Abstract
Background
The risk of coronavirus disease 2019 among people with multiple sclerosis with different disease-modifying therapies is not well established.
Objective
To investigate the occurrence of coronavirus disease 2019 and the remaining symptoms among people with multiple sclerosis and the associations with different disease-modifying therapies.
Methods
Individuals aged 20–50 listed in the Swedish Multiple Sclerosis Registry were invited to participate in a survey in 2021. Information on reported coronavirus disease 2019 infection and remaining symptoms were linked to individual-level register data. The risks by disease-modifying therapy of having coronavirus disease 2019 or having remaining symptoms were estimated with logistic regression.
Results
Of the 4393 participants, 1030 (23.4%) self-reported coronavirus disease 2019 (749 confirmed and 281 suspected). The observed odds for coronavirus disease 2019 did not differ by disease-modifying therapy (
Conclusion
Despite the initial concerns of differing infection risks by MS treatments, we observed no differences in coronavirus disease 2019 occurrence or remaining symptoms among those who had coronavirus disease 2019. Nonetheless, exercising caution in interpreting our findings, it remains implicit that people with multiple sclerosis are particularly susceptible to infection and that lingering symptoms may persist beyond the initial infection.
Keywords
Introduction
Many people with multiple sclerosis (PwMS) faced uncertainty regarding their risk of coronavirus disease 2019 (COVID-19). 1 These concerns are related both to reducing their risk of infection with COVID-19 and also regarding receiving treatment or care for multiple sclerosis (MS). 2 PwMS generally have an elevated risk for serious infections, especially those with progressive forms of the disease. 3 Furthermore, general risks of infection are also partly dependent on MS treatment choice, 4 owing to the different mechanisms to suppress the immune response that disease-modifying therapies (DMTs) for MS invoke. 5 Differences in dosing frequencies and locations for administration of DMTs can also affect contagion risk. Hence, raising questions over whether the risk of infection of COVID-19 differed by DMT.
COVID-19 symptoms vary from asymptomatic to severe, 6 with severe morbidity often attributed to an exaggerated immune response rather than unchecked viral replication. 7 Long-COVID includes the presence of new or lingering symptoms beyond initial infection. 8 While long-COVID only affects a proportion of individuals, it imposes an increasing burden on individuals and society.8–10
Our knowledge of COVID-19 and its impact on PwMS has evolved over time.7,11 While early insights suggested slight differences in the risk of infection and disease severity related to COVID for PwMS compared to the general population, 7 recent studies suggest similar rates of COVID-19 infection. 12 Factors such as older age, obesity, comorbidities, progressive MS, severe physical disability, and recent steroid use contribute to worse outcomes.13–16 Findings regarding the use of DMTs are mixed but suggest fewer differences in risk of infection or severity than initially hypothesised based on mode of action and pre-existing knowledge on general infection risks by DMT.7,11,17 Recent studies suggest that only anti-CD20 monoclonal antibodies may moderately increase COVID-19 severity.1,14,15 Treatment considerations are crucial for MS management and treatment protocols in Sweden, where there is a high off-label use of rituximab, an anti-CD20 monoclonal antibody. 18
In Sweden, where MS prevalence is high, 19 a unique strategy was adopted during the pandemic, primarily relying on public health recommendations and voluntary behaviour change. 20 Despite studies already being published, variations in COVID-19 incidence rates across countries exist owing to implemented non-pharmaceutical interventions and MS clinical management approaches. 21 Accordingly, there is an interest in exploring the impact of COVID-19 on PwMS in different contexts.
Hence, we aimed to investigate the proportion of working-aged PwMS in Sweden having had confirmed or suspected COVID-19 as well as any remaining symptoms and whether the type of MS treatment was associated with the occurrence of COVID-19 or remaining symptoms.
Materials and methods
A cross-sectional study was conducted using questions related to COVID-19 infection and remaining symptoms from a survey as well as individual-level linked register data.
Data sources and study population
All individuals listed in the Swedish MS Registry (SMSreg) 22 that were 20–50 years old and living in Sweden, were invited to participate between May and September 2021 in an online survey administered by Statistics Sweden. Of the 8458 PwMS invited, 4412 (52%) answered the survey. Further details of the study population are published elsewhere. 23 Statistics Sweden linked the survey responses to individual-level clinical data from the SMSreg and demographic data from the Longitudinal Integrated Database for Health Insurance and Labor Market Studies (LISA). 24 The anonymised data was then delivered to the researchers.
