Abstract
Background
Mental disorders (MDs) in multiple sclerosis (MS) patients decreases treatment adherence and quality of life, and increases the risk of disability progression and care consumption.
Objective
This study was to assess the prevalence of MDs in MS patients compared with healthy controls (HC) of the French general population and rheumatoid arthritis (RA) patients.
Methods
The 2015 prevalence of MDs for MS patients, RA patients and general population was estimated using a random population-based data sample from ‘National Inter-Scheme Information System on Health Insurance’ in the 2011–2015 period. Two control groups (1:5 ratio for the HC and 1:1 for the RA group) were matched to the MS group for year of birth, gender, area of residence and health insurance scheme.
Results
A total of 1145 MS patients were identified (sex ratio of 2.5 (F/M); median age 50 years). The prevalence of MDs was higher in the population of patients with MS (37.3%) than in the French general population (13.6%), and to a lesser extent in the RA group (21.1%) leading to the prevalence ratios of 2.8 (95% confidence intervals (CIs) 2.5–3.0) and 1.9 (95% CI 1.7–2.3), respectively.
Conclusions
This study confirmed that MS patients are at a higher risk of MDs than the French general population or RA patients.
Introduction
Multiple sclerosis (MS) is a demyelinating autoimmune disorder of the central nervous system that mostly starts in people in their mid-20s to 40s. This long term disease affects 2.3 million individuals worldwide, 1 and while it only mildly reduces life expectancy, 2 it does lead to a progressive accumulation of various impairments (such as motor, sensitive and cognitive functions). 3 Currently, 12 disease-modifying therapies (DMTs) have received marketing authorization for MS patients in France. These treatments aim to reduce the occurrence of relapses and the progression of disability.
Mental disorders (MDs) have been reported to be more frequent in MS patients than in the general population.4,5 This may be related to common physiopathological pathways in MS or may be related to the consequences it has on quality of life and disability. The presence of MDs in MS patients can delay diagnosis,6,7 increase disability progression, 8 decrease quality of life9,10 and the adherence to DMTs. 11 To our knowledge, only one study has been published, to date, regarding comorbidities of MS patients in France, however, the study was not specific to MDs. 12
To describe MDs associated with MS in France, the use of health administrative databases are relevant. National health insurance databases are exhaustive with a 98% coverage 13 and are unbiased as there are no recruitment criteria in terms of wealth or age. Being population based, the databases also provide an opportunity to select controls from the general population such as healthy controls (HCs) or individuals with other chronic diseases such as rheumatoid arthritis (RA). RA is a chronic non-brain related disease that affects more than 15 million individuals worldwide 14 and shares similar commonalities as MS. Both are auto-immune diseases, affecting both men and women, that can lead to a range of physical symptoms or impairments such as fatigue, pain and motor disabilities, and can have varying levels of severity over time.15,16
The objective of this study was to assess the prevalence of MDs in MS patients compared with a group of HC and patients with RA from the general population in France.
Materials and methods
Study design and data sources
A matched cohort study was conducted to compare the prevalence of MDs between MS patients and two control groups (HC and patients with RA).
Data from the ‘National Inter-Scheme Information System on Health Insurance’ (SNIIRAM) were compiled regarding care reimbursements for each individual covered by the health insurance system. This data accounted for 98% of the French general population. 13 All data regarding outpatient care in private practice were included and linked to the national hospital discharge databases of all public and private hospitals. 17 For this study, we used the Echantillon Généraliste des Bénéficiaires (EGB), a 1/97th random sample of the SNIIRAM including more than 20 years of follow-up. 18 It contains anonymous sociodemographic characteristics (date of birth/death, gender, area of residence) and medical data such as the presence of a long-term disease (Affection de Longue Durée [ALD]), ambulatory care reimbursement (including visits to a general practitioner [GP], specialists and drug deliveries) and in-hospital data (which are all admissions including 1-day hospitalizations). Unlike the SNIIRAM database, the EGB sample did not contain data regarding the different types of hospitals and only included data regarding medicine, surgery and obstetrics (MCO) wards, while psychiatric hospitals and follow-up and rehabilitation care hospitals are not included.
Study population
MS patients between 1 January 2011 and 31 December 2015 were included in the study. The selection period was set to 5 years due to the potential absence of MS activity for several years and consequent lack of use for health services.
