Abstract
Background
Comorbidities in people with multiple sclerosis (PwMS) can affect disease course and quality of life.
Objectives
To investigate comorbidities in the five years after diagnosis, timing of comorbidity occurrence, age and sex effects, and differences between multiple sclerosis (MS) and other chronic autoimmune diseases (AIDs).
Methods
In this retrospective cohort study, we systematically assessed differences in diagnosis frequencies in newly diagnosed PwMS (n = 9,880) compared to matched controls (noAID, n = 29,640) and individuals with other AIDs (psoriasis, n = 29,640; Crohn's disease, n = 9,880).
Results
Some comorbidities of PwMS are similarly frequent in other AIDs, while others, such as depression, are more prevalent in PwMS (odds ratio (OR) vs noAID = 2.03(1.94–2.13)). We found that personality disorders are more frequently recorded in PwMS before (OR = 1.34(1.21–1.49)) and after MS diagnosis (OR = 1.32(1.16–1.5)), especially in women (OR = 1.39(1.2–1.6)). PwMS are more frequently diagnosed with Lyme disease (OR = 1.98(1.69–2.33)), which was predominantly recorded by general practitioners after presentation with neurological symptoms. We observed lower acute tonsillitis frequencies in PwMS (OR = 0.8(0.75–0.85)).
Conclusions
Our results suggest that PwMS might have a generally increased risk for specific personality disorders. More frequent Lyme disease recordings for PwMS suggest misdiagnoses of MS symptoms. Lower tonsillitis frequencies suggest a link between MS and protection from specific infections.
Keywords
Introduction
Multiple sclerosis (MS) is a chronic inflammatory disease of the central nervous system (CNS) that typically manifests in early adulthood. Many factors may influence the disease course, including age, race, socioeconomic status, genetic factors, and comorbidities.1,2 Previously described comorbidities in people with MS (PwMS) include psychiatric disorders, cardiovascular and metabolic diseases, other autoimmune diseases (AIDs), and infections.3–12 Comorbidities have been associated with higher disease activity and reduction of quality of life,12–14 and can delay diagnosis and treatment initiation. 15 Here, we investigated the occurrence of comorbidities in PwMS in the first five years after diagnosis by systematically analyzing recordings of International Statistical Classification of Diseases and Related Health Problems, Tenth Revision (ICD-10) codes. We hypothesized that some comorbidities might be observable already in early disease stages and focused on this time frame as understanding early occurring comorbidities could potentially help optimize treatment choices, predict disease progression, and improve quality of life by addressing additional health conditions early. We further hypothesized that some comorbidities might not be specific to MS and therefore compared PwMS with persons with other chronic AIDs. We further investigated whether age and sex influence comorbidity occurrence.
Materials and methods
We conducted a retrospective cohort study on anonymous ambulatory claims data provided by the Bavarian Association of Statutory Health Insurance Physicians (BASHIP). Health insurance is mandatory in Germany, and the BASHIP data covers all individuals with public health insurance, encompassing 85% of Bavaria's population, which was approximately 13,140,000 in 2020.16,17
Data
Data were provided for quarterly billing periods between 2005 and 2020. They include ICD-10 codes, including the certainty of diagnoses and the coding physician's specialty, medical encounters, and procedures coded according to the uniform assessment standard (einheitlicher Bewertungsmaßstab; EBM).
We defined a cohort of newly diagnosed PwMS (ICD-10 code G35) and control cohorts with persons either newly diagnosed with Crohn's disease (CD, K50), psoriasis (L40) or without these AIDs (noAID) (Table 1, Figure S1A). To improve case definition validity, inclusion required at least two recordings for the respective code in separate quarters, and, for PwMS, a documented neurologist visit. We excluded persons belonging to multiple cohorts. To conservatively exclude PwMS with potentially longer standing disease, we excluded individuals with a confirmed diagnosis of secondary progressive MS (SPMS, G35.3), a first diagnosis after age 50, or recordings of a defined set of ICD-10 codes possibly indicating a demyelinating event prior to MS diagnosis (Table S1). The last exclusion criterion was also applied to the control cohorts to remove individuals with potential demyelinating diseases. We further excluded individuals aged <21 at first diagnosis due to small sample sizes in this age group. Controls were matched to the MS cohort by sex, age, and time of first diagnosis in a 3:1 ratio, which was the maximum feasible given the data provided, where possible or a lower ratio. We defined a synthetic index date for each individual from the noAID cohort based on that of their matching partner.
