Abstract
Background
Persistent symptoms after acute COVID-19, formally recognized as post-COVID-19 condition (PCC), are prevalent and disabling. The World Health Organization has emphasized early, multidisciplinary rehabilitation using accessible and hybrid care models. However, no standard protocol currently exists, and few studies have evaluated digital formats in this context.
Objective
To examine the effects of two digital rehabilitation formats—immersive virtual reality (IVR) and a self-guided online program—on cognitive and functional outcomes in individuals with PCC.
Methods
The study used a nonrandomized, quasiexperimental pre–post design. We enrolled 172 adults with PCC into a 12-week multimodal program combining cognitive training, physical activity, and mindfulness; participants chose IVR or an online self-guided program, and usual-care controls were recruited at a separate site. Outcomes were assessed before and after across cognitive, psychological, lifestyle, functional, and quality-of-life domains.
Results
Significant group-by-time interactions were observed across multiple domains. IVR was associated with improvements in global cognition, immediate recall, recognition memory, and response inhibition. The online intervention showed improvements in verbal recognition, mindfulness, and perceived health-related quality of life. Additional outcomes showed significant group-by-time interactions without postintervention differences, indicating differential trajectories over time rather than discrete between-group effects.
Conclusions
Both IVR and online multimodal interventions were associated with domain-specific cognitive and functional benefits in individuals with PCC. These findings support the potential value of multicomponent rehabilitation approaches, while highlighting that some effects reflect differential trajectories rather than definitive postintervention group differences.
Study Registration
www.ClinicalTrials.gov, identifier NCT05846126 (date of registration 04 May 2023).
Keywords
Introduction
Many individuals recovering from acute COVID-19 continue to experience persistent symptoms, often compromising daily functioning and quality of life (QoL). This condition is formally recognized as post-COVID-19 condition (PCC). It is defined as the continuation or onset of new symptoms at least 3 months after the initial SARS-CoV-2 infection, lasting for at least 2 months and not explained by other diagnoses. 1 Persistent fatigue and cognitive impairment are among the most common symptoms reported in individuals with PCC, often affecting memory, executive functioning, and processing speed. 2 These difficulties are frequently accompanied by emotional symptoms, such as anxiety and depression, which may in some cases reach clinically significant levels.3,4 Other manifestations—such as sleep disturbances, autonomic dysfunction, or residual respiratory complaints—have also been described in this population.5,6
In response, the World Health Organization (WHO) has identified the rehabilitation of medium- and long-term post-COVID sequelae as a priority area, recommending early assessment and multidisciplinary, personalized care. 7 Hybrid care models—combining remote and in-person modalities—are particularly emphasized to ensure accessibility and adaptability across healthcare systems. 7 Although no standard rehabilitation protocol currently exists for PCC, there is growing interest in adapting evidence-based interventions from other clinical populations.
Cognitive training consists of structured, repeated mental exercises that inherently engage multiple cognitive processes, irrespective of the specific cognitive outcomes later assessed
Physical exercise contributes to physical fitness and has been associated with cognitive and emotional benefits in several clinical populations, including chronic fatigue syndrome, 16 fibromyalgia, 17 acquired brain injury, 18 MCI and Alzheimer's disease, 19 and Parkinson's disease. 20 Virtual reality (VR)-based exercise has been associated with improvements in working memory performance. 21 Some evidence also suggests associations with gains in physical performance (e.g., muscular strength and aerobic capacity) and with psychological and physiological exercise-related outcomes, including reduced perceived exertion and improvements in motivation and self-efficacy. 22 However, these effects are typically modest and domain-specific, supporting its integration as one component within broader multimodal rehabilitation approaches. In PCC, emerging evidence indicates that closely supervised and fatigue-adapted exercise programs are safe and can improve physical capacity and overall well-being without exacerbating post-exertional symptoms. 23
Mindfulness-based interventions (MBIs), defined as “paying attention in a specific way: intentionally, in the present moment, and nonjudgmentally” 24 have shown moderate effects in improving well-being and psychological functioning across a variety of medical conditions. 25 Evidence also supports their efficacy in clinical populations with anxiety and depression,26–28 with some MBIs demonstrating short-term effects comparable to cognitive behavioral therapy in anxiety disorders. 29 Furthermore, MBIs have produced sustained improvements in individuals with stress or subclinical anxiety symptoms, lasting up to several months postintervention. 30 Mindfulness training in VR has been associated with significant mindfulness improvements in individuals with mental health disorders, but not in those with chronic physical conditions. 31 Online MBIs have been shown to reduce depression, anxiety, and stress in post-COVID populations.32,33
Studies in healthy adults and clinical populations suggest that combined programs outperform single-modality interventions in cognitive performance and well-being. This advantage is commonly attributed to the multidimensional nature of symptoms, the complementary mechanisms targeted by different components, and higher engagement compared with isolated interventions.34–38 In healthy young adults, combining physical exercise with cognitive training has been associated with more selective memory benefits than exercise alone. 34 In patients with chronic stroke, adding of mindfulness or physical exercise to cognitive training has been linked to modality-specific emotional or physical benefits. 37 Consistently, a recent meta-analysis indicates that combining physical activity with mindfulness is effective in improving mental health and well-being. 38 In PCC, a pilot study integrating cognitive training, physical exercise, and mindfulness, delivered using IVR, reported preliminary improvements in cognitive and mental health outcomes compared with control groups. 39
Beyond the intervention components, considerations regarding implementation and access are particularly relevant in PCC, a condition that affects millions worldwide. Expanding access to rehabilitation requires offering alternatives to traditional, hospital-based programs. Effective formats must be both scalable and generalizable to patients’ daily life activities. In practice, this can be achieved through approaches such as web-based platforms or IVR. IVR refers to environments that provide a first-person perspective and a sense of presence, including 360° scenarios delivered through head-mounted displays or projection-based systems, regardless of the level of user interactivity.
Web-based platforms are typically designed to facilitate remote and flexible access to care, thereby reducing geographical and logistical barriers,40,41 and have been implemented in interventions in both healthy and clinical populations, with reported good feasibility and adherence across settings.10,12,13,32 IVR, in contrast, has been primarily developed as a delivery format to enhance engagement and real-life applicability through immersive environments that simulate everyday activities42,43 and has been applied in rehabilitation across a range of intervention approaches.14,15,21,22,31,39
Importantly, these characteristics are not exclusive to either format. IVR interventions have also been delivered remotely in other contexts, including PCC, demonstrating the feasibility of home-based VR programs, 44 while web-based platforms have incorporated interactive, multimedia, and persuasive design strategies to promote engagement and adherence in daily life. 41 Despite these advances, routine clinical implementation of VR remains limited by implementation and infrastructure constraints. 43
In PCC, rehabilitation research has not yet established reference programs or criteria to guide intervention design or delivery format. The primary objective was to evaluate the effectiveness of two multimodal intervention programs, based on shared cognitive, physical, and mindfulness components but delivered through different digital formats, in improving cognitive functioning and psychological well-being in individuals with PCC, compared with a passive control group.
