Abstract
Rationale
To determine the prevalence of epileptic seizures in multiple sclerosis (MS) at an Australian tertiary hospital and to define their clinical features.
Methods
We retrospectively analysed adult patients at the Royal Melbourne Hospital electronically identified to have ICD codes for MS and seizures and/or epilepsy between 1996 to 2019, utilising paper and electronic-based records.
Results
Of the 2,125 MS patients identified, 16 (0.75%) experienced epileptic seizures during a mean follow-up period of 12.9 years. Median age of MS diagnosis (SD) was 38 (9.3) years. Four patients had relapsing remitting MS (25%), 10 secondary progressive MS (63.5%), and 2 primary progressive MS (12.5%). More than two-thirds of patients had seizure onset following the diagnosis of MS, and the majority of these had advanced disease (approximate EDSS >6) at the time of seizure onset. Focal onset-seizures occurred in 87.5% of patients with seizures.
Conclusion
The estimated prevalence of seizures in our cohort was lower than in previous studies (0.75 vs 2–4%). In most cases, seizures occurred after the diagnosis of MS in the context of advanced disease. Further studies are required to determine if MS disease modifying treatments reduce the risk of seizures in this cohort.
Introduction
Previous studies suggest that the prevalence of epileptic seizures in multiple sclerosis (MS) is 2–4%.1–3 Although the risk of seizures is thought to be higher in patients with more active MS and longer disease duration than in other MS groups, seizures have been reported in all MS types (relapsing remitting and progressive forms). However, they are uncommonly the presenting symptom of MS.1,4 Imaging-based studies have demonstrated a higher number of cortical lesions and greater cortical atrophy in MS patients with epilepsy that in those without, postulating that epileptogenicity may arise from juxtacortical and grey matter damage.5,6 However, the exact pathophysiology of epileptogenicity in MS has not been fully elucidated, and there is likely a dynamic interplay between cortical and juxtacortical inflammation, demyelination, cortical grey matter damage and neuroanatomical factors 7 in the development of seizures and epilepsy. It has been shown recently in a large population-based register that the risk of epilepsy increases with more advanced types of MS. 3 However, despite a greater cortical lesion load being a risk factor for developing seizures, not all patients with cortical lesions have epilepsy8,9 and previous studies have not consistently shown that a greater proportion of patients with advanced MS types have epilepsy.2,6,8,10,11 This raises the possibility that cortical lesions may be a surrogate risk factor for epilepsy rather than being directly related to the development of epilepsy. In addition, more recent studies have also shown a lower prevalence than older studies, speculating that newer, higher efficacy disease modifying therapies may affect epileptogenesis.12,13
In this first study of an Australian MS cohort, we aim to estimate the prevalence of seizures/epilepsy in patients with MS in a large tertiary hospital. We also characterize seizure types and assess the relationship between the seizure/epilepsy occurrence and MS disease course.
Subjects and methods
After approval by the Human Research Ethics Committee at our institution (Approval number QA2019096), a cohort of MS patients who met the International Classification of Disease (ICD) codes for ‘MS’ (G35) and ‘epilepsy’ (G40), and ‘MS’ and ‘seizures (G41)’ were identified from a single tertiary health centre between 1996 and 2019, and were analysed retrospectively. A combination of electronic and paper based medical records, including iMED (www.imed.org) and NeuroBase databases (for multiple sclerosis and epilepsy respectively) were used to obtain clinical information. After confirming the diagnosis of MS, seizures and epilepsy by medical records' review, we collected: demographic data, age at seizure onset, age at MS and seizure/epilepsy diagnosis, MS classification (relapsing remitting multiple sclerosis, RRMS; secondary progressive multiple sclerosis, SPMS; primary progressive multiple sclerosis, PPMS), MRI characteristics (e.g. presence of atrophy on MRI report), and electroencephalogram (EEG) characteristics. Seizures were classified according the 2017 International League Against Epilepsy (ILAE) classification of seizure types. 14 Due to incomplete data, it was not always possible to determine if the diagnoses of MS were made according to the Poser 15 or McDonald 16 criteria. We only included patients with MS, and excluded patients with clinically isolated syndrome or ‘probable MS’. Data collection was deidentified and collated using Microsoft Excel version 16.29.
Results
Between 1996 and 2019, a total of 2125 patients met ICD codes for MS, and of these, 22 were identified to also have an ICD code for seizures or epilepsy. Six patients were subsequently excluded: five did not actually have evidence of MS (n = 1) or seizures (n = 4) on medical records’ review, and one patient was found to have psychogenic non-epileptic events (demonstrated on video-EEG monitoring) rather than epileptic seizures.