Cohort overview.

AID: autoimmune disease; CD: Crohn's disease; IQR: interquartile range; MS: multiple sclerosis; SD: standard deviation; ICD: International Statistical Classification of Diseases and Related Health Problems. Regional distributions describe the area in which most of the individuals doctor appointments took place.
To investigate differences in comorbidity occurrence between treated and untreated PwMS, we used a separate, smaller data set that included information on redeemed drug prescriptions coded according to the Anatomical Therapeutic Chemical Classification (ATC) for quarterly billing periods between 2012 and 2022 and applied the same filtering criteria, apart from excluding PwMS that never visited a neurologist because this information was unavailable. We classified PwMS as treated if they redeemed at least one prescription for a disease-modifying treatment (DMT) (Table S2).
We analyzed three-character ICD-10 codes marked as secured and recorded in ≥1% of the persons in the MS and noAID cohorts and excluded any sex-specific ICD-10 codes, which resulted in 338 ICD-10 codes.
Analysis
Primary analysis
We analyzed associations of 338 ICD-10 codes with MS in the 5 years after first diagnosis, comparing PwMS to three control cohorts using a binarized outcome indicating whether the code was recorded at least once. We considered all recordings in the investigated time frame. To validate this approach, we performed a more conservative analysis requiring at least two ICD-10 code recordings and compared the effect sizes. We used unconditional logistic regression models to investigate the relation of diagnosis (MS/Psoriasis/CD/noAID) to the recorded ICD-10 codes, including age group (5-year bins) and sex as covariates, quantifying the effects by odds ratios (ORs), and Firth bias-reduced logistic regression in the case of complete or quasi-complete separation. We used the MS cohort as baseline, and resulting ORs were reciprocated, so values above 1 represent higher recorded rates in PwMS. We excluded the first quarter after diagnosis to rule out effects that might represent disease onset rather than comorbidities. We used Sidak correction to control the family-wise error rate at a 5% significance level, accounting for multiple testing.
Secondary analyses
For ICD-10 codes significantly associated with MS compared to noAID, we performed analyses on the five years before first diagnosis as described above but excluding the last quarter before diagnosis. Here, we also performed a sensitivity analysis for which we aimed to exclude all individuals with evidence of possible MS symptoms in the years before the first MS diagnosis, represented by ICD-10 codes we previously observed to be more frequently recorded before an MS diagnosis and suggestive of possible MS symptoms (G25, H35, H81, N31, R20, R26, R32, R39, R42). 18
We further used generalized estimating equations, including the interaction with the time point as a covariate, to compare the effect sizes for single years and before and after first diagnosis. We investigated differences between sex and age groups by including respective interaction terms in the models. For selected ICD-10 codes, we performed analyses on four- and five-character ICD-10 code subcategories to identify the specific diagnoses contributing to the observed associations.
To further investigate an observed negative association between acute tonsillitis and MS, we used EBM data to analyze differences in tonsillectomy rates (Gebührenverordnungspositionen—GOP codes 31231-31233, 36231-36233). To investigate the effects of DMTs, we compared treated PwMS to untreated PwMS and matched controls. Descriptive statistics were composed for the frequency of encounters and diagnoses made by different medical specialists.
Analyses were performed using R, version 4.3.0.
Results
We report the descriptive statistics of the analyzed cohorts in Table 1. We found no statistically significant differences in age and sex distribution between PwMS and individuals with other AIDs, but the noAID cohort had a slightly higher age (p = 0.0004) and fewer distinct recorded ICD-10 codes. Untreated PwMS were older than treated PwMS (p < 2.2 × 10−12).
