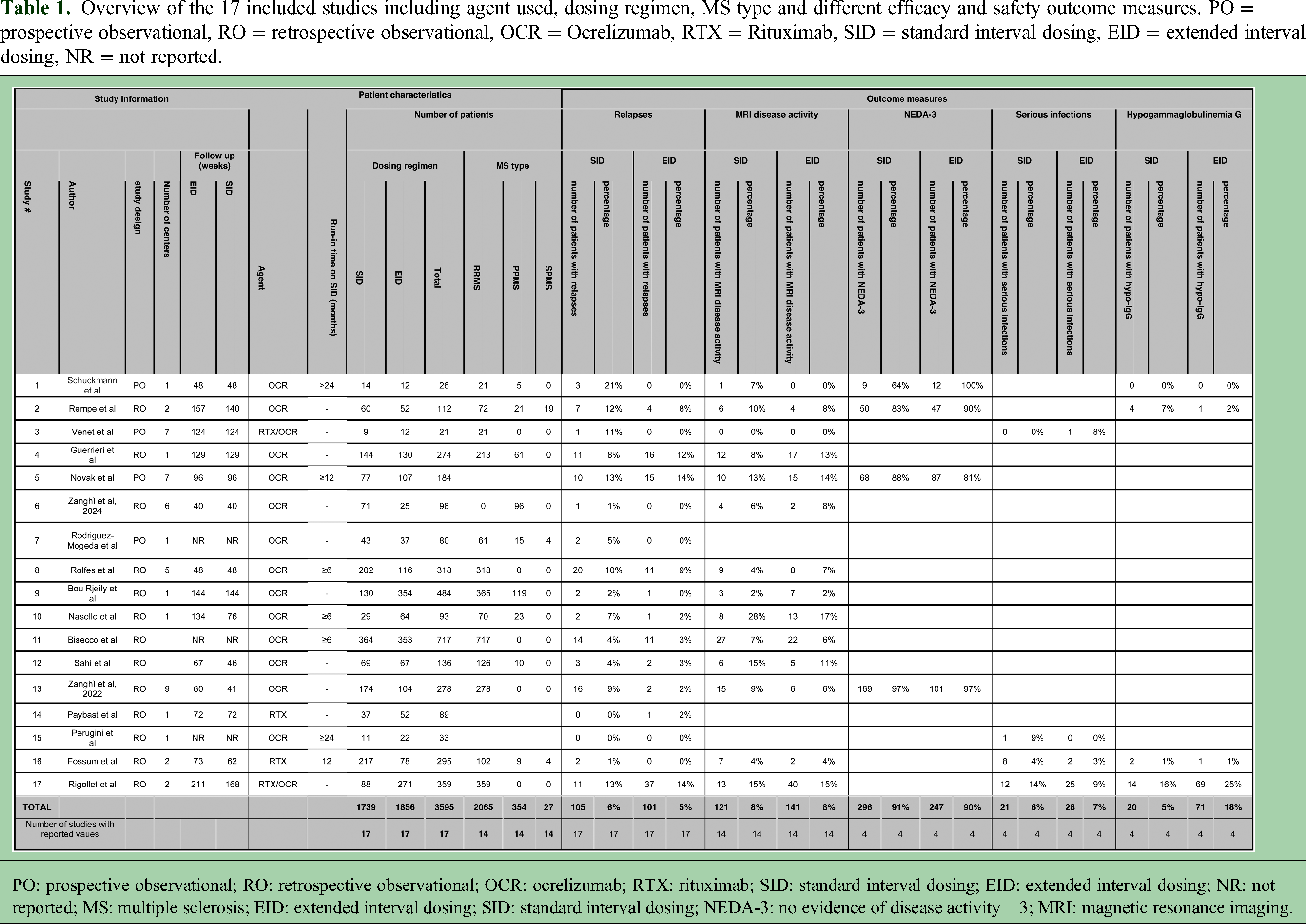
PO: prospective observational; RO: retrospective observational; OCR: ocrelizumab; RTX: rituximab; SID: standard interval dosing; EID: extended interval dosing; NR: not reported; MS: multiple sclerosis; EID: extended interval dosing; SID: standard interval dosing; NEDA-3: no evidence of disease activity – 3; MRI: magnetic resonance imaging.
