Abstract
Tumefactive demyelinating lesions (TDLs) are rare in older populations. In this scoping review we conducted a PUBMED search for published cases of TDLs in patients aged ≥55 years. We identified 31 cases and report data on their presentation, diagnosis, treatment, and outcomes. Motor symptoms were the most common presenting symptoms. Investigations including magnetic resonance imaging (MRI) of the brain and spinal cord, CSF studies, serum autoantibodies and brain positron emission tomography (PET) were used to differentiate TDLs from key differentials including malignancy. Key diagnostic findings were similar to those in younger patients with TDLs, with a slightly higher occurrence of open ring enhancement on MRI (11 cases, 35% in present study; 21% in non-age specified populations), and a lower frequency of unmatched oligoclonal bands (6 cases, 19%; 62% in non-age specified populations). The median time to diagnosis was 30 days with 68% (21 cases) of cases requiring biopsy. Initial treatment involved high dose corticosteroids in 90% of patients (29 cases), with 26% (8 cases) started on an MS disease modifying therapy after the index attack. The median Expanded Disability Status Scale score at presentation was 3 and at last follow up was 2.5 indicating potential for limited recovery from TDL in older patients.
Keywords
Introduction
Tumefactive demyelinating lesions (TDLs) are large demyelinating lesions in the brain which commonly resemble neoplastic lesions on neuroimaging.1,2 Multiple sclerosis (MS) is the most common underlying cause, but TDLs may occur as a clinically isolated syndrome or a radiologically isolated syndrome, as a paraneoplastic phenomenon, or as part of another inflammatory demyelinating disease such as acute disseminated encephalomyelitis (ADEM), neuromyelitis optica spectrum disorder, or myelin oligodendrocyte glycoprotein antibody-associated disease (MOGAD).2–4
TDLs can present in individuals with or without previous demyelinating disease and, similar to MS, occur more commonly in females. 1 The clinical presentation of TDLs can resemble a typical MS relapse. Depending on the location of the lesion in the brain, there may be focal weakness, sensory disturbance or ataxia. A TDL may also present with symptoms and signs atypical for MS-related demyelination, such as headache, seizures, aphasia or confusion – features that reflect the larger size of the lesion, and which are more likely if the lesion is accompanied by oedema and mass effect. 2
Diagnostic uncertainty about TDLs can lead to significant unease in patients and clinicians and result in unnecessary invasive investigations such as brain biopsy or resection.2,5 Ancillary testing including MRI of the spine, CT brain, CSF white cell count and oligoclonal bands, and FDG-PET study may provide clues that suggest a demyelinating lesion over a neoplasm, while serological testing for MOG IgG or aquaporin-4 IgG can help to identify non-MS causes of TDLs. 2
TDLs occur most commonly in the fourth and fifth decade of life with a mean age of onset of 38.5 +/-15 years. 6 Therefore, when a TDL presents in an older patient, it is often not even considered as a diagnostic possibility, yet TDLs can occur late in life. 7 We present 31 cases of TDL in adults aged ≥55 years taken from 27 reports in the literature. The aim of this study is to better understand the demographics, presentation, investigation findings, treatment, and outcomes of patients aged ≥55 with TDLs and to promote awareness of TDLs in older patients.
Methods
Study design and ethics
We conducted a retrospective study of published cases identified in PubMed following PRISMA scoping review guidelines including 4 cases recently published by the authors. 7 The data contained within the studies were already publicly available and anonymised.
Information sources
We searched PubMed with the search terms “tumefactive multiple sclerosis” OR “tumefactive demyelinating lesion” OR “pseudotumoral multiple sclerosis” OR “pseudotumoral demyelinating lesion” OR “tumefactive demyelination” OR “pseudotumoral demyelination” AND “case report” or “case series” OR “case study” from database inception until August 2024, limiting the results to studies available in English. The search results yielded 412 studies. Search results were imported into Covidence (Veritas Health Innovation, Melbourne, Australia), an online software for managing systematic reviews. Each abstract was screened by one reviewer against pre-specified inclusion and exclusion criteria. Forty-three full texts were subsequently screened, and 26 studies identified (Figure 1). One further study known to the reviewers but not yet published was added.