We identified 76 ICD-10 codes recorded more frequently for PwMS compared to noAID in the 5 years after first diagnosis (Figure S1B, Table 2, Tables S3 and S4). Three ICD-10 codes for acute tonsillitis, hemorrhoids, and perianal venous thrombosis, and enthesopathies were recorded less frequently for PwMS. The more conservative approach requiring at least two recordings of an ICD-10 code yielded very similar results (Figure S1C, D).
Selected ICD-10 codes recorded more or less frequently in people with MS (PwMS) compared to controls.
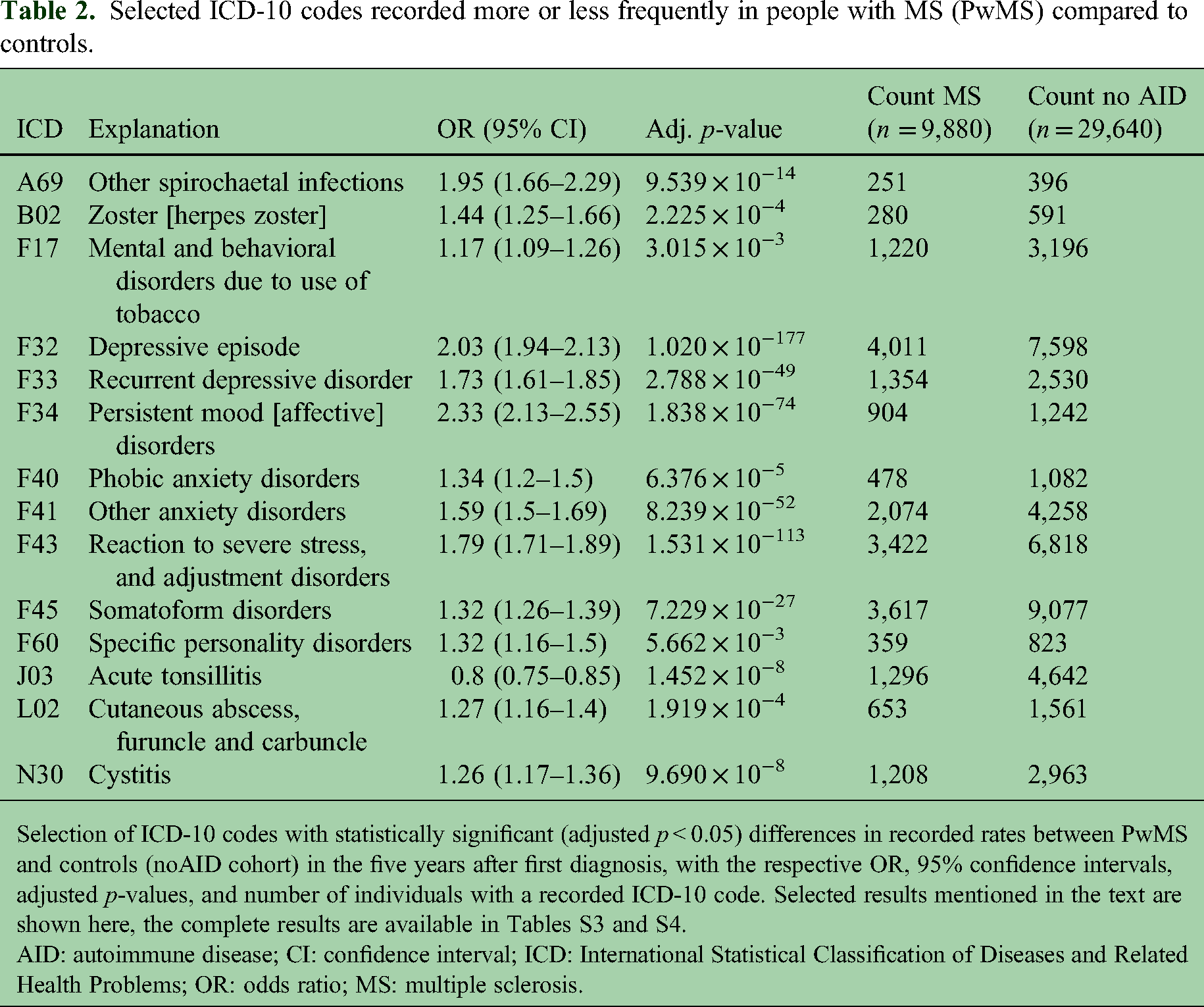
Selection of ICD-10 codes with statistically significant (adjusted p < 0.05) differences in recorded rates between PwMS and controls (noAID cohort) in the five years after first diagnosis, with the respective OR, 95% confidence intervals, adjusted p-values, and number of individuals with a recorded ICD-10 code. Selected results mentioned in the text are shown here, the complete results are available in Tables S3 and S4.