Methods
Design, participants, and setting
The RehabCOVID project was a three-arm, parallel, quasiexperimental study conducted by the Consorci Sanitari de Terrassa–University Hospital (CST) in collaboration with the Universitat de Barcelona, the Universitat de Girona, the Universitat Politècnica de Catalunya, and the Hospital Universitari Santa Maria. It followed a pragmatic, effectiveness-oriented design. Rather than aiming to establish experimental efficacy under highly controlled conditions, the study prioritized ecological validity and real-world applicability in routine clinical practice. This design choice reflects the absence of standardized rehabilitation protocols for PCC and the need to evaluate feasible and scalable interventions that can be implemented in real-world healthcare settings. No a priori hypothesis was formulated regarding the superiority of one intervention delivery format over the other; however, it was hypothesized that participants in the intervention groups would show improvements or maintain their functioning relative to controls.
A total of 172 participants with PCC participated in one of three study arms: an online intervention, an IVR intervention, or a passive control group. Interventions lasted 12 weeks, with pre- and postintervention assessments. Participants were recruited via clinician referral and self-referral using convenience sampling; no systematic sampling plan was implemented. Prior to study inclusion, all potential participants underwent an initial screening and eligibility assessment to confirm PCC and compliance with inclusion and exclusion criteria. Eligibility criteria operated exclusively at the individual level, with no site- or city-level restrictions across CST and Hospital Universitari Santa Maria.
Participants in the intervention arms completed their baseline and follow-up assessments, as well as the IVR sessions, at the Neuropsychology Unit of the CST. For logistical reasons, most control assessments were conducted at a different location, the Hospital Santa Maria (Lleida, Spain), although a subset was assessed at the main study site. Participants were not necessarily patients at these hospitals; rather, they were referred or self-referred based on proximity and interest in participating. Site assignment was based on logistical feasibility and availability of intervention facilities.
The study was conducted between May 2023 and December 2024, with recruitment running from May 2023 to September 2024 and postintervention follow-up completed by December 2024. It was approved by the Drug Research Ethics Committee (CEIm) of the Consorci Sanitari de Terrassa (reference code: 01-21-107-111) and the Clinical Research Ethics Committee (CEIC) of the Hospital Santa Maria (reference code: CEIC-2897). All participants provided written informed consent, and all procedures were conducted in accordance with relevant ethical guidelines and regulations. The trial was registered at ClinicalTrials.gov (NCT05846126, date of registration: 4 May 2023). The implemented design deviated from the original protocol. Deviations included nonrandom, preference-based allocation with an imbalanced ∼1:2:1 group ratio; absence of assessor blinding due to cross-site logistics; and a pragmatic increase in enrollment to mitigate expected attrition. Further details on this deviation are provided in the Supplementary Material (Table S1).
The study was designed as a pragmatic effectiveness trial aimed at evaluating the real-world applicability and feasibility of two technology-delivered multimodal rehabilitation programs for individuals with PCC.
Inclusion criteria
Inclusion criteria were: (a) men and women aged 18–65 years; (b) confirmed SARS-CoV-2 infection documented by a positive polymerase chain reaction (PCR) or rapid antigen test; (c) reporting subjective cognitive complaints at least 12 weeks after acute infection, expressed in lay terms and not restricted to a specific cognitive domain, and confirmed during an initial screening conducted by a trained neuropsychologist; and (d) consent from a physician to engage in a physical exercise intervention.
Exclusion criteria were: (a) established diagnosis before COVID-19 disease of psychiatric, neurological, developmental disorder, or systemic pathologies known to cause cognitive deficits; and (b) motor or sensory alterations that impede the rehabilitation program.
Sample size
Sample size estimation was conducted using G*Power (version 3.1.9.6), 45 based on a repeated-measures analysis of variance (ANOVA) design with three groups (control, online intervention, and IVR intervention) and two time points (pre and post). Assuming a small-to-moderate effect size (f = 0.20), informed by effect sizes reported in previous cognitive and rehabilitation intervention studies in related populations, a correlation of 0.5 between repeated measures, an alpha risk of 0.05, and 80% power, the required sample size was approximately 52 participants per group (total N = 156). Based on prior clinical experience with this population, an attrition rate of approximately 10–15% was anticipated at the design stage. To mitigate the impact of potential losses during the intervention period and to preserve the planned number of evaluable participants at follow-up, the recruitment target was pragmatically increased beyond the minimum required sample size, resulting in a final recruitment target of 172 participants. No interim analyses were planned, and no formal stopping rules were specified; recruitment continued until the prespecified study end.
Participant allocation across groups
The unit of assignment was the individual participant. Although the original protocol specified random assignment to the study groups, this procedure was never implemented. Recruitment revealed that participation was only feasible if participants were allowed to choose the intervention format. Accordingly, participants were recruited but not immediately assigned to any group and remained on hold, while the allocation procedure was reconsidered.
No randomization procedure was applied prior to group assignment. Allocation therefore deviated from the published protocol and followed a nonrandomized process defined prior to group assignment. Participants eligible for an intervention were allowed to choose between the IVR and the online formats according to their availability for in-person sessions, access to technology, and scheduling constraints. Participants in the control group did not choose their allocation; they were directly invited to participate as controls and were not offered access to any intervention format. This approach was adopted to ensure feasibility and maintain participation in a population with marked fatigue, symptom variability, and frequent logistical constraints, including transportation difficulties, health-related instability, and competing work or caregiving demands.
Blinding (masking)
Given the nature of the interventions (IVR, self-guided online, and control), participants and intervention staff were not blinded. Outcome assessors were not blinded to group assignment. For data analysis, group labels were numerically coded with the intent to mask allocation; however, participant identifiers encoded the treatment format, so masking of analysts was incomplete. No formal assessment of blinding was performed.
Intervention
The study included three arms: a passive control group, an online multi modal program, and an IVR multimodal program. Both intervention arms comprised 24 sessions over 12 weeks (two sessions per week; ∼60 min each) with a fixed sequence of mindfulness practice, cognitive training, and multicomponent physical exercise, adapted to the delivery mode.
Online multimodal program
The online program was delivered via a customized instance of the iCF platform, a patient-oriented e-training environment developed by Corporación Fisiogestión. 46 This platform allows the integration of multimedia content, progression tracking, and user-specific prescription. Each session followed the same three-component structure—mindfulness training, cognitive stimulation, and multicomponent physical exercise—and was completed consecutively. The aerobic training component was not integrated into the platform sessions but was recommended separately as an independent activity. Participants accessed the materials at home or another preferred environment with internet access, and sessions were self-administered without a live facilitator.
Mindfulness training
The first part of each session was dedicated to mindfulness practice, with the aim of preparing the patient mentally and emotionally for the rest of the training. The content was based on a structured introductory mindfulness training program and included guided videos and audio recordings designed to support formal and informal mindfulness practice. 47 These materials introduced patients to the essential theoretical foundations of mindfulness, offering both formal techniques (e.g., breathing, body scan, meditation) and informal practices for everyday integration. Each session day introduced a new guided mindfulness practice of approximately 10 min. Practices were delivered in a progressive sequence, and participants were encouraged to apply the learned exercises independently during daily activities outside the guided sessions to support consolidation. Completion and access to each session were recorded automatically by the platform.
Cognitive training
Immediately following the mindfulness component, patients engaged in a 20-min cognitive training segment using the Cerebral Training platform. The aim was to stimulate neural circuits involved in attention, working memory, and executive functions, thereby enhancing cognitive efficiency in daily life. Each session included tasks across four domains: attention and reaction, working memory, visual and auditory memory, and executive control. A default level of training was prescribed unless significant physical or cognitive impairments were identified, in which case a low-level variant was assigned. An adaptive algorithm dynamically adjusted task difficulty based on real-time performance, and a virtual monitoring system flagged any irregularities requiring clinician review. Detailed performance data were available throughout the program to facilitate ongoing monitoring.