Thus, the prevalence of seizures/epilepsy in our MS cohort was 0.75% (n = 16/2125), with a mean follow-up (SD) of 12.9 (8.4) years. Of these 16 patients, 15 had epilepsy, yielding a prevalence of epilepsy in our MS cohort of 0.70% (15/2125).
Demographic and clinical data for the 16 patients with seizures/epilepsy are presented in Tables 1 and 2 Most cases were female (n = 12, 75%). Median age of MS diagnosis (standard deviation, SD) was 38 (9.3) years. EDSS data was available for 10 patients, with a median EDSS (SD) of 6.4 (2.5). The remainder had a clinical description suggestive of advanced MS disease. Of the 16 patients, 5 patients had RRMS (31%), 10 SPMS (62.5%), and 2 PPMS (12.5%). Eleven patients had seizures following MS diagnosis with a median time (SD) to seizure onset of 9.0 (6.9) years, whereas six had antecedent seizures, with a median time (SD) from seizure onset to MS diagnosis of 10.0 (11.1) years. A breakdown of demographic and clinical characteristics of the patients can be found in Tables 1 and 2, and a summary of the clinical characteristics can be found in Table 3.
Demographic and clinical characteristics of patients with MS and seizures/epilepsy.
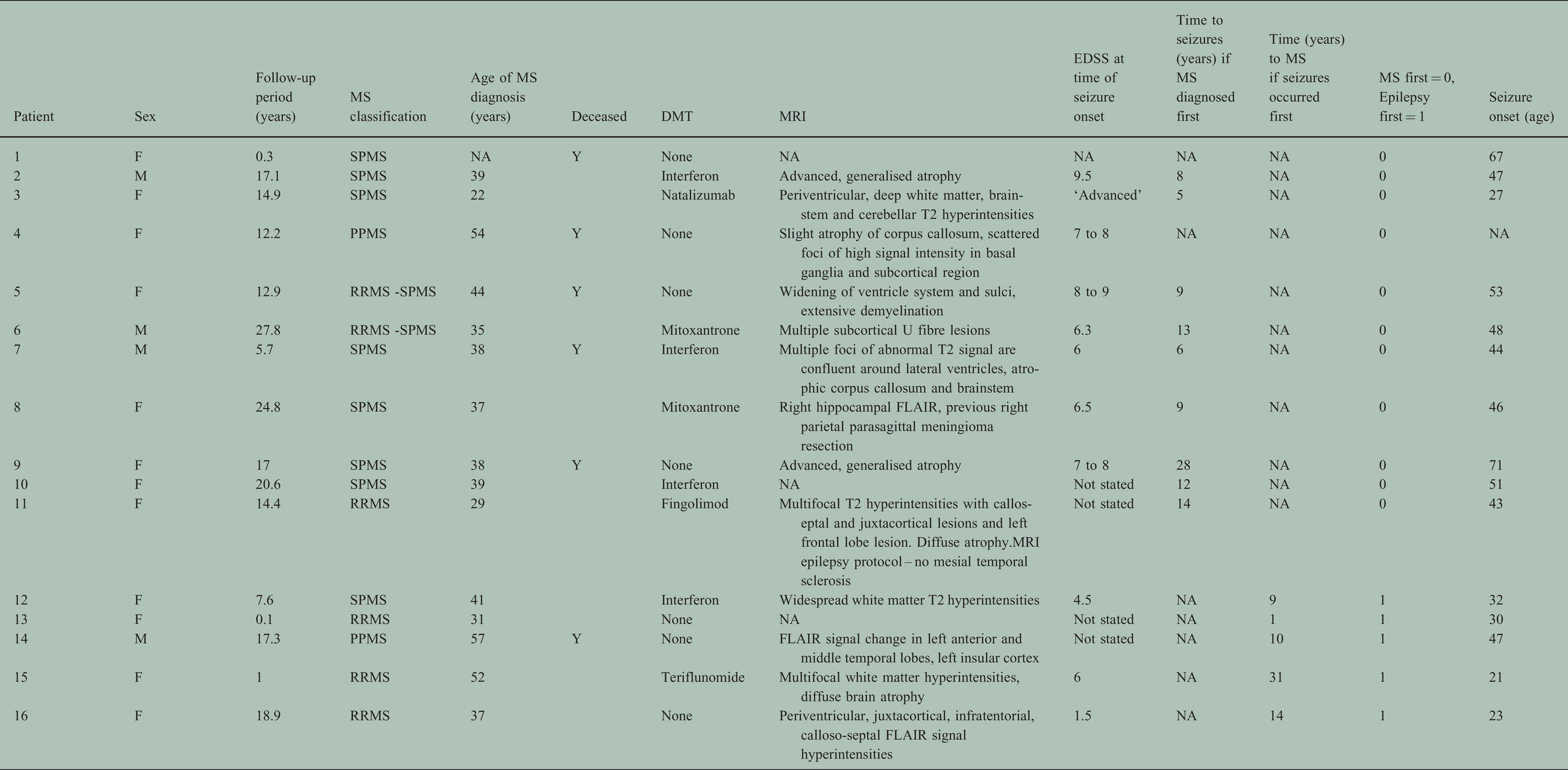

In Table 2, * denotes that patients in whom epilepsy/seizures occurred prior to MS diagnosis.