An individual was considered to have MS if at least one of the following criteria was met19,20,21: (a) an MS ALD status (ALD with an International Classification of Diseases 10th version (ICD-10) with a diagnosis code ‘G35’) 21 ; (b) at least one DMT specific for MS (such as beta interferon, glatiramer acetate, dimethyl fumarate, fingolimod, teriflunomide, or natalizumab); (c) at least one hospital admission with an ICD-10 diagnosis code ‘G35’.
For each MS patient, up to five HC from the general population and 1 RA patient (due to the limited prevalence of RA 22 ) were identified. The matching criteria were: gender, year of birth, area of residence and health insurance scheme17 (which were under general scheme, agricultural workers, self-employed workers and other). Anyone with a demyelinating disease (optic neuritis, transverse myelitis, unspecified demyelinating disease, or neuromyelitis, with ICD-10 codes ‘H46’, ‘G36’, or ‘G37’) was excluded from the study to eliminate the risk of misclassification (the possibility of MS incorrectly considered as a control). MS patients with RA were also excluded (n = 5). Some criteria combinations increased the difficulty to identify the controls. When five controls could not be found, the criterion regarding the health insurance scheme was relaxed. For the RA group, the insurance scheme criterion was deleted, and if the match was still unfeasible, the age criterion was relaxed to ± 5 years instead of ± 1 year, and the area of residence was extended to a region rather than a department.
Prevalence of mental disorders
On 31 December 2015, an MD was deemed to be present, when at least one of the three following criteria was met in 2015:
an ALD status for a psychiatric affection (an ALD diagnosis with ICD-10 corresponding to F2X for schizophrenia, schizotypal and delusional disorders; F3X for mood (affective) disorders; F4X for neurotic, stress-related, and somatoform disorders; and F50 for eating disorders); at least one hospital admission in relation to an MD between 2011 and 2015 (the ICD-10 codes were the same as for the ALD status: F2X, F3X, F4X, and F50); at least two reimbursements for treatment associated with an MD. The specific codes for the treatments were identified with the Anatomical Therapeutic Chemical (ATC) classification and are specified in Table 1. A list of antidepressants (N06A), antipsychotics (N05A) and anticonvulsants in combination with mood-stabilizing agents (N03A and N05A) were compiled in this study. The choice was based on medications specific to a MD. Anxiolytics were excluded as they entailed a significant risk of misclassification due to their frequent use for stress or insomnia. In the case of anticonvulsants, anyone exhibiting epilepsy was also excluded to avoid misclassification.
List of the ATC codes considered to identify treatments related to mental disorders.

The criteria were defined based on scientific literature23,24 and after consulting with a psychiatrist to adapt the choices derived from previous studies, for instance, the inclusion or exclusion of treatments in France and Canada when they had different indications. 25
In each group, the prevalence of MDs was defined as the number of individuals with an MD in 2015, divided by the total number of people in the group. The prevalence ratio (PR) was estimated by dividing MS prevalence by each control group (HC and RA, respectively) with the associated 95% confidence intervals (CI). Venn diagrams were made to document the prevalence of MDs according to three different sources of identification (ALD status, hospital admission or reimbursed treatments). Moreover, each group was described in terms of global health consumption with descriptive statistics (proportions or medians with interquartile range and associated statistical tests). For each type of care, two indicators were computed, which were the number of patients receiving the care at least once over the study period and the frequency or duration of care. PRs were estimated for all groups, and then stratified by gender and age subgroups.
Sensitivity analyses were conducted to explore the impact of possible misclassification on estimates and assess the impact of more restrictive and specific definitions in order to exclude potential misclassification bias and over-estimation. Several definitions for MDs were developed by varying the number of prescriptions or the list of treatments, to assess the impact they had on the results against the initial definition. The minimum number of treatment reimbursements were firstly increased to four, instead of two. Treatments that were also indicated for treatment of neuropathic pains (N06 codes in the ATC classification) were then excluded. The PRs between two age groups (0–65 and ≥65 years of age) were finally compared, to explore the impact of antipsychotic treatments (N05A codes) that might have been prescribed for elderly individuals, in case of behavioural problems and/or agitation.
