Abstract
Background
Siponimod is approved for use in people with secondary progressive multiple sclerosis (pwSPMS). An integrated digital platform, MSGo, was developed for pwSPMS and clinicians to help navigate the multiple steps of the pre-siponimod work-up.
Objective
To explore real-world onboarding experiences of siponimod amongst pwSPMS in Australia.
Methods
Retrospective, non-interventional, longitudinal, secondary analysis of data extracted from MSGo (20 April 2022). The primary endpoint was the average time for siponimod onboarding; secondary endpoints were adherence and sub-group analyses of variables influencing onboarding.
Results
Mixed-cure modelling estimated that 58% of participants (N = 368, females 71%, median age of 59 years) registered in MSGo would ever initiate siponimod. The median time to initiation was 56 days (95% CI [47–59] days). Half of the participants cited ‘waiting for vaccination’ as the reason for initiation delay. Cox regression analyses found participants with a nominated care partner had faster onboarding (HR 2.1, 95% CI [1.5–3.0]) and were more likely to continue self-reporting daily siponimod dosing than were those without a care partner (HR 2.2, 95% CI [1.3–3.7]).
Conclusions
Despite the limitations of self-reported data and the challenges of the COVID-19 pandemic, this study provides insights into siponimod onboarding in Australia and demonstrates the positive impact of care partner support.
Keywords
Introduction
Most people with relapsing-remitting MS (RRMS) eventually develop secondary progressive MS (SPMS), 1 characterised by the steady accumulation of disability. Responses to available treatments differ between RRMS and SPMS. 2
Until recently, there were no disease-modifying therapies (DMTs) available for people with SPMS (pwSPMS) that could delay disability progression. 3 Siponimod was the first DMT to meet its primary endpoint in a randomised controlled trial of pwSPMS by demonstrating a reduction in the risk of 3-month confirmed disability progression (using the Expanded Disability Status Scale [EDSS]). 4 Siponimod (MAYZENT®) is the only oral DMT listed on the Pharmaceutical Benefits Scheme in Australia specifically for the treatment of SPMS. 5
Initiating siponimod involves a pre-screen work-up and requires that pwSPMS undergo a 6-day titration prior to maintenance. 3 MSGo, an integrated digital platform that also functions as a patient support service, was specifically developed to support both healthcare professionals and pwSPMS with siponimod onboarding. This study utilised data exclusively derived from MSGo to understand how pwSPMS in Australia titrate and adhere to siponimod during the onboarding and early maintenance phase of therapy.
Materials and methods
Study design and endpoints
The Mayzent MSGo platform captures standard-of-care information about individual pwSPMS who plan to initiate treatment with siponimod in Australia. It enables sharing of results and status of specific laboratory tests and assessments (CYP2C9 genotype test, maintenance dose selection, requirement for first dose observation [FDO], ophthalmologic assessment [optical coherence tomography, OCT], varicella zoster virus [VZV] serology, pregnancy test [where applicable]), titration outcomes (adherence during titration, progression through to maintenance, FDO outcome) and ongoing treatment adherence during maintenance. A feature of MSGo is that during the titration period, in-built system-triggered warnings in the HCP/support portal enable real-time investigation of patient app reporting errors.
This was a retrospective, non-interventional, longitudinal, descriptive study based on secondary analysis of pooled, de-identified data derived from the MSGo platform. The primary endpoint was the average time for siponimod onboarding, defined as the number of days between the period of participant registration on MSGo to taking the first dose of siponimod. Secondary endpoints were analyses of the rate of adherence to the siponimod titration protocol and to siponimod treatment during maintenance therapy. Safety endpoints were the occurrence of adverse events and reasons for siponimod cessation.
The study protocol and programme-specific information and consent forms were reviewed by appropriate independent human research and ethics committees (Sydney Local Health District [Concord] Human Research Ethics Committee, Concord NSW; CH62/6/2020-129 and Bellberry Human Research Ethics Committee, Eastwood, SA; 2020-07-650).
Setting and participants
Adult pwSPMS being managed by a neurologist in either the public hospital system or private clinics in Australia were included. Participants were required to have an EDSS of 3.0 to 6.5 (inclusive) and to meet the prescribing criteria for siponimod and additional criteria for compassionate use or reimbursed siponimod supply (Supplemental Table 1). Participants were excluded if they had a contraindication to treatment with siponimod, 5 and/or were diagnosed with clinically isolated syndrome or primary progressive MS.
