Abstract
Disability accrual is mainly driven by progression independent of relapse activity, which is present even in early stages of relapsing-remitting multiple sclerosis (RRMS) and sometimes overlooked. This multicenter, non-interventional study evaluated whether patient-reported outcomes measures (PROMs) could capture disability in 189 early-stage RRMS patients (mean age: 36.1 ± 9.4 years, 71.4% female, mean disease duration: 1.4 ± 0.8 years, median EDSS: 1.0). The 9-Hole Peg Test (9-HPT), NeuroQoL Upper Extremity (NeuroQoL-UE), Timed 25-Foot Walk (T25-FW), Multiple Sclerosis Walking Scale (MSWS-12), Symbol Digit Modalities Test (SDMT), and Perceived Deficits Questionnaire (PDQ-5) were used to assess hand function, gait, and cognition, respectively. These functions were at least mildly affected in this early-stage population, finding significant correlations between PROMs and clinical assessments. PROMs could enable early-stage RRMS patients to communicate their perceived disability in different domains, assisting clinicians in disease monitoring and decision making.
Keywords
Introduction
Irreversible disability accumulation in multiple sclerosis can occur through relapse associated worsening and progression independent of relapse activity, being the latter pointed out as the main contributor even in early stages of relapsing-remitting multiple sclerosis (RRMS), and sometimes underestimated at this point.1,2 Previous studies have shown that balance, walking endurance, and manual dexterity are impaired in one-third to half of these patients, 3 whereas 25–57% present cognitive decline in first few years after diagnosis. 4 Patient-reported outcome measures (PROMs) could help clinicians when assessing these signs of disability progression. 5 The aim of this study was to evaluate whether PROMs are able to capture disability in early-stage RRMS patients.
Methods
Study design
We conducted a multicenter, non-interventional, cross-sectional study. Eligibility criteria included age ≥18 years, diagnosis of RRMS, 6 disease duration ≤3 years, and Expanded Disability Status Scale (EDSS) score between 0 and 5.5. Patients not able to understand or complete the study questionnaires according to physician's criteria, including those who had a relapse close to the study visit or who were not stable on their treatment, were excluded. Patients were consecutively recruited at 21 hospital-based neuroimmunology clinics between November 2020 and March 2021. This study was approved by the investigational review board of Hospital Universitari Arnau de Vilanova (Lleida, Spain). All participants provided written informed consent.
Outcome measures
Neurologists collected patients’ sociodemographic and clinical characteristics. The 9-Hole Peg Test (9-HPT), 7 Timed 25-Foot Walk (T25-FW), 8 and Symbol Digit Modalities Test (SDMT) 9 were used by neurologists to assess hand function, gait, and cognition, respectively, whereas the NeuroQoL Upper Extremity (NeuroQoL-UE), 10 Multiple Sclerosis Walking Scale (MSWS-12), 11 and Perceived Deficits Questionnaire (PDQ-5) 12 were the corresponding PROMs completed by patients to assess the same functions (Table 1).
Outcome measure definitions, scoring, and ranges.
