Abstract
Objectives and aims
Ocrelizumab (OCR) is disease-modifying therapy that depletes B cells for the treatment of patients with relapsing and progressive multiple sclerosis (MS). The aim of this study was to evaluate occurrence of hypogammaglobulinemia and infection rates in patients with MS treated with OCR for up to 5.9 years in a real-world, single-center study.
Methods
We assessed immunoglobulins IgG, IgM, IgA; CD19, CD4, CD8 cell counts, anti-JCV antibodies, at baseline and every six months prior to each OCR infusion. Mixed effects regression models controlling for random patient intercept were used to analyze immunological trends. Infections requiring oral or intravenous antibiotics were also recorded and analyzed.
Results
A total of 238 patients were followed for up to 5.9 years and received a mean of 7.6 (range, 2-13) OCR infusions. IgG declined on average by 3.7% (p < .001) and IgM by 11.8% (p < .001) annually. At last follow-up, 14.6% of patients had IgG < 600 mg/dL and 51.1% had IgM M < 40 mg/dL. Patients with IgG < 600 mg/dL experienced higher rates of infections requiring oral antibiotics (1.17 vs 0.99 per year, p = .004) and IV antibiotics (0.147 vs 0.066 per year, p = .009) compared to those with normal IgG. White race was associated with reduced IgG (p = .008) and IgA (p < .001) levels, while female sex was associated with increased CD4 (p < .001) and CD8 (p = .015) counts. Anti-JCV antibody index declined by 1.9% annually (p < .001). Linear trend analysis predicted that 14.9 years of treatment would be required for at least 50% of patients to have IgG levels <600 mg/dL.
Conclusions
Continuous OCR treatment results in increasing hypogammaglobulinemia associated with increasing risk of moderate and severe infections. The clinical significance of these immunological findings and the influences of demographic characteristics highlight the importance of regular, personalized immunoglobulin monitoring during long-term OCR therapy.
Keywords
Introduction
Treatment for multiple sclerosis (MS) has evolved significantly with the introduction of highly effective disease-modifying therapies.1–3 Ocrelizumab (OCR), a humanized anti-CD20 monoclonal antibody, is a well-tolerated and highly effective treatment option for both relapsing MS and primary progressive MS (PPMS). 4 As the first approved therapy for PPMS, OCR has become one of the most prescribed disease-modifying therapies for MS in the United States. 5
OCR achieves clinical efficacy through B-cell depletion, which can also lead to decreased antibody production. 6 Thus, in addition to its well-established efficacy, a common and clinically significant side effect is infections secondary to immunosuppression.7–9 Data from seven-year analyses of the OPERA and ORATORIO trials show that OCR leads to a sustained decrease in circulating immunoglobulins (hypogammaglobulinemia), which correlates with infection risk.8,9
For patients on OCR, there's a small but real risk of developing progressive multifocal leukoencephalopathy (PML) especially among those who have been treated with DMTs linked to PML. As of June 2024, there have been 16 confirmed cases of PML in more than 350,000 patients treated with OCR globally. Of these, 12 were carry-over cases attributed to a prior DMT (Genentech, data on file).10–13 When we began our study in 2018, just a few months after the drug was approved, the incidence of PML related to OCR in a real-world setting was unknown, so we decided to measure and keep a track of anti-JCV antibodies and to determine if B cell-depleting therapy would affect the index and whether we could rely on that level as we do in natalizumab-treated patients for PML risk. Monitoring immunoglobulin levels throughout treatment is important, as their decline may influence treatment decisions, particularly in patients starting with lower baseline levels. 8 Guidelines regarding the duration of safe continuous treatment or validated markers for optimal monitoring of patients treated with OCR have yet to be established.
Real-world evidence is crucial for understanding the long-term safety profile of OCR and establishing monitoring guidelines. Real-world data consistently suggest anti-CD20 therapies lead to hypogammaglobulinemia that worsens over time, particularly decreased levels of IgM and IgG; however, the association with serious infections is not yet well-defined.14–17 Considering the chronic nature of MS and the need for ongoing therapy, long-term assessments into the association between hypogammaglobulinemia and infection risk data are needed. Furthermore, the influence of patient characteristics on outcomes, such as the potential association between BMI and the duration of treatment efficacy, require further investigation.18–20
In this study, we examined a real-world cohort of patients with MS who were treated with OCR at our center for up to 5.9 years. The primary aim of was to identify the incidence and risk factors for hypogammaglobulinemia and moderate to serious infections.