Abbreviations: M – male, F – female, NA – data not available, RRMS – relapsing remitting multiple sclerosis, SPMS – secondary progressive multiple sclerosis, PPMS – primary progressive multiple sclerosis, DMT – disease modifying therapy, FLAIR – fluid attenuated inversion recovery, EDSS – expanded disability status score, FIAS – focal impaired awareness seizures, FBTCS – focal to bilateral tonic-clonic seizure, FMS – focal motor seizure; PNES – psychogenic non epileptic seizure, GTCS – generalised tonic clonic seizure, NCSE – non-convulsive status epilepticus, NOS – not otherwise specified, PHT – phenytoin; LEV – levetiracetam; CZP– clonazepam; TPM – topiramate;CLB – clobazam; CBZ – carbamazepine; VPA – valproate, EEG – electroencephalogram, UTI – urinary tract infection, DVT – deep vein thrombosis, SPC – suprapubic catheter, GORD – gastro-oesophaegeal reflux disease, IHD – ischaemic heart disease.
Summary of characteristics of patients with MS and seizures.

Abbreviations: MS – multiple sclerosis, RRMS – relapsing remitting multiple sclerosis, SPMS – secondary progressive multiple sclerosis, PPMS – primary progressive multiple sclerosis, DMT – disease modifying therapy, FIAS – focal impaired awareness seizure, FAS – focal aware seizure, NOS – not otherwise specified, PNES – psychogenic non-epileptic seizures, EEG – electroencephalogram, AED – anti-epileptic drug.
Fourteen patients (87.5% of all those with seizures) experienced focal-onset seizures, with focal impaired awareness seizures (FIAS) being the most common seizure type (7/14 patients). Two patients had focal status epilepticus and six had focal to bilateral generalised tonic-clonic seizures. One patient had both focal-onset seizures and psychogenic non-epileptic events. Of the two patients without focal-onset seizures, one patient had generalised-onset seizures and the other had unclassified seizures. Of the 11 patients who experienced seizures after a diagnosis of MS, 8 had epilepsy presumably related to MS (7 of whom had focal epilepsy, 1 had unclassified epilepsy), one had an acute symptomatic seizure in the setting of sepsis, one had post-stroke epilepsy and one had tumour-associated epilepsy (Table 2). Of these patients, seven had been on MS disease modifying therapy (DMT): interferon (3 patients), mitoxantrone (2 patients), fingolimod (1 patient) and natalizumab (1 patient). Of the five patients who had seizures prior to the diagnosis of MS, three had non-lesional focal epilepsy, one had acquired focal epilepsy due to brain infection, and one had generalised epilepsy. A breakdown of the seizure and epilepsy diagnoses can be found in Figure 1

Epilepsy and seizure diagnosis.
EEG results was available for 10 of the 16 patients with seizures. Of these, seven had focal abnormalities such as focal slowing or interictal epileptiform discharges; in six of these cases, these abnormalities were found over the temporal or fronto-temporal regions. Fourteen patients were on anti-epileptic drugs (AEDs), of whom seven were on 1 AED, five on 2 AEDs and two on 3 AEDs.
Discussion
We present the first Australian study examining the prevalence of seizures/epilepsy in MS patients, ascertained at a large tertiary health centre. The estimated prevalence was 0.75%, which is lower than previously reported (2-4%),1,2 with an average follow-up time of 12.9 ± 8.4 years. In most cases, the diagnosis of MS preceded the onset seizures/epilepsy, and focal seizures were the most common broad seizure type.
