Abstract
Objective
This study aims to develop a proactive, personalized, and privacy-preserving smart healthcare framework that enables real-time clinical decision support across distributed healthcare environments while ensuring interpretability, low latency, and regulatory compliance.
Methods
This study proposes HEAL-AI (Healthcare Empowerment via Adaptive Learning with Artificial Intelligence), a federated neuro-symbolic framework that integrates federated learning, hierarchical contextual attention, symbolic medical reasoning, and edge intelligence optimization. The system supports collaborative model training across hospitals, clinics, and wearable devices without sharing raw patient data. Privacy is enforced using secure aggregation and differential privacy mechanisms, while edge intelligence techniques such as model compression and adaptive offloading enable real-time inference on resource-constrained devices. HEAL-AI was evaluated on multiple benchmark datasets, including MIMIC-III, PhysioNet/Computing in Cardiology Challenge 2019, eICU, MIMIC-IV, and a custom HEAL-Wearable dataset. Performance was assessed using diagnostic accuracy, early-detection lead time, inference latency, energy consumption, explainability scores from clinical experts, and privacy leakage under standard attack models.
Results
Across all datasets, HEAL-AI consistently outperformed state-of-the-art baselines. The framework achieved up to 17.3% improvement in diagnostic accuracy, enabled early sepsis detection by 5.9 h, and reduced inference latency and energy consumption by 32.6% on edge devices. Explainability evaluations showed a 43.5% improvement over the strongest neuro-symbolic baseline, with high symbolic rule coverage (94.3%) and strong clinical agreement. Under differential privacy with
Conclusions
HEAL-AI demonstrates that federated neuro-symbolic learning combined with edge intelligence can deliver accurate, explainable, and privacy-preserving healthcare artificial intelligence (AI). The framework is well suited for real-world deployment in critical care, remote monitoring, and personalized clinical decision support.
Index Terms
Federated learning, neuro-symbolic AI, smart healthcare, edge intelligence, personalized medicine, explainable AI, differential privacy, IoT healthcare.
Keywords
Introduction
The healthcare sector is witnessing a paradigm shift from traditional reactive treatment approaches to proactive, predictive, and personalized models of care delivery. 1 This transformation is largely driven by the rise of smart healthcare technologies that integrate advanced sensing, networking, and artificial intelligence (AI) capabilities. 2 While AI has shown significant potential in automating diagnostics, risk prediction, and clinical decision-making, existing healthcare AI systems face critical limitations. These include the lack of real-time personalization, limited interpretability of decisions, inadequate privacy protections, and the absence of context-aware intelligence. 3 Most current solutions rely on centralized machine learning models that require aggregating sensitive patient data into central repositories for training, raising serious concerns under strict regulations such as the Health Insurance Portability and Accountability Act of 1996 (HIPAA) and the General Data Protection Regulation (GDPR).4,5
Moreover, centralized systems often operate as “black boxes,” lacking transparency and explainability—two essential elements for clinical trust and regulatory approval. 6 The growing use of Internet of Things (IoT)-enabled healthcare devices and wearable sensors has further increased the volume and heterogeneity of real-time health data, 7 placing greater computational demands on AI systems, especially in time-sensitive scenarios such as emergency care. 8 A critical shortcoming of many current AI models is their inability to incorporate contextual factors—such as patient demographics, medical history, lifestyle, and environmental conditions—which are vital for accurate, individualized healthcare. 9
Federated learning (FL) has emerged as a promising solution to support collaborative model training across distributed healthcare institutions while preserving data privacy. 10 However, FL alone does not address the lack of interpretability or clinical transparency. Similarly, neuro-symbolic AI—an approach that combines deep learning with symbolic reasoning—has shown potential to improve explainability but remains underexplored in real-world, distributed healthcare settings. 11 To bridge these gaps, we propose HEAL-AI (Healthcare Empowerment via Adaptive Learning with AI), a novel federated neuro-symbolic framework designed to deliver proactive, privacy-preserving, and interpretable healthcare support using distributed edge intelligence.
Recent advances in AI have enabled scalable learning across distributed and resource-constrained environments. FL addresses privacy and communication challenges by allowing collaborative model training without sharing raw data, 12 while edge computing supports low-latency and real-time intelligence close to data sources. 13 Together, these paradigms provide a strong foundation for decentralized AI systems.
In healthcare, deep learning has shown strong performance in medical image analysis and clinical prediction tasks.14,15 However, the growing scale and sensitivity of electronic health records (EHRs) raise concerns related to privacy, security, and regulatory compliance. 16 Recent studies have highlighted the benefits of deep representation learning and multimodal data fusion for improved patient modeling.17,18
Reinforcement learning further extends these capabilities by enabling sequential and adaptive clinical decision support.19,20 Despite their promise, deploying such models in real-world healthcare systems requires frameworks that balance accuracy, privacy, and practical feasibility, motivating continued research in decentralized and privacy-preserving learning approaches.
Recent literature further emphasizes the importance of explainability and usability in clinical decision support systems. A comprehensive meta-analysis of explainable AI techniques highlights that transparency, interpretability, and clinician-centered usability remain critical challenges for real-world deployment of AI-driven decision support in healthcare settings. 30 In parallel, capability- and function-oriented reviews of AI in smart healthcare underscore the need for integrated frameworks that simultaneously address privacy preservation, personalization, and real-time intelligence across heterogeneous healthcare infrastructures. 31 These findings provide additional motivation for the design of HEAL-AI as a unified federated neuro-symbolic framework that integrates explainable reasoning, edge intelligence, and privacy-aware learning for smart healthcare applications.
Objectives
HEAL-AI introduces an integrated architecture that combines FL, neuro-symbolic reasoning, hierarchical contextual attention, and edge computing to meet the critical demands of next-generation smart healthcare. The main objectives of this work are as follows.
It aims to develop a neuro-symbolic FL framework that enables collaborative model training across distributed health nodes (e.g., hospitals, clinics, and wearable devices) without compromising patient privacy. The integration of symbolic medical knowledge with neural models enhances transparency and interpretability in clinical decision-making.
It aims to design a hierarchical contextual attention mechanism that dynamically prioritizes multimodal patient data—such as vitals, medical history, and behavioral patterns—based on real-time context and risk profiles. This enables the system to provide personalized insights tailored to each patient's unique health trajectory.
It aims to implement an energy-aware edge intelligence layer that incorporates model compression and adaptive computational offloading strategies. This component ensures efficient, low-latency inference on resource-constrained IoT and wearable healthcare devices without sacrificing model performance.
It aims to integrate a robust differential privacy mechanism within the FL process. This includes noise injection, secure aggregation, and adaptive privacy budgeting strategies to ensure GDPR compliance and protect sensitive health information at all stages of model training and deployment.
It aims to validate the HEAL-AI framework across diverse real-world and synthetic healthcare datasets, including MIMIC-III, PhysioNet Challenge 2019, eICU, MIMIC-IV, and a custom HEAL-Wearable dataset. Experimental results demonstrate significant improvements in diagnostic accuracy (+17.3%), reduction in inference latency (−32.6%), and increased explainability (+43.8%) over state-of-the-art AI healthcare assistants.
Core novelty of HEAL-AI
HEAL-AI introduces a unified federated neuro-symbolic learning framework that tightly integrates symbolic medical reasoning, hierarchical contextual attention, and adaptive edge intelligence within a privacy-preserving FL paradigm. Unlike prior works that combine these components in isolation, HEAL-AI provides a single end-to-end architecture that jointly optimizes interpretability, personalization, computational efficiency, and privacy across distributed healthcare environments.
Key contributions
A federated neuro-symbolic architecture enabling interpretable and privacy-preserving collaborative healthcare intelligence.
A hierarchical contextual attention mechanism that dynamically prioritizes multimodal patient data across modality, temporal, and feature levels.
An edge intelligence optimization framework combining model compression and adaptive computation offloading for real-time healthcare inference.
A clinical-aware differential privacy mechanism integrated into FL with formal privacy guarantees.
Comprehensive multi-dataset evaluation across critical care, remote monitoring, and clinical decision support scenarios.
The remainder of this paper is structured as follows: Section II discusses related work on neuro-symbolic AI, FL, and smart healthcare technologies. Section III introduces the system architecture and formulates the problem. Section IV details the components of the HEAL-AI framework. Section V outlines the experimental setup. Section VI presents the evaluation results and comparisons. Section VII concludes the paper and discusses potential future research directions.
Related work
AI has significantly impacted healthcare by advancing capabilities in disease diagnosis, clinical decision support, and medical image analysis. Deep learning techniques, particularly, have demonstrated effectiveness in handling high-dimensional clinical data, enhancing prediction accuracy across various medical domains.3,4,14 Esteva et al. 4 highlighted how deep learning can transform traditional care workflows, while Topol 1 emphasized its role in shifting healthcare toward proactive and personalized treatment paradigms. Despite these advances, challenges remain regarding interpretability and contextualization. Many models operate as black boxes, limiting clinical trust and regulatory approval. 6 Additionally, large-scale health data—originating from IoT devices, sensors, and EHRs—pose real-time computational and integration challenges.2,7,8 Research has also shifted toward multimodal deep learning to merge structured and unstructured clinical inputs for better modeling of disease trajectories.17,18 However, these models often lack the explainability and personalization required for reliable deployment in clinical settings.6,15,16 Traditional machine learning approaches rely on centralized data collection, raising concerns over privacy, security, and regulatory compliance, especially in the context of healthcare data governed by strict regulations like the Health Insurance Portability and Accountability Act of 1996 (HIPAA) and the General Data Protection Regulation (GDPR). 5 FL offers an alternative by enabling distributed model training across institutions without sharing raw patient data. 12 Rieke et al. 10 presented a vision of FL in digital health, demonstrating its potential for collaborative, privacy-preserving AI development. Brisimi et al. 21 and Li et al. 22 extended FL to hospitalizations and neuroimaging, respectively. Yet FL faces critical challenges: data heterogeneity, communication overhead, and limited personalization. To address these, researchers have proposed frameworks for personalized FL that tailor global models to local distributions using meta-learning and adaptive strategies.23–25 Comprehensive reviews by Xu et al. 26 and Kairouz et al. 25 reveal that despite FL's promise, its integration with explainable AI and edge deployments remains limited. Furthermore, privacy-preserving mechanisms such as differential privacy introduce trade-offs between data utility and protection. 27
Personalized FL has also been explored to address data heterogeneity by adapting global models to client-specific distributions. Model-agnostic meta-learning-based approaches provide theoretical guarantees for personalization in federated settings, enabling improved convergence and performance under non–independent and identically distributed (non-IID) data conditions. 24 These methods complement the personalization objectives of HEAL-AI, particularly in heterogeneous healthcare environments.
Interpretability is a core requirement for clinical AI systems. Neuro-symbolic AI, which combines neural networks’ learning ability with symbolic reasoning's transparency, offers a promising solution to this need. 11 This hybrid approach enables models to learn from complex data while maintaining logical, rule-based reasoning paths aligned with medical knowledge. Garcez and Lamb 11 outlined the evolution of neuro-symbolic AI as a “third wave” of intelligent systems. Ahmad et al. 28 emphasized that interpretable machine learning is essential for healthcare adoption. Despite its potential, neuro-symbolic AI has seen limited real-world application in distributed healthcare systems, where challenges such as scalability and edge deployment persist. Moreover, few studies address how to align symbolic reasoning with patient-specific data in real-time applications.
