Abstract
Background
Early life stress (ELS) has been implicated in the risk of developing autoimmune disorders including multiple sclerosis (MS), but less is known about its relationship to the clinical course of MS.
Objective
Investigate the relationship between ELS and MS patient-reported outcomes (PRO)
Methods
In a single-center, cross-sectional study, persons with MS completed a composite of surveys assessing ELS and PRO including the adverse childhood experiences (ACE) survey, the Childhood Trauma Questionnaire, MS Performance Scales, MS Impact Scale (MSIS-29) and MS Quality of Life (MSQOL-54). Multiple linear regression models were used to assess the associations between ELS and outcomes while adjusting for potential confounding variables.
Results
A total of 133 persons with MS completed the study. ELS was significantly associated with worse outcomes on the MSQOL-54. Persons with MS with ACE score ≥ 4 were 25.6 points lower (95% CI −15.7 to −35.6) on the Mental Health Composite and 24.6 points lower (95% CI −14.4 to −34.8) on the Physical Health Composite scores of the MSQOL-54. ELS was also associated with worse outcomes on the MS Performance Scale and the MSIS-29.
Conclusion
In persons with MS, a history of ELS is associated with significantly worse MS symptom burden and quality of life.
Introduction
The association between early life stress (ELS) and multiple sclerosis (MS) is gaining attention but remains understudied. 1 ELS results from toxic experiences that occur during the formative childhood years such as neglect or abuse. ELS is common with an estimated 45% of children in the United States experiencing at least one adverse event during their childhood or adolescence. 2 ELS may be associated with an increased lifetime risk of several health conditions including immune-mediated inflammatory diseases such as MS3–5. The physiologic underpinnings of the association between ELS and immune-mediated inflammatory diseases is suggested by the finding that ELS can lead to persistent changes in inflammatory pathways6,7 and hypothalamic–pituitary–adrenal (HPA) axis dysfunction, 8 which are both implicated in the pathobiology of MS.9,10 Prior studies have examined the impact of ELS on the disease course and symptom burden of MS. In persons with MS, exposure to ELS was associated with higher relapse rates, 5 earlier onset of disease 11 and worse fatigue, 12 suggesting that ELS negatively impacts MS. Whether ELS impacts MS disability remains unsettled with varying results from different studies.13,14 One weakness of the prior research on ELS and MS has been the relative lack of a comprehensive analysis of patient-reported outcomes (PRO). To address this gap in knowledge, we assessed the relationship between ELS and PRO in a cross-sectional observational study of persons with MS.
Methods
Participants
Eligible participants were aged 18 to 75, had a neurologist confirmed diagnosis of MS and received care at the Virginia Commonwealth University Health Department of Neurology. Participants were recruited during routine clinic visits or through invitations sent via a secure online portal. Online participants were identified using the electronic medical record and had been seen in the outpatient MS clinic within a 24-month period prior to the survey. In-person and online participants were asked to participate in a study about “the relationship between early life stress (ELS) and multiple sclerosis (MS).” A history of ELS was not an inclusion or an exclusion criterion. The anonymous online survey invitation also included the statement “Understanding the relationship between ELS and MS requires information from a wide range of individuals, including those without early life stress.” In-person participants completed the surveys on paper in an unsupervised private clinic room. Online participants completed surveys online at a place and time of their choosing. Data from the in-person and online surveys were pooled for analysis. Demographic information collected included age, race, date of MS symptom onset and diagnosis, current MS disease-modifying therapy (DMT), clinical phenotype (relapsing vs. progressive) and patient-reported comorbid illnesses. DMT was categorized as none; low to moderate efficacy (cladribine, fumarates, glatiramer, beta-interferons, sphingosine 1 phosphate receptor modulators or teriflunomide); or high efficacy (alemtuzumab, natalizumab, ocrelizumab or ofatumumab). Socioeconomic status was measured using the Barratt simplified measure of social status (BSMSS). 15 Institutional approval was obtained from the Virginia Commonwealth University Institutional Review Board and all participants provided written informed consent prior to participating in any study-related procedures.