Data (Figure 1) were collected in the MSGo platform (RxPx® Camperdown, NSW, Australia) between 7 July 2020 and 11 April 2022. De-identified participant data were extracted on 20 April 2022 for analysis (Datalytics Pty Ltd, Kingston, ACT, Australia). Eligibility for inclusion in the data analysis required agreement from the participating clinician, confirmation that institutional requirements had been met, and informed patient consent.

Overview of data obtained in the MSGo programme.
Sample size and statistical analyses
During the data extraction period of approximately 21 months, it was anticipated that MSGo would capture between 350 and 500 pwSPMS. It was estimated that for an exponential survival distribution, with 80% of participants followed until the first dose of siponimod, 500 pwSPMS would be required to enable the mean time to the first dose of siponimod to be estimated with a maximum margin of error of 10% at a 95% level of confidence.
Sub-groups were established for analysis of secondary onboarding and adherence endpoints. These comprised participant demographic sub-groups (gender, age, location, fingolimod switch status, care partner status) and pre-screening sub-groups (CYP2C9 genotype, VZV serology, OCT test, FDO). To gain insight into any potential differences between pwSPMS who chose to start on siponimod and those who chose not to, data for baseline characteristics were presented for two sub-populations: all eligible participants and participants who had started on siponimod.
The primary endpoint of time for siponimod onboarding was analysed as survival data. This enabled data from pwSPMS who had yet to take their first dose of siponimod at the time of the analysis to be censored at the time of data extraction. The distribution of the time for siponimod onboarding was estimated using a Kaplan–Meier curve, which enabled estimation of the median and quartile times as well as the proportion of participants who had onboarded at specific times since registration. The proportion of participants who had registered but who would never start siponimod was also estimated from the Kaplan–Meier curve. All estimates included 95% confidence intervals (CI). For the secondary endpoints, the proportion of participants still on treatment at specific times since the start of treatment was estimated using the Kaplan–Meier method. Adherence in specified time periods was summarised as the proportion of participants who were adherent, with a 95% CI calculated assuming a binomial distribution for the proportion. Comparisons of survival distributions (for time to onboarding and persistence) between participant sub-groups were displayed as Kaplan–Meier curves and differences between sub-groups were compared using Cox regression. For categorical variables/outcomes, absolute counts and/or rates within a particular sub-group were presented. Where appropriate and possible, chi-squared tests were used for comparative analyses. No imputation was made for missing values and no sensitivity analyses were conducted. All analyses were conducted using STATA Version 16 or above (StataCorp LLC, College Station, TX, USA).
Results
The analysis included 368 pwSPMS, mostly females (71%) with a median age of 59 years (Table 1). Acceptance of the terms of service in the MSGo app occurred rapidly; one-third of participants accepted on the day of registration, and this had risen to 50% by the following day. Most participants had not previously had fingolimod treatment (97%) and did not nominate a care partner (89%) in MSGo. Demographic profiles were comparable between the overall population and those participants who proceeded to siponimod initiation (Table 1).
Participant demographics.

Patient previously taking fingolimod and switching to siponimod.
Initiation comprises either starting siponimod via titration or starting directly on siponimod maintenance.
Most participants for whom CYP2C9 genotype data were available (62% [182/294]) were reported to have the *1*1 genotype (Table 2). Siponimod maintenance doses 2 mg (n = 166) and 1 mg (n = 27) were aligned with the recommendations in the prescribing information (Supplemental Table 2). A maintenance dose of 1 mg was assigned by the treating neurologist to participants with rare allele genotypes for which no guidance was available. Within 6 months of registration, 84% of participants had used MSGo to complete CYP2C9 genotyping. The median time from registration to complete testing was 19 days (95% CI [17–21]).
Pre-screening characteristics.

VZV: varicella zoster virus.
Denominator = number of participants tested.
Initiation comprises either starting siponimod via titration or starting directly on siponimod maintenance.
All pwSMPS undergo genotyping prior to commencing siponimod; this reflects patients who either joined MSGo for patient support only (i.e. were on maintenance therapy when they enrolled) or had a previously obtained genotype result outside of the MSGo PSP.