AID: autoimmune disease; CI: confidence interval; ICD: International Statistical Classification of Diseases and Related Health Problems; OR: odds ratio; MS: multiple sclerosis.
For several ICD-10 codes, effect sizes decreased with age (Table S5), including codes for neurological conditions and possible MS symptoms, such as sensory and visual disturbances. Most associations were consistent across genders, but some showed gender-specific variations: the association between MS and epilepsy was more pronounced in women, while associations with vitamin D deficiency, fatigue, and urinary tract disorders were stronger in men (Table S6). However, the ICD-10 codes with more pronounced associations with MS in men were significantly less frequent in men compared to women in the noAID cohort (Table S6), while the differences were less pronounced in the MS cohort. Epilepsy was less frequent in women than men in noAID.
In the following sections, we describe the findings we consider most interesting; a complete list of the results can be found in Tables S3 and S4. Summarized findings on other previously reported comorbidities are reported in Text S1 and Table S7.
Mental and behavioral disorders
Eleven mental and behavioral disorders (Figure 1A, Table 2) were recorded more frequently for PwMS compared to the noAID cohort, such as depression (OR = 2.03(1.94–2.13), adj.p-value = 1.02 × 10−177), anxiety disorders (OR = 1.59(1.5–1.69), adj.p-value = 2.5 × 10−152), somatoform (OR = 1.32(1.26–1.39), adj.p-value = 7.22 × 10−27), and adjustment disorders (OR = 1.79(1.71–1.89), adj.p-value = 1.53 × 10−113), as well as nicotine dependence (OR = 1.17(1.09–1.26), adj.p-value = 3.02 × 10−3). Comparing PwMS to persons with other AIDs, we found that affective and adjustment disorders were still more frequently recorded for PwMS (Figure 1A, Table S8), while this was not the case for nicotine dependence, somatoform, and anxiety disorders. Apart from nicotine dependence, all codes were recorded significantly more frequently for women than men (Table S6).