The ICD cohort of patients we studied appeared to fall into two broad clinical groups. The first group, consisting of 8 of the 16 patients (50%), represent those who have focal epilepsy or unclassified epilepsy without any other competing aetiology aside from MS. In this group of patients, EEGs showed focal abnormalities such as slowing or interictal epileptiform discharges in 4 of the 8 (50%) patients. The majority (7 of 8 patients, 87.5%) of patients whose focal epilepsy could have been due to MS had SPMS, suggesting a trend towards an increased risk for the development of focal epileptogenic lesions with progressive disease. In line with previous post-mortem and imaging studies that have found a higher proportion of cortical lesion load in the temporal regions in those with MS and seizures,6,17 3 of 8 patients had EEG evidence of temporal lobe dysfunction or epileptogenicity. The remainder of 3 patients had generalised or non-temporal region slowing, and the remaining two did not have EEG. However, somatotopic analysis in our cohort was limited by high quality, epilepsy-specific MRI for assessment of cortical lesions, and although 2 patients also had epilepsy-specific MRI, they did not yield additional information. Previous studies have shown that 65% to 100% of patients have focal-onset seizures,4,5,10,13,18 not dissimilar to our cohort of patients who had MS preceding their seizure onset, of whom 87.5% had focal-onset seizures. Together with the EEG findings in this group that showed that half of the patients had focal abnormalities on their EEG, our study supports the notion that focal epilepsy is the most common type of epilepsy in those who have epilepsy attributable to MS.
The second group of patients represented those who have another established cause of epilepsy which either predated or followed their diagnosis of MS. Most had focal epilepsies, which were attributable to acquired brain insults, namely stroke, tumour, and encephalitis. Three patients had non-lesional temporal lobe epilepsy had epilepsy diagnosed before MS diagnosis and one had generalised epilepsy with variable interval between first seizure onset and MS diagnosis (median 10, SD 11 years) and with no evidence to attribute MS as the cause.
More than 80% of patients were on AED therapy, and of these patients, half were on one AED only. It is difficult to ascertain who achieved seizure freedom due to inadequate follow-up information. Carbamazepine was the most commonly used AED, followed by phenytoin. This is reflective of a typically older cohort that was studied as AED selection has evolved and other agents with improved tolerability are now available. Given that carbamazepine is also used for other non-epileptic paroxysmal symptoms in MS such as tonic spasms and trigeminal neuralgia, we were cautious in the medical record review to ensure that the indication of carbamazepine was epilepsy. It is noted that previous studies in tertiary centres have shown that approximately half of patients are not seizure-free on two or more agents.4,12 In our cohort, 47% of patients needed two or more AEDs, but data are lacking as to whether this suggests drug resistance per the current accepted definition. 19 In our study, although neuroimaging is limited by its assessment of cortical lesions, the presence of MS related subcortico-cortical inflammation and atrophy may be associated with focal and recurrent predisposition to seizures. In our cohort, all 8 patients with focal or unclassified epilepsy with no competing diagnosis but comorbid MS were on at least one AED.
Older studies have reported higher prevalence of seizures in patients with MS compared to our investigation, although more recent studies have shown a reduced prevalence compared to older studies.2,5,12,13 In our study, we were able to determine which patients had an acute provoked symptomatic seizure compared to those who had epilepsy, which is a brain disease characterised by an enduring predisposition to generate seizures. 14 Excluding the one patient with acute symptomatic seizures, the estimated prevalence of epilepsy in our cohort is 0.70%, which is lower than previous studies,2,5,12,13 many of which combined epilepsy and seizures (including acute symptomatic seizures) in the calculation of prevalence estimates. A recent meta-analysis found the lifetime prevalence of epilepsy of 0.76% (76 per 1000 persons), suggesting that our estimated prevalence is similar to the prevalence of epilepsy in the general population. However, this comparison must be made with caution given methodical differences between our study and the literature, and that Australian studies were excluded in this meta-analysis. 20
There are several reasons why we found a lower prevalence of epilepsy/seizures in MS patients. Firstly, as the largest tertiary centre for MS in the state, our MS cohort includes patients that span the entire spectrum of MS disability from mild disease, low EDSS, to advanced disease. Many of our patients are on DMT, raising the question of the potential influence of DMT on preventing seizures. It remains unclear as to how seizures are generated in MS, although it has been proposed that increased levels of IL-18 and its effect in increasing interferon gamma may play a role in epileptogenicity 21 and other autoimmune conditions have been demonstrated an increased odds of developing epilepsy, 22 suggesting an underlying inflammatory mechanism to seizure generation. Although not specifically evaluated in this study, it is possible that controlling MS related inflammation may have beneficial effects on reducing risk of developing epilepsy and the low prevalence may be indirectly related to a proactive approach in using DMTs where appropriate.