Early life stress assessment
ELS was assessed using two validated surveys: adverse childhood experiences (ACE) and Childhood Trauma Questionnaire (CTQ). Consistent with the NIH Stress Measurement Toolbox, we collected the ACE survey, 16 utilizing the original 10-item survey which is divided into three categories: (1) childhood abuse—emotional, physical and sexual, (2) childhood neglect—emotional and physical and (3) growing up with household dysfunction—substance abuse, mental illness, domestic violence, criminal household member and parental marital discord.17,18 For the purposes of analysis, participants were grouped based on their ACE scores: ACE = 0, ACE = 1, ACE = 2 or 3, and ACE ≥ 4. Participants also completed the CTQ, 19 a 28-item psychometrically validated, self-report inventory which has five subscales: emotional abuse, physical abuse, sexual abuse, emotional neglect and physical neglect. 20 The CTQ provides a continuous variable that has been validated to assess the intensity of traumatic experience, and provides additional data on ELS not captured by ACE survey including data on the frequency of ELS exposure and a higher sensitivity to identifying physical abuse and neglect. 21
Multiple sclerosis-relevant outcomes
To quantify the level of disability, we used the patient-determined disease steps (PDDS)22,23 and the MS Performance Scales.24,25 The MS Performance Scales is a self-reported measure for MS-associated disability, assessing mobility, bowel/bladder, fatigue, sensory, vision, cognition, spasticity, hand function, tremor and coordination. The MS Impact Scale (MSIS-29) and the MS Quality of Life (MSQOL-54) instrument were used to capture MS-relevant PRO on symptom burden and quality of life. The MSIS-29 is a validated measure that includes two subscales measuring the physical (20 items) and psychological (9 items) impact of the disease. 26 The MSQOL-54 is a measure that includes 12 subscales: physical function, physical role limitations, emotional role limitations, pain, emotional well-being, energy, health perceptions, social function, cognitive function, health distress, overall quality of life and sexual function.27,28 The Physical and Mental Health Composite scores are derived from a weighted combination of the MSQOL-54 subscales. Participants also completed the Beck's Depression Inventory (BDI) 29 and the Generalized Anxiety Disorder (GAD) survey 30 as measures of depression and anxiety, respectively. Finally, participants completed the Pittsburgh Sleep Quality Index (PSQI), a measure of sleep quality. 31
Statistical analysis
We summarized continuous variables by mean and standard deviations, and categorical variables by proportions. For continuous variables with asymmetric distributions, we also determined the median, minimum and maximum. We utilized Pearson's correlations among predictor variables (ACE score, CTQ total score and CTQ subscale scores) and outcome variables of interest (MSIS-29, MSQOL-54, GAD, BDI and PSQI), and visualized the correlation matrix in a correlation plot. The ACE and CTQ were assessed for their convergent validity using Pearson's correlation. We also assessed the omnibus group differences among the four ACE groups using the one-way analysis of variance (ANOVA). To test our primary hypotheses on whether ELS is associated with MS outcomes, we fit multiple linear regression models to assess the relationship between ACE score group or CTQ total score and MS outcomes (MSIS-29 and MSQOL-54), anxiety (GAD), depression (BDI), and sleep (PSQI), adjusting for age, age at diagnosis, sex, race and study type (in-person vs. online). The relationships between CTQ subscales and the MSQOL-54 Mental Health Composite and the MSQOL-54 Physical Health Composite scores were also assessed using multiple linear regression models. Multivariate normality, linearity and homogeneity of variance assumptions were checked after modeling fitting. Proportional hazard assumptions were checked after model fitting. To assess potential multicollinearity among independent variables, we calculated the variance inflation factor (VIF) for each predictor in the regression model. A VIF greater than 5 was considered indicative of moderate collinearity, while a VIF above 10 suggested serious multicollinearity concerns. None of the variables in the analysis had a VIF greater than 5. All analyses were done by complete-case analysis using R Studio (R Version 4.4.1).