Pregnancy tests were conducted in women of childbearing age at the discretion of the treating doctor.
Although pwSPMS initiating siponimod is expected to have an ophthalmologic examination, this was not required to be recorded in MSGo.
Time to siponimod onboarding
Almost two-thirds (64%, 237/368) of participants were initiated on siponimod at the time of the data analysis cut-off and almost half (125/237, 53%) of those who initiated treatment had done so by about 8 weeks after registration in MSGo. The initial Kaplan–Meier survival curve suggested that a proportion of participants would never initiate siponimod. To account for this, a mixed-cure model was fitted (Supplemental Table 3), from which it was estimated that 58% of participants would ever initiate siponimod, with a median time to initiation of 56 days (95% CI [47–59]) from registration (Figure 2). A small proportion of participants (14%; 53/368) had a recorded reason for delay in initiating siponimod (Figure 3). Among those who initiated siponimod, the most common reported reason for delayed initiation was ‘waiting for vaccination’ (52%), whilst amongst those who did not initiate the most common reason was ‘patient choice’ (31%).
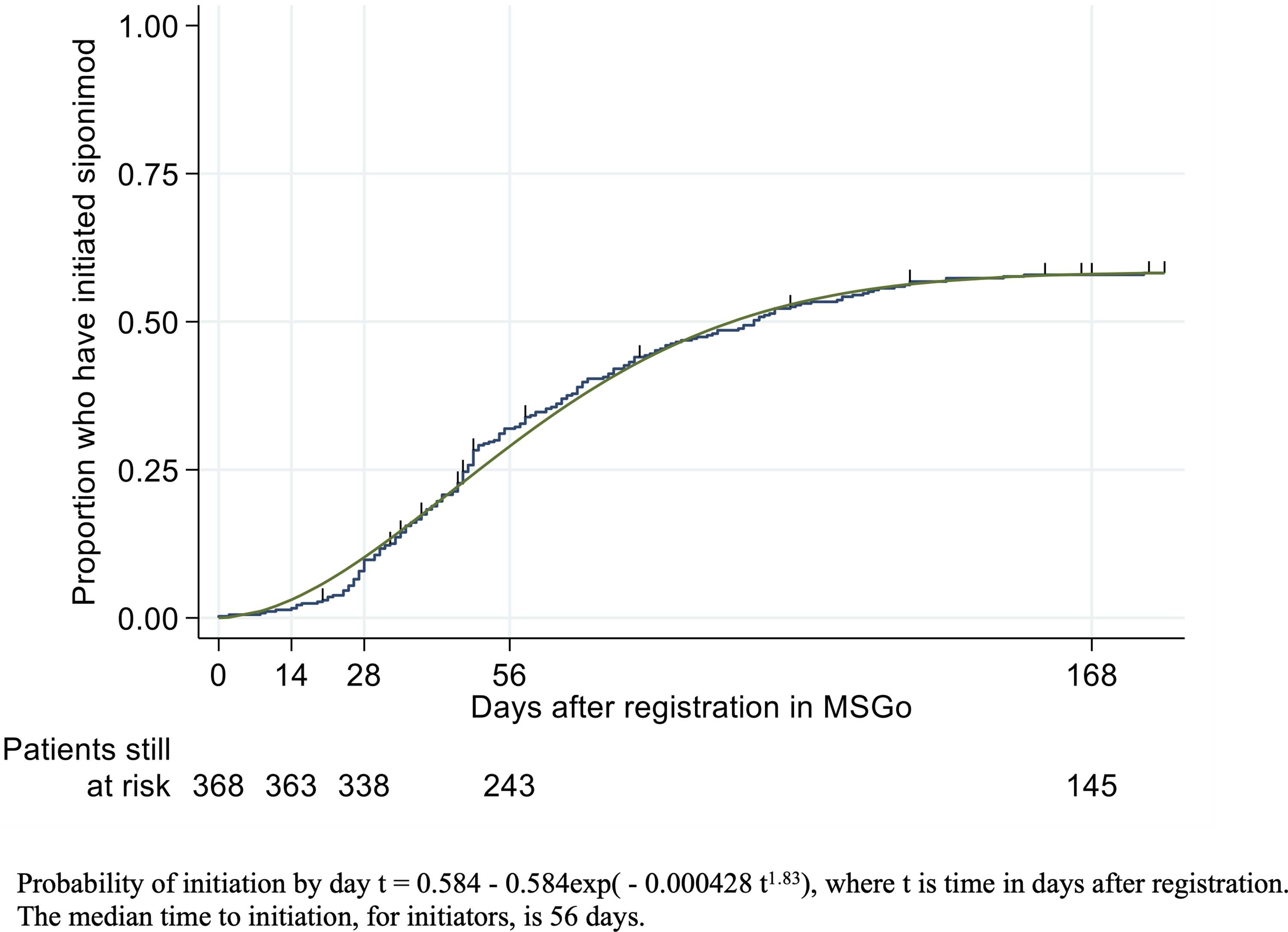
Time (days) from registration to siponimod initiation.