Methods
Study population
The study population consisted of all the MS patients at our center who had received at least one infusion of OCR. The first infusion was divided into two 300 mg intravenous doses two weeks apart, and the subsequent doses were 600 mg per six months. Quantitative immunoglobulins, CD19, CD4 and CD8 counts and JCV antibody levels were obtained at baseline and then every six months prior to each subsequent OCR infusion. In addition to obtaining standard baseline clinical characteristics, BMI was also calculated for all at baseline. Based on a previous study, infections were recorded as moderate (requiring oral antibiotics) or severe (requiring IV antibiotics). 21
Hypogammaglobulinemia was defined as IgG < 600 mg/dL and IgM < 40 mg/dL. This was based on the previously established normal ranges (IgG 700–1600 mg/dL and IgM 40–230 mg/dL 22 ) and the definition of hypogammaglobulinemia from. 23
Approval of this study was obtained from our local institutional research review board.
Statistical analysis
Distributions of continuous variables were reported by sample means and standard deviations, as well as by 0.25, 0.5, and 0.75 level quantiles. Distributions of categorical variables were summarized by relative frequencies and category-specific counts. Fisher's exact test and Wilcoxon–Mann–Whitney tests were used for comparing distributions of categorical and continuous variables between groups of patients (Tables 1 and 2). COVID-19 pandemic occurred two years into our study, and we decided to collect vaccination data against COVID-19, especially since they were on B cell-depleting therapy. Due to prevailing health concerns regarding COVID-19, we tested the association between death and confirmed vaccination status with Fisher's exact test. Mixed effects regressions were used to model linear over-time trends of IgG, IgM, IgA, JCV, CD4, and CD8. These models controlled for random patient intercept and always included baseline values of outcomes. To reduce the effect of skewness all observed values of IgG, IgM, IgA, JCV, CD4, and CD8 including their baseline values were transformed with natural logarithm. Normality was assessed with Shapiro–Wilk tests, histograms, and quantile-quantile plots. While means differ between original and log-transformed scales, medians remain equivalent after transformation; therefore, results are presented as changes in medians on the original scale to maintain clinical interpretability. All regression analyses were adjusted for age, sex, white race, duration of disease, Expanded Disability Status Scale (EDSS), race and BMI when significant. The log-transformed baseline values were always included in the set of covariates. CD19 were dichotomized into above and below specific threshold values and tabulated by year and BMI categories. Infections, which needed to be treated with either oral or intravenous antibiotics, were also recorded and analyzed. A traditional cutoff (p < .05) was used to declare statistical significance.
Clinical characteristics and outcomes among patients stratified by IgG level <600 b vs ≥600 mg/dL.

Note: BMI: body mass index; EDSS: Expanded Disability Status Scale; IgG: immunoglobulin G; IV: intravenous; UTI: urinary tract infection.
Calculated as number of infections per year per patient.
The group of patients with IgG < 600 was defined by patient with at least one observed IgG value below 600 at any time point.
Data are summarized by sample means ± standard deviation [median, range].
Clinical characteristics and outcomes among patients stratified by IgM levels <40 vs ≥40 mg/dL.