Results
A total of 133 participants completed all study surveys, with 87 participants completing the surveys online and 46 participants completing the surveys during in-person visits. Online surveys were sent to 300 persons with MS with 109 entering into the online survey (36% response rate) among whom 87 completed all surveys in the study link (20% incomplete/abandon rate). The online and in-person groups were similar in demographics except for race, socioeconomic status and MS clinical phenotype with the in-person group having a higher proportion identifying as Black and a higher proportion with progressive MS (eTable 1). There was no difference in the prevalence of ELS between the in-person and online participants. Table 1 provides participant characteristics for the entire cohort and by ACE score groups. The mean age for the cohort was 48 (±14). The majority (82%) of the study cohort was female. Participants were 78% white, 19% Black and 3% other/mixed race. The mean age at symptom onset and at diagnosis were 34.1 (±11) and 37.6 (±12), respectively. A history of ELS (ACE score > 0) was found in 60% (n = 80) of participants. There were no significant differences among the ACE score groups with respect to age, sex, race, disease duration, MS phenotype or socioeconomic status (Table 1). A higher proportion (28%) in the ACE = 0 group (i.e. no history of ELS) were not on DMT and a higher proportion (63%) in the ACE ≥ 4 group were on high efficacy therapy, but the differences in DMT use did not reach statistical significance. Psychiatric comorbidities were higher among those with a history of ELS (ACE > 0) and more prevalent with increasing ACE scores. Among participants with no ELS history (ACE = 0), 21% reported a diagnosis of anxiety and 11% reported depression, compared with 74% reporting anxiety and 78% depression in the ACE ≥ 4 group (Table 1).