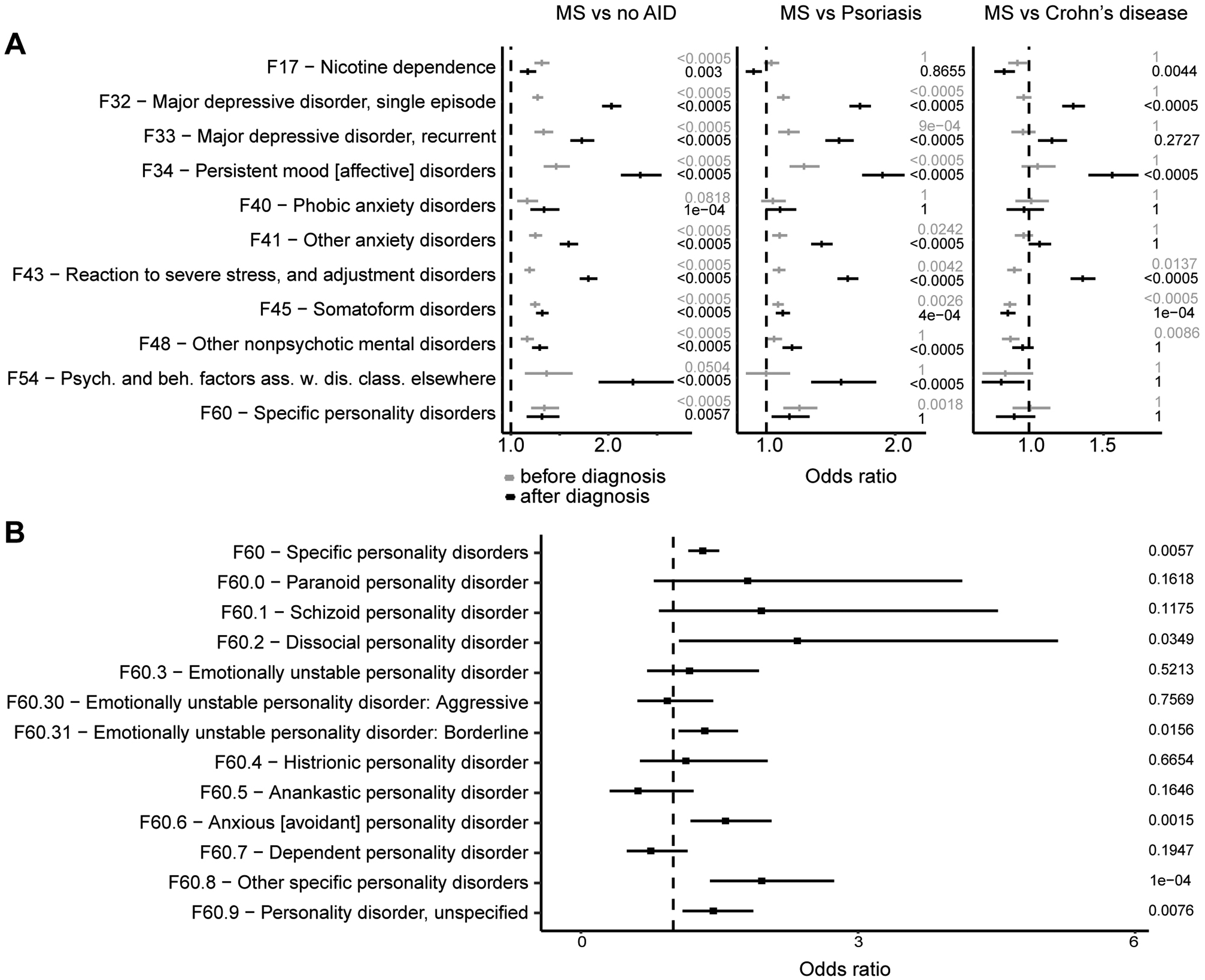
Mental and behavioral disorders were recorded more frequently for people with MS (PwMS) compared to controls. (A) Results for psychiatric ICD-10 codes recorded in PwMS in comparison to different control groups and for the five years before (gray) and after the diagnosis (black) with the respective adjusted p-values. (B) Results for F60 ICD-10 code subcategories after diagnosis in PwMS compared to the noAID cohort.
Our analyses further revealed a higher frequency of recordings of personality disorders (F60) for PwMS (Figure 1A, Table 2) compared to noAID (OR = 1.32(1.16–1.5), adj.p-value = 5.66 × 10−3) and compared to people with psoriasis, but not compared to people with CD. Personality disorders were more common in women than men across all cohorts, with a stronger association with MS compared to noAID in women (OR(women) = 1.39(1.2–1.6) and OR(men) = 1.13(0.87–1.46)). However, this difference was not statistically significant (p = 0.17, Table S6). We analyzed ICD-10 code subcategories and found that codes for anxious personality disorder, the dissocial type, the emotionally unstable borderline type, as well as unspecific codes (F60.8, F60.9) were recorded more frequently in PwMS (Figure 1B, Table S9).
In the five years before first diagnosis, the recorded frequencies of most psychiatric ICD-10 codes were still higher PwMS compared to noAID, but with less pronounced differences (attenuation 5–39%). The effect size was however similar for personality disorders (OR = 1.34(1.21–1.49), adj.p-value = 0.004, Figure 1A, Table S10) and higher for nicotine dependence (OR = 1.32(1.24–1.4), adj.p-value = 2.17 × 10−15). After excluding individuals with potential demyelinating events before diagnosis, the differences were even less pronounced for most diagnoses (Table S10; attenuation 17–48%).