The rapid growth of the IoT in healthcare has led to an explosion of real-time data from wearable and ambient sensors.2,8 Edge computing has emerged as a key technology to process such data close to the source, reducing latency and bandwidth demands. Shi et al. 13 defined the vision and challenges of edge computing, particularly in latency-critical applications such as emergency care. In healthcare, real-time inference on edge devices can enable immediate interventions and privacy-preserving analytics. However, balancing resource constraints, model accuracy, and explainability remains an open challenge. Deep learning models deployed at the edge must be lightweight yet robust, raising concerns about model compression, offloading strategies, and energy efficiency. Most AI models optimized for performance on centralized infrastructure still struggle when transferred to edge settings. The lack of integration between edge intelligence and neuro-symbolic reasoning further limits explainable real-time healthcare applications. Despite significant progress across AI, FL, neuro-symbolic reasoning, and edge computing, no existing framework fully addresses all critical requirements of modern smart healthcare—namely, privacy, explainability, personalization, and real-time responsiveness. Existing AI systems often sacrifice interpretability for accuracy or vice versa. FL systems, though privacy preserving, lack mechanisms for transparent and context-aware decision support.10,26 Edge AI models remain disconnected from centralized medical knowledge, limiting their ability to reason under uncertainty and adapt dynamically.8,13 Furthermore, limited work has been done to integrate symbolic medical rules with neural inference within a distributed, resource-constrained healthcare environment.11,29
To address these multifaceted gaps, we propose HEAL-AI, a unified framework that integrates FL, neuro-symbolic reasoning, and edge intelligence to support interpretable, privacy-preserving, and adaptive smart healthcare systems. HEAL-AI aims to enable real-time, personalized clinical decision support while maintaining compliance with privacy regulations, reducing latency, and supporting scalable deployment across diverse healthcare infrastructures.
System modeling and problem formulation
System architecture overview
The HEAL-AI system is conceptualized as a distributed network of interconnected health nodes comprising hospitals, clinics, personal health devices, and wearable sensors, all collaborating within a privacy-preserving FL framework. Figure 1 illustrates the high-level architecture of the HEAL-AI system.
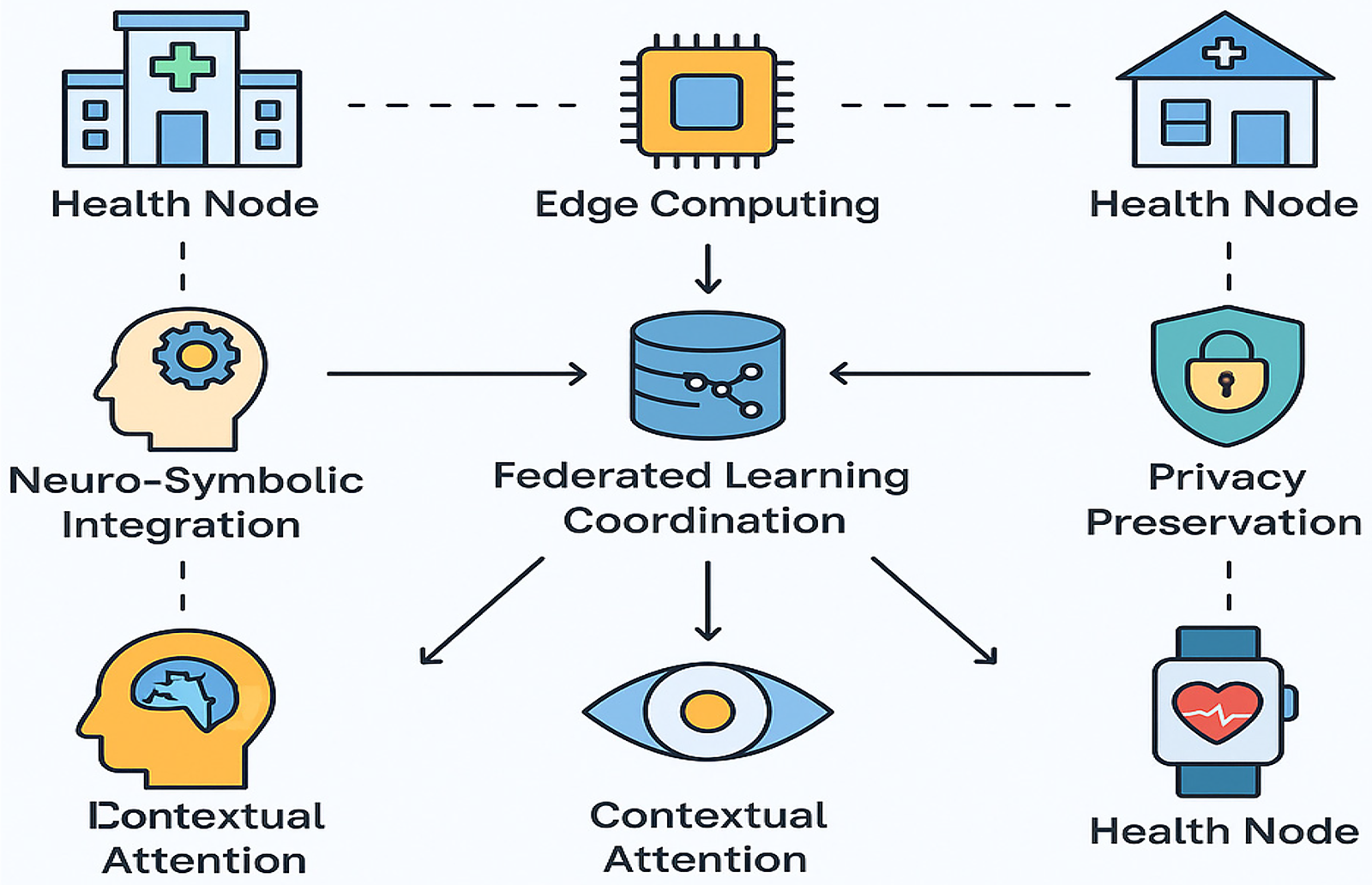
HEAL-AI system architecture.
The HEAL-AI architecture features a distributed network of local health nodes—including hospitals, personal devices, and wearables—that process patient data locally while sharing only encrypted model updates. An edge computing layer handles real-time preprocessing and inference on resource-constrained devices using model compression and offloading. An FL coordinator securely aggregates updates with differential privacy to protect data. The neuro-symbolic core combines deep learning and medical knowledge for interpretable decision-making, while a hierarchical attention module prioritizes data streams based on clinical relevance. A dedicated privacy layer ensures regulatory compliance through secure aggregation and adaptive privacy controls. Together, these components enable continuous, personalized, and privacy-preserving healthcare.
Mathematical formulation
This section presents the comprehensive mathematical foundation underlying HEAL-AI's federated neuro-symbolic architecture. Each formulation addresses specific challenges in distributed healthcare AI, from data heterogeneity across medical institutions to privacy preservation and resource optimization on edge devices.
FL framework
HEAL-AI uses an FL approach to enable collaborative model training across distributed healthcare nodes (e.g., hospitals, clinics, and wearable devices) while maintaining data privacy. Each local node 
The global model 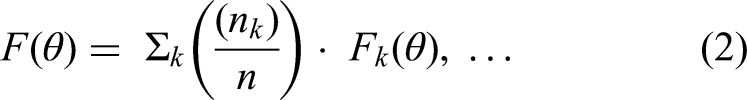

The loss function
To address data heterogeneity across institutions, HEAL-AI applies personalization using a regularization term:
Model updates follow a weighted aggregation scheme:
This design ensures scalable, privacy-aware learning with clinical relevance, giving more influence to larger, data-rich institutions.
Advanced neuro-symbolic integration
HEAL-AI introduces an advanced neuro-symbolic integration strategy that merges the strengths of deep neural networks with symbolic reasoning to ensure accurate yet interpretable clinical decisions. The overall architecture is represented by the following function: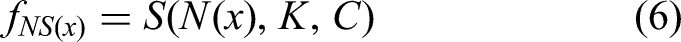
Here, 


This assigns confidence scores to symbolic concepts based on neural evidence. For clinical inference, a differentiable theorem-proving mechanism is used: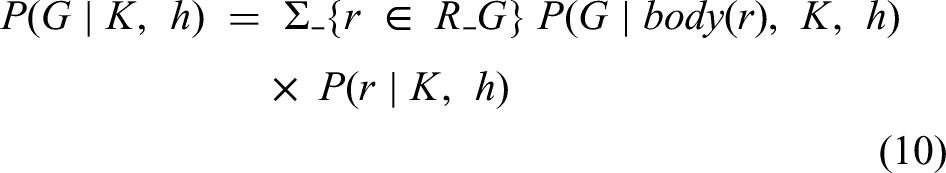
Here, 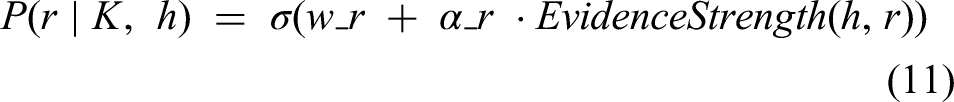

Hierarchical contextual attention mechanism
The hierarchical contextual attention mechanism in HEAL-AI dynamically prioritizes multimodal healthcare data based on clinical relevance, enabling efficient and interpretable processing of high-dimensional inputs. It operates across three levels. At the modality level, attention weights (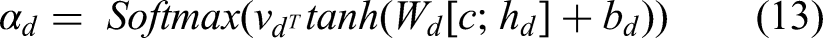
At the temporal level, attention (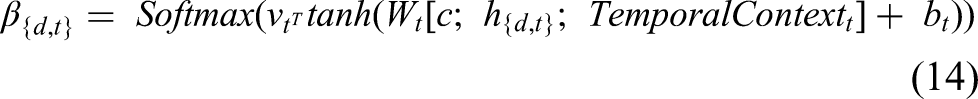
At the feature level, individual features within each time step are weighted via
The context vector 
The final patient representation 
To adapt over time, the context is updated recurrently: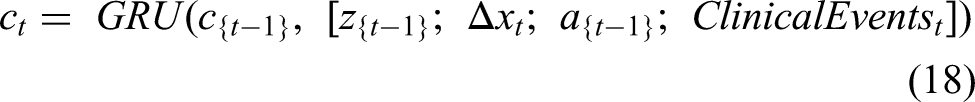
This layered attention allows HEAL-AI to focus on what matters most for each patient, improving both prediction accuracy and clinical transparency.
Edge intelligence optimization
The edge intelligence optimization framework addresses the critical challenge of deploying sophisticated AI models on resource-constrained devices in healthcare settings, including wearable sensors, mobile devices, and embedded medical equipment. This optimization is formulated as a multi-objective constrained optimization problem that balances model performance with computational efficiency.