Participant characteristics.
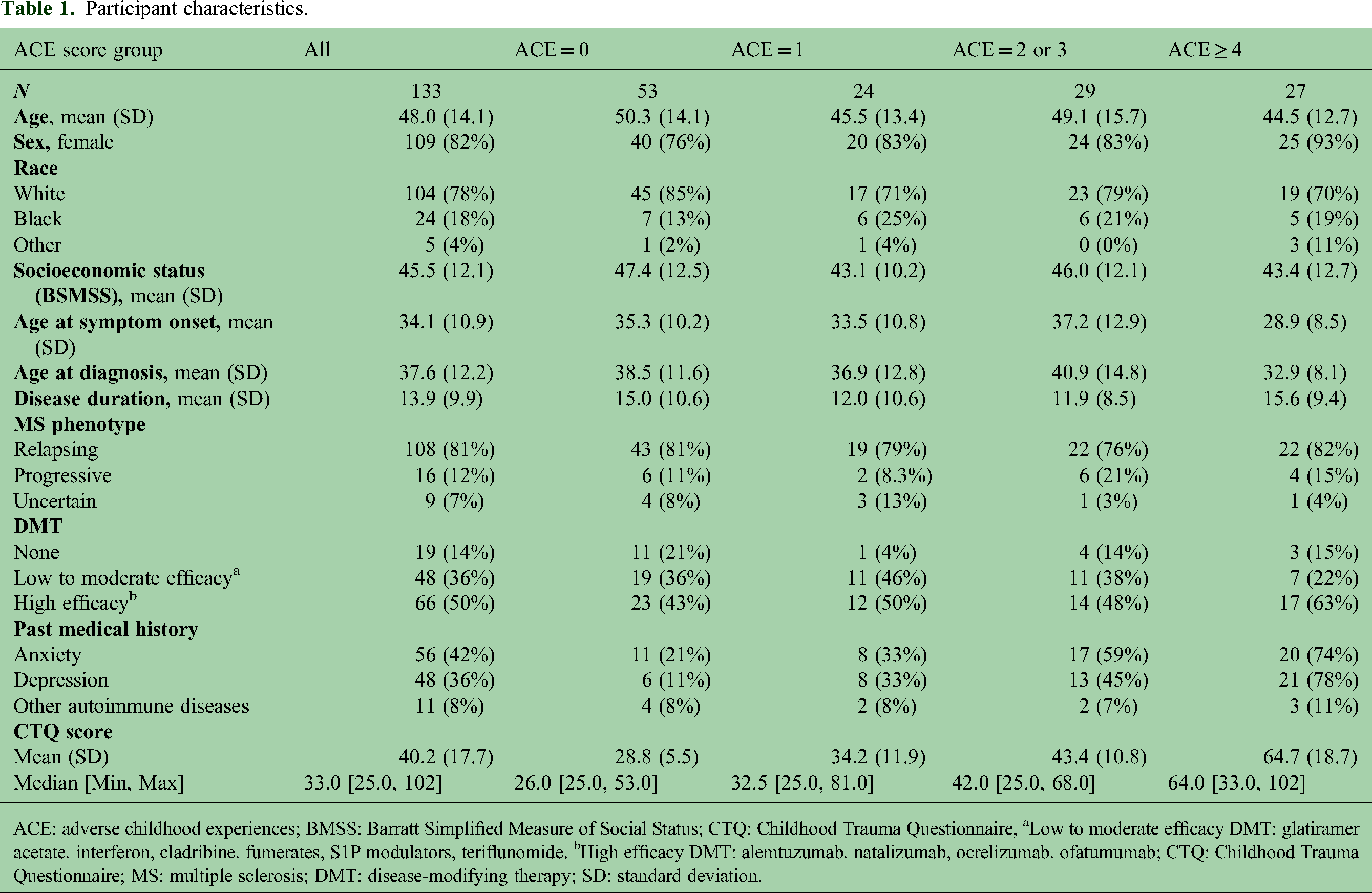
ACE: adverse childhood experiences; BMSS: Barratt Simplified Measure of Social Status; CTQ: Childhood Trauma Questionnaire, aLow to moderate efficacy DMT: glatiramer acetate, interferon, cladribine, fumerates, S1P modulators, teriflunomide. bHigh efficacy DMT: alemtuzumab, natalizumab, ocrelizumab, ofatumumab; CTQ: Childhood Trauma Questionnaire; MS: multiple sclerosis; DMT: disease-modifying therapy; SD: standard deviation.
Correlation analysis among variables
Figure 1 shows the correlation plot of ELS measures and PRO. ACE scores were highly correlated with the CTQ scores, demonstrating a good convergent validity and external consistency between the ACE and CTQ surveys for assessing ELS. The five CTQ subscales (i.e. emotional abuse, emotional neglect, physical abuse, physical neglect and sexual abuse) showed good correlations with CTQ total, indicating a good internal consistency of the CTQ survey. In persons with MS, ACE and CTQ scores were positively correlated with MSIS-29 scores, indicating a greater negative impact of MS in those with ELS. ACE and CTQ scores were negatively correlated with MSQOL-54 scores, indicating a worse quality of life in persons with MS with a history of ELS. Higher scores on measures of ELS also correlated with severity of depression (BDI), anxiety (GAD) and poor sleep quality (PSQI).

Correlation matrix of ELS measures and MS outcome measures. Pearson correlations were used and only significant correlations are displayed (p < 0.05). ELS: early life stress; MS: multiple sclerosis.