Multicomponent physical exercise
The final segment of each session involved a structured set of physical exercises targeting strength, balance, and flexibility. Exercises were selected from the WHO guide Support for Rehabilitation Self-Management after COVID-19-Related Illness 48 and organized into three progressive series, each including three difficulty levels. Initial prescription was based on dyspnea and fatigue using the mMRC Dyspnea Scale, 49 and progression was guided by the Borg Rating of Perceived Exertion 50 administered at sessions 8 and 16. Each exercise table was customized with parameters such as number of repetitions, sets, resistance, or elevation, and could be adjusted across the intervention period based on individual tolerance and performance. A progression log was maintained by the clinical team.
Aerobic training
In addition to the structured platform-based components, patients were instructed to perform aerobic activity on their own, choosing a modality that suited their preferences and functional status—such as walking, cycling, or running. This training was not included in the formal session structure and could be completed at any time during the day. The recommendation was to engage in aerobic activity for approximately 30 min per day, 5 days per week, with the goal of improving cardiovascular endurance and reducing postviral fatigue. No performance monitoring system was implemented for this component, but adherence was encouraged during routine follow-ups.
IVR multimodal program
The IVR multimodal intervention was delivered face-to-face in an immersive projection room using a 360° audiovisual system (MK2 projector, BroomX), in small groups of five participants, and was supervised by a neuropsychologist. Each 60-min session was organized into three consecutive blocks with a fixed order and duration: mindfulness training (10 min), cognitive training (30 min), and physical exercise (20 min). While the specific content varied across sessions, the overall structure and sequencing were consistent throughout the program. At the beginning of each session, the facilitator briefly introduced the objectives of the session and highlighted the relevance of transferring trained cognitive, emotional, and physical strategies to daily functioning.
The mindfulness component was based on established mindfulness-based approaches and focused on cultivating attentional regulation and body awareness. Short guided practices were used to promote a stable attentional and emotional state prior to the cognitively and physically demanding activities and included breathing-focused exercises, body awareness practices, and brief meditative techniques.
Cognitive training was delivered within ecologically valid virtual environments simulating real-life situations (e.g., walking in parks or urban settings) and sequentially targeted attention, processing speed, episodic memory, and executive functions, with task difficulty increasing progressively across sessions in the group IVR setting through systematic modifications of task demands within the same scenarios. Cognitive training activities included naturalistic observation-and-recall tasks designed to engage everyday cognitive demands, such as attending to complex scenes and recalling contextual details; numerical sequence tracking tasks involving the monitoring of color-coded elements; and visual search or counting tasks requiring identification or enumeration of target stimuli within the environment. Task difficulty increased progressively across sessions at the group level by systematically modifying task demands within the same scenarios, including the amount of information to be monitored or recalled, the number of stimuli presented, and the complexity of the required responses. No individual adaptive algorithms were applied.
The physical exercise component consisted of a structured functional training program aimed at activating large muscle groups and promoting basic mobility within a group-based immersive setting. Exercises were organized as continuous, low-impact repetitions and focused on balance, flexibility, and progressive muscle strengthening. Participants performed repeated sit-to-stand transitions from a chair, stationary pedaling movements, and step-based exercises involving repeated ascent and descent. The exercise block was delivered as a distinct segment within each session and was supervised by the facilitator to ensure appropriate execution and tolerance.
While the immersive projection device is a commercially available system, the intervention content—including the design of the exercises, session structure, and clinical application—was developed by the authors for this specific clinical purpose. The intervention does not rely on a conventional VR application such as a web-based platform, mobile app, or head-mounted display. Instead, it is delivered through a room-based 360-degree immersive projection system that creates a shared immersive environment without the use of headsets or wearable devices. The immersive system does not record interaction data or patient-generated metrics.
Control group
Participants assigned to the control group received treatment-as-usual, consisting of general health recommendations in line with standard clinical practice. Specifically, they were advised to engage in regular physical activity adapted to their condition, maintain cognitive stimulation through daily activities, and implement basic strategies for emotional self-regulation and stress management. No structured or supervised intervention was provided, and participants did not have access to the digital or immersive programs described above. All control participants were informed that they would be offered access to the online program after completing the final assessment.
Intervention adherence and safety
Adherence to the in-person IVR intervention was monitored directly by the research team, who recorded attendance at each session. Adherence was defined as the proportion of complete sessions attended out of the total scheduled sessions.
In the online program, the three structured components (mindfulness-based content, cognitive training, and physical exercise) were accessed independently through the digital platform. Adherence to each component was tracked automatically: the iCF platform registered access and completion of mindfulness-based content and physical exercise sessions, while the cognitive training platform recorded engagement and performance. As the online intervention did not consist of discrete, supervised sessions but of parallel components with independent usage patterns, overall adherence was operationalized as the arithmetic mean of adherence rates across the three structured components, reflecting global exposure to the multimodal program rather than completion of each component individually.
The research team reviewed usage/attendance logs on a regular basis and, when inactivity or missed components were detected, proactively contacted participants (phone/email) to troubleshoot barriers (e.g., technical issues, scheduling), provide brief assistance, and arrange rescheduling as needed. No financial incentives were provided.
Participants were considered adherent if they completed at least 80% of the scheduled sessions (IVR) or maintained a mean adherence of ≥80% across the structured components (online). Participants who did not reach ≥80% adherence were excluded from the primary (per-protocol) analysis.
Participants in both intervention arms were instructed to report any adverse events spontaneously throughout the intervention period. In the IVR group, adverse events could be identified and addressed on site by the supervising neuropsychologist. The online platform included an alert system that notified the clinical team of any irregularities or user-reported incidents.
Assessment procedures
Baseline information on sociodemographic characteristics, medical history, and COVID-19-related variables was collected during the initial clinical assessment. Participants were asked about their age, sex, years of formal education, nationality, ethnicity, profession, and income. The interview also covered preexisting medical conditions and health-related behaviors. Additionally, participants provided detailed information regarding their experience with COVID-19, including symptoms, treatments received, hospitalization, and time since diagnosis. Persistent post-COVID symptoms, including cognitive complaints, were also documented.