The comparatively lower prevalence relative to other single centre studies may have in part been due to the use of ICD codes for MS which may have inadvertently inflated the denominator by capturing patients who may not actually have ‘definite MS’. With reliance on medical documentation, there is a chance of under-reporting and also over-reporting with difficulty separating non-epileptic paroxysmal conditions related to MS from seizures, such as vertigo, tonic spasms and somatosensory symptoms. In our dataset, one patient with tonic spasms of the leg also had seizures and were therefore included in the ICD analysis. Indeed, when more stringent diagnostic criteria are used and other causes of seizures are excluded, Nyquist et al showed in an population-based epidemiological study that seizures do not occur at an increased incidence in MS. 8
Although none of our patients were confirmed to have seizures as a presenting feature of MS or relapse, seizures have been suggested to be a potential presenting symptom of MS relapse. By performing a brain MRI within 72 hours of seizure occurrence, Calabrese et al found new intracortical lesions in 5 out of 6 patients with known MS and epilepsy. 6 However, MRI Is typically not an urgent inpatient investigation in patients presenting with provoked acute symptomatic seizures or single unprovoked self-limiting seizures with or without a history of MS, thus identification of focal epileptogenic lesions due to MS may not be identified in this setting.
There are several limitations of this study. Firstly, the ICD data analysed only captured the subset of patients with both MS and epilepsy who have presented to the emergency department or those who were admitted as inpatients. Most patients seen in our MS centre are managed as outpatients. Similarly, the ICD may not capture all patients with an established diagnosis of epilepsy who present with a first seizure, as their MS neurologists may manage their seizures or they may be referred directly internally to the epilepsy unit. This raises the possibility of selection bias as those in the ambulatory setting were not included. As a result, it is likely that patients with both MS and epilepsy at our institution were not included in our dataset, which would have impacted on the estimated prevalence. The ICD may also selectively capture patients higher EDSS and those with progressive forms of MS, as these patients were more likely to present to the emergency department or require inpatient treatment for a range of complications related to advanced MS (e.g. infections, malfunction of indwelling catheters or percutaneous feeding tubes). Furthermore, although all patient records from the ICD dataset were examined to ensure they met the inclusion criteria, the identification of patients to include in the dataset was firstly dependent on the correct ICD designation for MS, epilepsy and seizures by the coder, and patients who were incorrectly coded would have been missed in our dataset. Thus, despite demonstrating a trend towards an increased prevalence of epilepsy with progressive forms of MS, due to the small sample size and heterogeneity of patients, results from our study cannot be generalised to all populations of patients with MS.
A further limitation of this study was the availability of neuroimaging of our patients. All MRI protocols and sequences are intrinsically poor at the evaluation of cortical grey matter,17,23 and conventional T2 weighted MR sequences are not sensitive to intracortical lesions due their longer relaxation times and low contrast resolution compared to grey matter. 8 Cortical lesions have been found to be more common in those with RRMS with seizures compared to RRMS patients without seizures.24,25 Neuropathology studies have also found that patients with PPMS are more likely to have a greater percentage of cortical demyelinating lesions compared to relapsing remitting forms. 9 Although, our study is not dissimilar in suggesting that advanced disease is associated with a greater propensity for seizures, it is likely that location of lesion is as important as MS type. Serial MRI would not have altered treatment decisions for many of the SPMS patients in our cohort given they were not on DMT. Although our centre now uses 3 T volumetric brain MRI protocol in all MS patients, only two patients had epilepsy-specific MRI, which would have better captured cortical lesions or provided fine slices of the hippocampal regions.
In conclusion, our study from a large Australian tertiary health centre which captured retrospective data for 10 years revealed a low prevalence of MS and epilepsy/seizures in this population. Whether MS lesions are directly linked to a developing a seizure focus and whether control of MS with DMTs can influence the propensity for epilepsy or seizures is unclear and warrants further studies.
Footnotes
Acknowledgements
The authors are grateful to Smisha Thomas for extracting of ICD10 data.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SO declare no conflict of interest. TK served on scientific advisory boards for Roche, Sanofi Genzyme, Novartis, Merck and Biogen, steering committee for Brain Atrophy Initiative by Sanofi Genzyme, received conference travel support and/or speaker honoraria from WebMD Global, Novartis, Biogen, Sanofi-Genzyme, Teva, BioCSL and Merck and received research or educational event support from Biogen, Novartis, Genzyme, Roche, Celgene and Merck. PP is supported by the NHMRC (Early Career fellowship, APP1163708), the Royal Australasian College of Physicians, Melbourne Health, and the Australian Epilepsy Research Fund. His institution has received speaker honoraria or consultancy fees from Eisai, UCB Pharma, Sun Pharma, Novartis, and Supernus. He is an Associate Editor for Epilepsia Open. MM’s institution receives funding from Merck and from National Health and Medical Research Council. MM served on scientific boards for Merck and received conference and speaker honoraria from Merck and Biogen.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