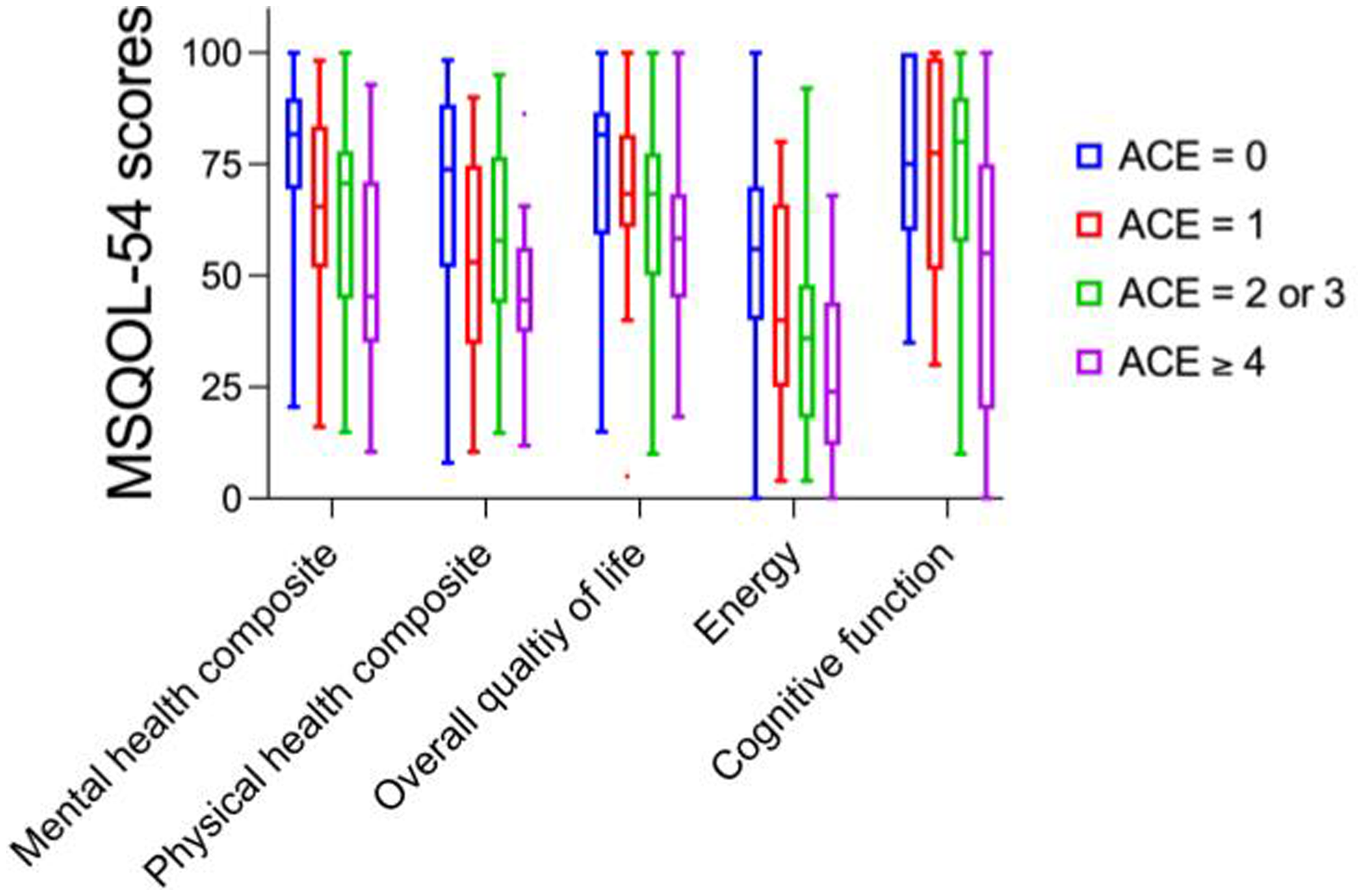
Box and whiskers (5 to 95 percentiles) plot showing MSQOL-54 component scores by ACE score groups. ACE: adverse childhood experiences; MSQOL: MS Quality of Life; MS: multiple sclerosis.