Outcomes
Here we present the prespecified primary and part of the secondary outcomes of the RehabCOVID project, which included a comprehensive evaluation of cognitive functioning as the primary outcome and selected psychological, lifestyle-related, and QoL and functioning measures as secondary outcomes. Assessments were conducted at baseline and immediately after the intervention, within 15 days before and after the training period. Participants were assessed at both time points by certified neuropsychologists, following standardized procedures
Cognitive performance measures
A comprehensive battery of standardized cognitive tests was administered to assess key aspects of cognitive functioning relevant to post-COVID recovery. General cognitive status was screened using the Montreal Cognitive Assessment (MoCA). 51 Verbal memory was evaluated with the Rey Auditory Verbal Learning Test (RAVLT), 52 which assesses learning, immediate and delayed recall, and recognition of verbal material across multiple trials. The Digit Span subtest from the Wechsler Adult Intelligence Scale (WAIS)-III 53 was used to measure verbal attention (forward span) and working memory (backward span), while the Digit Symbol Coding task 53 provided an index of processing speed and visuomotor coordination through rapid symbol-digit matching. The Trail Making Test (TMT A and B) 54 was administered to examine visual scanning, sequencing, motor speed, and cognitive flexibility. We derived the B–A difference score (TMT B–A), used as a proxy for cognitive flexibility while limiting speed-related variance. The Stroop Color and Word Test (SCWT) consists of three conditions: (a) reading color words printed in black ink (Word), (b) naming the color of colored squares (Color), and (c) naming the ink color of words that denote an incongruent color (Color–Word). In this study, the raw score of the Color–Word condition (i.e., the number of correct responses within a time limit) was used as a measure of cognitive inhibitory control. 55 Verbal fluency was assessed using phonemic fluency (Controlled Oral Word Association Test) 56 and semantic fluency (animal category), 57 reflecting lexical retrieval and executive language regulation. Sustained attention and inhibitory control were evaluated with the Conners’ Continuous Performance Test II (CPT-II), 58 a computerized go/no-go paradigm. The following indices were analyzed: detectability (d′), omission and commission errors, perseverative responses, and reaction time.
Psychological, functional, and lifestyle outcomes
Emotional symptoms were assessed using the Patient Health Questionnaire-9 (PHQ-9) and the Generalized Anxiety Disorder 7-item scale (GAD-7). The PHQ-9 59 evaluates the presence and severity of depressive symptoms over the previous 2 weeks, while the GAD-7 60 measures the severity of anxiety symptoms during the same period. For both instruments, higher scores indicate more severe emotional symptomatology. Fatigue was measured with the Chalder Fatigue Scale, 61 which captures both physical and mental dimensions of fatigue commonly observed in post-viral conditions.
Psychological flexibility was assessed using the Acceptance and Action Questionnaire-II (AAQ-II), a seven-item instrument that evaluates the tendency to avoid unwanted internal experiences and difficulties in acting in accordance with personal values. Higher scores reflect greater psychological inflexibility. 62 Dispositional mindfulness was measured with the Mindful Attention Awareness Scale (MAAS), a 15-item instrument that assesses the individual's ability to maintain present-moment awareness in a receptive and nonjudgmental manner. Higher scores indicate greater attentional presence and awareness in daily life. 63
Physical activity was evaluated using the International Physical Activity Questionnaire – Short Form (IPAQ-SF), which collects information on the frequency and duration of walking, moderate, and vigorous physical activity, as well as sedentary behavior over the previous 7 days. In the present study, we analyzed four separate indicators: weekly metabolic equivalent of task (MET)-minutes for walking, moderate activity, vigorous activity, and total physical activity, following the IPAQ scoring protocol. 64
Health-related quality of life (HRQoL) was assessed using the Spanish-validated version of the EQ-5D-3L, developed by the EuroQol Group. This instrument includes a descriptive system with five dimensions—mobility, self-care, usual activities, pain/discomfort, and anxiety/depression—each rated on three levels of severity. Responses were converted into a single index value using the Spanish value set, where higher scores indicate better perceived QoL. 65
Functioning was assessed using the 36-item Spanish version of the World Health Organization Disability Assessment Schedule 2.0 (WHODAS 2.0), which evaluates disability across six domains: cognition, mobility, self-care, getting along, life activities, and participation. Both domain-specific and total scores range from 0 to 100, with higher values indicating greater functional impairment. 66
Statistical analysis
Descriptive statistics were computed for all sociodemographic and clinical variables. Cognitive and behavioral outcomes were described and analyzed using standardized scores (z-scores). For each variable, baseline scores were standardized, and postintervention scores were expressed as standardized residuals from a linear regression predicting postscores from baseline values; for the time variables (TMT-A, TMT-B, and TMT B–A), scores were additionally sign-inverted so that higher values indicate better performance. Means and standard deviations were reported for continuous variables, and frequencies and percentages for categorical data. Between-group comparisons of clinical and sociodemographic variables at baseline were conducted to assess the comparability of study arms. For continuous variables, ANOVA or Kruskal–Wallis tests were used depending on the distribution of the data, and for categorical variables, chi-square or Fisher's exact tests were applied. In addition, comparisons were made between participants who completed the study and those who dropped out to examine potential attrition bias, using the same statistical procedures.
Group-by-time effects were analyzed using linear mixed-effects models (LMM), with fixed effects for group (control, online, IVR), time (pre, post), and their interaction. Schooling (years of education) was included as a covariate in the cognitive outcome models due to baseline group differences, while no covariates were included in the behavioral outcome models. Analyses used a per-protocol set (see Intervention adherence and safety); denominators vary by outcome/time and are reported with each table/figure; no intention-to-treat (ITT) analysis was performed. When the omnibus group-by-time F-test was significant, post hoc between-group comparisons at each time point were performed and multiplicity-adjusted within each outcome (Tukey for all pairwise contrasts and Dunnett-type for intervention-versus-control). No multiplicity adjustment was applied across outcomes; p-values across outcomes are reported unadjusted. No subgroup analyses were prespecified or conducted. No imputation was performed; analyses used all available observations under a missing-at-random assumption. Adjusted means, standard deviations, and confidence intervals were reported for each group and time point, along with marginal R2 values to assess model fit. For each significant interaction, the estimated β coefficient, 95% confidence interval, and p-value were reported to quantify the magnitude and direction of the effect. The unit of analysis was the individual participant, identical to the unit of assignment. All analyses were conducted in R version 4.4.0 (packages: lme4, lmerTest, emmeans; plus performance, broom.mixed, flextable, officer where applicable).
Results
Participants
Figure 1 presents the participant flow, including recruitment, group allocation, attrition, and the final sample. Of the 273 individuals initially screened, 172 were enrolled and assigned to one of three groups: 41 to control, 82 to online, and 49 to IVR. Attrition during the intervention ranged from 9.8% to 26.8%, and no adverse events or unintended effects were reported in any study arm. Five additional participants (one online, four IVR) did not meet adherence criteria and were excluded. The final sample included in the analyses comprised 129 participants, corresponding to a 75% retention rate. Baseline demographic and clinical characteristics are presented in Table 1.

CONSORT flow diagram of participant recruitment, allocation, follow-up, and analysis.
Demographic and clinical characteristics of participants.

ICU: intensive care unit; IVR: immersive virtual reality; PCC: post-COVID-19 condition.
Change in employment status resulting from PCC-related limitations, including medical leave, reduced working hours, or schedule modifications.
The three groups were comparable in age, sex, and time since COVID-19 diagnosis. Schooling levels were higher in the online group, and diabetes was more prevalent in the control group. Hospitalization during acute COVID-19 was slightly more common in the IVR group. Changes in employment status were more frequent in the intervention groups, potentially increasing their availability to engage in the program. No relevant between-group differences were observed in other comorbidities or income levels.