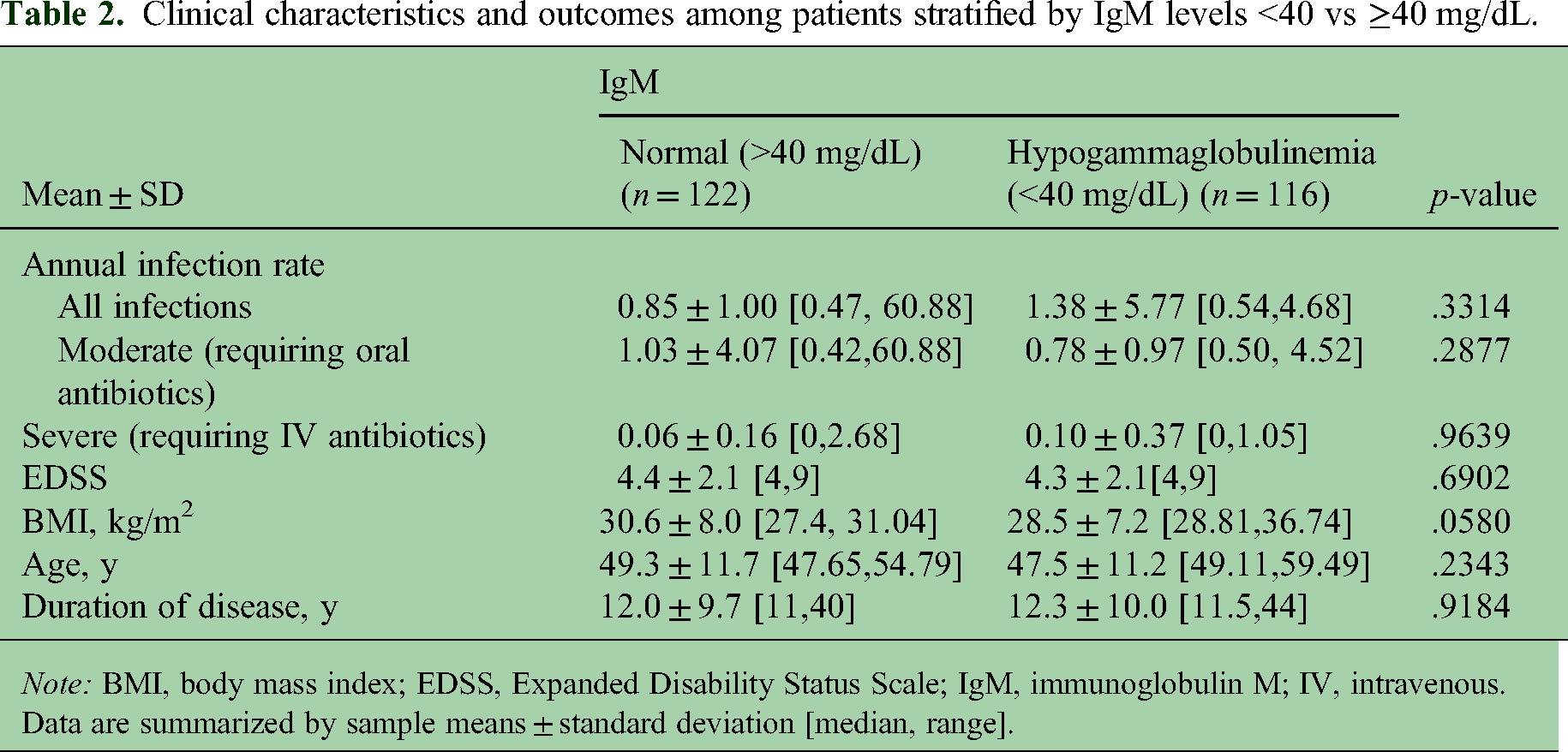
Note: BMI, body mass index; EDSS, Expanded Disability Status Scale; IgM, immunoglobulin M; IV, intravenous.
Data are summarized by sample means ± standard deviation [median, range].
R software was used for all data management and data analyses. 24
Results
Patient demographics
From an initial dataset of 244 patients, six were excluded due to no follow-up or no baseline blood tests, resulting in an analysis set of 238 patients. The mean (±SD) patient age at baseline was 48.4 ± 11.5 years (range, 18.9–78.4) (Table 3). The mean duration of disease was 12.2 ± 9.8 years, mean EDSS score was 4.4 ± 2.1, and mean patient BMI was 29.5 ± 7.6. Patients were followed for a mean duration of 3.3 years (range, 0–5.9). The average duration of follow-up was 3.3 years per patient. The mean patient age at the last visit was 51.7 ± 11.3 years. Nine patients died during this study. The causes of death: two due to COVID-19 complications; two related to cardiac events; one due to motor vehicle accident; one acute renal failure and drug overdose; one end-stage MS and respiratory failure; one pancreatic cancer; one unknown.
Baseline characteristics among all patients.

Note: BMI: body mass index; EDSS: Expanded Disability Status Scale; Ig: immunoglobulin.
A comparison of patient baseline characteristics between those who survived and those who died is summarized in Table 4.
Baseline characteristics among patients who survived or died during the study.

Note: BMI: body mass index; EDSS: Expanded Disability Status Scale; Ig: immunoglobulin.
Infection rates by IgG level
We compared patient baseline characteristics and outcomes based on whether or not patients had hypogammaglobulinemia, defined as either having IgG levels <600 mg/dL or IgM levels <40 mg/dL. Among patients with IgG data at baseline (n = 214) the mean (±SD) IgG level was 960.6 ± 259.6 mg/dL and 9.3% patients had IgG < 600 mg/dL. Of these 20 patients who had IgG < 600 at baseline, nine of them were on prior immunosuppressants/immunodepleting therapies, and they were older with a mean age of 55.4 (SD = 11.2), while the mean age of patients with the baseline IgG ≥ 600 was 48.9 (SD = 11.0).