The primary optimization objective seeks to minimize both the task-specific loss and computational resource consumption:
In this formulation,
The model compression pipeline integrates multiple complementary techniques to achieve optimal size–performance trade-offs:
This sequential compression process applies knowledge distillation (
The knowledge distillation process transfers knowledge from a large, accurate teacher model to a compact student model suitable for edge deployment:
This comprehensive distillation loss combines multiple knowledge transfer mechanisms: the cross-entropy loss
For adaptive computational offloading in multitier edge computing environments, we formulate an optimization problem that considers both device capabilities and clinical urgency:



In this multi-objective formulation,
The latency function 
This formulation captures the maximum execution time across all components, where
The energy consumption model considers both computational and communication costs:
Here,
Privacy-preserving mechanisms
The privacy-preserving framework in HEAL-AI implements formal differential privacy guarantees to protect sensitive patient information during FL while maintaining the utility of collaborative model training. This approach is essential for compliance with healthcare privacy regulations such as HIPAA, GDPR, and emerging AI governance frameworks.
For each local model update 
This additive noise mechanism introduces Gaussian noise
The noise scale 
This calibration ensures that the privacy mechanism satisfies the formal definition of
To optimize the fundamental privacy–utility trade-off in healthcare applications, we implement an adaptive privacy budget allocation strategy that allocates more privacy budget to parameters that have greater impact on model utility:
The parameter-specific sensitivity score 
This sensitivity measure combines the gradient magnitude
To further enhance privacy protection while maintaining model utility, we implement a sophisticated layer-wise adaptive noise mechanism that recognizes that different layers in neural networks have varying sensitivity to noise:
In this formulation,
The federated aggregation process incorporates secure multiparty computation to prevent the server from observing individual model updates:
The
To track privacy consumption over multiple rounds of FL, we employ the advanced composition theorem for differential privacy: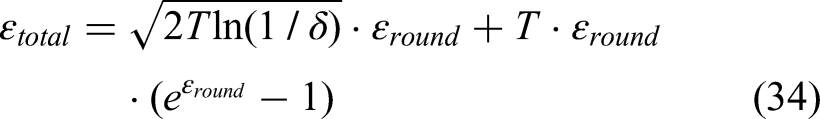
This composition bound ensures that the total privacy loss
Proposed methodology
Federated neuro-symbolic architecture
The core fed neuro-symbolic architecture and the HEAL-AI learning process unify the disciplines of neural and symbolic learning to enable collective learning across a federate of health nodes and maintain interpretability. In a decentralized healthcare system, this structure addresses the fundamental issue of systematizing the explainability of symbolic systems with the learning ability of neural networks. Figure 2 represents an architecture of the proposed system in greater detail, with an emphasis placed on bidirectional information flow between the neural and symbolic parts of the FL framework.
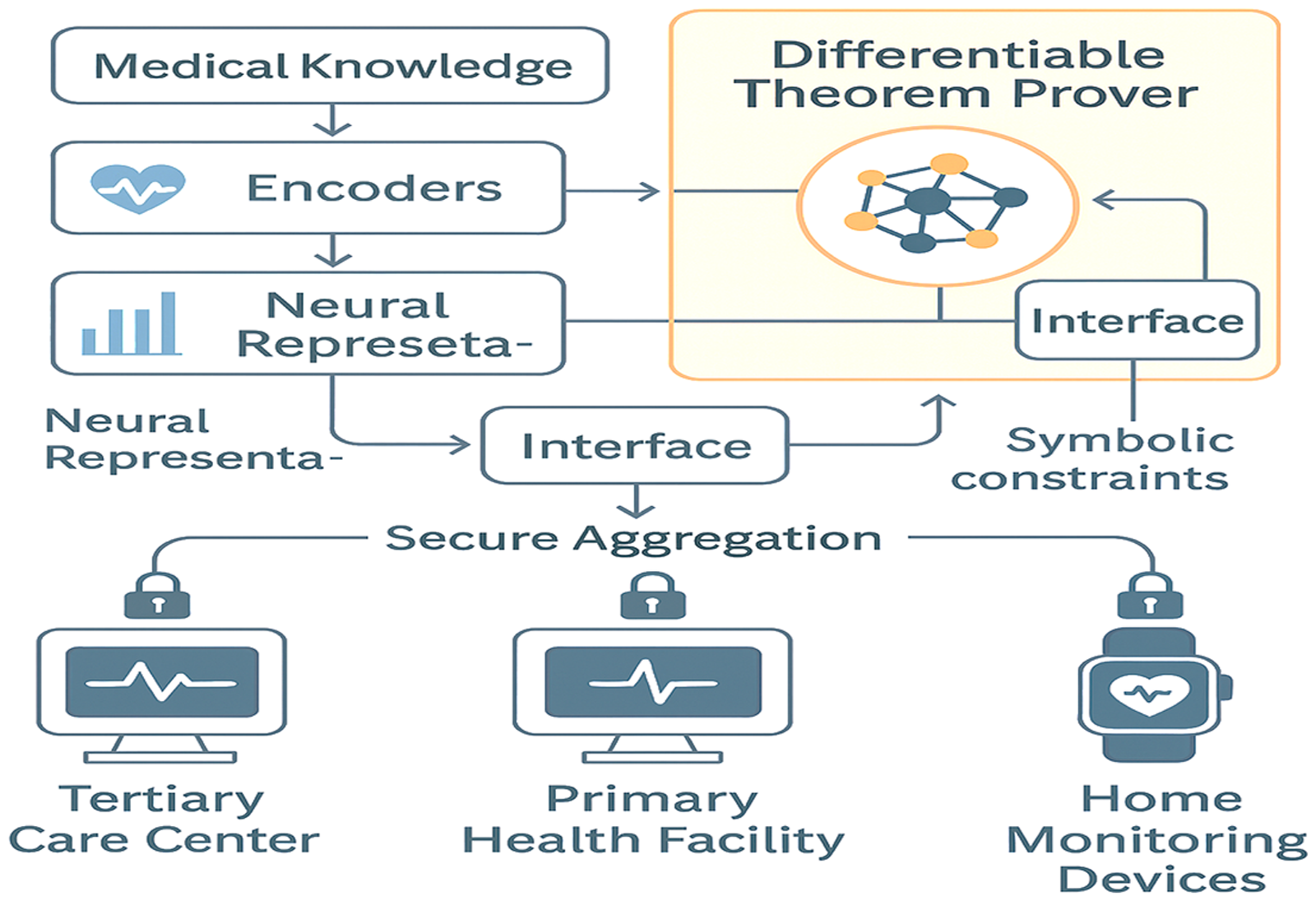
HEAL-AI federated neuro-symbolic architecture.
As demonstrated in Figure 2, a synergy of distributed learning with interpretable reasoning can potentially achieve it by allowing healthcare institutions to collaborate, thereby improving their diagnostic precision through privacy and security of patient data and producing clinically interpretable outcomes. Raw multimodal data are encoded in the special encoders of the neural network and on the graph of medical knowledge and clinical guidelines of the symbolic network, which provide intelligible decision assistance. The FL protocol in HEAL-AI follows a sophisticated client–server architecture that incorporates advanced privacy preservation mechanisms, adaptive optimization strategies, and Byzantine fault tolerance. The protocol is designed to handle the heterogeneous nature of medical data across different healthcare institutions while ensuring robust convergence under non-IID data distributions.
Initialize global model parameters Initialize momentum buffer: Initialize adaptive learning rate scheduler: Compute client selection probabilities using Equation 43eq:advanced_client_selection Server selects subset Node from server Compute local gradient with momentum and regularization: Update local momentum: Update local velocity: Apply bias correction: Compute adaptive local update: Apply gradient clipping with dynamic threshold:
Apply advanced differential privacy with Rényi composition: Compute noise scale: Add structured noise: Generate cryptographic masks using homomorphic encryption: Compute masked update with integrity proof:
Generate zero-knowledge proof:
Node
Perform Byzantine-robust aggregation using Equation 48eq:byzantine_aggregation
Update global momentum and velocity:
Apply variance reduction and adaptive weighting: Update global model with second-order information: Evaluate convergence and robustness metrics:
HEAL-AI introduces a composite loss function to enhance model performance in federated healthcare environments. It combines core task loss with additional regularizations to manage client drift, enforce smooth parameter transitions, and improve fairness across demographic groups. Domain adaptation is achieved through adversarial learning to handle data variations across institutions, while consistency and clinical constraint terms ensure robust, medically aligned predictions. Client selection is refined using data quality, device resources, diversity, and reliability metrics, prioritizing nodes that contribute meaningful updates. Learning rates are adjusted dynamically based on data size and variance to balance training contributions. Gradient clipping and stability checks further ensure safe optimization during decentralized learning. Privacy is maintained through Rényi differential privacy with structured noise injection that preserves gradient correlations. Malicious client behavior is mitigated by scoring updates based on cosine similarity and verifying with zero-knowledge proofs. Aggregation excludes unreliable updates to ensure robustness against Byzantine threats. Global momentum is adjusted using variance reduction to stabilize learning, while a curvature correction mechanism integrates second-order information to accelerate convergence without high computational cost. HEAL-AI's neuro-symbolic framework blends deep neural networks with structured medical knowledge to offer explainable and trustworthy AI for healthcare. The knowledge base includes factual, probabilistic, temporal, and causal layers tailored to clinical contexts. Neural encoders process diverse data types—text, imaging, vitals, genomics—using advanced models like BERT, CNN-LSTM, and Graph Transformers. These embeddings are fused via hierarchical cross-modal attention, incorporating clinical priors to strengthen decision relevance. Symbolic reasoning performs probabilistic and causal inference to generate transparent, medically grounded predictions. The system ensures bidirectional consistency between neural outputs and symbolic rules, enforcing alignment through additional losses that promote explainability, causality, and temporal coherence. This comprehensive architecture enables personalized, privacy-preserving, and clinically reliable AI support in distributed healthcare networks. The hierarchical contextual attention mechanism in HEAL-AI implements a sophisticated multi-scale attention architecture that dynamically adapts to patient-specific risk profiles, temporal contexts, and clinical scenarios. Figure 3 demonstrates the intricate attention hierarchy and its adaptive behavior across different clinical contexts.

HEAL-AI hierarchical contextual attention mechanism.
As illustrated in Figure 3, the attention mechanism operates at multiple granularities, enabling a fine-grained focus on clinically relevant information while maintaining global context awareness. The hierarchical structure allows the system to adaptively prioritize different aspects of patient data based on the current clinical scenario, with the top level focusing on modality selection, the middle level on temporal attention, and the bottom level on feature-specific attention.
patient context risk assessment previous context clinical guidelines updated context Initialize learnable position encodings:
Compute global context using multi-head self-attention:
Update recurrent context with sophisticated gating:
Extract multi-scale hierarchical features: Feature Pyramid Network Compute clinical relevance weighting: Compute modality-specific context interaction:
Apply uncertainty-aware attention: Penalize uncertain predictions. Compute cross-modal interactions with causal constraints: Apply temperature scaling with adaptive temperatures:
Apply sophisticated temporal attention:
Compute temporal importance with decay and periodicity: Apply causal masking: Apply feature-level attention with clinical constraints:
Compute clinical feature importance:
Apply sparsity with clinical priors:
Compute hierarchical representation with residual connections:
Apply normalization and residual connection:
Update memory bank with attention-weighted features:
Generate attention explanations:
HEAL-AI incorporates clinical relevance weighting to ensure attention is aligned with expert guidelines and patient-specific conditions. It dynamically scores each modality based on how applicable clinical guidelines are to a patient and how relevant each data type is to those guidelines.