Variables
Three survey questions were used as outcomes in this study.
First, the proportion of all study participants who had COVID-19 was investigated with the question “
Confirmed or suspected responses were also combined as a single alternative to represent having had COVID-19. This was based on the changing Swedish recommendations throughout the pandemic on who should be offered testing, as the default was not always mandatory mass testing and reporting if symptomatic
Second, among individuals responding that they have had confirmed or suspected COVID-19, there was a follow-up question about their recovery ‘How do you feel now?’ with three possible responses:
(a) Breathing (b) Physical (c) Cognition (d) Sensory/feel (e) Smell and taste (f) Impaired immune system/susceptibility to infection (g) Sensory/vision (h) Pain and aches (i) Fatigue/lack of energy (j) Other (k) Missing
Third, among the participants responding that they had remaining symptoms, an open-ended response was available to the question ‘What remaining symptoms do you have
The exposure of interest was the participants’ DMT. This was informed by the most recent DMT from the SMSreg
Lastly, the following covariates were included from register data: Age groups (20–29; 30–39; 40–49; and 50–51), legal sex (women, men), country of birth (Sweden: yes, no), level of education (primary school, high school, university), type of living area (city, town/suburb, rural area) and type of MS (relapsing-remitting MS, primary progressive MS, secondary progressive MS, missing).
Statistical analyses
The study population was described by frequencies and proportions for all, and by their reported COVID-19 status. Chi-square tests were then used to test for differences in proportions reporting COVID-19 comparing those with and without COVID-19, and in contrast to those responding do not know.
The risk of having COVID-19 by DMT was estimated using logistic regression in unadjusted and mutually-adjusted models including all covariates except type of MS and then lastly, all covariates including type of MS. Models were run with dimethyl fumarate as the reference DMT. An alternative model specified rituximab as the reference given its wide use in Sweden 18 and the particular interest owing to potentially heightened vulnerability to infection with this DMT. 27 The results are reported as odds ratios (ORs) with 95% confidence intervals (CIs). The main analysis included those reporting Yes, both confirmed with a test and suspected, with sensitivity analyses investigating only those reporting COVID-19 confirmed by a test.