At the last follow-up the mean IgG level among 235 patients with available data was 857.0 ± 259.5 mg/dL with 14.6% of patients having IgG < 600 mg/dL. Patients with IgG < 600 mg/dL had a significantly increased annual infection rate compared with those with IgG ≥ 600 mg/dL (1.32 ± 1.63 vs 1.06 ± 4.50; p < .001) (Table 1). This included both rates of infections requiring oral antibiotics (1.17 ± 1.61 vs 0.99 ± 4.48, p = .004) and IV antibiotics (0.15 ± 0.29 vs 0.07 ± 0.28; p = .009). Of the clinical baseline characteristics examined, severity of disability (based on EDSS), race, age, and BMI were identified as potential risk factors associated with hypogammaglobulinemia and infection rates and were included as covariates in regression modeling when significant (see below).
Mean IgM at baseline (n = 213) was 94.4 ± 59.9 mg/dL, with 10.8% patients having values <40 mg/dL. At the last follow-up, among the 235 patients with available data, mean IgM was 51.1 ± 38.4 mg/dL with 51.1% of patients having IgM < 40 mg/dL. As shown in Table 2, there were no significant differences in infection rates or other risk factors between IgM < 40 and IgM ≥ 40 groups; however, there was a trend suggesting a possible association with BMI (p = .0580). None of the patients dropped their IgG below 200 mg/dL.
We note that 37.5% of those who died were vaccinated whereas 78.3% of those who survived had been. Due to a small number of deaths (n = 9), we limited our analyses to summary statistics without any claims on statistical significance. Due to a small number of deaths (n = 9), we limited our analyses to summary statistics without any claims on statistical significance. Analysis of COVID-19 confirmed vaccinations showed that vaccinated patients had significantly lower odds of death compared to other patients (Odds ratio 0.148, 95% CI [0.023 to 0.723]; p = .008). We did not collect information on any other vaccinations.
Regression analyses
Annual rates of decline in immunoglobulins were 3.7% for IgG (p < .001) (Supplemental Table S1), 11.8% for IgM (p < .001) (Supplemental Table S2), and 3.4% for IgA (p < .0001) (Supplemental Table S3). At the last follow-up, mean IgG values were <600 mg/dL in 14.6% of patients and mean IgM values were <40 mg/dL in 51.1% of patients. Based on the linear trend in IgG levels, we predict a median of 14.9 years of treatment with OCR would be required to reduce IgG to <600 mg/dL in more than 50% percent of patients (Figure 1A).

Scatterplots of (A) IgG levels and (B) JCV Index scores show observations in all patients. Black lines draw linear trends of four randomly selected patients; blue lines connect observed values of the same four patients. The green trendlines are predicted population trends. The IgG trend (panel A) predicts that it takes 14.9 years of treatment with ocrelizumab to reduce IgG to <600 mg/dL in 50% of patients. The overall trend for JCV Index shows a significant annual decline of 1.9% (p < .001).
Annual rates of T-cell count increases were 3.7% for CD4 + cells (p < .001) (Supplemental Table S4) and 3.4% for CD8+ cells (p < .001) (Supplemental Table S5). Our analysis also revealed a significant 1.9% annual decline in anti-JCV antibody index over time (p < .001) (Supplemental Table S6 and Figure 1B).
Regression analysis supported our observations suggesting influence of demographic characteristics on immunological outcomes. Specifically, white race was associated with a significant reduction of IgG (p = .008) (Supplemental Table S1) and IgA (p < .001) (Supplemental Table S3). Female sex was associated with increased CD4 counts (p < .001) (Supplemental Table S4) and CD8 (p = .015) counts (Supplemental Table S5).
BMI and CD19 levels
Since we identified BMI as a potential risk factor for hypogammaglobulinemia and infection, and prior evidence suggests a potential association between BMI and a “wearing off” of OCR treatment effect mediated by CD19 B-cell counts, 20 we investigated whether there was an association between BMI and CD19 counts in our study population. As shown in Supplemental Table S8, we observed a trend suggesting that patients with BMI ≥30 kg/m2 were less likely to have reduced CD19 counts compared with those with BMI <30 kg/m2. CD19 cells decreased to <10 cells/µL in 87.0% of patients at one year of follow-up then remained stable at approximately 90% in consecutive years.