Adverse Childhood Experiences survey and multiple sclerosis outcomes
MS outcomes across the ACE score groups are shown in Table 2, with omnibus group differences assessed by one-way ANOVA. The median PDDS score was 1.0, indicating mild disability, with a range from 0 to 7. There were no significant differences among the ACE score groups with respect to mean PDDS (p = 0.72). However, fewer proportions had PDDS scores of 0 in the ELS groups compared to those without ELS (p = 0.040). With respect to domain-specific disability, higher ACE scores were associated with higher scores on several MS Performance Scales (Table 2). Significant differences were found among the ACE groups on Performance Scales for Fatigue (p = 0.009), Pain (p = 0.027) and Tremor/Loss of Coordination (p = 0.023). The MSIS-29 scores were significantly higher in the higher ACE score groups. The MSQOL-54 scores were significantly lower in the higher ACE score groups (Table 2 and Figure 2). Differences were also seen in the ACE score subgroups on the BDI, GAD and PSQI.
MS-relevant outcomes by ACE score groups.

PDDS: patient-determined disease steps; MSIS-29: Multiple Sclerosis Impact Scale; MSQOL-54: Multiple Sclerosis Quality of Life Instrument; BDI: Beck's depression index; GAD: Generalized Anxiety Disorder Survey; PSQI: Pittsburgh Sleep Quality Index; ACE: adverse childhood experiences; MS: multiple sclerosis; SD: standard deviation. Bold values indicate p < 0.05.
The relationship between ACE scores and PRO was assessed by multiple linear regression analysis adjusting for potential confounding variables such as age, sex, age at diagnosis, race and study subtype (online vs. in-person, Table 3). The online survey participants had a small but statistically significant difference in their MSQOL-54 Physical Health Composite scores compared to the in-person participants (Table 3). Otherwise, the study types did not differ with respect to PRO or comorbidities. Adjusting for potential confounding variables including study type, we found that compared to patients reporting no ACE events (ACE = 0), patients with ACE ≥ 4 had significantly worse scores on the MSQOL-54 Mental Health Composite Score (25.6 points lower, 95% CI −15.67 to −35.62, p < 0.001) and the MSQOL-54 Physical Health Composite Score (24.6 points lower, 95% CI −14.39 to −34.75, p < 0.001). Patients with ACE ≥ 4 scored significantly worse on the MSIS-29 (25.8 points higher, p < 0.001). These results indicated that ELS is associated with worse quality of life, both mental and physical, and higher symptom burden in persons with MS. In addition, the association between ELS and BDI (10.7 points higher, p < 0.001), GAD (5.2 points higher, p < 0.001) and PSQI global scores (3.8 points higher, p < 0.001) remained significant after adjusting for potential confounding variables (Table 3).
Associations between ACE groups and MS outcomes in multiple regression models.

MSIS-29: Multiple Sclerosis Impact Scale; MSQOL-54: Multiple Sclerosis Quality of Life Instrument; MHC: mental health composite; PHC: physical health composite. BDI: Beck's Depression Index. GAD: Generalized Anxiety Disorder Survey. PSQI: Pittsburgh Sleep Quality Index; ACE: adverse childhood experiences: MS: multiple sclerosis. Bold values indicate p < 0.05.
Childhood Trauma Questionnaire survey and multiple sclerosis outcomes
The relationship between CTQ scores and MS outcomes was also assessed by multiple linear regression analysis, adjusting for potential confounding variables including age, sex, race, age at diagnosis and study type. We found that the CTQ total score was significantly associated with MSIS-29, MSQOL-54, BDI, GAD and PSQI global scores, with higher CTQ total score associated with worse PRO scores across these measures (Table 4). We next evaluated the relationship between CTQ subscales and MSQOL-54. Using multiple linear regression models, we found that all CTQ subscales were significantly associated with the MSQOL-54 Mental Health Composite (supplemental Table S2) and Physical Health Composite (supplemental Table S3) scores. The largest effect on the MSQOL-54 Mental Health Composite score was found with the CTQ-physical neglect (−2.72 points, p < 0.001) and CTQ-physical abuse (−2.48 points, p < 0.001). For the MSQOL-54 Physical Health Composite score, the strongest association was with the CTQ-physical abuse (−2.15 points, p-value <0.001).