Among the participants having reported that they have had COVID-19, the proportion having remaining symptoms and their symptom clusters were then explored with frequencies and proportions.
Lastly, the risks of having remaining symptoms (yes, no) with a given DMT were explored with logistic regression and reported as ORs with 95% CIs with unadjusted and adjusted models including all covariates.
All statistical analyses were performed using R version 4.2.2 and a p-value of <0.05 was considered statistically significant.
Results
Participants who answered the question regarding having had COVID-19 (
Study population characteristics for all participants and by reported COVID-19 status.
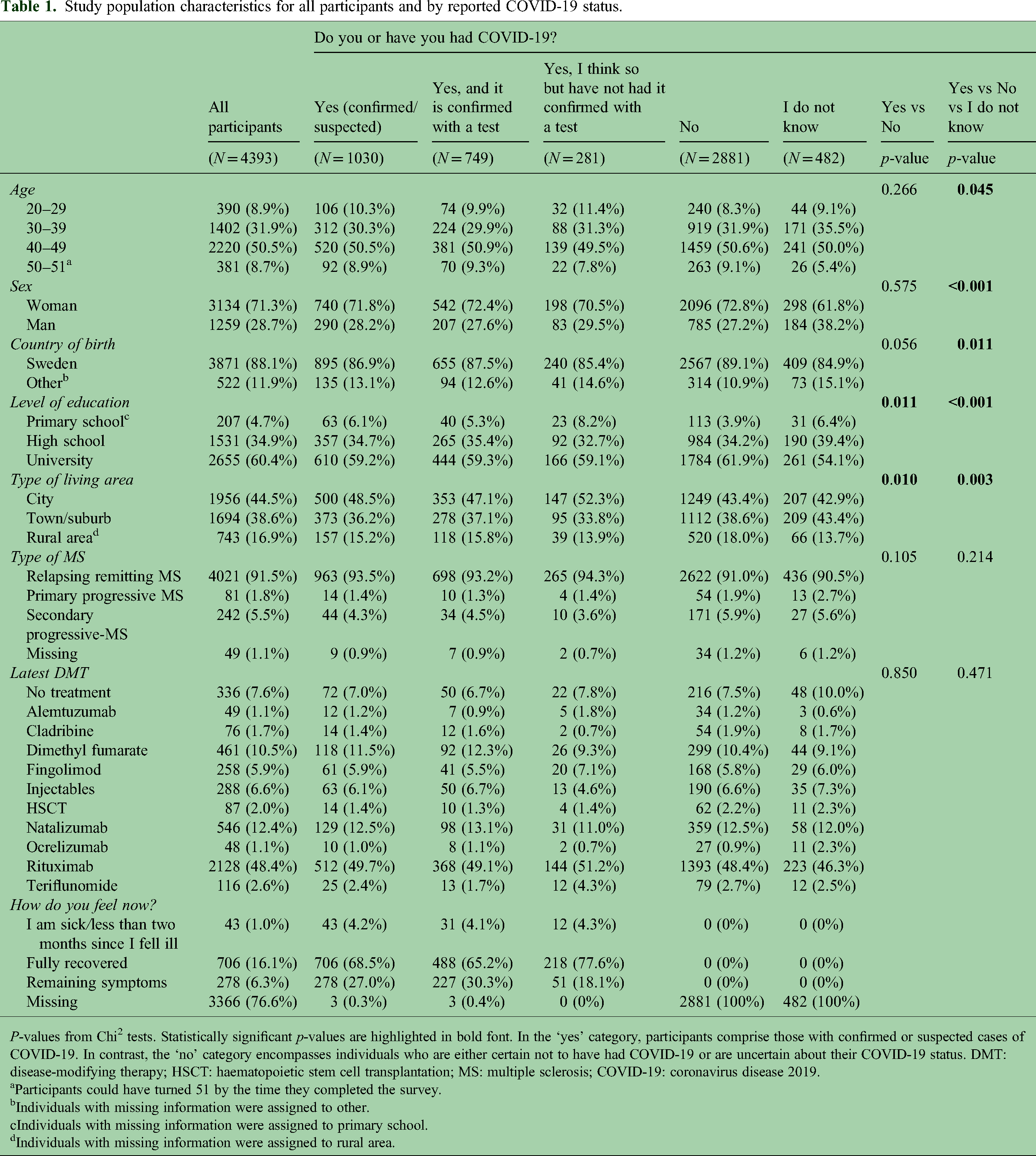
Participants could have turned 51 by the time they completed the survey.
Individuals with missing information were assigned to other.
Individuals with missing information were assigned to primary school.
Individuals with missing information were assigned to rural area.
In total, 1030 (23.4%) of the participants responded that they had COVID-19, whether confirmed (
The risk of COVID-19 was investigated among the 3911 participants who responded that they have had COVID-19 (confirmed or suspected) compared to those who had not had it. The ORs for having had COVID-19 were similar across the different DMTs (Table 2). Using dimethyl fumarate as the reference treatment, the observed ORs were all less than 1, although there were no statistically significant differences (Table 2). ORs closer to 1 were observed after adjusting for potential confounders. Similarly, using rituximab (the most common DMT) as the reference group, no differences in the risk of COVID-19 were observed by type of treatment (Supplemental Material, Table 1). The findings did not substantially change when investigating the risk only among the participants with confirmed COVID-19 compared to those not having had it (
Odds ratios for having had COVID-19 (both confirmed with a test or suspected) compared with not having had COVID-19 in participants with MS (

CI: confidence interval; DMT: disease-modifying therapy; HSCT: haematopoietic stem cell transplantation; MS: multiple sclerosis; OR: odds ratio; COVID-19: coronavirus disease 2019.
Mutually-adjusted for all other covariates except type of MS.
Mutually-adjusted for all other covariates including type of MS.
OR with 95% CIs from logistic regression models.