Summary of included studies
4/17 studies evaluated B-cell repopulation-guided EID:
Schuckmann et al.
33
conducted a prospective observational cohort study of PwMS treated with OCR SID for at least 24 months. At that time, patients chose whether to continue SID (n = 14) or to switch to B-cell-adapted EID (n = 12) based on clinical and radiological disease stability. The authors’ rationale to conduct the study was to assess longitudinal safety and efficacy data of B-cell repopulation-guided EID, as only minorities show B-cell repopulation during the 6-month period between infusions. There was no difference between the two groups in different efficacy measures. While no patient developed hypogammaglobulinemia G, the authors described a decrease in IgG, IgM, and IgA in the SID group that was not observed in EID. Rempe et al.
39
previously conducted a two-center retrospective study comparing rates of “no evidence of disease activity – 3” (NEDA-3) and hypogammaglobulinemia between patients treated with B-cell repopulation-guided EID versus SID. The selection of the dosing interval was based on provider preference. We did not find a significant difference in NEDA-3 rates (EID: 47/52 [90.4%] and SID 50/60 [83.3%]) or hypo-IgG. However, hypogammaglobulinemia M was significantly less frequent in the EID group (17.3%) than in the SD group (55%, p < 0.001). Venet et al.
35
conducted a prospective study of 21 pediatric PwMS to assess the efficacy of RTX/OCR SID (n = 9) versus EID (n = 12) guided by B cell repopulation. Only one clinical relapse occurred in the SID group. No disability worsening or new T2 lesion was observed in both groups. Rodriguez-Mogeda et al.
34
used mass cytometry to compare peripheral B cell repopulation in 80 PwMS treated with either SID (n = 43) or EID (n = 37) of OCR, along with a control group (n = 28). No differences in disease activity among both groups.
10/17 studies reported variable EID regimens utilized during the COVID-19 pandemic:
Rolfes et al.
40
conducted a retrospective multicenter cohort study of 318 people with RRMS in Germany to assess the effects of EID of ocrelizumab during the COVID-19 pandemic. Patients receiving EID (≥4-week delay) were compared to those with SID. There were no significant differences in efficacy measures between groups (loss of NEDA, confirmed progression of disability, and MRI progression). Both groups showed persistent CD19+ B-cell depletion, and B-cell repopulation was not associated with re-emergence of clinical (relapses) or radiographic (MRI progression) disease activity. Guerrieri et al.
41
retrospectively analyzed 274 PwMS (213 RMS and 61 PPMS) to assess the effects of OCR EID during the COVID-19 pandemic. PwMS were grouped into SID or EID cohorts based on whether their infusion was delayed by ≥4 weeks. No significant differences in efficacy measures were observed between SID and EID. Zanghì et al.
42
compared OCR EID to SID in people with RRMS from January 2020 to June 2021. While there was no difference in most evaluated efficacy outcome measures, the study found that PwMS on EID had an increased risk of MRI activity. Zanghì et al.
43
also reported real-world data on 96 PPMS patients treated with ocrelizumab during the COVID-19 pandemic, comparing SID and EID. Over a median follow-up of 10 months, confirmed disability progression occurred in 4.2% of SID patients and 8% of EID patients, with no statistically significant difference (p = 0.167). The study concluded that EID did not increase the risk of disability progression in this cohort. Bou Rjeily et al.
16
reviewed records of 361 PwMS to assess the impact of OCR EID (infusion intervals ≥8 months in 64%; ≥ 12 months in 34%) during the COVID-19 pandemic. No significant differences in breakthrough disease activity were observed between EID and SID groups. Bisecco et al.