Cognitive outcomes
Table 2 presents the LMM results (cognitive models adjusted for years of education), and Figure 2 illustrates the model-adjusted z-scores at baseline and postintervention for each cognitive outcome exhibiting a significant group-by-time interaction. RAVLT immediate recall showed a significant interaction, with greater improvement in the IVR group. A similar pattern was observed for recognition, where both the IVR and online groups demonstrated larger gains than the control. In the TMT B–A, the omnibus interaction reached significance, although no post hoc contrast was significant. As illustrated in Figure 2(D), adjusted postintervention scores increased in the online group and decreased in the IVR and control groups. For CPT commission errors, the interaction reached significance, with the IVR group showing a greater reduction in errors relative to the control. Finally, in the MoCA, which assesses global cognitive functioning, the time-by-group interaction approached significance. A significant group effect was observed, with post hoc contrasts revealing higher postintervention scores in the IVR group compared to the control group.

Cognitive outcomes with significant group-by-time interaction. Each panel (A–E) represents the estimated marginal means obtained from linear mixed models, adjusted for years of education, and plotted separately for the three study groups (control, online, and IVR): (A) Rey's Auditory Verbal Learning Test (RAVLT) immediate recall; (B) RAVLT recognition; (C) Continuous Performance Test (CPT) commission errors; (D) Trail Making Test (TMT) B minus A; (E) Montreal Cognitive Assessment (MoCA). Note: For panel D (TMT B–A), scores were standardized and sign-inverted; higher values indicate better performance.
Linear mixed model results for cognitive outcomes.
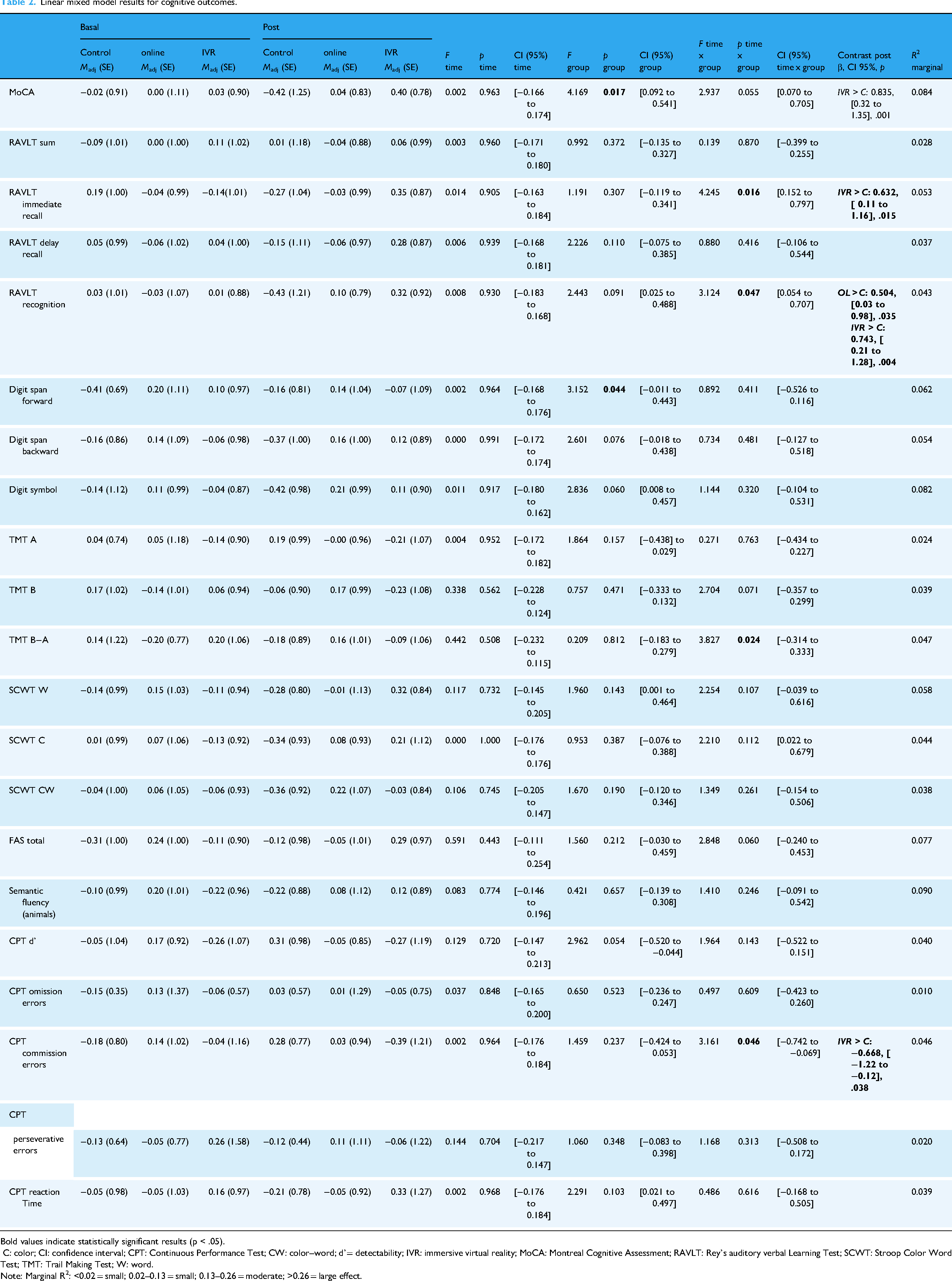
Bold values indicate statistically significant results (p < .05).
C: color; CI: confidence interval; CPT: Continuous Performance Test; CW: color–word; d’= detectability; IVR: immersive virtual reality; MoCA: Montreal Cognitive Assessment; RAVLT: Rey's auditory verbal Learning Test; SCWT: Stroop Color Word Test; TMT: Trail Making Test; W: word.
Note: Marginal R2: <0.02 = small; 0.02–0.13 = small; 0.13–0.26 = moderate; >0.26 = large effect.
Psychological, lifestyle, QoL, and functional outcomes
Table 3 presents the results of the LMM for the psychological, lifestyle, QoL, and functional variables. Likewise, Figure 3 displays the adjusted z-scores at baseline and postintervention for each outcome within these domains showing a significant group-by-time interaction. The MAAS showed a significant group-by-time interaction, with online participants outperforming the control group in post hoc analysis. For psychological flexibility (AAQ), the group-by-time interaction was significant, although post hoc contrasts did not reach significance. Adjusted postintervention scores increased in the IVR group, remained relatively stable in the online group, and decreased in the control group. For moderate physical activity (IPAQ Moderate), the group-by-time interaction was significant, although no post hoc contrast reached significance. However, adjusted scores increased in the IVR group, remained relatively stable in the online group, and decreased from pre- to postintervention in the control group. The EQ-5D health index revealed in turn a significant group-by-time interaction, with lower baseline scores in the online (β = −0.67, 95% confidence interval [CI] [−1.13, −0.21], p = .002) and IVR (β = −0.73, 95% CI [−1.28, −0.18], p = .007) groups compared to controls. Post hoc contrasts indicated a greater improvement in the online group compared to the control group. Finally, functional status as measured by the WHODAS also showed a significant group-by-time interaction. At baseline, the online group presented significantly higher disability than controls (β = 0.60, 95% CI [0.09, 1.12], p = .018), although post hoc comparisons at follow-up did not identify significant differences between individual groups. However, while the online group showed a marked reduction in scores from baseline to postintervention, the IVR group remained relatively stable, whereas the control group exhibited a substantial increase in postintervention scores. Figure 3 displays the adjusted z-scores at baseline and postintervention for each psychological, lifestyle, QoL, and functional outcome showing a significant group × time interaction.