Nevertheless, differences in the occurrence of COVID-19 were observed for characteristics other than DMT choice. Smaller ORs for COVID-19 by increasing levels of education and less populated living areas (
Among 1030 who reported that they have had COVID-19 (both confirmed or suspected), the majority had fully recovered (68.5%), 4.2% were sick or it had been less than 2 months when they responded, and 27.0% reported that they had remaining symptoms (Table 1). The contents of the participants’ statements of their remaining symptoms were categorised into 11 symptom clusters (Table 3). The three most frequent remaining symptoms among related to the sense of smell or taste (37%), fatigue/lack of energy (20.0%), and breathing (12.0%). No differences in the risk of having any remaining symptoms (
Clusters of symptoms identified from the statements of participants who reported having had coronavirus disease 2019 (COVID-19) and having remaining symptoms at least 2 months later, for all (

*Impaired immune system/susceptibility to infection (
Risk of remaining symptoms after COVID-19 (confirmed with a test or suspected) (

CI: confidence interval; DMT: disease-modifying therapy; HSCT: haematopoietic stem cell transplantation; MS: multiple sclerosis; OR: odds ratio; COVID-19: coronavirus disease 2019.
Mutually-adjusted for all other covariates including type of MS.
OR with 95% CIs from logistic regression models.
Discussion
In this cross-sectional study of working-aged PwMS in Sweden, almost a quarter (23.4%) self-reported having had COVID-19 by summer 2021. No differences were observed among PwMS reporting COVID-19 by DMT, although higher education levels, living in less populous areas, and secondary-progressive MS were associated with a lower occurrence of COVID-19. A third (27.0%) of the participants with MS reporting COVID-19 also reported having symptoms remaining at least 2 months after their acute infection. Importantly, no differences were observed by DMT in the odds of reporting remaining symptoms. Eleven clusters of remaining symptoms were identified, the three most frequently reported related to the sense of smell or taste (37%), fatigue/lack of energy (20.0%), and breathing (12.0%). This real-world observational study on outcomes of COVID-19 in young and actively treated PwMS in Sweden adds to the body of knowledge on the impact of COVID-19 in the MS population.
The proportion of self-reported COVID-19 cases among PwMS participating in the study, even if only considering those with a confirmed positive test (17.1%), is higher than what was estimated for the whole Swedish population (11%) – based on the available data of the accumulated confirmed cases of all age groups and Swedish population on the 30 September, 2021.28,29 These findings could suggest a potential susceptibility for COVID-19 infection among PwMS, at least among those with the characteristics of the participants in this study (i.e., working-age population, living in highly and moderately populated-density areas, having relapsing-remitting MS, and actively treated – half of whom had rituximab). However, it's important to approach these findings with caution, as age groups are not directly comparable and younger populations in general, are recognised to face elevated risks against COVID-19. 30 Additionally, and contrary to our findings, a recent systematic review and meta-analysis suggested a pooled infection rate of COVID-19 of 4% among PwMS with mean age ranged from 35 to 54 years. 31 Despite this, the authors stated that incidence among PwMS differed significantly among countries, and Sweden, with its unique approach, was not included in this review.
Nevertheless, we found no statistically significant indications of a difference in risk of having COVID-19 by DMT, irrespective of the comparator. Our findings are consistent with other studies where the DMT was not a predictor for a severe COVID-19 course. 32 Further, although not statistically significant, COVID-19 infection (confirmed by a test or clinically suspected) was most frequently observed among those on rituximab. 33 Regardless, it seems that the choice of treatment among PwMS does not have a significant impact on the risk of contracting COVID-19. 7
Besides, the participants may have also adjusted their life situation to correspond to their perceived elevated risk by adopting safer behaviour, e.g., physical distancing during the pandemic. This could partly explain our finding that participants with secondary-progressive MS had a decreased occurrence of COVID-19 compared with participants with relapsing-remitting. However, we did not capture such lifestyle changes in activities or health behaviours in the survey. Nonetheless, it can be hypothesised that individuals could not entirely control or mitigate their exposure owing to other factors, for example, having to go to their physical workplace or living with children, irrespective of self-identifying as belonging to a risk group. 34 Further studies regarding family life and work-related situations among PwMS during the pandemic could aid in unravelling these questions.