Analyses were performed with SAS Enterprise Guide 4.3 software. The study received data management approval (authorization DE-2017-026) by the French Data Protection Authority (Commission Nationale de l'Informatique et des Libertés, CNIL).
Results
Characteristics of the three study groups
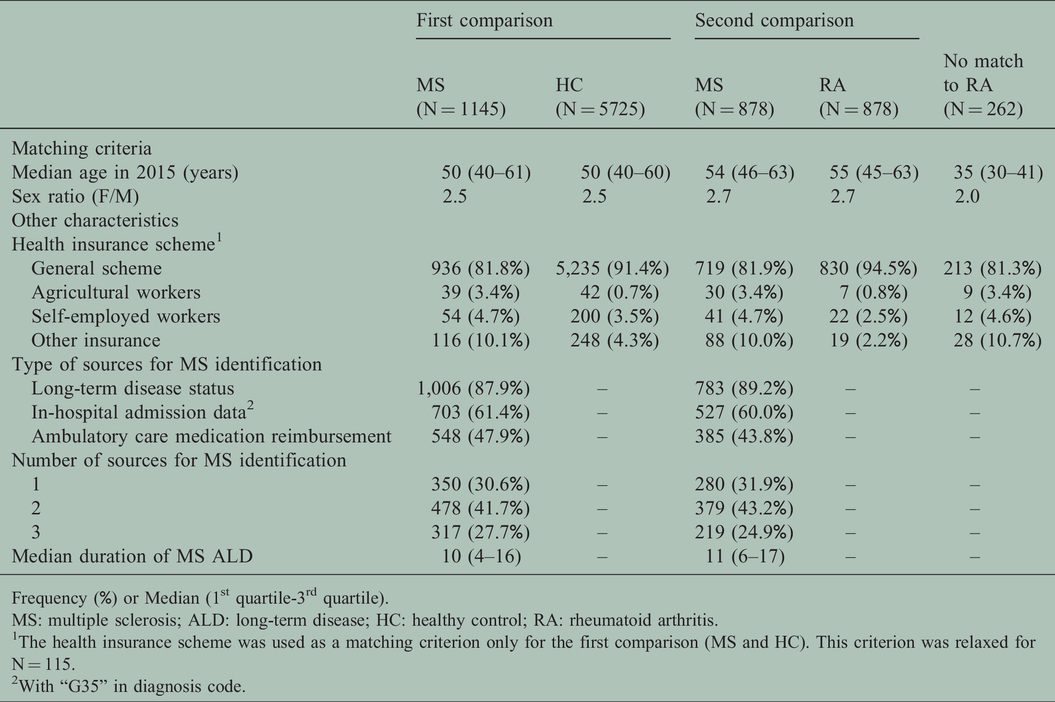
As shown in Table 2, 1145 MS patients were identified and matched to the 5725 HCs (the criterion regarding the health insurance scheme was relaxed for 115 MS cases); 878 MS patients matched with 878 RA patients (704 MS patients matched with the relaxed criterion). In the MS population, the sex ratio (F/M) was 2.5 and the median age was 50 years. Most individuals had an MS ALD status (n = 1006; 87.9%) for a median duration of 10 years. Half of them received at least one DMT (n = 548) over the study period, and 69.4% were identified by at least two data sources. MS patients that could not be matched with the patients with RA group (n = 262) were younger (median age 35 years) and with a sex ratio (F/M) of 2.0.
Characteristics of the three study groups: MS patients, HC, and patients with RA.
Frequency (%) or Median (1st quartile-3rd quartile).
MS: multiple sclerosis; ALD: long-term disease; HC: healthy control; RA: rheumatoid arthritis.
1The health insurance scheme was used as a matching criterion only for the first comparison (MS and HC). This criterion was relaxed for N = 115.
2With “G35” in diagnosis code.
Prevalence of mental disorders
The prevalence of MDs in the three study groups are shown in Table 3 and the PRs are presented in Figure 1(a) and (b). Prevalence was higher among the MS patients than in the HC (37.3 vs. 13.6%; p-value <0.0001) and to a lesser extent in the patients with RA (41.1 vs. 21.1%; p-value <0.0001). Therefore, the PRs of MDs were 2.8 (95% CI 2.5–3.0) and 1.9 (95% CI 1.7–2.3) compared with the HC and patients with RA group, respectively.