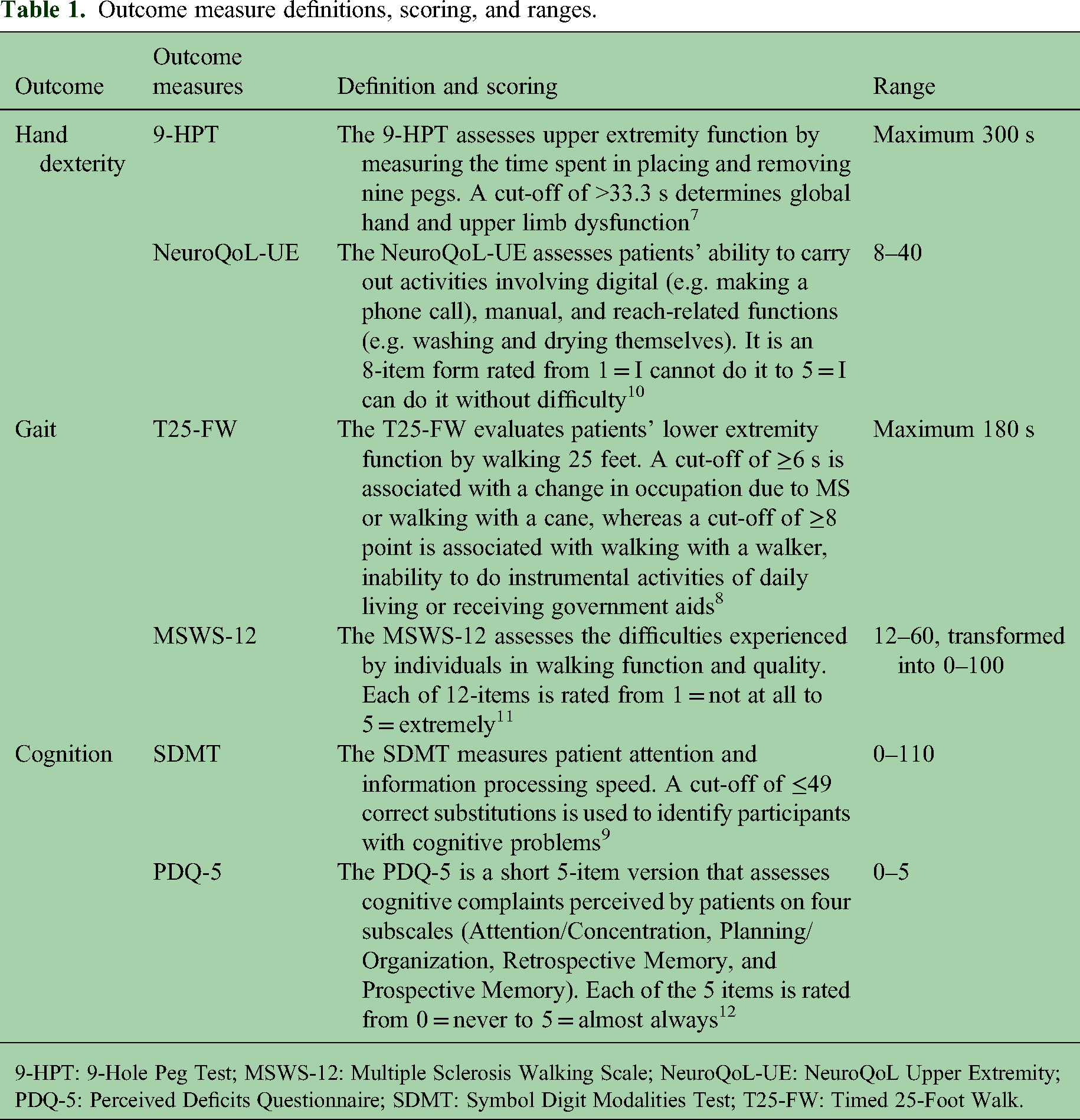
9-HPT: 9-Hole Peg Test; MSWS-12: Multiple Sclerosis Walking Scale; NeuroQoL-UE: NeuroQoL Upper Extremity; PDQ-5: Perceived Deficits Questionnaire; SDMT: Symbol Digit Modalities Test; T25-FW: Timed 25-Foot Walk.
Methodological approach
For the descriptive analysis, categorical variables were described as the total number of available values and relative percentage per subgroup of interest. Continuous variables were described by the number of available values, mean, standard deviation, and median, Q1, Q3, minimum and maximum.
Outcome measures associations were analyzed using Fisher’s exact test correlation, Kruskal-Wallis test, and Mann-Whitney U test. Correlations were analyzed categorizing the sample according to cut-off points in each outcome described in Table 1.