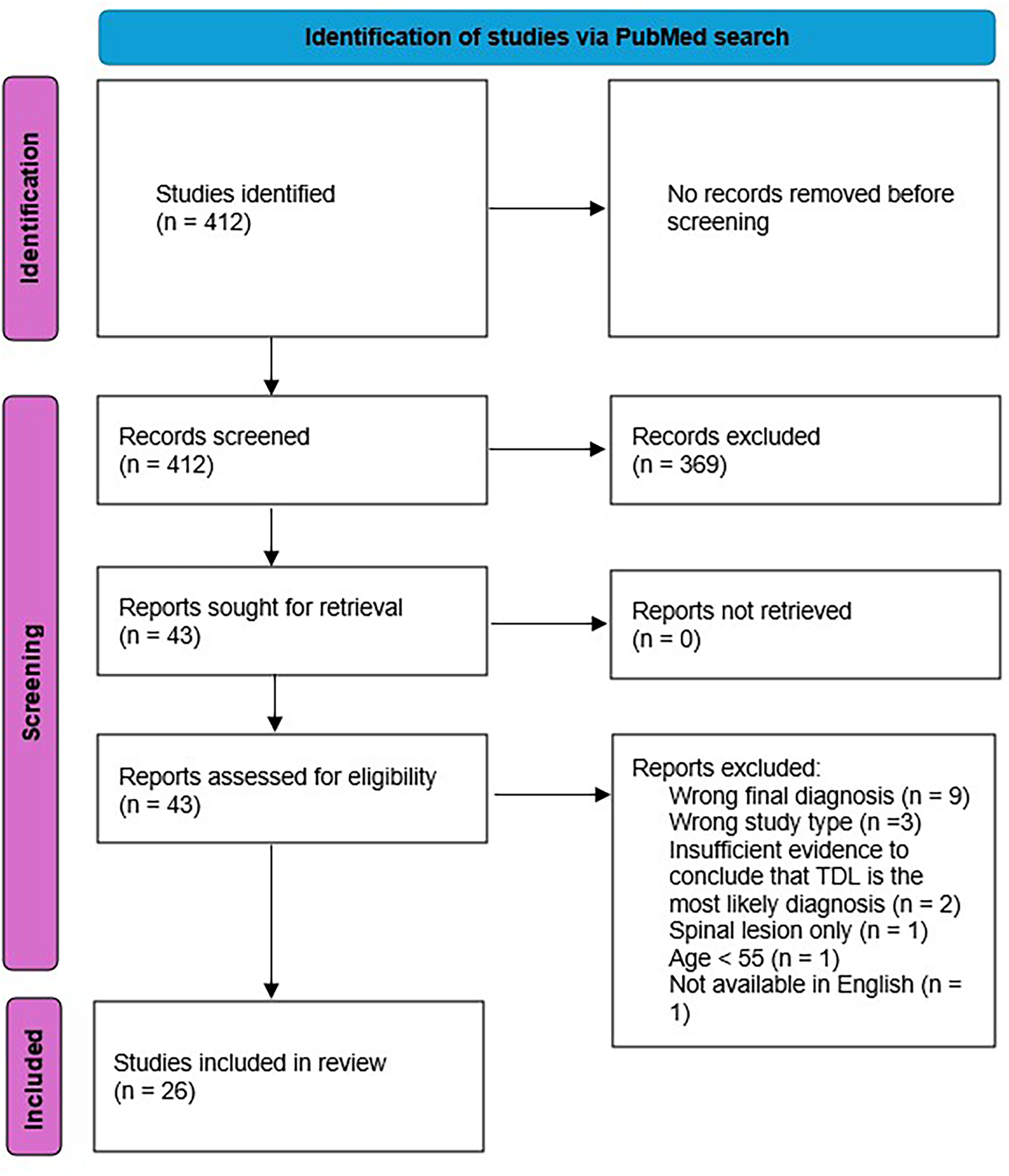
Study selection flowchart.