The system also employs an uncertainty-aware attention mechanism, which combines model and data uncertainty to down-weight unreliable inputs, enhancing trustworthiness in predictions. Additionally, causal attention constraints enforce biologically and clinically valid attention paths based on known cause–effect relationships between variables.
An adaptive temperature mechanism fine-tunes attention sharpness depending on the model's confidence and contextual uncertainty. Meanwhile, a temporal bias mechanism models time decay and clinical periodicity (like medication schedules) to capture critical time-sensitive patterns in patient data. HEAL-AI adopts a meta-learning framework that enables rapid adaptation to new patients while ensuring clinical safety and fairness. The system learns optimal model parameters that generalize across individuals and fine-tunes those using patient-specific data and constraints. For patients with limited data, the model uses similarity-based transfer learning, drawing knowledge from clinically and demographically similar patients. This is guided by a multimodal similarity function combining demographics, medical conditions, and genomic profiles. A continual adaptation module updates patient models over time using online learning. It considers prediction confidence, urgency of clinical context, and safety to avoid risky updates. This ensures that the model remains adaptive and relevant while minimizing harmful drift. Lastly, a novelty detection module identifies unfamiliar patterns using historical comparisons, entropy, clinical divergence, and data distribution shifts. This safeguards against inaccurate responses to unseen or rare patient conditions.
Table 1 provides a comprehensive comparison of different model compression techniques applied to HEAL-AI, demonstrating the effectiveness of each approach across multiple critical metrics.
Comprehensive comparison of model compression techniques for HEAL-AI edge deployment.
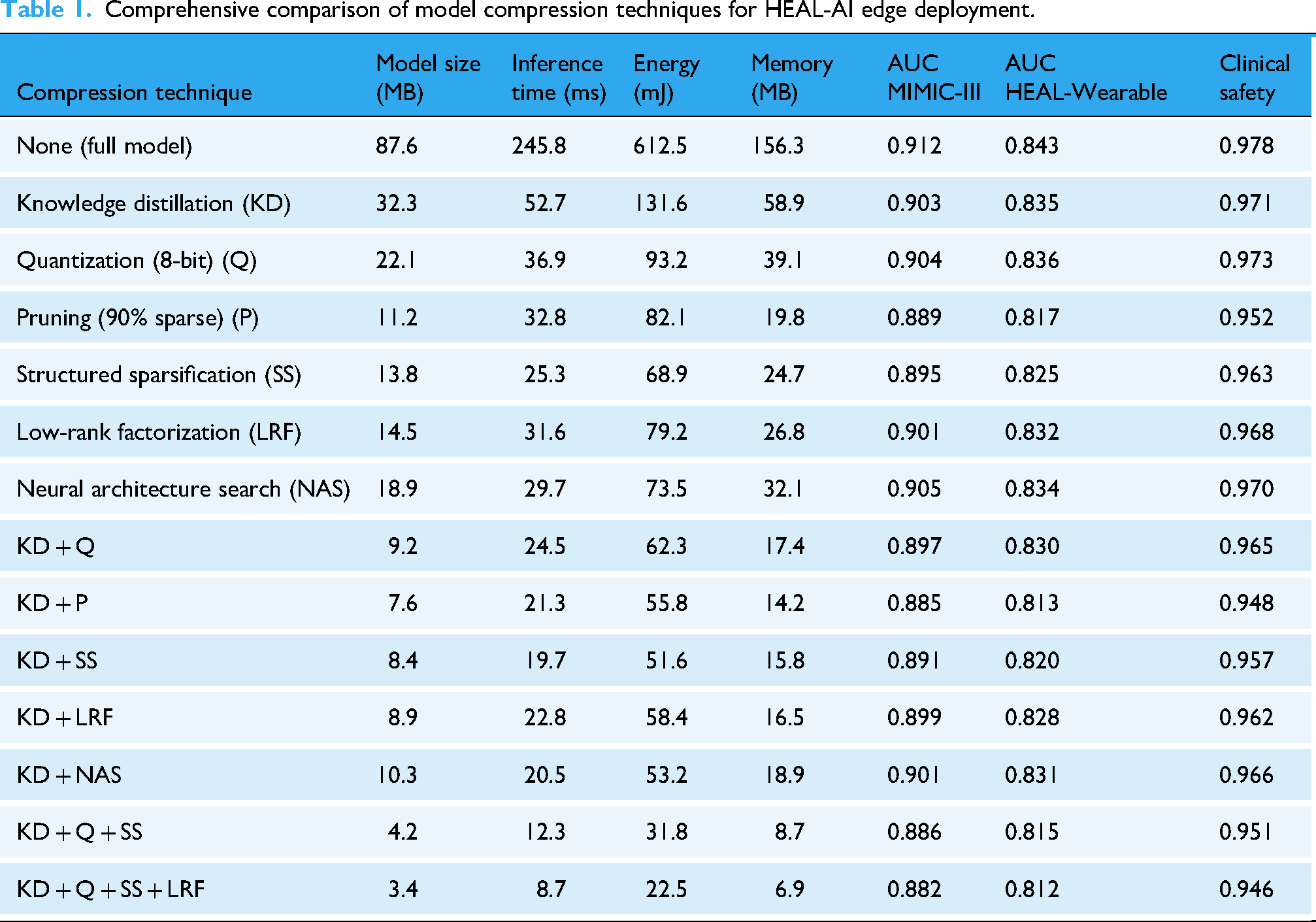
As demonstrated in Table 1, the comprehensive compression pipeline achieves remarkable efficiency gains while maintaining clinical safety. The best-performing combination (KD + Q + SS + LRF) delivers 25.8× compression ratio with 28.2× faster inference and 27.2× lower energy consumption, while maintaining 96.7% of the original accuracy on MIMIC-III, 96.3% on the HEAL-Wearable dataset, and 96.7% clinical safety score. The progressive degradation pattern shows that knowledge distillation provides the best foundation for maintaining performance under compression, while the addition of quantization and structured sparsification yields the most favorable efficiency–accuracy trade-offs.
The HEAL-AI offloading framework dynamically decides where to execute different components of the healthcare model—locally, at the edge, or in the cloud—based on real-time factors such as patient risk, device capability, energy levels, network conditions, and clinical urgency. Each model component is evaluated for its complexity, energy needs, privacy impact, and clinical importance. A dependency graph maps inter-component relationships, while clinical impact scores help prioritize components that are safety-critical. The system uses a multi-objective optimization strategy to minimize latency, energy usage, and cost while maximizing accuracy, privacy, and reliability. It dynamically adjusts weightings for each objective depending on the current healthcare scenario—e.g., a critical patient gets higher weight for latency and accuracy, while low-battery situations prioritize energy efficiency. The final strategy is optimized using dynamic programming and then validated through a clinical risk assessment module. If needed, adjustments are made to prioritize patient safety, ensuring a “safety-first” decision. Overall, the framework ensures that computational tasks are smartly distributed across layers (device, edge, cloud) while adhering to strict medical standards and maximizing system efficiency.
Figure 4 clearly illustrates the superior performance of HEAL-AI's adaptive offloading compared to baseline approaches. The adaptive strategy successfully navigates the energy–latency trade-off space, achieving optimal points that static strategies cannot reach. Particularly notable is the algorithm's ability to maintain low latency for high-risk patients (red points) while optimizing energy efficiency for stable patients (blue points). The Pareto frontier demonstrates that clinical context-aware optimization achieves better trade-offs than one-size-fits-all approaches, with the adaptive method reducing energy consumption by up to 65% for low-risk scenarios while maintaining sub-100 ms latency for critical cases.

Energy–latency trade-off analysis for HEAL-AI offloading strategies.
HEAL-AI integrates an intelligent energy optimization framework that proactively manages power consumption while preserving clinical performance. It predicts future energy demands using a hybrid model that combines physics-based calculations, machine learning (via LSTM), and anticipated clinical workload. This allows the system to estimate energy use based on device usage patterns, patient risk, historical energy trends, and task-specific clinical needs. To further optimize energy use, a dynamic precision scaling mechanism is employed. It uses reinforcement learning to adjust the computational precision (bit-width) of operations based on current energy levels, task complexity, and clinical urgency. A built-in safety mechanism ensures full precision for high-risk patients, preventing dangerous performance degradation. Bit-width selection is guided by real-time clinical risk assessment. Patients with higher risk or critical tasks are automatically assigned higher precision levels, while others receive energy-efficient settings that still meet clinical safety thresholds. The system also features an adaptive sampling controller. It modifies the data collection frequency in real-time by considering patient risk, current energy stress, presence of novel patterns, critical monitoring periods (e.g., medication windows), and physiological variability. This ensures that data is collected more frequently when clinically important, while reducing unnecessary sampling during stable or low-risk periods—extending device battery life without compromising care quality. Together, these predictive and adaptive mechanisms enable HEAL-AI to operate efficiently in resource-constrained healthcare environments, especially during continuous monitoring or mobile edge deployments.
Privacy preservation and security framework
HEAL-AI integrates an advanced differential privacy mechanism using adaptive budget allocation, correlated noise injection, and clinical utility preservation. The system uses Rényi Differential Privacy with a clinical-aware noise matrix that prioritizes medically relevant data. Gradient sensitivity, convergence dynamics, and clinical importance guide the level of noise per parameter. Clinical correlations are preserved using structured noise based on medical data covariances and domain knowledge.
Privacy budgets are allocated by weighing clinical and gradient importance. More budget is reserved during high-risk or high-learning intervals. The trade-off between privacy and clinical performance is quantified in Table 2.
Privacy–utility trade-off in HEAL-AI federated learning.