An important contribution of our study is the insights on the proportion of PwMS having remaining symptoms after the acute infection. Similar to other studies, we identified the most frequent clusters of symptoms experienced included respiratory manifestations, loss of sense of smell or taste, and fatigue or brain fog.8,9 Our findings indicate that just under a third of the PwMS who had COVID-19 self-reported remaining symptoms after 2 months
There are several methodological aspects to consider with our findings. First, we included both individuals with COVID-19 confirmed by a test result and suspected, like in other studies. 14 Furthermore, we found no differences when limiting the risk of infection to only those with test results confirming COVID-19, as in a recent study. 33 Our outcome of COVID-19 infection likely includes a higher proportion of less severe cases than in many other studies using clinically identified COVID-19. By including self-reported suspected COVID-19, our study inclusion was not dependent upon requiring healthcare and introducing ascertainment bias. 16 In addition, no fatal cases were possible in our study as all were alive post-COVID-19 to respond to the survey. Nevertheless, this study comprises a representative selection of the population with MS in Sweden, considering the high coverage of MS patients in the SMSreg, 22 and compared to other studies that are primarily focused on clinical settings with more active MS cases. While we lacked information regarding the vaccination status of the participants, the survey was conducted in the early stages of the vaccine roll-out. In addition, the strains of the virus circulating, and immune response are also relevant to consider. Finally, residual confounding is possible. We did not have information on all clinical and behavioural factors that could have affected the risk of COVID-19 infection at the time including obesity, hypertension, smoking, and other treatments such as recent steroid use.
To conclude, in this cross-sectional survey with linked register data, we observed no major differences in the risks of infection with COVID-19 by MS treatment, nor did we observe differences by MS treatment among participants reporting remaining symptoms following their acute infection. In total, a quarter of the participants with MS living in Sweden reported having had suspected or confirmed COVID-19 by summer 2021, a higher estimate than the accumulated cases reported in the entire Swedish population at a similar time. Our findings support the accumulation of knowledge suggesting that the risk that DMTs pose is smaller than previously presumed, however, our findings also suggest that PwMS, in general, are more vulnerable to infection. While further research on risk assessment for this population is still needed, tailored disease prevention and management should be considered for PwMS as a group at elevated risk given the likelihood of future pandemics.
Supplemental Material
sj-docx-1-mso-10.1177_20552173241248293 - Supplemental material for Coronavirus disease 2019 infection among working-aged people with multiple sclerosis and the impact of disease-modifying therapies
Supplemental material, sj-docx-1-mso-10.1177_20552173241248293 for Coronavirus disease 2019 infection among working-aged people with multiple sclerosis and the impact of disease-modifying therapies by Chantelle Murley, Emma Pettersson, Jan Hillert, Alejandra Machado and Emilie Friberg in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Authors’ contributions
CM contributed to the conceptualisation, methodology, writing-original draft. EP contributed to the methodology, formal analysis, writing-review, and editing. JH contributed to the conceptualisation, methodology, writing-review, and editing. AM contributed to the conceptualisation, methodology, writing-review and editing. EF contributed to the conceptualisation, methodology, writing-review, editing, funding acquisition and supervision.
Data availability statement
The data used in the study cannot be made public. Such data can only be made available, after legal review, to researchers who meet the criteria to access such sensitive and confidential data, according to the General Data Protection Regulation, the Swedish Data Protection Act, the Swedish Ethical Review Act, and the Swedish Public Access to Information and Secrecy Act. Readers may contact Associate Professor Emilie Friberg (emilie.friberg@ki.se) regarding these data.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CM was previously funded partly by unrestricted research grants from Biogen as well as from Celgene/Bristol-Myers Squibb. Today, CM is employed by Macanda AB and works as a market access consultant for various pharmaceutical companies. EP declares no conflict of interest. JH has received honoraria for serving on advisory boards for Biogen, Celgene, Sanofi-Genzyme, Merck KGaA, Novartis and Sandoz and speaker's fees from Biogen, Novartis, Merck KGaA, Teva and Sanofi-Genzyme, has served as principal investigator for projects, or received unrestricted research support from Biogen, Celgene, Merck KGaA, Novartis, Roche and Sanofi-Genzyme, and his MS research was funded by the Swedish Research Council and the Swedish Brain Foundation. AM is funded partly by unrestricted research grants from Biogen as well as from Celgene/Bristol-Myers Squibb. EF is funded partly by an unrestricted research grant from Biogen and has received unrestricted research grants from Celgene/Bristol-Myers Squibb and speaker's fees from Merck.
Ethics approval
The survey received approval from the Swedish Ethical Review Authority (Dnr 2020-04996). Participants in the survey provided informed consent by submitting the survey.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was supported by research grants from Celgene/Bristol-Myers Squibb. The design of the study, data collection, analyses, interpretations of data, and manuscript drafting were performed without the involvement of the funding bodies. Celgene/Bristol-Myers Squibb was given the opportunity to comment on the manuscript before submission. The authors made the final decision on the submission of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