Results
A total of 189 patients were included in the study. The mean age was 36.1 years and 71.4% were female. Mean disease duration was 1.4 years and median EDSS score was 1.0. Hand dexterity, gait, and cognition impairment were present in 3.7%, 24.6%, and 43.1% of patients, respectively. Sociodemographic and clinical characteristics are shown in Table 2.
Sociodemographic and clinical characteristics of patients.

9-HPT: 9-Hole Peg Test; EDSS: Expanded Disability Status Scale; IQR: Interquartile Range; MSWS-12: Multiple Sclerosis Walking Scale; NeuroQoL-UE: NeuroQoL Upper Extremity; PDQ-5: Perceived Deficits Questionnaire; SD: Standard deviation; SDMT: Symbol Digit Modalities Scale; T25-FW: Timed 25-Foot Walk.
n = 188, bn = 187, cn = 183.
MSWS-12 and T25-FW
Moderate-to-extreme limitation on the MSWS-12 was reported by 24.3%, 17.5%, and 18.5% of patients in running, balance, and endurance abilities, respectively. Of patients, 66.1% reported some impact on walking ability. All individual item scores but one were significantly correlated with T25-FW score, the need for support when walking indoors being non-significant (p = 0.054). Total MSWS-12 score was significantly correlated with categorized T25-FW score (Table 3).
Relationships between PROMs and clinical assessments (n = 189).
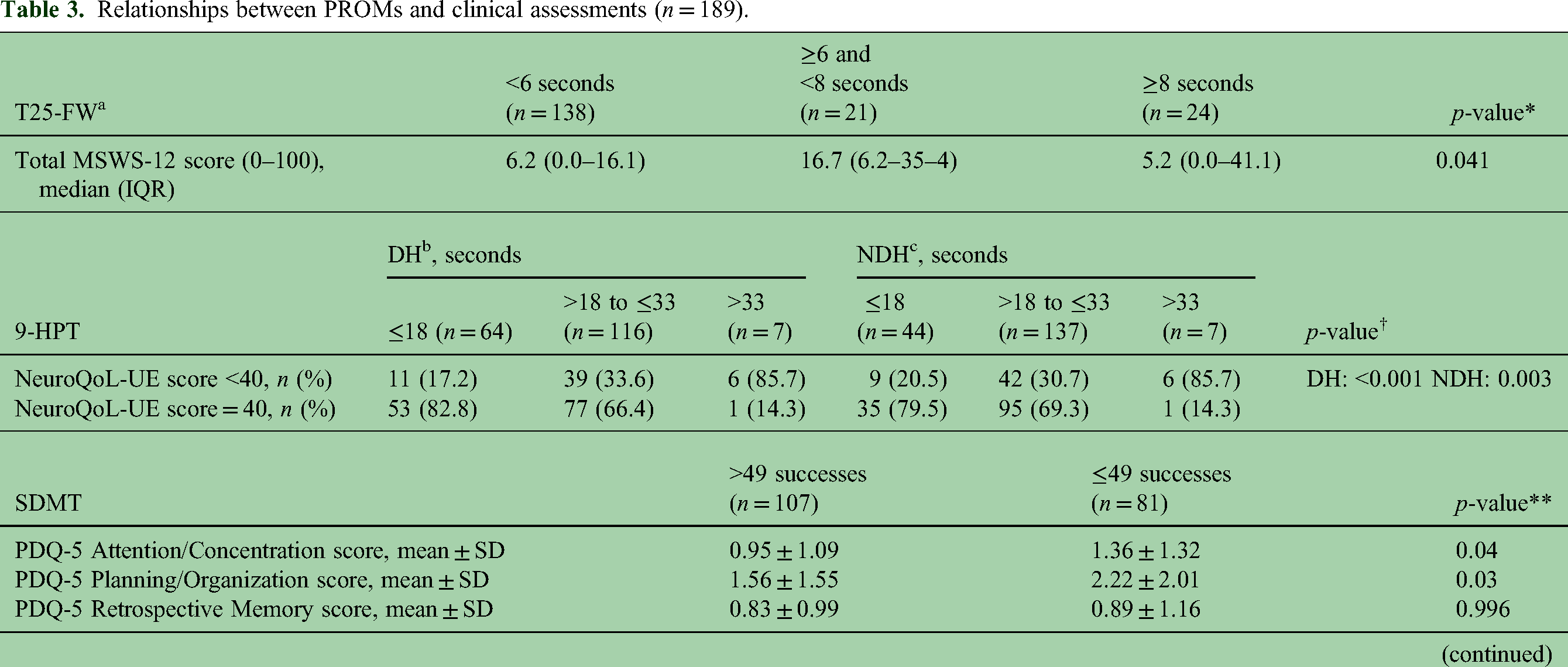
9-HPT: 9-Hole Peg Test DH: Dominant hand; IQR: interquartile range; MSWS-12: Multiple Sclerosis Walking Scale; NDH: Non-Dominant Hand; NeuroQoL-UE: NeuroQoL Upper Extremity; PDQ-5: Perceived Deficits Questionnaire; PROMs: Patient Reported Outcome Measurements; SD: standard deviation; SDMT: Symbol Digit Modalities Test; T25-FW: Timed 25-Foot Walk.
n = 183, bn = 187, cn = 188.
*Kruskal-Wallis test; †Fisher's exact test; **Mann-Whitney U-test. A cut-off of <40 is used to categorize patients with some limitation in at least one of the activities listed in the NeuroQoL-UE questionnaire versus patients with no limitation.
NeuroQoL-UE and 9-HPT
Writing with a pen/pencil and picking up coins from a table top were the tasks with the highest proportion of patients (8.5%) reporting some or a lot of difficulty in performance. A proportion of 30.2% of patients had some limitation in at least one of the questionnaire activities. Each individual NeuroQoL-UE item had a significant correlation with dominant and non-dominant 9-HPT categorized scores (p < 0.001). Total NeuroQoL-UE score was significantly correlated with 9-HPT dominant and non-dominant hands (Table 3).
PDQ-5 and SDMT