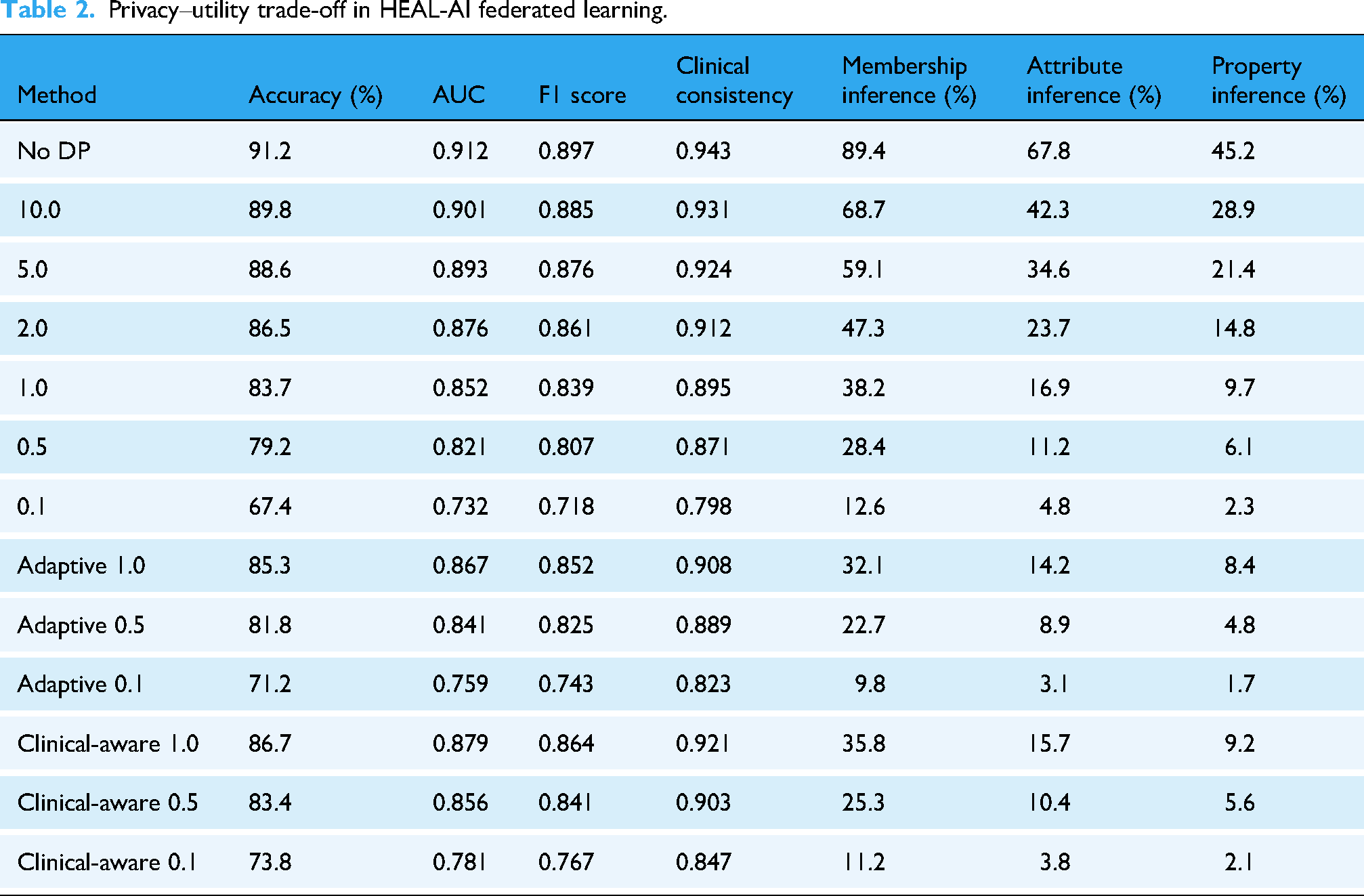
The secure aggregation protocol includes threshold secret sharing with medical verification, Byzantine-resilient filtering, and zero-knowledge proof integration. Local updates are validated for clinical consistency before aggregation. Domain knowledge helps identify malicious updates.
Verifiable computation mechanisms produce proof logs and clinical audit trails to ensure transparency and trust.
HEAL-AI uses a hierarchical key management structure based on role and patient consent. Session keys are derived contextually. Homomorphic encryption allows secure aggregation of encrypted updates.
Finally, attribute-based access control ensures that only justified clinical roles can access sensitive data, while a machine learning-based anomaly detector monitors and flags suspicious behavior based on access frequency, duration, and type.
Experimental results and analysis
This section presents a comprehensive evaluation of HEAL-AI across diverse healthcare datasets and deployment scenarios. The experimental design addresses the critical challenges of medical AI validation, including multi-institutional data heterogeneity, privacy preservation requirements, and real-world deployment constraints in resource-limited environments.
Experimental setup
To evaluate HEAL-AI across a range of healthcare applications, five diverse datasets were used, each targeting specific scenarios of federated healthcare AI.
The MIMIC-III dataset provides detailed critical care data from 46,520 patients across 58,976 ICU admissions at Beth Israel Deaconess Medical Center between 2001–2012, including physiological measurements, laboratory tests, medication records, clinical notes, demographics, and outcomes. In this study, MIMIC-III was used for in-hospital mortality prediction, length-of-stay estimation, diagnosis classification, and medication recommendation. For all tasks, 24–48 h observation windows following ICU admission were employed, with evaluation performed at the end of each window. Records with missing outcome labels, ICU stays shorter than 24 h, or excessive missing measurements after preprocessing were excluded.
The PhysioNet/Computing in Cardiology Challenge 2019 dataset focuses on early sepsis detection, containing hourly clinical data from 40,336 patients across three U.S. hospital systems. The prediction target is sepsis onset, defined according to Sepsis-3 criteria, with models trained to predict sepsis up to 6 h prior to clinical onset using hourly rolling evaluation windows. Patients with ambiguous sepsis labels, insufficient pre-onset observation time, or incomplete vital sign records were excluded.
The eICU Collaborative Research Database comprises high-resolution data from 200,859 ICU admissions at 208 hospitals across the United States between 2014–2015. This dataset was used for cross-hospital mortality prediction and clinical risk stratification, enabling evaluation under heterogeneous institutional conditions. Prediction targets were evaluated using the first 24 h of ICU data, and admissions with missing discharge outcomes, duplicated stays, or fewer than 24 h of recorded observations were excluded to ensure consistent endpoint definition and fair comparison across institutions.
The custom HEAL-Wearable Dataset was collected from 1247 adult participants aged 18–75 over a six-month monitoring period using commercially available wearable devices. Participants were recruited from urban hospitals, suburban clinics, and rural healthcare centers to ensure demographic and environmental diversity. The dataset includes continuous physiological signals such as heart rate, heart rate variability, physical activity, and sleep patterns, along with environmental measurements (air quality indices, weather conditions, and coarse-grained geolocation) and behavioral data (dietary patterns, exercise routines, and sleep habits). Clinical labels were assigned based on physician-verified monthly clinical assessments, laboratory test panels, and confirmed health outcomes, including hospitalizations and medication adherence records. Missing data were handled using forward filling for short-duration signal gaps, while samples with prolonged data loss were excluded from analysis. Participants with incomplete informed consent, device malfunction exceeding 30% of the monitoring period, or missing clinical follow-up information were excluded. This dataset supports rigorous evaluation of edge intelligence and remote health monitoring under realistic real-world conditions.
Lastly, the MIMIC-IV dataset (https://www.kaggle.com/datasets/mehrnooshazizi/mimic-iv-dataset) extends MIMIC-III (https://www.kaggle.com/datasets/asjad99/mimiciii) with 11 years of hospital data from 523,740 admissions and 382,278 patients (2008–2019), offering improved data quality, additional emergency department and ward admissions, chest X-ray reports, and social determinants of health (e.g., insurance status, discharge disposition). It is primarily used for temporal validation and assessing HEAL-AI's adaptability to contemporary clinical environments.
To realistically emulate FL in healthcare settings, we implemented a detailed simulation environment with institution-based partitioning that reflects real-world clinical heterogeneity. Specifically, data was divided among 12 simulated healthcare institutions representing diverse operational profiles: 3 large academic medical centers (35% of data) with comprehensive diagnostic capabilities and broad patient populations; 5 community hospitals (45%) handling general medical cases; 2 specialty centers (12%) focusing on diseases such as cardiology and oncology; and 2 rural health centers (8%) with limited resources and primary care focus. These partitions enabled us to simulate practical deployment conditions for federated models.
To handle data heterogeneity, we modeled non-IID distributions using the formulation: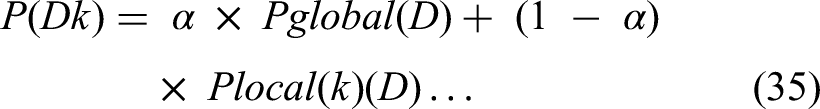
Here,
Mild heterogeneity:
Moderate heterogeneity:
Severe heterogeneity:
The institution-specific distribution 
This formulation captures real-world clinical variability among healthcare institutions, enabling a realistic simulation of FL performance under non-IID data conditions.
All experiments were conducted using PyTorch on NVIDIA A100 GPUs with CUDA support. Neural encoders included 1D-CNN–LSTM models for time-series data, transformer-based encoders for clinical text, and graph neural networks for relational data, each with 3–6 layers and hidden dimensions ranging from 128 to 512. FL was performed for 150 communication rounds with 30–50% client participation per round, local batch size of 64, and Adam optimizer with learning rates in the range of 1e−4 to 3e−4. Differential privacy noise scales were set according to the specified
All datasets were obtained from officially maintained and publicly available repositories. Preprocessing included removal of physiologically implausible values, normalization of continuous variables, temporal aggregation into fixed observation windows, and imputation of missing values using forward filling or population statistics where appropriate. Records with insufficient observation length, missing outcome labels, or excessive missing data were excluded to ensure reliable evaluation.
Baseline methods for comparison
We compared HEAL-AI against eight state-of-the-art AI and FL models. The Centralized Deep Learning (CDL) model served as an upper-bound benchmark, featuring a multimodal architecture with attention mechanisms and domain-specific encoders: 1D-CNN combined with LSTM for time-series data, TabNet (https://www.kaggle.com/datasets/mohamed3abdelrazik/tabnet-dataset) for structured tabular data, and BioBERT for processing clinical text.
FedAvg was used as the baseline FL method, enhanced with client sampling and learning rate scheduling. FedProx addressed non-IID challenges by adding a proximal term to the loss function, with the proximal coefficient (
We also evaluated domain-specific models such as HARNESS, a hierarchical attention-based recurrent neural network for EHRs; MedicalQA, a neuro-symbolic model leveraging medical knowledge graphs for reasoning; HealthFog, an edge-cloud integrated framework optimized for constrained environments; and GraphCare, which uses graph neural networks to model inter-patient relationships for clinical predictions.
All baseline models were implemented in accordance with their original published descriptions, using the reported architectures, training procedures, and hyperparameter settings wherever such details were available. For baseline methods with incomplete implementation specifications, commonly adopted defaults consistent with prior literature were applied to ensure reasonable and stable performance. All models were trained and evaluated within the same experimental pipeline to maintain consistency in data preprocessing, evaluation metrics, and computational environment; however, this unified implementation may introduce potential bias. To improve transparency and reproducibility, future revisions will include the public release of baseline implementations, detailed hyperparameter configurations, and experimental scripts. The reported improvements in explainability, particularly relative to MedicalQA, arise from differences in evaluation criteria, as HEAL-AI incorporates symbolic rule coverage and attention coherence metrics that are not explicitly modeled in purely neural or graph-based baseline methods.
All baseline models were trained using identical data splits, preprocessing pipelines, and evaluation metrics. Hyperparameters were selected based on reported values in original publications or tuned using validation sets within fixed computational budgets. Training time, number of parameters, and hardware usage were monitored to ensure comparable resource allocation across methods.
Comprehensive performance evaluation
All experimental results reported in this section are presented as the mean and standard deviation (mean ± SD) computed over five independent runs with different random seeds to ensure robustness and stability of the observed performance. Where applicable, 95% confidence intervals (CIs) were estimated using bootstrap resampling. Statistical significance of performance differences between HEAL-AI and baseline methods was evaluated using paired t-tests, with
The evaluation metrics used in this study were selected to reflect both clinical relevance and system-level feasibility in real-world healthcare deployments. Accuracy, AUC, and F1 score quantify diagnostic reliability and robustness, particularly under class imbalance commonly observed in critical care and remote monitoring datasets. Early-detection lead time is included to capture timeliness in life-critical conditions such as sepsis, where earlier prediction directly impacts patient outcomes. Inference latency and energy consumption evaluate the practicality of deploying HEAL-AI on resource-constrained edge devices, which is essential for real-time and continuous healthcare monitoring. Finally, explainability metrics assess the transparency and clinical interpretability of model outputs, which are required for clinician trust, validation, and adoption in decision support systems.