Characteristics of patients with MDs in the three different study groups (MS patients, HC, and patients with RA.

Frequency (%) or Median (1st quartile-3rd quartile).
MS: multiple sclerosis; MD: mental disorder; ALD: long-term diseases; HC: healthy control; and RA: rheumatoid arthritis.
1 Several diagnoses were made over the same hospital admission (three types of diagnosis: principal, related, or associated with admission).
2 Number of boxes delivered with prescription.
3 Number of distinct active substances (drugs) among all of the reimbursements.
4 Median number of visits between 2011 and 2015.
5 Statistical significance was set at p-value = 0.004 (Bonferroni correction for 13 tests of association).
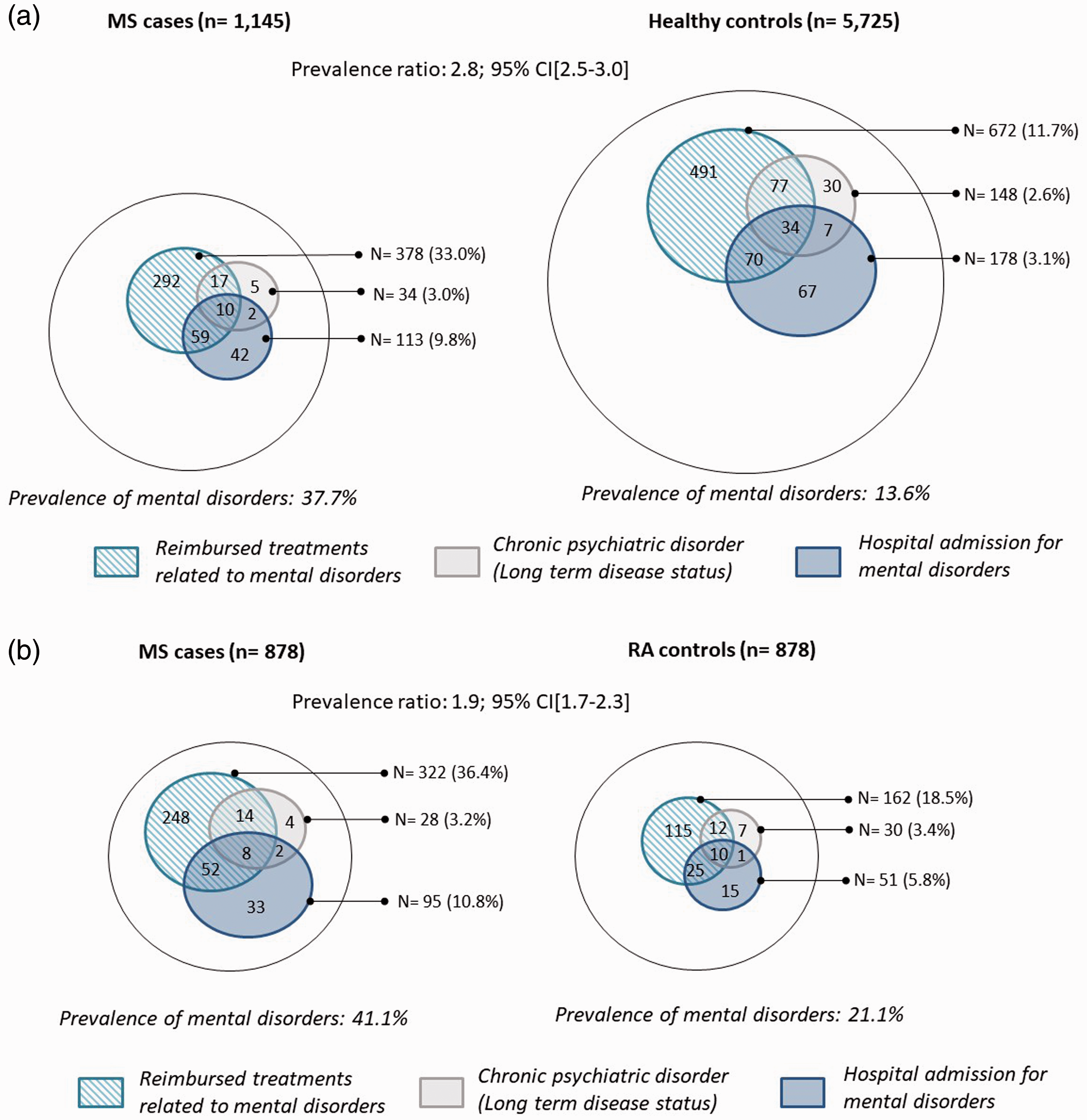
(a) Venn diagrams illustrating the frequency of mental disorders (MDs) for multiple sclerosis (MS) patients and the healthy controls (HC), according to various data sources in 2015. (b) Venn diagram illustrating the frequency of mental disorders (MDs) for multiple sclerosis (MS) patients and the rheumatoid arthritis (RA) controls, according to various data sources in 2015.