Inclusion and exclusion criteria
Inclusion criteria
Case study/case report/case series Adult patient aged 55 years or older Final diagnosis of tumefactive demyelinating lesion(s) in the brain Full text available English language study
Exclusion criteria
Articles in which case details were not presented e.g., narrative review articles Patient aged less than 55 years Large demyelinating lesion only present in spinal cord Final diagnosis of Balo's concentric sclerosis, neuromyelitis optica spectrum disorder, myelin oligodendrocyte glycoprotein antibody-associated disease Full text not available (including posters, abstracts) Text not available in English Insufficient evidence presented in the text to conclude that the TDL is the most likely diagnosis
Data extraction
The extracted data from these cases for analysis included demographics, clinical presentation, diagnosis, MRI lesion appearance and other investigation results, treatment and outcomes. The initial and final Expanded Disability Status Scale (EDSS) score was estimated by the authors using the information provided in each case.
Statistical analysis
Continuous data was calculated as mean, median and range. Categorical data was expressed as a proportion of the total number of patients.
Results
The data from individual cases is recorded in Table 1 (presentation and diagnosis), Table 3 (Investigations), and Table 4 (treatment and outcome).
Patient demographics and initial diagnosis.

CNS: central nervous system; CSF: cerebrospinal fluid; EDSS: expanded disability status scale; IV: intravenous; IVIg: intravenous immunoglobulin; MRI: magnetic resonance imaging; mRNA: messenger ribonucleic acid; MS: multiple sclerosis; PET: positron emission tomography; T2DM: type 2 diabetes mellitus; TD: tumefactive demyelination; TDL: tumefactive demyelinating lesion; WCC: white cell count
Patient demographics
Thirty-one cases were identified from a total of 27 reports. The median age at presentation was 62 years (range 55–87 years). Fifteen patients (48%) were female. Two patients (6%) had a pre-existing diagnosis of MS. Five cases (16%) noted vaccination in the preceding three months. Four cases (13%) had underlying or concomitant active malignancy – seminoma, renal cell carcinoma, retroperitoneal germ cell tumour, and glioma (Table 1, cases 1, 6, 11, and 20 respectively). The patient demographics are summarised in Table 2.
Patient demographics.

Presentation
The most common presenting signs and symptoms were motor (17 cases, 55%), aphasia (10, 32%), cognitive (8, 26%) and cerebellar (8, 26%) (Table 1; Figure 2.A). In 16 cases (52%), the initial differential diagnosis included malignancy, and in 10 cases (32%) the initial diagnosis included demyelination. In the 19 studies that specified the time from presentation to diagnosis the median time to diagnosis was 30 days (SD = 24.5 days), with 10 (32%) of cases requiring >1 month to arrive at a diagnosis (Figure 2.B).

(A) Presenting symptoms; (B) time from presentation to diagnosis.
MRI findings
The most affected brain areas seen on MRI were the parietal (13 lesions, 42%) and frontal lobes (12 lesions, 35%) (Figure 3.A). Enhancement with intravenous gadolinium contrast was seen in 28 lesions (90%) with 13 of these (46%) noting ring enhancement, 11 of which specified open or incomplete ring enhancement (39%). The median lesion size in the 15 cases where dimensions were reported was 33mm (SD = 4.7, range 14–70). Other lesions were present in 10 cases (32%). MRI spine at presentation was reported in 9 cases (29%), 4 of which identified a spinal lesion (13% of all patients).

(A) Lesion location on MRI; (B) oligoclonal bands of IgG in CSF analysis.
CSF findings
CSF results were included in 30 cases (97%), with 21 of these negative for OCBs (68%), 6 were positive and unmatched in the serum and CSF (19%) and 3 were positive and matched (10%) (Figure 3.B). Protein was elevated in 17 of the 26 studies in which it was reported (65%). In the 18 studies which reported the CSF protein value, the median was elevated at 695 mg/L (SD = 89.8 mg/L, range 210–1960 mg/L). WCC was elevated in 11 of the 23 (46%) studies in which it was reported. (Using New South Wales Health reference range 150–450 for protein, <1 for WCC). Two studies specified CSF lymphocyte predominance (Table 3, cases 6 and 15) and one specified neutrophil predominance (Table 3, case 18). Where a value was reported, the median WCC was 0 /mm3 (SD 32.5, range 0–718 /mm3); the median WCC among studies in which WCs were elevated at 1/mm3 or more was 1.5 /mm3 (SD = 51.0, range 0–718).