Infections
Four infections were recorded more frequently for PwMS compared to noAID (Table 2, Figure 2A): cystitis (OR = 1.26(1.17–1.36), adj.p-value = 9.69 × 10−10), zoster (OR = 1.44(1.25–1.66), adj.p-value = 2.23 × 10−4), cutaneous abscesses (OR = 1.27(1.16–1.4), adj.p-value = 1.92 × 10−4), and spirochetal infections (OR = 1.95(1.66–2.29), adj.p-value = 9.54 × 10−14). Comparing PwMS to individuals with other AIDs, we found no statistically significant differences for zoster and cystitis, while cutaneous abscesses were less frequently recorded in PwMS compared to individuals with CD. All four infections were still positively associated with MS in a subanalysis of untreated PwMS (Figure 2B, Table S11).

Four infections were recorded more frequently for people with MS (PwMS) compared to controls. (A) Single-year analysis for four infections in PwMS compared to the noAID group. (B) Results for analyses performed on treated and untreated PwMS. (C) A69 recordings in PwMS were made by different specialists (all contributions with less than 5% not made by neurologists (Neurol) or general practitioners (GPs) are grouped into “Other”). (D) Percentage of PwMS diagnosed with A69 presenting with neurological ICD-10 codes related to demyelination (“rel. to dem.”; G04, G05, G09, G36, G37, H46, H47, H48, R90), previously reported possible early MS symptoms 15 (“pos. MS-symp.”), or codes from the ICD-10 code category G (“ICD-10 cat. G”) in the two years before the A69 recording.
The higher frequency of spirochetal infections (A69) for PwMS, however, was evident in comparison to all three control groups (Table S8). This association appeared to be driven by a higher frequency of recordings of the subcategory A69.2—Lyme disease (Table S9). In most cases, we observed that A69 was recorded by general practitioners (GPs) (74%) and rarely by neurologists (8%, Figure 2C). Approximately 16% of PwMS with an A69 diagnosis had received an ICD-10 code related to demyelination (Table S1) in the two years before the A69 recording, 56% received at least one ICD-10 code possibly representing early MS symptoms, and 62% received at least one neurological ICD-10 code (Figure 2D). The differences in recorded frequencies of A69 were more pronounced for untreated than for treated PwMS (Figure 2B, Table S11).
We observed lower frequencies of acute tonsillitis (J03; OR = 0.8(0.75–0.85), adj.p-value = 1.45 × 10−8) for PwMS (Figure 3A and B, Table 2) in comparison to noAID, and to individuals with other AIDs (Table S8). The ORs declined in the years before and slightly increased after diagnosis (Figure 3A). The effect was more pronounced in older individuals (Figure 3C, Table S5), men had lower recorded rates than women (Table S6), and lower frequencies appeared to be more prominent in treated compared to untreated PwMS (Table S11).

Acute tonsillitis was recorded less frequently for people with MS (PwMS) compared to controls. (A) Single-year analysis for acute tonsillitis in PwMS compared to the noAID group. (B) Results for acute tonsillitis and tonsillectomies in comparison to different control groups for the five years before (gray) and after first diagnosis (black), indicating the respective p-values, for J03 after adjustment for multiple testing. (C) Age-group analysis for acute tonsillitis in PwMS compared to the noAID group.
We analyzed reimbursement claims for tonsillectomies and found no statistically significant differences between noAID and PwMS after diagnosis, but a higher frequency of tonsillectomies in PwMS before diagnosis (Figure 3B). Tonsillitis rates were still lower in PwMS after diagnosis when adjusting for tonsillectomies before diagnosis (OR = 0.8(0.75–0.85), adj.p-value = 3.32 × 10−9).
Discussion
We systematically assessed differences in recorded rates of medical diagnoses between PwMS and controls in the 5 years after first diagnosis and investigated the timing of comorbidity occurrence, age, and sex effects, and differences between MS and other AIDs.
We found higher recorded rates for over 70 ICD-10 codes in PwMS, including previously reported comorbidities3,10,11 such as psychiatric disorders, 6 epilepsy, migraine, infections, 7 and codes likely representing MS symptoms such as fatigue, and visual and skin sensation disturbances. 19 We observed increased rates for neurological ICD-10 codes, for which we previously found that they might represent misdiagnosed MS symptoms. 18 Other associations might be explained by more frequent medical encounters or diagnostic procedures performed for PwMS, such as higher recorded rates of vitamin D deficiency, disc disorders, and spondylosis.