Discussion
In this real-world prospective analysis of OCR-treated patients with MS followed for up to 5.9 years, we found statistically significant and progressive development of hypogammaglobulinemia that was associated with increased infection risk over time. By the last follow-up, 14.6% of patients had IgG levels below 600 mg/dL and 51.1% had IgM levels below 40 mg/dL, representing significant declines from baseline. Notably, patients with IgG < 600 mg/dL experienced significantly higher rates of both moderate and severe infections than those with IgG ≥ 600 mg/dL. In patients with low IgG serum levels, respiratory infections are more common than urinary tract infections (UTIs). However, in our study, we encountered more UTIs than respiratory infections. We do not have a good explanation for this finding, but certain IgG subclasses are linked to increased susceptibility to recurrent respiratory and UTIs. Studies Beattie-Spanjol et al. 25 and Barton et al. 26 show that low levels of subclass IgG2 can be associated with UTIs and decreased IgG1 with an increased risk of recurrent respiratory infections. Reduced levels of IgG1 and IgG2, particularly when isolated, can significantly increase susceptibility to various infections, especially those affecting the respiratory and sinus systems. IgG2 is particularly important in combating encapsulated bacteria, and its deficiency can lead to recurrent infections such as pneumonia, otitis media, and sinusitis. IgG1 also plays a role in the immune response to UTIs, and secretory IgA (sIgA) is directly involved in mucosal defense. Some evidence suggests that recurrent UTIs may be associated with defective sIgA excretion, underscoring the importance of a robust humoral immune response in preventing such infections. 27 We did not measure the subclasses of IgG or sIgA in this study; however, future investigations incorporating these parameters may help explain the higher incidence of UTIs among IgG-deficient patients compared with respiratory infections.
Furthermore, innate immunity plays a more dominant role in defending against infection than IgG. OCR can affect the innate immune system. Study 26 suggests that OCR treatment can induce inflammatory toll-like receptor (TLR) signaling pathways in innate immune cells, like monocytes, macrophages, dendritic cells, and NK cells and leads to increased expression of proinflammatory TLR genes (TLR2, TLR4, TLR5, and TLR8) and decreased expression of antiinflammatory genes (TRAF4 and TLR10). None of our patients reported any symptoms suggestive of GI infections. Viral infections were mild and did not require any treatments and were not included in this study. No opportunistic or fungal infections were reported. We also identified several patient characteristics associated with immunological outcomes, including race, sex, age, use of prior immunodepleting drugs and potentially BMI. Overall, these findings provide important insights into the long-term safety profile of OCR and highlight the need for careful monitoring of immunoglobulin levels during treatment.
Our results are consistent with previous findings showing an increasing risk of hypogammaglobulinemia over time with ongoing anti-CD20 therapy. For example, an integrated safety analysis of 11 clinical trials with up to seven years of follow-up, which demonstrated average annual decreases of 0.3 g/L in IgG and 0.8 g/L in IgM. 8 Real-world studies have also consistently found that anti-CD20 therapies lead to hypogammaglobulinemia that worsens over time, particularly affecting IgM and IgG levels, though the association with serious infections has varied across studies.14–17 Our modeling predictions suggest that half of patients would develop IgG levels below 600 mg/dL within 15 years of continuous OCR treatment, potentially placing them at increasing risk for infections over time. This has important implications for clinical decision making for patients who require long-term therapy and may experience substantial relief from MS symptomatology with OCR treatment.
The relationship we observed between hypogammaglobulinemia and infection risk adds to a complex body of evidence on the safety of anti-CD20 therapies. Based on our results, an IgG 600 mg/dL represents an important threshold for the risk of moderate and severe infections, the rates of which were both significantly increased for patients above vs below this level. Prior real-world studies on patients treated with OCR have shown mixed results regarding immunoglobulin levels and infection risk. Notably, one study found that higher serum IgG levels were associated with reduced odds of infection, 28 whereas a separate study found no correlation between serum IgM or IgG levels and risk of infection. 29 However, it is important to note that both of those studies had relatively short follow-up periods of up to approximately 2.5 years. A more recent analysis with longer follow-up found that although IgG levels generally decreased over 12 years of treatment, more than 80% of patients maintained normal IgG levels with transient periods of low IgG. 17 Importantly, decreased IgG levels were not independently associated with serious infections in multivariate analyses, concluding that OCR is associated with a manageable infection risk profile overall. 17 These mixed results highlight the importance of long-term assessment and effective study design.