Investigations.

Key: ADEM: acute disseminated encephalomyelitis; CD: cluster of differentiation; CNS: central nervous system; CSF: cerebrospinal fluid; EDSS: expanded disability status scale; GFAP: glial fibrillary acidic protein; IV: intravenous; IVIg: intravenous immunoglobulin; MRI: magnetic resonance imaging; MS: multiple sclerosis; PET: positron emission tomography; TD: tumefactive demyelination; TDL: tumefactive demyelinating lesion; WCC: white cell count
Other testing
Investigation for serum MOG IgG and aquaporin 4 IgG were tested in 11 and 15 cases respectively (35%, 43%), all of which were negative. Brain PET was included in 9 cases (29%), 4 of which showed a hypometabolic lesion (44%), 2 showed a hypermetabolic lesion (22%), and 3 were normal (33%). The lesion was biopsied in 21 cases (68%).
Initial treatment
Corticosteroids were the most common initial treatment, utilised in 28 cases (90%), with 18 cases (58%) specifying the use of high dose intravenous corticosteroids. The lesion was resected in 4 cases (13%).
Longer term treatment and follow up
The median length of available follow up was 12.5 months. Eight patients (26%) experienced further demyelinating lesions at last follow up., 5 of these were symptomatic (63%), 2 were identified on follow up MRI (25%), and 1 was not specified (13%). The median duration of follow up in patients who had further lesions was 16 months (SD = 2.6 months, range 0–48 months). Twelve patients (39%) received long-term immunosuppression of some description (Table 4); eight of which were started after the first episode (25%), three after a second episode (10%), and one was pre-existing (3%) for another indication (rheumatoid arthritis) with 8 of these (26%) started on disease modifying MS therapy (DMT). The median initial EDSS was 3.0 (SD = 0.34, range 0–6.5); nineteen patients (61%) demonstrated an improved EDSS at last available follow up; six patients (19%) demonstrated a worsened EDSS, and three (10%) demonstrated no change. Where estimates were available, the median change in EDSS from presentation to last follow up was an improvement to an EDSS of 1.0 (SD = 0.48, range 0–11.5).
Discussion
We present data collected from 31 published cases of TDLs in patients aged ≥55 years. In our study, we saw slightly more male than female cases which differs from the slight female predominance seen in non-age specified tumefactive demyelination, but is in keeping with studies that have identified a less striking female predominance of older patients presenting with late onset MS (LOMS) compared to early onset MS. 8 There appeared to be a lower index of suspicion for TDLs in older patients, with only 32% of the initial differential diagnoses including demyelination, compared to 70% among patients of all ages. 6 Again, this is mirrored in cases of LOMS, where similar delays in diagnosis have been recognised. 9 Likely as a result, the frequency of patients undergoing brain biopsy was 68% among our cases which is significantly higher than the non-age specified rate of 30%.
There were minor differences in the investigation results in our cohort of older patients compared to 257 non-age specified TDL cases 6 (Table 5). Cerebral MRI findings were similar across both groups, with frontal and parietal lesions comprising the largest proportion of cases. Incomplete open ring enhancement; one of the most specific radiological signs for TDLs, was present in 35% of cases – higher than that of non-age specified cases (21%). 10 CSF-restricted oligoclonal bands were positive less frequently in the aged cohort (19% compared to 62% in a mixed aged cohort). CSF protein was more commonly elevated in older patients, although it is worth noting that CSF protein increases with age anyway. 11 We note that these findings have not been demonstrated in comparisons between LOMS and early onset MS (EOMS) patients, though this may be partially attributable to the exclusion of clinically isolated syndromes. 9 Brain PET was infrequently utilised. When used, 78.4% of cases showed a normal or hypometabolic lesion which is more typical of a TDL than a high-grade CNS malignancy. 12
Treatment and outcome.