An ALD status was attributed to approximately 3% of the individuals identified with MDs, with a median duration of 8 years for MS patients, 7 years for the HC group and 5.5 years for patients with RA. Mood disorders (F3X) were the most frequent diagnosis in all three groups. Regarding hospital admissions, MS patients were admitted more often for MDs than the HC (9.8% vs. 3.1%, respectively; p-value < 0.0001) and with a median length of stay of 5.5 vs. 3.2 days for the HC. Similar results were observed with patients with RA. The distribution of diagnoses was the same between MS patients and matched groups, except for mood disorders (F3X), which were more frequent in patients with RA.
Most MD cases were identified by the treatment source. MS patients were more often prescribed drugs for MDs (33.0 vs. 11.7% of HC and 18.5% of patients with RA; p-value < 0.0001). The frequency of reimbursements, as well as the number of drugs dispensed per patient did not differ between the three groups. The drug prescriber was usually a GP (approximately 77%), irrespective of the group. For MS patients, a neurologist generated a significant portion of prescriptions (13%), whereas a psychiatrist accounted for approximately 5% of the prescriptions. For both matched groups, a psychiatrist was the second prescriber (20% for the HC and 15% for patients with RA), and as expected, the neurologist generated less than 1% of the prescriptions. The number of GP consultations, which was a general proxy of healthcare consumption in each group, was higher for MS patients than for the HC (median of 25 vs. 19; p-value < 0.001), and was higher for patients with RA than for MS patients (median of 30 vs. 26; p-value < 0.001).
In the gender-stratified analysis (Figure 2), the PRs were higher in men than women, for both matched groups. Regarding age groups, the highest PR was observed in the youngest group (≤ 40 years) for MS patients compared with the HC, and the oldest age group (> 60 years) for MS patients compared with patients with RA (Figure 3).
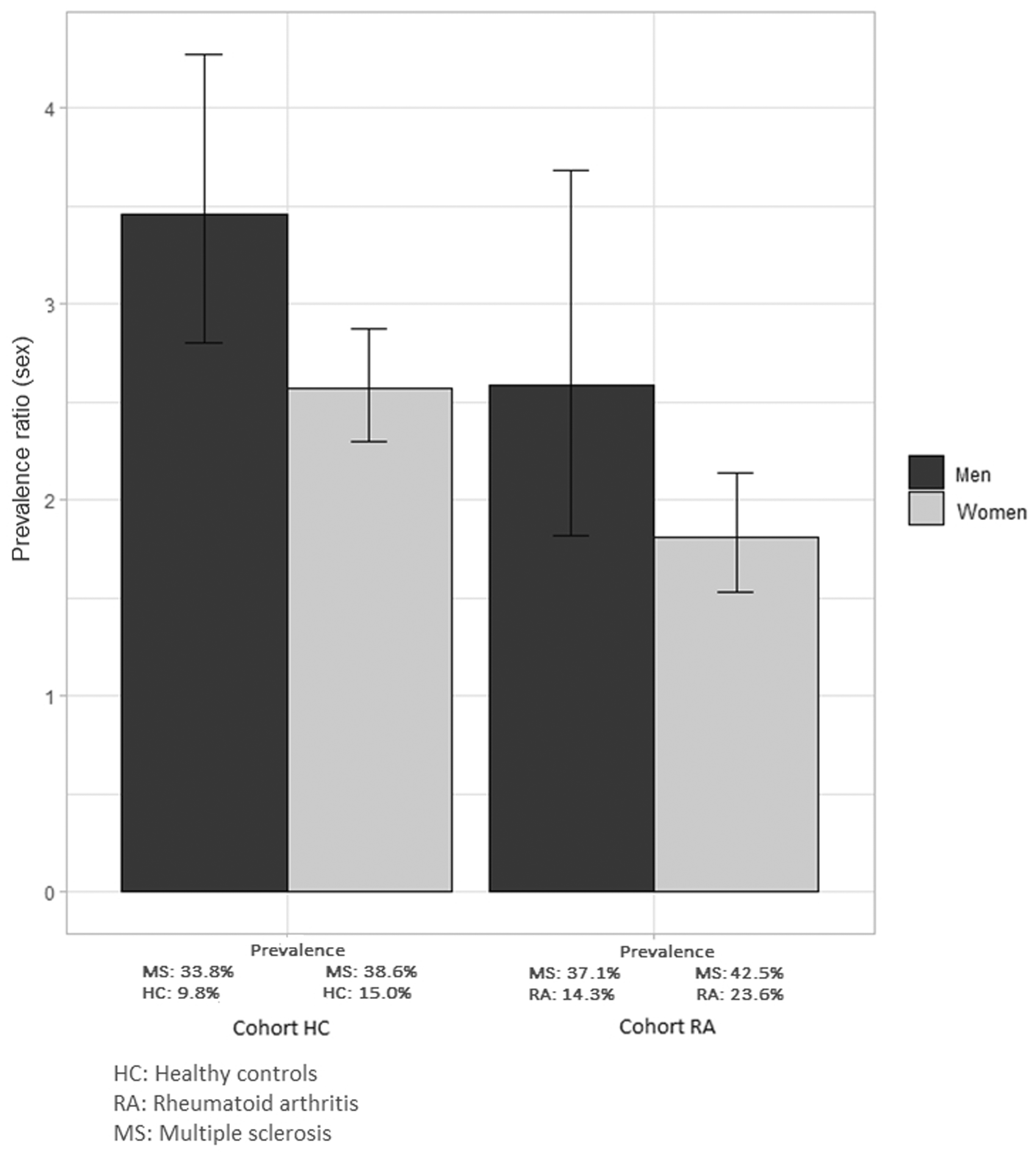
Prevalence ratios of mental disorders by gender in multiple sclerosis (MS) patients compared with the two control groups (the healthy controls [HC] and patients with rheumatoid arthritis [RA]).