44
assessed changes in OCR infusion schedules during the COVID-19 pandemic in Italy in PwMS with at least one prior cycle of OCR and evaluated the impact of delayed infusion on clinical and radiological endpoints. The mean delay between two OCR infusions was 2.74 weeks. The study concluded that short-term suspension of OCR was associated with durable efficacy and absence of rebound. Sahi et al.
45
conducted a retrospective study of 136 MS patients who received OCR during the COVID-19 pandemic found no significant difference in clinical relapse rate or radiological activity between those who received EID and SID. While EID patients had significantly higher CD19+ B-cell counts, with 94% of EID patients repleting >1% of peripheral lymphocytes by the next ocrelizumab cycle compared with 17% of the SID patients, repopulation not associated with clinical relapses or MRI disease activity. Novak et al.
32
conducted a prospective, open-label, multi-center study comparing SID and EID of OCR in 184 PwMS who had received SID with OCR ≥12 months. Participants in the EID group had an average delay of 9 weeks. There were no significant differences in clinical, radiological outcomes, or biomarker levels (NfL, glial fibrillary acidic protein [GFAP]). Paybast et al.
36
compared treatment outcomes of PwMS receiving RTX at EID (6-month infusion intervals + ≥ 4 weeks) versus SID and found no significant difference in EDSS progression, RRMS, and PMS. However, PwMS in the EID group were less likely to develop moderate/severe infectious adverse events after 6 and 12 months. Nasello et al.
46
retrospectively analyzed 93 PwMS (75.3% RRMS) that received the two induction, and at least one maintenance infusion, and subsequently either received OCR SID (n = 29) or EID (n = 64), defined as a ≥ 4-week delay. No significant differences were found between EID and SID in clinical or radiological outcomes. EID was associated with reduced B-cell depletion, while T-cell counts and immunoglobulin levels, including hypogammaglobulinemia incidence, were similar between groups.
3/17 studies evaluated set increased dosing intervals ranging from every 9 months to every 12 months:
Perugini et al.
47
compared the efficacy and safety of every-9-month OCR EID (n = 22) and SID (n = 11) in PwMS with ≥2 years of previous OCR treatment and found no significant differences in disease onset age, sex, treatment duration, conversion to progressive forms, progression index, and annualized relapse rate. Infection incidence was higher in the SID group, including one serious infection. Fossum et al.
37
compared RTX every-9-month EID to SID in PwMS treated at two hospitals in Norway using data from the Norwegian MS Registry. All PwMS initially received two every-6-month intervals of RTX with one hospital subsequently transitioning to every-9-month EID in response to the COVID-19 pandemic. A total of 295 PwMS (EID: n = 78 and SID: n = 217) were included. No significant differences were observed between EID and SID groups in relapse rates, new T2 lesions, or incidence of adverse events. Rigollet et al.
38
retrospectively analyzed 359 people with RRMS treated with RTX or OCR using either SID (n = 88) or every 12-month EID (n = 271) in a retrospective two-center study. EID was initiated in one of the centers with the goal of improving safety. No significant differences were found between groups in time to first relapse, sustained disability accumulation, or MRI activity. Time to first severe infectious event was shorter in the SID group (p = 0.005). Reduction in serum IgG was less pronounced in the EID group, and higher IgG levels were associated with reduced infection risk (HR = 0.77 per g/L, p = 0.006).
Efficacy outcomes
Clinical relapses
All (17/17) studies reported relapse rates with no significant difference between the two groups (SID: 105/1738 [6.04%]; EID: 101/1856 [5.44%]); pooled logOR = 0.15 [confidence interval (CI): −0.17–0.46] p = 0.37). Meta-regression showed that follow-up duration did not explain between-study heterogeneity (R2 = 0%) and substantial residual heterogeneity remained (τ2 = 4.93; I2 = 95.9%; Q(12) = 164.15, p < 0.001).