Associations between CTQ and MS outcomes in multiple regression models.

MSIS-29: Multiple Sclerosis Impact Scale; MSQOL-54: Multiple Sclerosis Quality of Life Instrument; MHC: mental health composite; PHC: physical health composite. BDI: Beck's Depression Index. GAD: Generalized Anxiety Disorder Survey. PSQI: Pittsburgh Sleep Quality Index; CTQ: Childhood Trauma Questionnaire; MS: multiple sclerosis. Bold values indicate p < 0.05.
Discussion
We report on the negative impact of ELS on MS-relevant outcomes across a broad suite of PRO in persons with MS. We found that both measures of ELS, the ACE and the CTQ, were significantly associated with worse outcomes pertaining to MS disease impact, symptom burden and quality of life. Persons with MS with a history of ELS were more likely to report higher symptom burden and worse quality of life on the MSIS-29 and the MSQOL-54. Both the physical and mental components of these PRO were worse for those with a history of ELS. Analysis of CTQ subscales indicated that physical abuse and physical neglect showed strongest association with worse PRO, with significant association also observed with emotional abuse/neglect and sexual abuse.
Our study corroborates and extends prior research with an in-depth assessment of the relationship between ELS and PRO in MS. Shaw et al. found a negative association between ELS and a measure of cognition in persons with MS, suggesting a relationship between ELS and MS outcomes. 11 Pust et al. used path analysis of online survey results to identify an association between ELS and fatigue in persons with MS. 32 Emotional neglect and emotional abuse scores of the CTQ were significantly associated with fatigue in MS in their study. We also show in the present study that ELS is associated with the fatigue component of the MS Performance Scale. Importantly, whereas prior studies focused on the impact of ELS on a narrower set of PRO, our study extends the prior research by identifying the broad impact of ELS on MS symptom burden and quality of life.
The association between ELS and MS PRO in our study was most broadly significant for ACE ≥ 4, which is a relatively high burden of ELS. A limitation of the ACE score is the lack of information about the frequency, intensity and chronicity of ELS exposure, and as a result may lead to under- or overestimation of risk conferred by ELS when using ACE score alone. 33 The CTQ score, included in our analysis, does provide information regarding the frequency of each ELS exposure. Overall, our data suggests that a relatively high burden of ELS is most reliably associated with negative impact on MS PRO.
ELS was associated with worse quality of sleep (PSQI) in our cohort of persons with MS. Our study is one of the few that have examined and identified relationships among ELS, sleep and MS. A study examining stress across the lifespan showed that poor sleep may be one of the factors mediating the negative impact of stress on MS severity. 34 These results suggest that a greater index of suspicion for sleep disorder may be clinically relevant for persons with MS with a history of ELS or lifespan of stress.
We did not find a significant relationship between ELS and PDDS, a patient-reported measure of disability in MS. However, a lower proportion had a PDDS of 0 (zero) in the ELS groups, suggesting that persons with MS with a history of ELS are less likely to have minimal disability. The individual MS Performance Scales indicated a higher degree of disability with increasing ACE scores in the domains of Fatigue, Pain and Tremor/coordination. Prior research on ELS and MS with respect to MS disease course or disability progression provided mixed results. Spitzer et al. reported a significant association between ELS (physical abuse and emotional neglect) and MS relapse rates but no association between ELS and EDSS in a cross-sectional study of persons with MS. 5 Horton, et al. reported a trend for increased risk of need for a walking aid in MS associated with increased ELS exposure that did not remain significant after adjusting for multiple testing comparisons. They found no association between ELS and the Multiple Sclerosis Severity Score (MSSS). 13 One possible conclusion is that ELS does not influence the accumulation of physical disability in MS. Alternatively, given that the EDSS and the related metrics PDDS and MSSS are largely determined by gait disability at the higher range of the scores, it remains possible that ELS may influence disability accumulation in MS in domains that are not well captured by these metrics in the higher range of disability. Other considerations include limitations of the instruments used to measure ELS and the influence of ELS on stress across the lifespan. Polick et al. reported an association between ELS and PDDS using the Stress and Adversity Inventory which captures both counts and severity of stressors across the lifespan. 14 The relationship between ELS and disability progression in MS warrants further study.