Age influenced several associations, including neurological ICD-10 codes as well as codes likely representing MS symptoms, with younger individuals showing more pronounced effects. Some symptoms, such as visual and skin sensation disturbances, have previously been reported to be more frequent first MS symptoms in young individuals.20,21
Some associations showed gender-specific differences, with stronger associations between MS and epilepsy in women, and stronger associations with vitamin D deficiency, fatigue, and urinary tract disorders in men. These differences are likely explained by differences in recorded rates in the noAID cohort, suggesting no direct influence of sex on these comorbidities.
Our results show that some comorbidities develop around the time of or after first MS diagnosis, such as psychiatric disorders and epilepsy, suggesting a more direct link between MS manifestation and these disorders. In other cases, we found that higher recorded rates precede first MS diagnosis. Among others, we observed this for nicotine dependence, which is in concordance with a potential causal link between smoking and disease manifestation,2,22 and for neurological ICD-10 codes, suggesting unrecognized disease activity before first diagnosis. 18
Comparing PwMS to individuals with two other AIDs, we found that some comorbidities such as migraine, infections, and nicotine dependence appear not to be specific to MS but shared by different AIDs. Others, including depression and epilepsy, were more prevalent in PwMS compared to individuals with other AIDs, suggesting that a higher risk for these might be explained by MS-specific mechanisms and not be related to autoimmunity in general. Many of the ICD-10 codes differing between MS and control cohorts were codes for neurological disorders or symptoms, potentially reflecting unrecognized or misdiagnosed MS symptoms.
We observed higher recorded rates of personality disorders in PwMS both before and after MS diagnosis, suggesting that PwMS have a generally higher risk for personality disorders, independent of psychological reactions or changes in behavior after MS diagnosis. We found no significant differences between individuals with MS and CD. Consistent with our findings, a previous study reported higher incidence rates of personality disorders in immune-mediated inflammatory diseases, including MS, before and after diagnosis. 23 Our results indicate that this association may be driven by specific personality disorders from the anxious, dissocial, and emotionally unstable borderline types. Furthermore, we observe a more pronounced association of personality disorders and MS in women.
We observed higher rates for specific infections, consistent with previous studies.7,9,24–26 Our results suggest that these associations are not specific to MS. While the risk for infection can be increased under DMTs, 27 we also observe higher infection rates in untreated PwMS, suggesting treatment-independent effects. We observed higher recorded rates of spirochetal infections, particularly Lyme disease, in PwMS. A potential link between MS and Lyme disease has been explored with no definite consensus.24,28 Here, spirochetal infections were mainly recorded by GPs, not neurologists, and most individuals had previously presented with neurological symptoms. These findings, and the increasing effect sizes before MS diagnosis and decreasing ones after suggest possible misdiagnoses of MS symptoms, potentially due to the similarity of symptoms. 29 Misdiagnoses could potentially lead to unnecessary antibiotic treatments and delay in MS diagnosis and initiation of appropriate treatment.
Somewhat unexpectedly, we found lower recorded rates of acute tonsillitis in PwMS. This negative association was evident also in the years before and most pronounced around the time of first MS diagnosis, aligning with our previous observation of lower rates of upper respiratory tract infections in the years before diagnosis. 18 We further observed higher recorded rates of tonsillectomies in PwMS, but this was apparent only in the years preceding MS diagnosis. We hypothesized that the higher rates of tonsillitis could be explained by higher rates of tonsillectomies in individuals who later develop MS. However, when adjusting for tonsillectomies, we still observed significantly lower recorded rates of tonsillitides in PwMS, suggesting other underlying mechanisms. The effect was even more pronounced in treated individuals, suggesting a possible link between lower recorded rates of tonsillitides and higher disease activity. The relationship between tonsillectomies and MS was previously investigated with no final consensus30–32, highlighting the need for further investigations.