Psychological, lifestyle, quality of life, and functional outcomes with significant group-by-time interaction. Each panel (A–F) presents the estimated marginal means obtained from linear mixed models and plotted separately for the three study groups (control, online, and IVR): (A) Mindful Attention Awareness Scale (MAAS); (B) Acceptance and Action Questionnaire-II (AAQ-II); (C) International Physical Activity Questionnaire – Moderate activity (IPAQ moderate); (D) IPAQ total; (E) EuroQol 5-Dimension Health Index (EQ-5D Index); (F) World Health Organization Disability Assessment Schedule 2.0 (WHODAS).
Linear mixed model results for psychological, lifestyle, functional and QoL outcomes.
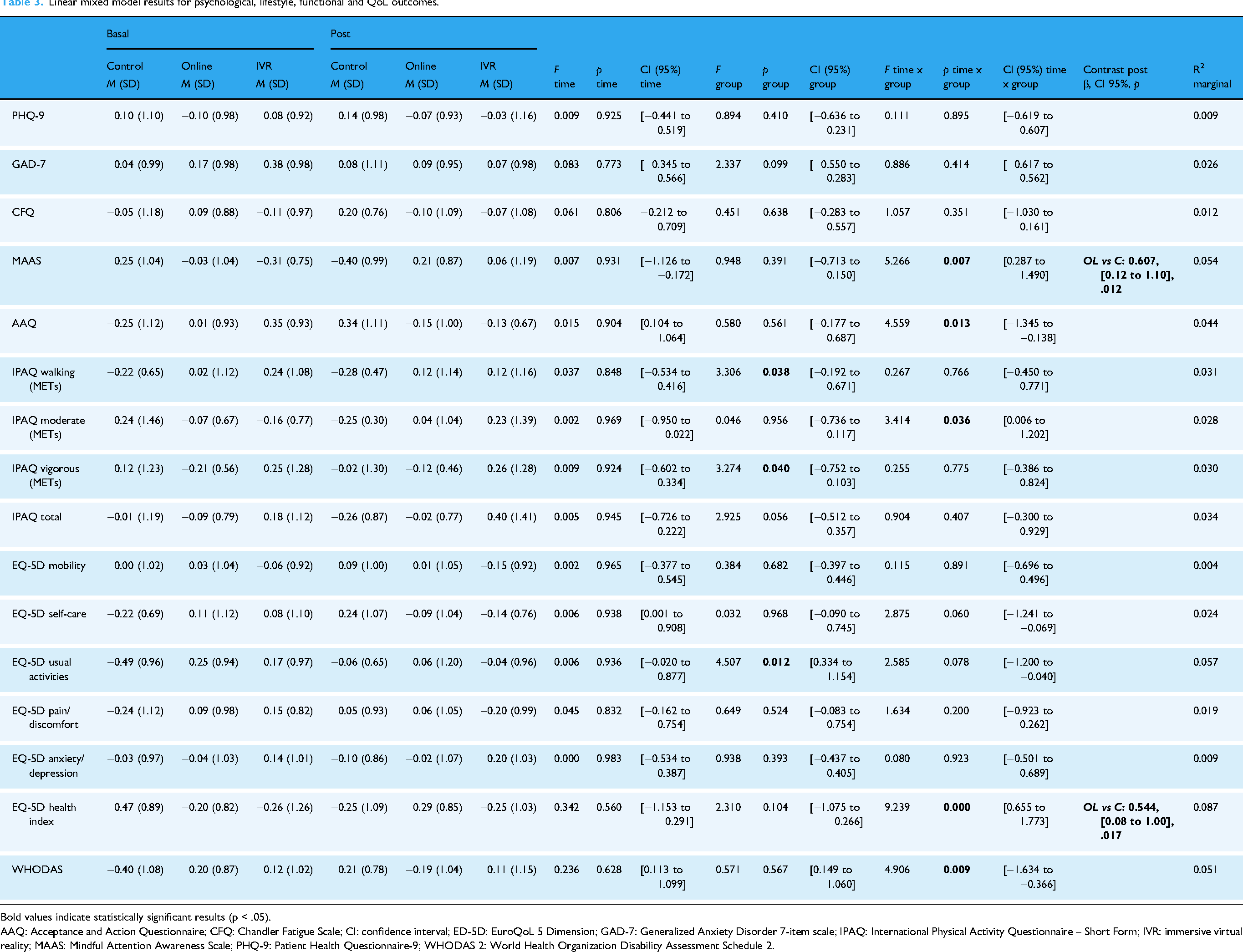
Bold values indicate statistically significant results (p < .05).
AAQ: Acceptance and Action Questionnaire; CFQ: Chandler Fatigue Scale; CI: confidence interval; ED-5D: EuroQoL 5 Dimension; GAD-7: Generalized Anxiety Disorder 7-item scale; IPAQ: International Physical Activity Questionnaire – Short Form; IVR: immersive virtual reality; MAAS: Mindful Attention Awareness Scale; PHQ-9: Patient Health Questionnaire-9; WHODAS 2: World Health Organization Disability Assessment Schedule 2.
Comparison between participants who completed the intervention and dropouts
To explore potential selection bias, we compared participants who completed the intervention with those who dropped out prior to the postintervention assessment (including participants who did not complete the treatment or did not meet adherence criteria). As shown in Supplementary Table S2, no statistically significant differences were found between groups regarding sociodemographic or clinical variables such as sex, ethnicity, years of education, COVID-19 severity, or time from diagnosis to assessment. However, a significant difference was observed in group allocation, with a higher proportion of dropouts belonging to the online group compared to the IVR and control groups. The most marked difference between completers and dropouts was observed in labor status. All participants who dropped out reported no change in their employment situation, whereas most completers in the intervention groups had experienced a change, such as medical leave or reduced working hours.
Discussion
Previous studies have shown that both web-based and IVR programs can be as effective as traditional face-to-face rehabilitation.67–69 We compared two digital formats that had been previously tested in other populations, applying them to individuals with PCC. Including a passive control group helped contextualize the results by capturing changes over time in the absence of any intervention. Our findings suggest that both multimodal interventions—delivered either online or via IVR—were associated with multidomain changes, including cognitive performance, psychological well-being, and perceived HRQoL in individuals with PCC. Compared to the control group, the IVR intervention led to improvements in global cognition, immediate recall and recognition memory, and response inhibition. The online format was also associated with better outcomes than the control in verbal recognition, mindfulness, and perceived HRQoL.
Additionally, although several outcomes showed significant group-by-time interactions without significant post hoc contrasts, the observed trajectories suggested group-specific patterns of change: more favorable changes over time in cognitive flexibility and functioning in the online group and more favorable changes in psychological flexibility and increased moderate physical activity (IPAQ moderate) in the IVR group.
Our results are in line with two previous studies that tested different types of digital interventions for individuals with PCC, each compared to no intervention. Victoria et al. evaluated a remotely delivered cognitive training program and reported improvements in processing speed, task-switching, cognitive fatigue, anxiety, and QoL. Their intervention was unimodal and focused exclusively on cognition. 13 In parallel, a pilot study conducted by our group 39 tested a similar IVR-based intervention and found significant improvements in global cognition, simple attention, processing speed, memory, and depressive symptoms, with a marginal effect for executive function. This intervention was multimodal—targeting cognitive, physical, and emotional domains—and thus aligned with the approach adopted in the current trial.