Of patients, 82% reported problems in at least one dimension of the PDQ-5 and Planning/Organization was the most affected domain. Attention, Planning/Organization, and Prospective memory domains were significantly correlated with SDMT categorized score (Table 3).
Discussion
Disability accrual in MS is present from the start and can be independent of relapses. Identifying it is important to establish rehabilitation programs and to adapt treatment regimens to achieve long-term outcomes. In our study, gait, hand dexterity, and cognition were functions frequently affected in early-stage RRMS. Short and easy-to-complete PROMs can be used as a complementary strategy to clinical assessments.
Previous studies have found significant associations between MSWS-12 and balance impairment, fatigue and increased gait asymmetry, and instability in early-stage RRMS patients, supporting its use in this population. 13 We found significant correlations between categorized T25-FW and all MSWS-12 dimensions but support needed indoors, probably because of the population's early-stage nature and low impairment reported. However, a greater percentage of patients reported some impact on their walking ability than that reflected by the T25-FW when using the cut-off point of 6 s, supporting the MSWS-12 as a complementary tool.
The 9-HPT has previously shown significant correlations with NeuroQoL-UE total score. 10 Not only total score but individual items showed significant correlations for both dominant and non-dominant hands in this study in early-stage RRMS, providing additional evidence.
Contrary to our results, previous investigators have been unable to find a correlation between total score or individual dimensions of the PDQ and SDMT. 12 However, the PDQ used was the 20-item version and participants were older, with longer time since diagnosis, and different forms of MS.
To our knowledge, this is the first study assessing simultaneous correlations between PROMs and clinical assessments regarding gait, hand dexterity, and cognition in patients with short disease duration and low physical disability, and it is the first study in demonstrating significant correlations between the PDQ-5 and SDMT. Nonetheless, several limitations should be mentioned, as we did not correct the SDMT scores according to age and education normative data, we did not assess the influence of symptoms (fatigue, depression, or anxiety) on outcomes assessed, and the small sample size and the cross-sectional design did not allow us to assess changes or causal relationships in correlations between PROMs and clinical assessments over time.