This study has several limitations. The BASHIP data does not cover privately insured individuals, primarily civil servants, self-employed persons, and individuals earning above a certain income threshold. The claims data used are not systematically audited, and their validity depends on the clinical judgment of the recording physicians. Potential confounding factors are missing information on hospital encounters, missing clinical confirmation of diagnoses and comorbidities, missing information on socioeconomic status, and potential access to healthcare, as well as stringent exclusion criteria. We observed an unexpectedly high percentage of more than 50% of PwMS without any DMT data, possibly due to missing information for hospital treatments. This might lead to substantial bias in our analyses of treated and untreated individuals. Furthermore, a detailed analysis of specific DMTs was impossible due to the low number of events. Lastly, the non-experimental study design based on retrospective ambulatory claims data might have induced potential confounding factors.
In conclusion, we replicated previously identified comorbidities and report higher recorded rates of specific personality disorders in PwMS both before and after diagnosis, suggesting a higher risk for these disorders, especially in women. Medical specialists treating PwMS should be aware of the higher risk for these and other psychiatric disorders, even in the early disease stages. We report lower recorded rates of acute tonsillitis in PwMS, suggesting a possible link of protection from certain infections and autoimmunity in MS. We observed high rates of Lyme disease recordings in PwMS possibly representing misdiagnoses of MS symptoms that require thorough investigation and attention. Lastly, we found that some comorbidities appear to be more prevalent in MS or possibly specific to MS, while others appear to be shared with other AIDs.
Supplemental Material
sj-docx-1-mso-10.1177_20552173251315458 - Supplemental material for Early comorbidities and diagnostic challenges in people with multiple sclerosis with possible impact on disease management
Supplemental material, sj-docx-1-mso-10.1177_20552173251315458 for Early comorbidities and diagnostic challenges in people with multiple sclerosis with possible impact on disease management by Christine Makarov, Ewan Donnachie, Alexander Hapfelmeier, Bernhard Hemmer and Christiane Gasperi in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Supplemental Material
sj-docx-2-mso-10.1177_20552173251315458 - Supplemental material for Early comorbidities and diagnostic challenges in people with multiple sclerosis with possible impact on disease management
Supplemental material, sj-docx-2-mso-10.1177_20552173251315458 for Early comorbidities and diagnostic challenges in people with multiple sclerosis with possible impact on disease management by Christine Makarov, Ewan Donnachie, Alexander Hapfelmeier, Bernhard Hemmer and Christiane Gasperi in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Consent to participate
According to the Guidelines and Recommendations for Good Practice of Secondary Data Analysis, 33 there was no need for written informed consent.
Data availability
Data protection regulations prohibit the distribution of the underlying data. Interested researchers may contact BASHIP or the corresponding author to request access.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: C. Makarov declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. E. Donnachie declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Dr A. Hapfelmeier received research funding from Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) and Innovationsausschuss beim G-BA (The Federal Joint Committee), not related to this project. Dr B. Hemmer has served on scientific advisory boards for Novartis; he has served as DMSC member for AllergyCare, Sandoz, Polpharma, Biocon, and TG therapeutics; his institution received research grants from Roche for MS research. He has received honoraria for counseling (Gerson Lehrmann Group). He holds part of two patents; one for the detection of antibodies against KIR4.1 in a subpopulation of patients with MS and one for genetic determinants of neutralizing antibodies to interferon. All conflicts are not relevant to the topic of the study. He is associated with DIFUTURE (Data Integration for Future Medicine) [BMBF 01ZZ1804[A-I]]. He received funding for the study by the European Union's Horizon 2020 Research and Innovation Program [grant MultipleMS, EU RIA 733161] and the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) under Germany's Excellence Strategy within the framework of the Munich Cluster for Systems Neurology [EXC 2145 SyNergy – ID 390857198]. Dr C. Gasperi received funding from Deutsche Forschungsgemeinschaft (DFG, German Research Foundation), not related to this project.
Ethical considerations
The retrospective cohort study uses anonymous claims data held by BASHIP. The data protection officer of BASHIP approved the study. According to the Guidelines and Recommendations for Good Practice of Secondary Data Analysis, 33 approval by an ethics committee was not needed.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Christiane Gasperi received funding from the German Federal Ministry of Education and Research (BMBF - 161L0216) and the Hertie Foundation (P1200018).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