The three-arm design used in this study provided a unique opportunity to examine not only the effects of each intervention relative to no treatment, but also the differential impact of two technology-delivered modalities offering identical content. While both the IVR and online groups showed improvement in verbal recognition compared to the control group, broader gains—including global cognition, immediate recall, and response inhibition—were observed only in the IVR group. These findings suggest that delivery format may influence the effectiveness of multimodal interventions even when cognitive, physical, and emotional components are content-matched, without implying inherent superiority of one format over the other across contexts or outcomes. In this study, delivery format is understood not only as the technological modality, but also as the broader implementation context, including supervision, structure, and social interaction. Accordingly, the IVR intervention prioritized functional relevance through supervised and ecologically valid delivery, whereas implementation characteristics—such as in-person attendance and the need for direct supervision—may limit scalability relative to the self-guided online format.
The immersive and supervised nature of the IVR sessions, combined with the structured and socially interactive setting, may have facilitated deeper engagement and more consistent activation of cognitive processes. 15 The cognitive demands of the IVR tasks—including sustained attention, rapid decision-making, and real-time shifting between stimuli—may have further reinforced executive functioning. By reproducing dynamic and complex environments that resemble everyday cognitive demands, IVR-based training also increases ecological validity and may facilitate the transfer of gains to real-world contexts.43,70 In contrast, the self-paced format of the online intervention, while accessible and convenient, may offer more limited support for participants with reduced initiative or difficulty sustaining engagement—features commonly reported in individuals with PCC. 71 However, the online format may nonetheless encourage greater autonomous control, potentially supporting executive processes such as cognitive flexibility, which require self-initiated strategy shifts in less structured environments. 72 The online group, which started from a lower baseline, was the only one to show improvement in cognitive flexibility, as both the IVR and control groups obtained lower scores at postintervention compared to baseline.
The IVR group reported showed more favorable trajectories and higher postintervention levels of moderate physical activity as measured by the IPAQ. This finding is consistent with previous evidence indicating that VR-based interventions may be associated with modest increases in physical activity–related outcomes across different populations. 73 However, given the self-reported nature of the IPAQ and the absence of objective activity monitoring, these results should be interpreted cautiously. Differences between intervention formats, including the in-person versus remote delivery context, may have influenced physical activity behavior beyond the structured exercise components of the programs. In contrast, the online intervention was completed remotely and independently, which may partly account for the relatively stable patterns observed in IPAQ scores, although no direct conclusions can be drawn regarding underlying mechanisms.
Scores on the MAAS scale increased in the online group compared to the control group, suggesting that participants who followed the online intervention experienced an overall increase in mindfulness. This change may reflect not just temporary effects of the intervention, but a more sustained incorporation of mindful awareness into daily life. Practicing mindfulness exercises in a familiar and private environment may have supported emotional comfort and helped participants stay engaged, making it easier to include these practices in their daily lives. Given that the MAAS reflects habitual mindful awareness, the more autonomous and everyday nature of the online format may have facilitated this generalization, whereas mindfulness practice in the IVR setting may have remained more closely linked to the intervention context.
Despite the increase in mindfulness, emotional symptom scales did not show significant changes in the online group. This finding is somewhat unexpected given the well-documented link between MBIs and reductions in depression and anxiety. Previous studies, including in individuals with acute COVID-19 infection,29,32,33 have shown that even brief MBIs can alleviate emotional distress. In the present study, several factors may explain the absence of detectable changes. Participants’ baseline scores on these scales were already close to normative values, limiting the potential for measurable improvement. Importantly, normative-range scores on emotional symptom scales do not preclude the presence of clinically relevant distress in chronic conditions such as PCC, nor do they fully capture long-standing emotional burden. Furthermore, the chronicity of their condition—over 2 years since infection on average—likely reflected persistent fatigue and psychological exhaustion, which may be less responsive to short interventions and may not be adequately detected by standard depression or anxiety symptom scales. In a previous online study, a reduction in depressive symptoms was observed following the same MBI, 33 but pre–post analyses were based on raw scores and participants presented with clinically elevated baseline depression that normalized after the intervention. In that context, change may have been easier to detect due to higher initial symptom severity. In contrast, in the present study, the increase in mindfulness was accompanied by improvements in perceived health status, suggesting changes in emotional adjustment not captured by conventional symptom scales. HRQoL measures may be more sensitive to gradual shifts in acceptance, emotional regulation, and functional coping, which may precede measurable reductions in emotional symptomatology.
In line with this, differences between formats in HRQoL and functioning suggest that broader outcomes may be particularly sensitive to the context in which the intervention is delivered. While the online program was associated with improvements in perceived health status and functional disability compared to the control group, the IVR format showed relative stability over time. These differences are unlikely to reflect the immersive technology per se, but rather how the intervention was embedded in participants’ everyday lives. Delivery at home may have supported greater autonomy and continuity in managing persistent symptoms, whereas the hospital-based IVR program may have been experienced as a more bounded, medicalized intervention, with perceived benefits remaining more closely tied to the treatment setting. Importantly, this interpretation does not question the potential of IVR but highlights the place of delivery as a modifiable factor that may shape the impact of digital interventions on HRQoL and functioning.
To our knowledge, this is the first study to directly compare two active digital delivery formats—online and IVR—for neurorehabilitation in individuals with PCC. This type of comparison is timely and necessary, as the WHO has called for evidence-based strategies to guide the digital transformation of healthcare systems globally, with an emphasis on accessibility, equity, and sustainability. 7 By evaluating well-established digital rehabilitation modalities within a single design, our study contributes to this agenda and informs future decisions on how best to implement digital rehabilitation for populations affected by PCC. Importantly, the results suggest that the benefits of these interventions may lie not only in promoting improvement but also in preventing the functional decline observed in the control group. This pattern reinforces the need for proactive rehabilitation programs in people with PCC, who might otherwise experience progressive deterioration in key areas such as cognition, psychological well-being, and daily functioning.
Although the study included two distinct delivery formats, these were not intended as competing models. Rather, the rationale for including both formats lies in their complementary value within healthcare systems. The IVR format offers greater ecological validity and may enhance engagement through immersive, realistic simulations. In contrast, the web-based format is more accessible, scalable, and cost-effective—particularly for populations facing geographical, logistical, or resource-related barriers.
Our results showed that both intervention formats were associated with beneficial outcomes across cognitive and neurobehavioral domains, with partially distinct profiles but no clear overall superiority. In practical terms, receiving any structured intervention appeared preferable to no treatment, given the signs of deterioration observed in the control group. From a practical perspective, the choice between formats may depend more on system capacity, logistical feasibility, and contextual priorities than on efficacy differences. This is reflected in the observed 1:2 ratio between IVR and web-based delivery, which emerged from participant choice and real-world feasibility constraints rather than from a prespecified allocation. This pattern suggests that scalable, low-resource formats can reach a larger proportion of individuals, while IVR may be more appropriately used as a targeted option for selected profiles. Despite overall attrition, nonadherence was uncommon, indicating adequate implementation fidelity, and participant feedback also indicated good acceptability (satisfaction data collected but not reported here). Overall, the online format may facilitate broader access while maintaining comparable outcomes on the measures assessed.