MRI activity
MRI activity frequencies were reported in 13/17 studies without a significant difference between groups (SID: 121/1567 [7.72%]; EID: 141/1713 [8.23%]; pooled logOR = 0.05 [CI: −0.22–0.32] p = 0.7) (Figure 2). Meta-regression showed no significant residual heterogeneity (Q(10) = 5.60, p = 0.847; τ2 = 0; I2 = 0%). Follow-up duration did not explain variability in effect size (R2 = 0%), although between-study heterogeneity was negligible.

Forest plots depicting the results of the meta-analysis: (a) relapse rates; (b) MRI activity in both groups; (c) NEDA-3 frequency in both groups; (d) frequencies of serious infections; and (e) frequencies of hypogammaglobulinemia G in both groups.
NEDA-3
NEDA-3 rates were reported in 4/17 studies with no significant difference (SID: 296/325 [91.08%]; EID: 247/275 [89.82%]; pooled logOR = 0.33 [CI: −0.33–0.98] p = 0.33)32,33,39,42 (Figure 2).
Safety outcomes
Serious infections
4/17 studies reported frequencies of serious infections with no significant difference (SID: (21/325 [6.46%]; EID 28/383 [7.31%]); pooled logOR = 0.46 [CI: −0.18–1.1] p = 0.16).35,37,38,47
Hypogammaglobulinemia G
4/17 studies reported frequencies of patients with development of hypogammaglobulinemia G, and there was no significant difference in between the two groups (20/365 [5.48%]) compared to SID (71/401 [17.71%]; pooled logOR = −0.24 [CI: −1.24–1.1] p = 0.76)37–39 (Figure 2). Meta-regression was not performed for outcomes with fewer than 10 included studies due to insufficient statistical power and unstable heterogeneity estimates.
Discussion
This review and meta-analysis of a total of 17 studies did not show statistically significant differences in EID and SID of RTX and OCR in both efficacy (relapses, MRI activity, and NEDA-3) and safety (serious infections and hypogammaglobulinemia G).
The most widely reported efficacy measures were frequencies of patients with clinical relapses and MRI activity, and the substantial number of available observational studies for these outcome measures does not suggest a significant impact on efficacy associated with extension of the dosing interval. Accordingly, EID of anti-CD20 therapy could potentially be a promising approach to mitigate safety concerns of prolonged B-cell depleting therapy. The current meta-analysis did also not show a significant difference in serious infections or hypogammaglobulinemia G rates. However, it is important to note that safety measures were only reported in a minority of studies.
While no statistically significant difference in NEDA-3 rates was observed, NEDA-3 was only reported in four studies, which may not be sufficient to discern the previously reported association between OCR exposure and disability progression. 15 However, recently published data from the MUSETTE (NCT04544436; relapsing MS) and GAVOTTE (NCT04548999; PPMS) 48 randomized-controlled clinical trials confirmed that higher exposure (1200 or 1800 mg of OCR compared to 600 mg standard dosing) did not lead to an additional benefit in confirmed disability progression. 49
It is important to note that the development of hypogammaglobulinemia G occurs over a long period of time 33 and that the cumulative time on treatment and the cumulative total dose are important factors associated with the emergence of anti-CD20 therapy-associated adverse events. 50 Accordingly, the observation period in the included studies may not have been long enough to detect differences between SID and EID in rates of serious infections and hypogammaglobulinemia.
At the time of this study, there was no data available regarding UTX and OFA, and while the mode of action of these medications is similar, current observations may not be reproducible in these medications.