Reasons given for delay in initiation to siponimod.
Sub-group analyses (Table 3) found no significant differences in siponimod onboarding time by gender or age group, but participants in Western Australia were less likely and slower to initiate siponimod compared to those in other states (Figure 4(a); hazard ratio [HR]: 0.3, 95% CI [0.3–0.4]). The small proportion of participants with a care partner (11% [42/368]) were more likely to initiate siponimod than those without a care partner (81% [34/42] vs 58% [189/326]), and their onboarding time occurred earlier (Figure 4(b); HR: 2.1, 95% CI [1.5–3.0]). Siponimod onboarding time was faster in participants who had CYP2C9 genotyping recorded in MSGo than those who did not (Figure 4(c); HR: 1.5, 95% CI [1.2–1.8]). Whilst participants who had an OCT test recorded in MSGo were more likely to initiate siponimod (70% [97/138] vs 55% [126/230]), there was no difference in the time to initiation (Figure 4(d); HR: 0.9, 95% CI [0.7–1.0]).

Time (days) from registration to siponimod initiation in pre-specified sub-groups.
Sub-group analyses: Proportion of patients initiated on siponimod by day 168 after registration.

All pwSMPS undergo genotyping prior to commencing siponimod; this reflects patients who either joined MSGo for patient support only (i.e. were on maintenance therapy when they enrolled) or had a previously obtained genotype result outside of the MSGo PSP.
Although pwSPMS initiating siponimod is expected to have an ophthalmologic examination, this was not required to be recorded in MSGo.
Adherence to titration protocol
Self-reported titration data (one or more titration days recorded in MSGo) were available for 93% (221/237) of the participants in whom siponimod had been initiated during the analysis period. Most (186/221, 84%) participants completed the standard titration period and there were no discernible differences in the number of recorded titration days between participants allocated to the 1 mg and the 2 mg maintenance dose groups (Figure 5). Non-standard titration periods were recorded in 35 participants. There were 24 participants (11%) with fewer than five recorded titration doses and 11 participants (5%) with more than six recorded titration doses. The majority of these cases represented incomplete recording within the MSGo database as opposed to a documented lack of adherence to the titration protocol.

Number of siponimod titration days recorded by participants, by planned siponimod maintenance dose.
Adherence during maintenance therapy
Self-reporting of daily treatment was available for 190 participants for whom siponimod maintenance had begun before 1 April 2022. There was a rapid drop-off after the first week of siponimod initiation (Figure 6(a)) and 34% (64/190) of these participants did not record maintenance past the first 14 days. Of the 126 participants who had continued to self-report daily treatment, half were recorded as remaining on maintenance dosing 6 months after the start of siponimod maintenance (Figure 6(b)). There were no significant differences in the proportion of participants self-reporting their siponimod maintenance dosing in any of the participant characteristic sub-groups, with the exception that participants who reported a care partner were more likely to continue self-reporting their daily siponimod medication than were those without a care partner (HR: 2.2, 95% CI [1.3–3.7]). While observed variation across age groups was not statistically significant (Cox regression 5.83, p = 0.2), the post-hoc comparison showed that the oldest age group (>70 years) had the highest rate of stopping maintenance dose self-reporting (HR compared with the other age groups of 1.8, 95% CI [1.1–3.1]).

Self-reporting of daily siponimod therapy in MSGo.