CNS: central nervous system; CSF: cerebrospinal fluid; EDSS: expanded disability status scale: IV: intravenous; IVIg: intravenous immunoglobulin; MRI: magnetic resonance imaging; MS: multiple sclerosis; PET: positron emission tomography; TD: tumefactive demyelination; TDL: tumefactive demyelinating lesion; WCC: white cell count
We suggest that the use of ancillary testing may provide opportunities to avoid invasive brain biopsy and the associated risks in older patients. 13 For example, in a patient with a solitary cerebral lesion suspicious for a TDL, and no prior history of MS, MRI of the spine to screen for demyelinating lesions, CSF examination for intrathecal oligoclonal bands, and FDG-PET imaging to help exclude high grade malignancy can provide clues as to whether a lesion is more likely to be a TDL or a malignancy. MR perfusion showing reduced cerebral blood volume within the lesion also favours a TDL over high grade neoplasm and should be considered in cases of diagnostic uncertainty. 14 Serum autoantibody screening, such as MOG IgG and aquaporin-4 IgG, can be used to diagnose MOGAD or NMOSD respectively. 2 Less than 50% of the presented cases included results for MOGAD or NMSOD testing respectively, which may also reflect that several of these reports predate the use of these screening assays. This represents a potential source of bias in these results.
Comparison of results in present study and a non-aged-specified cohort of TD cases.

CSF: cerebrospinal fluid; EDSS: expanded disability status scale score; MRI: magnetic resonance imaging; OCBs: oligoclonal bands; TD: tumefactive demyelination; WCC: white cell count
The most common initial treatment in our study was corticosteroids which were administered in 90% of cases. This is in keeping with recommendations of three-days of intravenous methylprednisolone followed by repeat imaging in 4–6 weeks to assess response in those cases where a TDL is difficult to exclude from a glioma.2,15,16 This approach is better avoided where the differential diagnosis lies between TDL and CNS lymphoma as corticosteroids before a diagnosis of CNS lymphoma is established can alter histopathology and lead to false reassurance that a biopsied lesion is not due to lymphoma. We also recommend close clinical and MRI follow-up of biopsy proven cases of TDL as cases of CNS lymphoma arising from sentinel demyelination have been described. 17 Cases of recurrent or relapsing TDLs should invoke a widespread search for an underlying malignancy. 18 In patients with a pre-existing diagnosis of malignancy, with the potential for metastasis to the brain, we would advocate a lower threshold for proceeding to biopsy.
The low rate of prescribing of DMTs might reflect that older patients with MS are less likely to have inflammatory disease activity requiring immune intervention,19,20 but also less frequently meet the diagnostic criteria for MS. Additionally, older patients may be more vulnerable to the adverse effects of these therapies and this may influence clinical decision making. 21 Almost two-thirds of cases had improvements in EDSS following their clinical nadir which indicates that older patients can make some degree of recovery from TDLs despite more limited brain reserve. We report radiological relapse in 26% of patients, as compared to 50% in a non-age specified cohort. 22 Relapse has been identified as more likely in patients with unmatched OCBs, which may be of significance given the low rates of both unmatched OCBs and relapse in the presented cases. 22 We also note that all of the eight cases with a further MS lesion at last follow up had negative OCBs at their index attack which suggests that OCBs are not a predictor of future relapse in this cohort.
There are several limitations to this study. Only a small number of published cases could be included. Additionally, case reports often contained incomplete datasets and details such as recent vaccination or detailed serological screening were missing. Data points such as time to diagnosis and EDSS were estimated based on the information provided. Whilst this discussion has compared our findings to that of a non-age specified cohort, this comparison is qualitative only and no statistical analysis has been completed.
This review highlights TDLs as a differential diagnosis in older people with a cerebral lesion, noting a broadly similar presentation and investigation findings to that of TDLs in younger people except for a lower frequency of intrathecal OCB synthesis and a higher proportion of open ring enhancing lesions. We suggest that appropriate use of neuroimaging and ancillary testing may help to avoid misdiagnosis and unnecessary biopsy in some patients.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Carla De Pasquale has no disclosures. Todd Hardy has received speaker honoraria, conference support and Scientific Advisory Board fees from Bayer-Schering, Bristol Myers Squibb, Biogen-Idec, Merck, Novartis, Roche, UCB, Sanofi-Genzyme, Alexion, Sandoz and Teva. There has been no prior presentation of this research.
Data availability
Data from this study is available upon reasonable request to the corresponding author.
Ethical considerations
Ethical approval was not required for this scoping review.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