Prevalence ratios of mental disorders by age group in multiple sclerosis (MS) compared with the two control groups (the healthy controls [HC] and patients with rheumatoid arthritis [RA]).
Sensitivity analyses
When the minimum number of reimbursements for treatments related to MDs was increased, no difference was found, with a PR of 2.8 (95% CI 2.5–3.1) compared with the HC and 2.1 (95% CI 1.8–2.5) with patients with RA. Similarly, regarding medications, removal of the N06 codes (which includes antidepressants with tricyclic antidepressants (TCAs) often prescribed for pain, a frequent symptom in MS 26 and RA) had little impact on the results (PR of 2.2 (95% CI 1.9–2.5) for the HC and 1.7 (95% CI 1.4–2.0) for patients with RA). In the same way, antipsychotics of the N05A group are also prescribed for the elderly, in case of behavioural problems or agitation, and led us to conduct an age-stratified analysis (0–65 vs. ≥65 years). Comparisons of age groups showed that there was no difference in the PR with a cohort matched to the HC, but had a slight difference with patients with RA (PR of 2.8 (95% CI 2.5–3.2) in the 0–65 years group vs. 2.4 (95% CI 2.0–2.9) in the >65 years group for the HC, and PR of 1.8 (95% CI 1.5–2.1) in the 0–65 years group vs. 2.7 (95% CI 1.9–3.8) in the >65 years group for patients with RA).
Discussion
In this study, a random sample of the French health insurance allowed us to conduct a large population-based study to describe the prevalence of MDs in MS patients. MDs were more frequent in the MS population than in the matched groups derived from the general population or patients with RA. This study showed that more than one-third of MS patients received care for MDs; similarly shown in a study from the Canadian administrative databases which identified 33.7% of MDs in the MS cohort. 25
The statistical association observed in this study did not establish a causal link between MS and MDs for the French population. Despite a plausible biological link found in previous studies,26,27 the increased prevalence may have been related to an increased healthcare consumption of MS patients leading to a potentially increased chance of being diagnosed for MDs. Nevertheless, prevalence was still higher in MS patients than for patients with RA, who similarly have high healthcare utilization as an ALD, indicating that MS may bring a specific mental burden.