In conclusion, disability accrual is already present in early-stage RRMS patients, affecting their hand dexterity, gait, and cognition. The NeuroQoL-UE, MSWS-12, and PDQ-5 are useful tools to screen for specific difficulties or quickly assess whether a patient needs a referral for more extensive testing, carry out neuropsychological batteries, or use portable widgets to monitor exact domains or movements. This approach could assist clinicians in disease monitoring and decision making, and would open the possibility of recommending early physical and cognitive rehabilitation.
Footnotes
Acknowledgements
Authors are most grateful to all patients, neurologists, and nurses participating in the study.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JM, NM, and RGB are employees of Roche Pharma Spain. SSM received payment for lecturing or travel expenses from Merck-Serono, Biogen, Sanofi-Genzyme, Roche, and Novartis. JMM has served on scientific advisory boards and/or has received speaking honoraria, research funding, and support to attend scientific meetings from Biogen, Merck, Novartis, Roche and Teva. JS has received speaking honoraria, compensation for consulting services and support to attend scientific meetings from Almirall, Bayer, Biogen, Merck, Novartis, Sanofi, Roche, and Teva. AA has received compensation for consulting services from Biogen, BMS, Sanofi, Roche, Janssen, and Novartis; and speaking honoraria from Biogen, BMS, Sanofi, Roche, Janssen, Merck, Almirall, and Novartis. ABC has received courses and honoraria for her participation as speaker/meeting moderator/symposia organizer from Alter, Almirall, Bayer, Bial, Biogen, Bristol-Myers-Squibb, Lilly, Merck-Serono, Mylan, Novartis, Roche, Sanofi-Genzyme, Teva, and UCB; and support to attend scientific meetings from Biogen, Bial, Merck-Serono, Novartis, Roche, Sanofi-Genzyme, and Teva. JLSM has received support to attend scientific meetings from Novartis, Merck, and Biogen; speaking honoraria from Biogen, Novartis, Sanofi, Merck, Almirall, Bayer, and Teva; and has participated in clinical trials from Biogen, Merck, and Roche. FJBH has received compensation for consulting services and speaking honoraria from Almirall, Biogen, Genzyme, Merck-Serono, Novartis, Roche, Sanofi, and Teva. CC has received compensation for consulting services, speaking honoraria and support to attend scientific meetings and courses from Merck, Teva, Sanofi-Genzyme, Novartis, Biogen, Roche, and Bristol-Myers-Squibb. LB has received compensation for consulting services, speaking honoraria and support to attend scientific meetings from Bayer, Celgene, Biogen, Genzyme, Merck, Novartis, Roche, Almirall, and Teva. JDGS has received compensation for consulting services and speaking honoraria from Biogen, Novartis, Merck, UCB, Sanofi-Genzyme, Roche, Almirall, and Teva. MCA has received compensation for consulting services from Genzyme, Roche, Novartis, Sanofi, and Biogen. LNC has received compensations from Sanofi-Genzyme, Merk, Biogen, and Roche. EA has received speaking honoraria from Roche, Novartis, Merck, Sanofi, and Biogen. MGR has received speaking honoraria from Biogen, Sanofi, Almirall, and Novartis. OC has participated in studies and has received speaking honoraria from Roche, Merck, Biogen, and Novartis. LGT has received speaking honoraria from Biogen, Novartis, Merck, Bayer, Sanofi-Genzyme, Almirall, Roche, and Teva. MH has participated in observational studies and has received compensation for consulting services and speaking honoraria from Roche, Merck, Sanofi, Biogen, Novartis, and Bayer. The rest of the authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Roche Medical Department, Spain (ML42064). The funding source had no role in the design of this study, data analysis and interpretation, review and approval of the manuscript or the decision to submit for publication.