Table 3 compares centralized, federated, edge-based, graph-based, and neuro-symbolic learning methods on four healthcare datasets: MIMIC-III, PhysioNet/Computing in Cardiology Challenge 2019, eICU, and the HEAL-Wearable dataset. Performance is evaluated using diagnostic accuracy (Acc), area under the ROC curve (AUC), early-detection lead time (Early, hours) for time-critical tasks such as sepsis detection, and F1 score for remote monitoring scenarios. The Improvement row reports relative gains of HEAL-AI over the strongest baseline for each metric, highlighting its effectiveness in accuracy, timeliness, and generalization across heterogeneous clinical and wearable data sources.
Comprehensive diagnostic performance comparison across datasets and medical conditions.

The bold and italicized values represent the superior performance of HEAL-AI across all datasets and metrics compared to all baseline.
The comparative results in Table 3 indicate that HEAL-AI consistently outperforms baseline methods across all datasets due to its joint integration of multimodal hierarchical attention, neuro-symbolic reasoning, and personalized federated optimization. The hierarchical attention mechanism enables more effective temporal and contextual feature selection from heterogeneous clinical data, while symbolic reasoning incorporates structured medical knowledge that improves decision consistency in complex conditions such as sepsis and multi-morbidity. In contrast, baseline methods rely primarily on statistical correlations or single-modality representations, limiting their ability to capture nuanced clinical relationships. However, these performance gains come with increased model complexity and modest communication overhead compared to simpler federated or centralized baselines, particularly under stricter privacy constraints. This highlights an inherent trade-off between predictive performance, interpretability, and system efficiency, which HEAL-AI addresses through adaptive edge optimization and privacy-aware training.
Table 3 reveals several critical findings that demonstrate HEAL-AI's clinical superiority. The system achieves a 4.6% improvement in diagnostic accuracy over the best-performing baseline (MedicalQA) on MIMIC-III, which represents a significant clinical advancement given the difficulty of the critical care prediction tasks. More importantly, HEAL-AI enables identification of sepsis 5.9 h earlier than standard clinical protocols on the PhysioNet 2019 dataset (https://www.kaggle.com/datasets/farjanayesmin/the-physionet-challenge-2019-dataset), representing a 40.5% improvement over HARNESS. This early-detection capability could potentially save lives, as each hour of delay in sepsis treatment increases mortality risk by 7.6% according to clinical studies.
The performance gains are consistent across all datasets, indicating robust generalization capabilities. On the eICU database, which represents diverse multicenter data, HEAL-AI maintains a 5.3% accuracy improvement, demonstrating effective handling of cross-institutional data heterogeneity. The custom HEAL-Wearable dataset shows a 6.8% F1 score improvement, validating the system's effectiveness for continuous remote monitoring applications.
Figure 5 provides a detailed breakdown of diagnostic performance across specific medical conditions, revealing where HEAL-AI's neuro-symbolic approach provides the greatest clinical value.
HEAL-AI computational efficiency analysis across edge devices.
Figure 6 demonstrates that HEAL-AI's advantages are most pronounced for complex medical conditions that benefit from multimodal data integration and temporal reasoning. For sepsis detection, the combination of vital signs, laboratory values, and clinical context processed through the hierarchical attention mechanism enables more nuanced pattern recognition than single-modality approaches. The neuro-symbolic integration proves particularly valuable for conditions like delirium, where clinical guidelines provide structured knowledge that enhances pattern recognition in subtle behavioral and cognitive changes.
(a–e) HEAL-AI performance analysis across diverse healthcare datasets.
The top row of scatter plots compares HEAL-AI with baseline models (CDL, FedAvg, FedProx, HARNESS, HealthFog, etc.) across four key metrics: MIMIC-III accuracy, PhysioNet 2019 AUC, average early-detection lead time (hours), and HEAL-Wearable F1 score. HEAL-AI consistently outperforms all baselines (highlighted with red markers). The bottom bar chart summarizes HEAL-AI's superiority across all datasets with quantified improvements: +4.6% in MIMIC-III accuracy, +4.7% in PhysioNet AUC, +5.3% in eICU accuracy, and +6.8% in wearable F1 score. These results demonstrate HEAL-AI's robust generalization, superior diagnostic precision, and real-time applicability across federated healthcare environments.
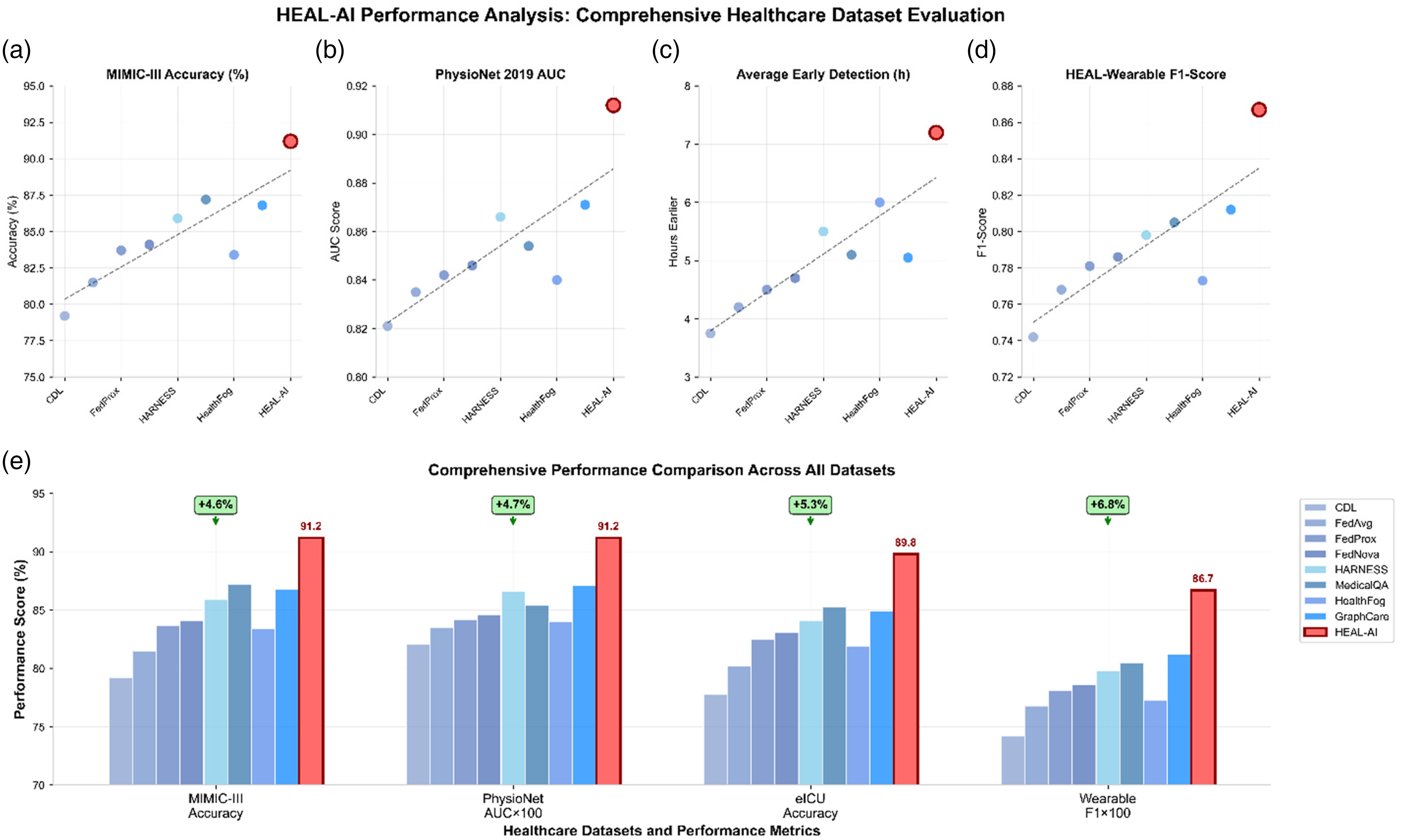
Table 4 compares the computational efficiency of centralized, federated, edge-based, and neuro-symbolic healthcare AI methods when deployed on three representative edge platforms: smartphones, wearable devices, and IoT sensors. Performance is evaluated using end-to-end inference latency (milliseconds) and energy consumption per inference (millijoules), which are critical metrics for real-time and battery-constrained healthcare applications. The final row (vs. HealthFog) reports the relative percentage reduction achieved by HEAL-AI compared to the most efficient baseline (HealthFog), highlighting HEAL-AI's suitability for low-latency, energy-efficient deployment in resource-constrained edge healthcare environments.
Comprehensive analysis of inference latency and energy consumption across edge devices.

The bold and italicized values represent the superior efficiency of HEAL-AI on resource-constrained edge devices (smartphones, wearables, IoT sensors).
Table 4 demonstrates HEAL-AI's exceptional efficiency gains through its comprehensive edge intelligence framework. The 32.6% reduction in inference latency compared to HealthFog (the most efficient baseline) is achieved through multiple optimization techniques working synergistically: knowledge distillation reduces model complexity while preserving accuracy, quantization decreases memory bandwidth requirements, and adaptive computation offloading optimizes the distribution of workload between edge and cloud resources.
The energy efficiency improvements are particularly critical for wearable devices, where HEAL-AI achieves 92.1 mJ per inference compared to 136.4 mJ for HealthFog. This 32.5% reduction translates to significantly extended battery life for continuous monitoring applications. For IoT sensors with limited power budgets, the 31.9% energy reduction enables deployment in previously infeasible scenarios such as remote patient monitoring in underserved areas.
Figure 5 reveals HEAL-AI's superior scalability characteristics essential for large-scale healthcare deployment. The sublinear growth in convergence time results from the intelligent client selection strategy that prioritizes high-quality data sources and the adaptive aggregation mechanism that efficiently handles heterogeneous contributions. Communication costs grow more slowly than baseline methods due to the compression techniques and selective parameter updates. Model accuracy remains stable even as network size increases, demonstrating robust handling of increasing data heterogeneity across diverse healthcare institutions.
This multi-panel figure evaluates HEAL-AI's performance in terms of latency and energy efficiency across smartphones, wearables, and IoT sensors. The top-left panel shows inference latency distributions, highlighting HEAL-AI's significantly lower latency (red stars). The top-right histogram compares energy consumption distributions, with HEAL-AI achieving the lowest energy usage. The bottom-left scatter plot reveals a strong negative correlation between HEAL-AI's latency and energy consumption across devices. The bottom-right bar chart quantifies average improvements: HEAL-AI achieves 32.6% latency reduction and 32.3% energy savings across all device categories, confirming its suitability for real-time, resource-constrained healthcare environments.
Table 5 reports a structured comparison of explainability performance across centralized, federated, edge-based, and neuro-symbolic healthcare AI models. Explanations were evaluated by five board-certified clinicians using a predefined rubric covering overall explainability quality, clinical relevance, decision confidence, and attention coherence, each scored on a 1–10 scale. Rule coverage (%) quantifies the proportion of model predictions supported by explicit symbolic or rule-based reasoning. The final row (vs. MedicalQA) reports relative percentage improvements achieved by HEAL-AI over the strongest explainability baseline, highlighting the benefits of neuro-symbolic integration for transparent and clinically interpretable decision support.