Safety outcomes
A total of 90 participants ceased participation in the MSGo programme during the analysis period; 48 prior to, and 42 after, siponimod exposure, predominantly due to the participant (25/90, 28%) or HCP (20/90, 22%) decision (Figure 7). Amongst participants with no exposure to siponimod recorded in MSGo, the mean time from registration to cessation was 144 days. The frequency of cessation prior to siponimod initiation was lower in participants with a nominated care partner than those without (2% [1/42] vs 14% [47/326]). Overall, 13 participants were noted to have ceased treatment due to an adverse event and seven participants ceased treatment due to a perceived lack of efficacy. All adverse events occurred in participants who had had exposure to siponimod.
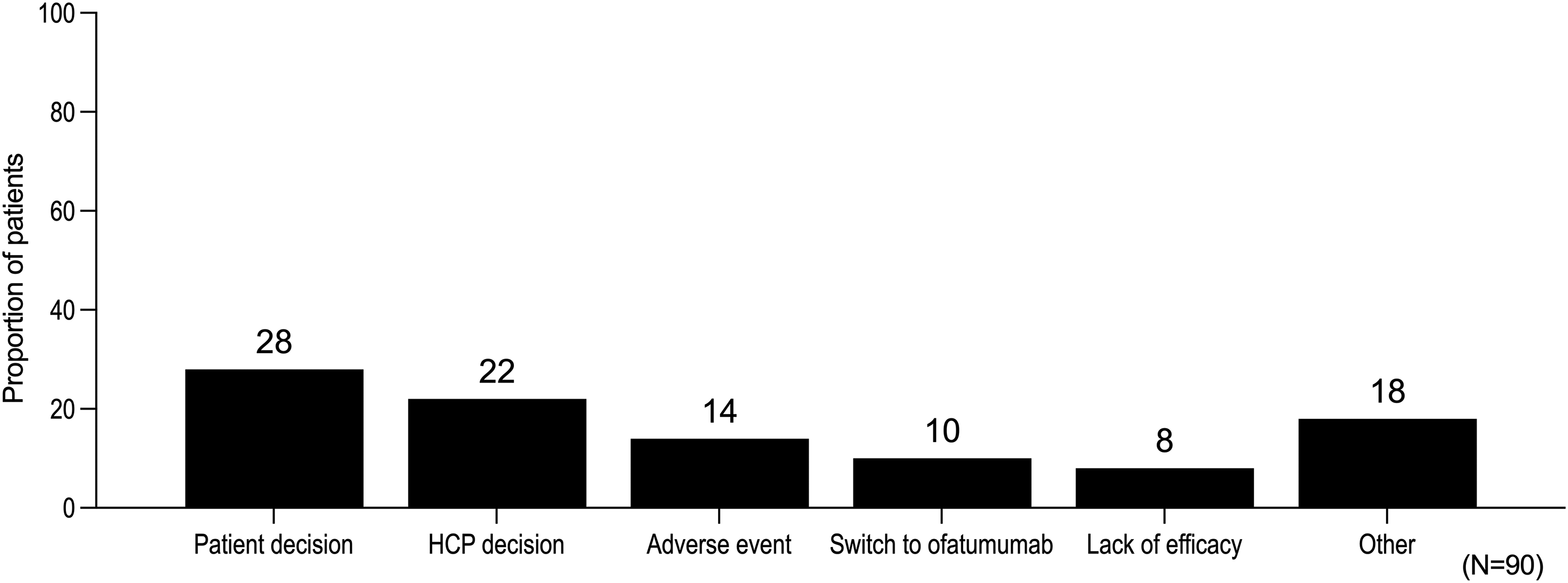
Reasons given for cessation of participation in the MSGo programme.
Discussion
This study provides the first real-world evidence regarding onboarding and adherence for pwSPMS treated with siponimod in Australia who utilised the MSGo platform. Key findings derived from using the MSGo platform in this study were that 58% of participants would ever initiate siponimod, there was a median of 8 weeks to establish a participant on siponimod, and having a nominated carer made participants more likely to initiate and to continue self-reporting. More than half of the participants who continued to self-report daily siponimod dosing in MSGo beyond the initial 14 days continued to do so beyond 6 months.