Future research should examine how delivery context influences the effects of immersive interventions in PCC. In particular, testing IVR approaches in home-based settings may help clarify whether combining immersive features with greater everyday integration enhances generalization to QoL and functional outcomes. Studies explicitly varying delivery context, supervision, and autonomy, as well as incorporating longer follow-up periods, will be important to determine how and when changes in these broader outcomes consolidate over time.
This study has several methodological limitations. First, participants self-selected into intervention formats rather than being randomly assigned, which improved recruitment but introduced selection bias and reduced internal validity. Second, the overall retention rate (∼75%) was suboptimal, though allowing self-selection likely helped minimize attrition. Third, the control group was mainly recruited at an external hospital due to feasibility constraints, introducing possible center-related confounding despite harmonized procedures. Fourth, specific measure of immersion or presence was not collected in this study, limiting the ability to directly assess participants’ subjective sense of immersion during the IVR sessions. Finally, group sizes were unequal, which—although manageable with LMM—may have affected the precision of some estimates, particularly for subtle between-group effects. In addition, the inclusion of two active intervention arms increased clinical relevance but also distributed statistical power across multiple contrasts, which may have limited the detection of small or uniform effects across all outcome domains. No adjustment for multiplicity was applied across outcomes; cross-outcome findings should be considered exploratory. Nonetheless, the study presents some strengths. It focuses on a clinically important yet understudied population—individuals with post-COVID cognitive symptoms—and adopts a pragmatic design that emphasizes ecological validity and reflects real-world clinical conditions and patient preferences, enhancing the generalizability of the findings. Furthermore, the use of a broad set of standardized, well-validated neurocognitive, behavioral, and functional measures allows for a comprehensive assessment of intervention effects.
Conclusions
Both digitally delivered interventions—IVR and an online self-guided format—were associated with multidomain changes in cognitive and functional outcomes in individuals with PCC. The IVR format was associated with improvements in global cognition, immediate recall, recognition memory, and response inhibition. For psychological flexibility and moderate physical activity, group-by-time interactions suggested differential change over time compared to the control group, despite no significant differences at the postintervention time point.
The online format was associated with improvements in verbal recognition, mindfulness, and perceived HRQoL. Significant group-by-time interactions were also observed for cognitive flexibility and functioning, suggesting differential patterns of change compared to the control group, despite no significant postintervention differences.
These results are most generalizable to adults with PCC receiving similar hospital-based services and able to access on-site IVR or home internet; effects reflect short-term change (∼12 weeks). Collectively, the pattern of effects supports the potential value of multicomponent rehabilitation programs for individuals with PCC. Future research should explore how delivery format interacts with individual characteristics to optimize intervention outcomes.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076261428205 - Supplemental material for Addressing persistent symptoms in post-COVID condition: Through a multimodal intervention delivered in two technology-based formats
Supplemental material, sj-docx-1-dhj-10.1177_20552076261428205 for Addressing persistent symptoms in post-COVID condition: Through a multimodal intervention delivered in two technology-based formats by Mar Ariza, Neus Cano, Anna Carnes-Vendrell, Olga Gelonch, Yemila Plana, Bruno Porras-Garcia, Elisabeth López-Soley, Silvia Moron, Gerard Piñol-Ripoll, Maite Garolera and in DIGITAL HEALTH
Supplemental Material
sj-pdf-2-dhj-10.1177_20552076261428205 - Supplemental material for Addressing persistent symptoms in post-COVID condition: Through a multimodal intervention delivered in two technology-based formats
Supplemental material, sj-pdf-2-dhj-10.1177_20552076261428205 for Addressing persistent symptoms in post-COVID condition: Through a multimodal intervention delivered in two technology-based formats by Mar Ariza, Neus Cano, Anna Carnes-Vendrell, Olga Gelonch, Yemila Plana, Bruno Porras-Garcia, Elisabeth López-Soley, Silvia Moron, Gerard Piñol-Ripoll, Maite Garolera and in DIGITAL HEALTH
Footnotes
Glossary
Acknowledgments
Mikel Aldaba and Meritxell Vilaseca: Centre for Sensors, Instruments and Systems Development, Universitat Politècnica de Catalunya (Barcelona), Spain.
Gemma Carreras-Badosa and Elisabet Kádár: Departament de Biologia, Facultat de Ciències, Universitat de Girona, Girona, Spain.
Javier Béjar and Cristian Barrué: Departament de Ciències de la Computació, Universitat Politècnica de Catalunya-BarcelonaTech, Barcelona, Spain.
Barbara Segura: Unitat de Psicologia Mèdica, Departament de Medicina, Universitat de Barcelona (UB), Barcelona, Spain. Institut d’Investigacions Biomèdiques August Pi i Sunyer (IDIBAPS), Barcelona, Spain. Institut de Neurociències, Universitat de Barcelona (UB), Barcelona, Spain.
Jose Antonio Bernia: Servei d’Anestesiologia, Reanimació i Teràpia del Dolor. Consorci Sanitari de Terrasa-Hospital Universitari, Terrassa (Barcelona), Spain.
Antonio Arevalo: Centre Atenció Primària Dr Joan Planas Castellbisbal. Consorci Sanitari de Terrassa)-Hospital Universitari. Castellbisbal (Barcelona), Spain.
Author's note
Yemila Plana, Departament de Psicologia, Facultat de Medicina i Ciències de la Salut, Universitat Internacional de Catalunya (UIC), Barcelona, Spain.
ORCID iDs
Ethical considerations
The study was approved by the Drug Research Ethics Committee (CEIm) of the Consorci Sanitari de Terrassa-Hospital Universitari (reference code: 01-21-107-111; approval date: 19 December 2022) and the Clinical Research Ethics Committee (CEIC) of the Hospital Santa Maria of Lleida (reference code: CEIC-2897).
Consent to participate
All participants provided written informed consent prior to enrollment in the study.
Author contributions
Mar Ariza: conceptualization, data curation, formal analysis, funding acquisition, investigation, methodology, resources, software, supervision, validation, visualization, writing–original draft, writing–review and editing.
Neus Cano: investigation, methodology, resources, supervision, writing–review and editing.
Anna Carnes-Vendrell: data curation, investigation, resources, supervision, writing–review and editing.
Olga Gelonch: data curation, investigation, resources, writing–review and editing.
Yemila Plana: investigation, writing–review and editing.
Bruno Porras-Garcia: investigation, writing–review and editing.
Elisabeth López-Soley: investigation, writing–review and editing.
Silvia Moron: investigation, writing–review and editing.
Gerard Piñol-Ripoll: data curation, investigation, resources, supervision, and writing–review and editing.
REHAB project collaborative group: funding acquisition, investigation, project administration, and writing–review and editing.
Maite Garolera: conceptualization, data curation, funding acquisition, investigation, project administration, methodology, resources, supervision, writing–review and editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministerio de Ciencia e Innovación/Agencia Española de Investigación, cofunded by the European Union NextGenerationEU/PRTR, through grants TED2021-130409B-C51 and JDC2022-048939-I.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of data and materials
The datasets used and/or analyzed during this study are available from the corresponding author upon reasonable request.
AI disclosure statement
The authors used AI-assisted tools to support English language editing and to assist with the development of R code for data processing and statistical analyses. No AI tools were used to generate or manipulate study data. All AI-assisted outputs were reviewed, validated, and finalized by the authors, who retain full responsibility for the integrity and accuracy of the work.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