Prior studies have reported an association between ELS and earlier age of MS onset. 11 We also analyzed the relationship between ELS and age at MS onset by Cox proportional hazard modeling (data not shown). Whereas ACE score ≥ 4 was significantly associated with an increased hazard of younger symptom onset (HR = 1.68, p = 0.041), the CTQ total score was not associated with an increased hazard of younger age at MS symptom onset (HR = 1.01, p = 0.27). Therefore, our data is inconclusive as to whether ELS is associated with earlier age of MS onset.
Strengths of this work include the diverse study population, including 19% Black persons with MS. The inclusion of two validated measures of ELS allowed for comparison and confirmation of our findings using both outcomes. The ascertainment of MS-relevant PROs that include evaluation of disease severity, symptom burden, as well as commonly associated comorbidities of depression and anxiety provide insight into the broad and multi-faceted impact of ELS on MS disease.
While the in-person and online groups were similar in many ways, we did identify differences in the demographics and a small but statistically significant difference in the MSQOL-54 score outcome. The lower MSQOL-54 in the online group is likely explained by a higher proportion of progressive MS participants in the online group compared to the in-person group. We therefore adjusted for study type in the multiple linear regression analyses. A limitation of this study is the lack of data on adult stress. Childhood stress could influence a person's experience of adult stress and therefore assessing stress across the lifespan may provide additional insight not provided by assessing ELS alone.14,34 We did not capture data on potential mitigating factors such as therapy for ELS or measures of resilience. Another limitation of this work is the lack of smoking data. ELS is associated with higher risk of smoking. 35 Smoking is a known risk factor for worsening progression in MS. 36 The extent to which smoking mediates the negative impact of ELS in MS remains an important question.
In summary, our study results indicate that ELS is common in persons with MS and may have a broad impact on the manifestations of the clinical disease. Screening for ELS in persons with MS may help clinicians identify and address comorbid conditions associated with ELS such as mood disorders, 37 smoking and/or sleep disorder. 34 Future studies are also needed to establish a clinically relevant threshold for the burden of ELS exposure at which further screening or intervention is indicated. Future studies are needed to elucidate the relationship between ELS and MS with respect to underlying pathophysiology common to both, i.e. systemic inflammation and HPA axis dysfunction.
Supplemental Material
sj-docx-1-mso-10.1177_20552173251387824 - Supplemental material for The impact of early life stress on patient-reported outcomes in multiple sclerosis
Supplemental material, sj-docx-1-mso-10.1177_20552173251387824 for The impact of early life stress on patient-reported outcomes in multiple sclerosis by Myla Goldman, Shanshan Chen, Rylan Pearsall, Gretchen Neigh and Unsong Oh in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Consent to participate
All participants provided written informed consent prior to participating in any study-related procedures.
Consent for publication
Not applicable.
Data availability
The authors had full access to the data used in the analyses and manuscript. The authors take full responsibility for the conduct of the research. De-identified participant data will be made available on request by any qualified researcher.
Declaration of conflicting interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article
Ethical considerations
Institutional approval was obtained from the Virginia Commonwealth University Institutional Review Board.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the ziMS Foundation and Mitchell Family Foundation.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