The redosing strategies utilized in the currently available anti-CD20 EID studies were highly variable. Besides fixed extension intervals and extensions due to the COVID-19 pandemic, four studies utilized B-cell repopulation-guided EID. Strict time intervals in both SID and EID do not consider the highly variable individual B-cell repopulation times. In fact, the duration for B-cell repletion can range from 27 to 175 weeks. Naïve CD19+/CD20+ B-cells are the initial B-cell subset to repopulate in anti-CD20 therapy but are generally not considered drivers of MS disease activity. Indeed, the benefits of anti-CD20 therapy were also demonstrated after B-cell repopulation previously.28,30,40 Repopulation of CD27+ central memory B-cells show a slow and delayed repopulation, and frequently do not recover numerically within 2 years.51,52 However, evidence of repopulation of naïve CD19+/CD20+ can serve as an indicator of subsequent repopulation of CD27+ central memory B-cells, indicating a promising potential to serve as a biomarker to determine the individual need for redosing of OCR therapy. Therefore, B-cell repopulation-guided EID, where redosing of the medication is delayed until there is serologic evidence of B-cell repopulation, could represent a promising approach for an individualized dosing strategy to mitigate OCR-associated risks. Such personalized dosing regimens can be easily incorporated into clinical practice because testing for lymphocyte subsets is readily available in most laboratories. 31 Given the absence of results of randomized-controlled clinical trials, there is no standard practice regarding specific redosing thresholds in B-cell repopulation-guided EID, but commonly used practices for redosing include any evidence of repopulation, 39 <10 cells/µL (NCT05296161), and <1% of total lymphocytes. 33 The laboratory monitoring of B-cell subsets to evaluate for repopulation of CD27+ B-cells could represent a more direct monitoring tool, but appears less feasible for routine clinical practice, as B-cell subsets are not widely available in commercial laboratories. There remains the question of whether anti-CD20 therapy could potentially even be discontinued completely, as there is evidence that B-cell depletion has potential immune induction properties with absent activated memory B-cells. 53 Accordingly, the discontinuation of OCR is currently evaluated in a randomized-controlled clinical trial (NCT05285891).
The main limitation of the currently available data is that only observational studies are available, with the absence of randomized-controlled clinical trials. Accordingly, multiple randomized-controlled trials have been designed to further investigate this topic. These include (1) WINDCORE (NCT05999604), which investigates the effects of OCR every-6-month SID versus every-12-month EID on radiological disease activity (new or enlarged T2 lesion), (2) BLOOMS (NCT05296161) which evaluates B-cell repopulation-guided EID of OCR compared to SID, and (3) RIDOSE, which compared RTX 500 mg every-6-month and every-12-month dosing with recently reported similar efficacy and safety of both dosing regimens. 54 Observational data is subject to selection bias and to confounding by indication. It is likely that PwMS selected for the EID regimen may have had lower risks of inflammatory activity and potentially higher risks of infectious complications. Furthermore, a thorough assessment of the occurred infections is difficult in retrospective studies due to missing data.
Additional limitations of the studies included the usage of OR instead of hazard ratios due to the outcome type. Furthermore, there was only a small number of prospective studies with a weaker effect on the results. Moreover, there is significant heterogeneity in between the different studies regarding the definition of the EID and the specific durations of the regimens. While meta-regression can explore whether follow-up duration may explain between-study variability, its statistical power is limited when the number of included studies is small. In this analysis, meta-regression was only performed for outcomes with ≥10 studies, as fewer studies lead to unstable and unreliable estimates.
In conclusion, the currently available data does not show any significant difference in between SID and EID, but the current level of evidence for EID of anti-CD20 therapy remains low overall (Class III). Currently available data on infection and immunoglobulin levels appear inadequate, and longer prospective assessments are needed to evaluate potential safety benefits of EID in preventing infectious complications. A randomized-controlled trial is needed to further evaluate the safety and efficacy of EID in anti-CD20 therapy.
Footnotes
Data availability statement
The data sets generated and analyzed during this study are available from the corresponding author on reasonable request and with necessary approvals.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Mahmoud Elkhooly and Rhea Jacob report no disclosures. Torge Rempe received grant funding from the National Multiple Sclerosis Society. He served on an advisory board for Genentech, Alexion, EMD-Serono, Amgen, and Sanofi-Genzyme. Dr Rempe has contract research support from Sanofi-Genzyme, Novartis, Celgene, EMD Serono, and Genentech.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.