Digital health technologies can record, store and report healthcare information that can then be used to promote self-management and/or facilitate HCP communication, with the ultimate aim of enhancing patient care. 6 They can reduce patient barriers (distance, time, costs) while also improving the efficiency of healthcare delivery. 7 MSGo has been designed to provide a seamless digital platform accessible by HCPs (MSGo HCP Portal, HCP App) and pwSPMS (MSGo Patient App), with the aim of integrating data into formal clinical care. People with MS are highly likely to adopt and engage with digital health technologies.7,8 In accordance with this, participants were shown to be quick to adopt the MSGo App.
Onboarding of siponimod took an average of 8 weeks. The reasons given for delaying initiation varied between those who did initiate and those who did not. Amongst the participants who initiated siponimod, over half noted that delays were due to vaccination delays and a further quarter were due to their HCP's choice. Advice to people with MS with regard to treatment and vaccinations was rapidly evolving during the COVID-19 pandemic, which impacted this study.9,10 Notably, non-selective sphingosine 1 phosphate (S1P) receptor modulators have been observed to potentially blunt COVID-19 vaccination responses.11,12 This could have resulted in delays in the initiation of all S1P receptor modulators, including siponimod, protracting onboarding time and limiting the interpretation of this data. While the numbers were small, amongst participants that had previously been treated with fingolimod, the conversion rate to siponimod was high (92%), but protracted, taking >6 months in over half of these patients. Amongst participants who did not initiate, reasons were predominantly patient and process-oriented (patient choice, waiting for an outpatient appointment, waiting for external test results). Others report that the COVID-19 pandemic was a period in which there was minimal change to treatment due to uncertainty about the safety of different therapies for people with MS.13,14 While we have no directly supporting data, the COVID-19 pandemic may have prompted pwSPMS to be more cautious about making changes to their existing treatment plans.
While challenging to achieve, patient adherence above 80% is crucial for optimal treatment outcomes. 3 Pooled real-world adherence and persistence to oral DMTs in people with RRMS report that around 20% of patients do not adhere to daily dosing and 25% discontinue treatment within 1 year. 15 Poor patient-HCP communication, particularly around treatment goals has been implicated as a driver of reduced treatment adherence. 16 Using self-reported daily dosing entries in the MSGo App, we defined a participant as adherent unless they missed one or more consecutive days of treatment during titration or unless they missed four or more consecutive days of treatment during the first 3 months of maintenance. Interpretation of this data is limited because non-adherence could include participants who were adherent to treatment, but who were not accurately reporting this in the MSGo App. Nevertheless, the data were favourable; 84% of participants were reported as adherent to the titration protocol and 66% were reported as adherent to recording daily maintenance dosing beyond 14 days. The observed cessation rate for using MSGo was 23% (90 participants). Again, while this does not directly correlate with treatment cessation, because it is possible that these participants ceased to use MSGo while continuing siponimod treatment, they are not dissimilar to the cessation rate of 31% observed amongst pwSPMS treated with siponimod in Germany over an 18-month period. 17 The exploration of relative onboarding time and adherence rates was limited by the lack of participant numbers in some sub-groups, most notably the fingolimod switch group. Utilising dose self-reporting data in the real world may provide an estimate of true adherence, but in this case, it was hampered by a high proportion of opt-outs for reminders during maintenance.
The study was not without its limitations. The MSGo platform does not collect data on disease status and therefore the analysis was unable to provide insights into the impact of delays in treatment initiation on clinical status. Eligibility for inclusion in the study was based on the current prescribing information for siponimod in Australia, 5 of which are derived from the siponimod pivotal study (EXPAND). 4 There is a potential for bias based on the analysis population having been limited to consented participants who were registered in MSGo. Enrollment in the MSGo platform is not mandatory. At the time of the study data cut-off, 74% of the total population of patients in Australia who had initiated siponimod therapy were using MSGo of whom 32% were represented in our study. Published demographical data suggest that real-world SPMS populations have a wider range of patient characteristics than those enrolled in EXPAND. 18 Despite this, many aspects of the study population (71% female, median age 59 years, 3% reporting prior fingolimod therapy) reflect the demographics of other publications, providing confidence in the generalisability of the data.