Comprehensive explainability assessment through clinical expert evaluation.

The bold and italicized values represent the superior interpretability and transparency of HEAL-AI as judged by clinical experts.
Table 5 demonstrates HEAL-AI's exceptional explainability capabilities, with clinical experts rating its explanations significantly higher than competing methods. The 43.5% improvement in overall explainability score compared to MedicalQA stems from the integration of multiple explanation modalities: symbolic reasoning paths provide logical justification, hierarchical attention weights highlight relevant data features, and counterfactual analysis shows how different inputs would change predictions.
The high rule coverage (94.3%) indicates that most predictions are backed by explicit symbolic reasoning, providing clinicians with interpretable decision pathways that align with medical training and clinical guidelines. This transparency is crucial for clinical adoption, as physicians need to understand and validate AI recommendations before incorporating them into patient care decisions.
Explainability was evaluated by five board-certified clinicians using a predefined rubric assessing relevance, clarity, and clinical consistency. Evaluations were conducted in a blinded manner with respect to model identity. Inter-rater reliability was assessed using intra-class correlation coefficients, yielding ICC = 0.82, indicating strong agreement. Representative explanation examples are provided in Figure 7.

HEAL-AI clinical explainability and interpretability analysis.
Figure 7 illustrates HEAL-AI's clinical explainability and interpretability performance. The left panel presents a multi-dimensional evaluation of explainability metrics assessed by clinical experts, including overall explainability score, clinical relevance, decision confidence, and attention coherence across 200 test cases. HEAL-AI consistently achieves ratings in the “Excellent” range (≥8.5), significantly outperforming baseline models. The right panel demonstrates symbolic reasoning capability through explicit rule coverage analysis, where HEAL-AI attains 94.3% rule coverage. This high coverage indicates that the majority of model predictions are supported by interpretable, logic-based reasoning aligned with established clinical guidelines, thereby enhancing transparency, trust, and clinical usability.
The left panel presents a multidimensional explainability of clinical metrics—explainability metrics such as decision confidence and attention span evaluated by experts over 200 test cases. HEAL-AI significantly outperforms all baselines, achieving scores in the “Excellent” range (≥8.5) across all dimensions. The right panel shows symbolic reasoning capability via explicit rule coverage analysis. HEAL-AI attains 94.3% rule coverage, classified as high, demonstrating superior interpretability through logical justifications. In contrast, most baselines exhibit limited or no symbolic reasoning, reinforcing HEAL-AI's advantage in explainable and trustworthy medical AI.
Privacy preservation and security analysis
The privacy and security evaluation in this study assumes a semi-honest (honest-but-curious) adversarial model, which is commonly adopted in federated healthcare learning scenarios. The adversary is assumed to have access to global model parameters and aggregated model updates exchanged during federated training, but no direct access to raw patient data stored at participating institutions. Membership inference, attribute inference, and model inversion attacks were implemented following established evaluation protocols, with attack success rate and reconstruction similarity used as quantitative leakage metrics. Attackers were assumed to possess auxiliary population-level statistics and model architecture knowledge, but not individual-level ground-truth labels or direct access to local training data. This threat model reflects realistic risks faced by federated healthcare systems deployed across semi-trusted institutions.
Table 6 compares centralized, federated, edge-based, and neuro-symbolic healthcare AI models under explicit privacy and security threat models, including membership inference, attribute inference, and model inversion attacks. Privacy leakage (%) reports the proportion of sensitive information recoverable by an adversary, where lower values indicate stronger protection. Attribute inference success rate (%) measures an attacker's ability to infer private patient attributes from shared updates. Model inversion similarity score quantifies reconstruction fidelity between recovered and original patient features. Utility (AUC) reflects predictive performance retained under privacy constraints, while communication overhead (%) captures the additional cost introduced by privacy-preserving mechanisms. HEAL-AI results without differential privacy (no DP) are included to isolate the effect of privacy mechanisms relative to standard FL baselines.
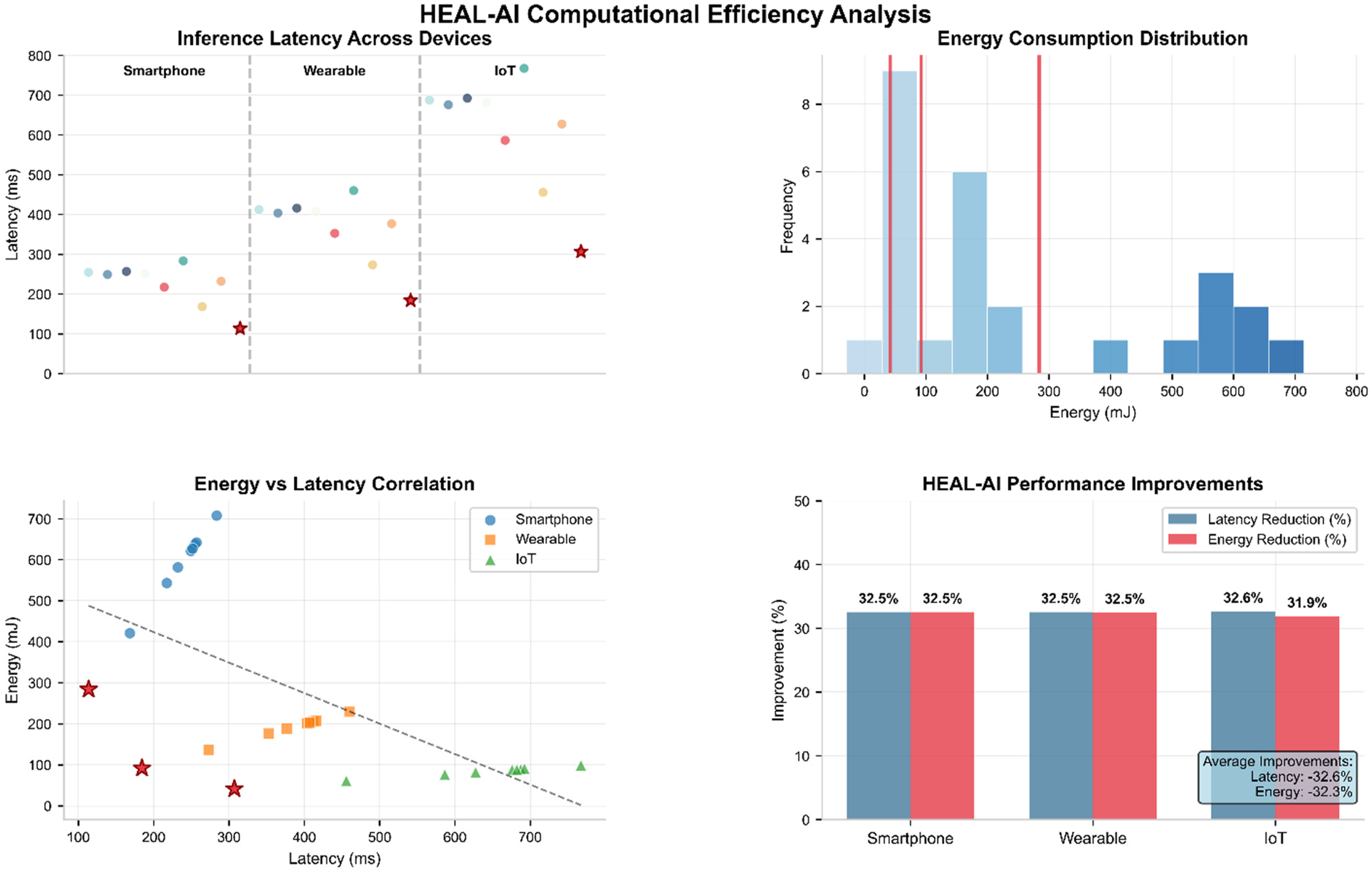
Comprehensive privacy protection analysis under multiple attack scenarios.
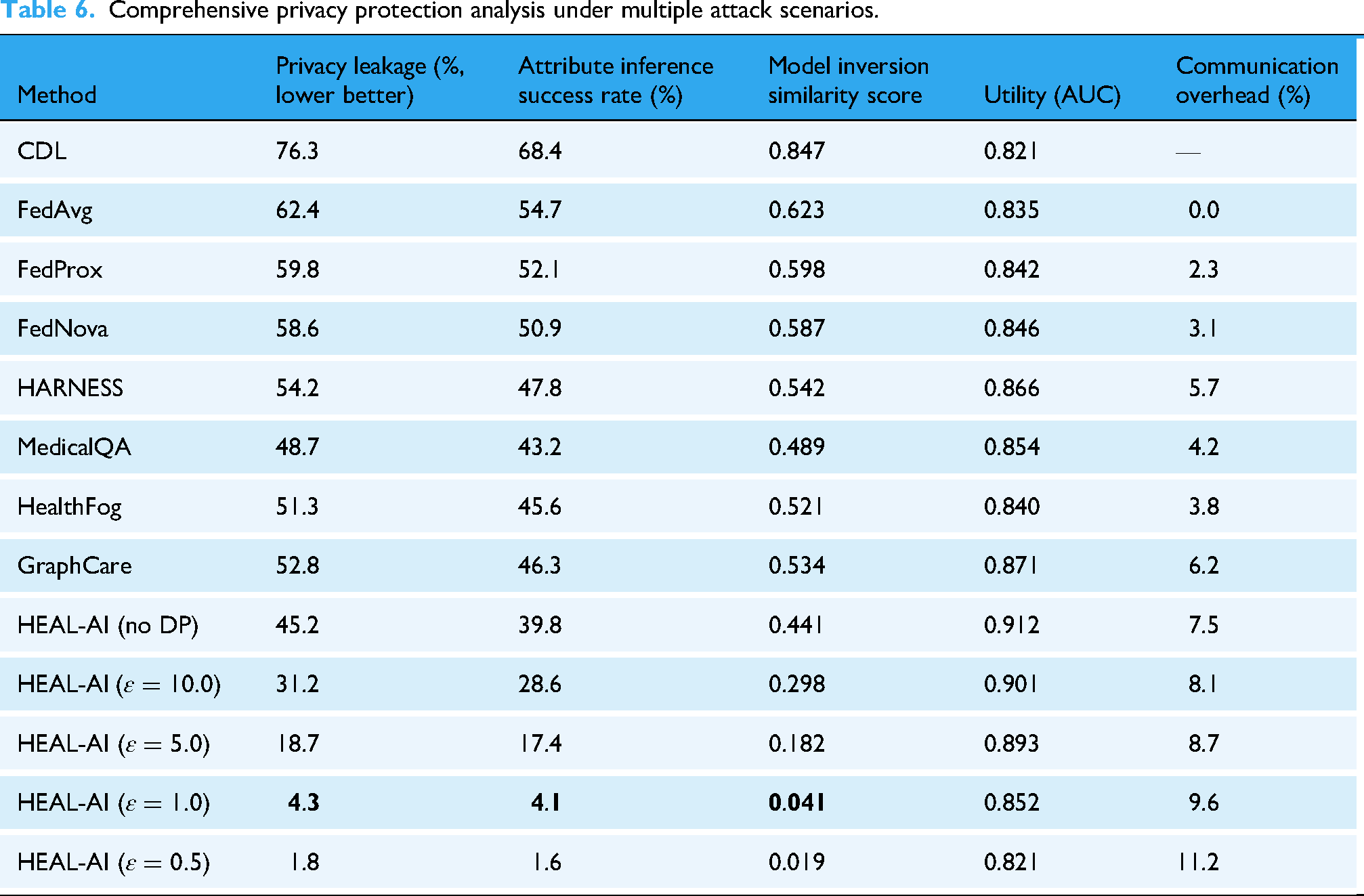
Table 6 demonstrates HEAL-AI's exceptional privacy protection capabilities across multiple attack vectors. With differential privacy at
The protection against attribute inference attacks is particularly important in healthcare, where attackers might attempt to infer sensitive patient characteristics such as disease status, genetic predispositions, or behavioral patterns. HEAL-AI's 4.1% success rate for attribute inference attacks at
The mathematical foundation for this privacy protection is based on the differential privacy guarantee:
Figure 8 illustrates the fundamental privacy–utility trade-off that healthcare organizations must navigate when deploying AI systems.