Prevalence in this study was higher than the previous estimations from a French study that was the first to describe comorbidities in MS patients. 12 This study was only based on the ALD status and estimated 1.1% of ALD for psychiatric disorders in MS patients of less than 45 years of age. The 1.1% estimate was similar to the results found in our study of 3% on the ALD status. The difference between the two studies was largely due to the criteria related to healthcare utilization used in our study, where 33% of MS patients had ambulatory care for an MD (11.7% for HC) and 9.8% had in-hospital care for an MD (3.1% for HC). The prevalence of MDs was higher for females than males, in the three study groups (15 vs. 9% in the HC, 39 vs. 34% in MS patients and 43 vs. 37% in patients with RA). This was in accordance with literature for the general population, 28 although such results have not been clear in MS patients to date.5,8,29
The use of a national health administrative database with a representative random sample of the French population, as well as the use of two matched groups, allowed our results to be obtained. The matching ensured the controls had similar characteristics as the MS patients. Health administrative databases offer several advantages regarding unbiased recruitment of patients and non-self-reported information on diseases and symptoms. Furthermore, they are representative of the national population, in addition to its power due to the large numbers involved. The several sensitivity analyses performed additionally generated a prevalence estimation of MDs in each group, limiting the risk of over- or under-estimating the prevalence.
This study presented several methodological limitations. The identification of MDs was based on the use of healthcare services, which depend on other characteristics of individuals and may not reflect the true differences in prevalence due to differential and non-differential misclassification biases. Therefore, there may have been a risk of under-diagnosis of MDs in MS patients 4 or in patients with RA 30 if they don’t see health care providers or did not receive any specific medication. Similarly, under-diagnosis of MDs in the HC may have been related to lower utilization of healthcare services. A lack of hospital admissions in psychiatric wards in the EGB dataset compared with the medical needs may have underestimated the global prevalence of MDs, irrespective of the study group.
Another limitation was that the administrative databases used in this study were not created for research purposes and lacked clinical information and accuracy. In ambulatory care, the identification of MDs was based on treatment prescriptions but data on the diagnoses associated with these prescriptions were not available. Only information regarding the prescriber was available. No information was available in the database to know if an individual took their prescribed medication. MD prevalence was studied globally (not considering MDs diagnoses separately), using multiple sources to limit the risk of an inaccurate estimation due to misclassification. In addition, in-hospital coding of clinical information based on ICD-10 diagnosis codes can lack accuracy because the coding quality may vary depending on doctors and hospitals missing or incorrectly entering codes.
There may have also been an under declaration of ALD for MDs for those that may have already had an ALD in the general population, such as cancer. ALD status was attributed according to stringent criteria, however, the information relating to the co-occurrence of an ALD status for a given individual was of questionable quality. As the ALD status provided 100% reimbursement of the care specifically related to a disease it was requested for, it is generally acknowledged in France that when people have an ALD and they declare a second pathology, some GPs may not ask for additional ALDs.
In conclusion, this study confirmed that MDs were prevalent in MS patients and more frequent in the age- and sex-matched HC, as well as in patients with RA. These findings may raise the question of specificities of MS patients. The management of MDs for MS patients is therefore of high importance and should be systematically screened in order to reduce the consequences of such comorbidities in the management of MS.
Footnotes
Acknowledgements
The authors would like to thank La Fondation pour l’Aide à la Recherche sur la Sclérose en Plaques (Fondation ARSEP) and Sarina Yaghobian from Acacia Tools for reviewing the article.
Conflict of Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AG, JR and DT declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. EL declares consulting and lecture fees, and travel grants from Biogen, Genzyme, MedDay Pharmaceuticals, Merck Serono, Novartis, and Roche.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported through research grants from (Fondation ARSEP). The funding agency had no role in the design nor execution of the study, including the collection, management, analysis or interpretation of the data; the preparation, review or approval of the manuscript; or decision to submit the manuscript for publication.