Real-world data from 277 pwSPMS newly initiated on siponimod in the U.S. report a very similar age (55 years) and sex (73% female) breakdown to our cohort, with slightly higher (∼10%) prior fingolimod use. 19 Similarly, real-world data from a German cohort of 227 pwSPMS treated with siponimod report a mean age of 53.4 years, but a slightly lower female preponderance (56%). 17 In a German registry-based study designed to examine the characteristics of pwSPMS, 70% were female, the mean age at visit was 57.6 years and only 5% had previously been treated with fingolimod. 20 This is further reinforced in registry data showing an average age for SPMS conversion of 51.7 to 57.9 years depending on the classification method used. 21 Amongst the participants identifying as female, 23 (9%) underwent pregnancy testing. The high proportion of women beyond childbearing age enrolled in the MSGo programme also aligns with data supporting that the mean age of onset of SPMS occurs for most women during the perimenopausal period. 22
Only a small number (11%) of participants recorded that they had a nominated care partner in the MSGo App. Nevertheless, the positive role of the nominated care provider emerged as a key finding from the study. Compared to participants without a nominated care partner, these participants were more likely to initiate siponimod and to continue self-reporting daily dosing data. People with SPMS are often cognitively and physically impaired. 23 Having a care partner may have facilitated siponimod onboarding by helping with organising appointments/tests and ensuring attendance in a timely manner. Lack of peer support has previously been highlighted as a potential factor driving medication non-adherence in people with MS. 3 Further exploration of this relationship could be used to guide future personal support requirements in this population.
The results from this analysis provide the first insights into siponimod onboarding for pwSPMS in Australia during a period challenged by COVID-19 and related vaccination considerations. Within the context of managing pwSPMS specifically through the MSGo programme, age, gender and geographical location had little impact on siponimod initiations. However, siponimod onboarding occurred earlier in the small sub-group of participants with a nominated care partner and in those whose pre-screen CYP2C9 genotyping results were known at the time of registration in MSGo.
Supplemental Material
sj-docx-1-mso-10.1177_20552173231226106 - Supplemental material for Onboarding of siponimod in secondary progressive multiple sclerosis patients in Australia: Novel, real-world evidence from the MSGo digital support programme
Supplemental material, sj-docx-1-mso-10.1177_20552173231226106 for Onboarding of siponimod in secondary progressive multiple sclerosis patients in Australia: Novel, real-world evidence from the MSGo digital support programme by TA Hardy, P Aouad, MH Barnett, S Blum, S Broadley, WM Carroll, D Crimmins, D Griffiths, S Hodgkinson, J Lechner-Scott, A Lee, R Malhotra, P McCombe, J Parratt, C Plummer, A Van der Walt, K Martel and RA Walker in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Acknowledgements
The authors wish to acknowledge the neurologists and their patients who participated in this research, study management support provided by Leonie Bates (Novartis Pharmaceuticals Australia Pty Ltd), MSGo database management provided by Emily Daley, Fiona Hammond and Louise Hatter (RxMx, Sydney, NSW, Australia). Dr Michael Adena (Datalytics Pty Ltd, Kingston, ACT, Australia) conducted the statistical analyses and Hazel Palmer (Scriptix Pty Ltd, Sydney, NSW, Australia), provided professional writing assistance in the preparation of this manuscript; Novartis Pharmaceuticals Australia Pty Ltd funded both.
Contributor statement
TAH, PA, KM, and RAW designed the protocol upon which this research is based. All authors were involved in the conduct of the study. TAH and PA directed the analysis of the data and contributed to the interpretation of the results. All authors contributed to an outline from which the manuscript was drafted, revised the manuscript critically for important intellectual content and gave final approval of the version to be published. TAH accepts full responsibility for this work, has access to the data, and controls the decision to publish.
Declaration of conflicting interests
TAH, PA, SB, and JLS have been an investigator, speaker, advisor, consultant, and/or report having received travel grants from Novartis Pharmaceuticals Australia Pty Ltd. KM and RAW are employees of Novartis Pharmaceuticals Australia Pty Ltd. All other authors report no conflicts of interest.
Financial support
This study was supported by Novartis Pharmaceuticals Australia Pty Ltd. The authors were responsible for all content, interpretation of the data and the decision to publish the results; they received no honoraria related to the development of this manuscript. The sponsor funded statistical and editorial assistance and reviewed the final draft before submission.
Data sharing
The data that support the findings of this study are available on request from the corresponding author, (TAH). The data are not publicly available because the participants of this study did not give written consent for their data to be shared publicly.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