In our demographics assessment we found that white race was significantly associated with reduced IgG and IgA levels, in agreement with previous findings showing a link between white race and increased risk of hypogammaglobulinemia during B-cell therapy. 30 Our observation that female sex was significantly associated with increases in CD4 and CD8 cell counts over time is interesting. However, the CD4 to CD8 ratio is far more important as it has been shown to correlate with infection rates. 31
Thus, we also explored the time trend of CD4/CD8 ratio (Supplemental Table S7), which was not significantly different between males and females. A relationship between BMI and CD19 kinetics suggests that body mass may influence the pharmacodynamics of OCR. 23 Supplemental Table S8 suggests that patients with BMI ≥30 kg/m² were less likely to have reduced CD19 counts than those with lower BMI. This is consistent with the finding that patients with higher BMI show fast repopulation of B cells. 32 The clinical significance of BMI is further heightened by the observation that high BMI, irrespective of B-cell count, is a significant predictor of wearing-off phenomenon. This phenomenon is observed in more than half of patients on OCR. 20
A unique aspect of our study was the longitudinal monitoring of JCV antibody index, which showed a statistically significant annual decline of 1.9%. Notably, no patients in our study converted from seropositive to seronegative or vice versa during follow-up, despite reported annual JCV seroconversion rates of 2–15% in MS patients not treated with natalizumab.32,33 Our study, therefore, concluded that the index cannot be relied upon, and this is very crucial for previously treated natalizumab patients who are JCV seropositive and now transitioning to OCR.
This observation raises important considerations for PML risk assessment in OCR-treated patients. The JCV antibody index was developed and validated for monitoring patients during treatment with natalizumab, 34 and our findings suggest that B-cell depletion may potentially limit the use of this biomarker by reducing antibody production, leading to false negative results in JCV antibody testing. This has important implications for PML risk stratification, especially for seropositive natalizumab patients transitioning to OCR therapy.
The results of our study have several important clinical implications. First, they emphasize the need for regular monitoring of immunoglobulin levels throughout OCR treatment, particularly given the cumulative nature of hypogammaglobulinemia risk. Second, they suggest that certain patient characteristics, including race, sex, age, prior immunodepleting therapies, and BMI, may influence immunological responses to treatment and should be considered in individualized monitoring strategies. Finally, they highlight the complexity of using traditional risk biomarkers like JCV antibody index in the context of B-cell depleting therapy.
Despite the strengths of our study, which included a sizable patient population and duration of follow-up, there were some limitations. First, although our follow-up period of up to 5.9 years is substantial, given the extended time scale for effective risk stratification revealed in our modeling, longer-term data will be needed to validate our projections about the cumulative effect of hypogammaglobulinemia and infection risk. Furthermore, despite our relatively diverse patient population, our single-center design may limit generalizability. We also should mention that binary COVID-19 vaccination does not fully capture the degree of vaccination exposure because patients may have received one or several boosters, as well as different vaccines.
In conclusion, our findings demonstrate that continuous OCR treatment leads to progressive hypogammaglobulinemia, with a reduction of IgG levels to below 600 mg/dL being associated with increased infection risk. The complex interplay between patient characteristics, immunological parameters, and clinical outcomes highlights the importance of individualized monitoring approaches for patients to safely experience the efficacy benefits of OCR. As MS often requires long-term therapy, these results should inform discussions about the optimal duration of OCR treatment and the potential need for alternative strategies in patients who develop significant hypogammaglobulinemia. Future research will be essential for identifying reliable predictors of hypogammaglobulinemia and infection (including PML) risk and developing evidence-based monitoring guidelines for long-term OCR therapy.
Supplemental Material
sj-docx-1-mso-10.1177_20552173251391975 - Supplemental material for Hypogammaglobulinemia and infection rates in patients with multiple sclerosis treated with ocrelizumab for up to six years: A real-world single-center study
Supplemental material, sj-docx-1-mso-10.1177_20552173251391975 for Hypogammaglobulinemia and infection rates in patients with multiple sclerosis treated with ocrelizumab for up to six years: A real-world single-center study by B. Khatri, P. Van Zealand, S. Tarima, S. Schutten, A. Baker and T. Perea in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the CENTER FOR NEUROLOGIAL DISORDERS REGIONAL MS CENTER.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