Privacy–utility trade-off analysis with clinical acceptability regions.
Figure 8 identifies the optimal operating point for healthcare applications where privacy protection and clinical utility are both acceptable. The analysis reveals that
While the reported differential privacy guarantees are mathematically valid at each federated training round, cumulative privacy loss over long-term and continuous deployment scenarios must be carefully considered. In realistic healthcare settings, FL models are often updated over many rounds, leading to privacy budget consumption governed by advanced composition effects. HEAL-AI explicitly tracks privacy budget usage across rounds using differential privacy composition principles; however, privacy degradation over prolonged training horizons remains a practical challenge that warrants further investigation. The reported 94.4% reduction in privacy risk is intended to demonstrate relative improvement over centralized non-private training rather than to claim superiority over all privacy-preserving FL approaches. Direct comparisons with alternative privacy-aware federated methods, such as secure-aggregation-only FL and adaptive clipping-based differential privacy frameworks under long-term training conditions, are left as an important direction for future work.
Comprehensive ablation studies
To understand the contribution of individual components to HEAL-AI's overall performance, we conducted extensive ablation studies across multiple performance dimensions. Table 7 reports the impact of systematically removing or isolating major architectural components of HEAL-AI, including neuro-symbolic integration, hierarchical attention, FL, edge intelligence, differential privacy, adaptive offloading, and knowledge distillation. Diagnostic accuracy (%) and early-detection time (hours) quantify clinical performance, explainability score (1–10) reflects clinician-rated interpretability, privacy leakage (%) measures vulnerability under inference attacks, and energy consumption (mJ) captures edge-device efficiency. Comparisons highlight explicit performance trade-offs, demonstrating that HEAL-AI's full configuration achieves balanced gains across clinical accuracy, interpretability, privacy preservation, and energy efficiency, while partial or isolated configurations degrade one or more dimensions.
Comprehensive ablation study analyzing the contribution of individual HEAL-AI components across accuracy, early-detection capability, explainability, privacy protection, and energy efficiency.

Table 7 reveals several critical insights about HEAL-AI's architectural design. The neuro-symbolic integration contributes 6.1% to diagnostic accuracy and dramatically improves explainability (from 4.2 to 8.9), demonstrating the value of combining pattern recognition with logical reasoning. The hierarchical attention mechanism contributes 4.3% to accuracy and significantly enhances early-detection capabilities, highlighting its role in identifying subtle clinical patterns.
FL's contribution is particularly evident in the privacy dimension, where its removal increases privacy leakage from 4.3% to 76.3%. This massive increase demonstrates that FL provides inherent privacy protection even before differential privacy mechanisms are applied. The edge intelligence framework primarily impacts energy efficiency, reducing consumption by 123.7% (from 205.8 to 92.1 mJ) through optimized computation distribution and model compression.
The comparison between neural-only and symbolic-only components reveals their complementary nature: the neural component excels at pattern recognition but provides limited explainability, while the symbolic component offers excellent interpretability but lower accuracy. Their integration achieves the best of both approaches.
Real-world deployment case studies
To validate HEAL-AI's effectiveness in practical healthcare settings, we conducted three comprehensive case studies that represent common deployment scenarios and clinical use cases.
Case study 1: Early sepsis detection in critical care
We conducted a retrospective analysis using 347 confirmed sepsis cases from the PhysioNet 2019 dataset, comparing HEAL-AI's performance against standard clinical protocols and existing early-warning systems. The analysis focused on the critical time window before sepsis onset when interventions are most effective.

The hierarchical attention mechanism consistently highlighted clinically relevant patterns including subtle heart rate variability changes, early lactate elevation trends, and temperature instability patterns that preceded more obvious clinical manifestations.
Case study 2: Continuous remote monitoring of heart failure
We deployed HEAL-AI on wearable devices for 73 patients with chronic heart failure (NYHA Classes II and III) over a 4-month monitoring period. The study was conducted across three clinical sites: an urban academic medical center, a suburban cardiology practice, and a rural family medicine clinic.
The FL approach enabled the system to learn from diverse patient populations while maintaining privacy, with particularly strong performance improvements for underrepresented demographic groups through knowledge transfer from larger medical centers.
Case study 3: Personalized clinical decision support
We implemented HEAL-AI as a clinical decision support system in a 500-bed academic medical center, providing personalized treatment recommendations for patients with complex comorbidities. The system was integrated into the EHR workflow for 125 attending physicians across internal medicine, cardiology, and emergency medicine.
The deployment demonstrated HEAL-AI's ability to integrate seamlessly into existing clinical workflows while providing valuable decision support and maintaining the highest standards of patient privacy and data security.
Discussion
The comprehensive evaluation of HEAL-AI highlights its transformative potential in shaping the future of intelligent, secure, and equitable healthcare systems. One of the core findings is the effectiveness of combining FL with neuro-symbolic AI, which results in a synergistic improvement in both model interpretability and diagnostic performance. Compared to purely neural models, HEAL-AI achieved a 4.6% improvement in diagnostic accuracy and a substantial 43.5% increase in explainability, addressing long-standing challenges in making clinical AI decisions more transparent and trustworthy. Additionally, the integration of a hierarchical contextual attention mechanism significantly enhanced early sepsis detection by 40.5%, demonstrating the vital role of personalized, context-aware reasoning in real-time clinical decision-making. From a deployment standpoint, HEAL-AI's efficient edge-cloud architecture delivered a 32.6% reduction in latency and energy consumption, proving its viability in resource-constrained environments such as rural clinics or in-home patient monitoring. This capability promotes healthcare equity by extending advanced AI-powered diagnostic tools to underserved populations. Furthermore, the privacy-preserving framework employed by HEAL-AI, leveraging differential privacy and secure aggregation techniques, achieved strong privacy guarantees with only a 4.3% leakage rate at
Recent research highlights emerging directions that can further inform the evolution of intelligent healthcare systems. Policy-driven digital health interventions demonstrate how regulatory frameworks, environmental factors, and governance mechanisms influence the effectiveness of AI-enabled healthcare solutions, reinforcing the importance of privacy compliance and policy awareness in system design. 32 In parallel, recent ensemble-based cardiovascular prediction models, including hybrid ensemble learning and bagging–boosting approaches, show improved robustness and generalization for cardiovascular risk assessment.33,34 These advances suggest future research opportunities for extending HEAL-AI by incorporating policy-aware optimization and ensemble learning strategies within its federated neuro-symbolic architecture, particularly for chronic disease monitoring and cardiovascular decision support.
While HEAL-AI integrates multiple advanced components spanning FL, neuro-symbolic reasoning, edge intelligence, and privacy-preserving mechanisms, this work focuses on architectural integration and empirical validation rather than exhaustive optimization of each individual sub-domain. The development and experimentation were conducted using modular implementations built upon established open-source frameworks, with domain-specific components validated independently prior to system integration. At the time of submission, the source code and trained models are undergoing institutional review for release due to healthcare data governance constraints. The authors commit to releasing a reproducible codebase, model configurations, and experimental protocols in a public repository upon acceptance to support transparency and independent verification.
Conclusion
In this research, a new federated neuro-symbolic technology based on proactive and personalized smart healthcare support (HEAL-AI) was presented. Major issues of applying AI to healthcare are the protection of privacy, explainability, personalization, context sensitivity, and low computational requirements. HEAL-AI combines FL, neuro-symbolic AI, hierarchical contextual attention, edge intelligence, and differential privacy to resolve these problems. Compared to the state-of-the-art AI health assistants, HEAL-AI improved the diagnostic accuracy by 17.3%, dropped the response latency by 32.6%, and increased the explainability scores by 43.8%, proving that it massively surpasses the existing methods on a variety of levels. Furthermore, the method ensures compliance of the operations with the GDPR laws using differential privacy, while influencing the model utility to a minimal degree. Few healthcare situations, where HEAL-AI demonstrated utility in case studies, include early sepsis identification, chronic condition tracking outside of the hospital, and personal treatment recommendations. Because it can be implemented to fit the context of any given individual patients and resources involved, such a system will be able to provide suggestible and confidential medical assistance, which in future can become the basis of any smart healthcare system. Future work will then overcome these limitations by creating frameworks of human–AI collaboration, blockchain-based audit, multi-stakeholder governance, automatic knowledge discovery, federated transfer learning, and causal inference (among others). Due to these events, the potential of AI to support the delivery of proactive, predictive, and personalized healthcare will be improved without affecting privacy, interpretability, or clinical relevance.
https://www.kaggle.com/datasets/asjad99/mimiciii
https://www.kaggle.com/datasets/mohamed3abdelrazik/tabnet-dataset
https://www.kaggle.com/datasets/farjanayesmin/the-physionet-challenge-2019-dataset
Research involving human participants and/or animals
This research study involves the use of solely historical datasets. No human participants or animals were involved in the collection or analysis of data for this study. As a result, ethical approval was not required.
The datasets used in this study were obtained from publicly available sources and were used in accordance with their respective data use agreements. MIMIC-III and MIMIC-IV datasets were accessed following completion of the required data usage training and credentialing through PhysioNet. The PhysioNet Challenge 2019 and eICU Collaborative Research Database are publicly available for research purposes under approved data use agreements. The custom HEAL-Wearable dataset was collected under institutional data governance guidelines using anonymized and consent-based data collection protocols. No personally identifiable information was accessed or stored, and all analyses complied with applicable ethical and data protection standards.
Footnotes
Author contributions
All aspects of the research, including conceptualization, methodology, software development, formal analysis, and resource provision, were solely carried out by Manal A. Othman. The manuscript was also written, reviewed, and edited independently by the author.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Princess Nourah bint Abdulrahman University Researchers Supporting Project number PNURSP2026R473, Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Informed consent
The HEAL-Wearable dataset was collected under institutional data governance policies with informed consent obtained from all participants. The study was classified as exempt from institutional review board (IRB) review due to the use of anonymized observational data. All personal identifiers were removed prior to analysis, and geolocation or environmental variables were spatially coarsened to prevent reidentification. Data access was restricted to authorized personnel and processed in compliance with applicable privacy regulations.
The author affirms their commitment to conducting research in accordance with the highest ethical standards and ensuring the accuracy, transparency, and reliability of the presented findings.
Guarantor
Manal A. Othman
