Abstract
Objective
Telerehabilitation is a rapidly growing area within telehealth, providing clinical rehabilitation services through telecommunication technologies to enhance accessibility, improve care quality, and support patient functionality. Pediatric motor rehabilitation has gained increasing recognition for its vital role in promoting neuromotor development, coordination, and independence in children with congenital or acquired motor disabilities. This systematic review analyzed home-based telerehabilitation interventions for pediatric neurological motor disorders, from the neonatal period onward, aiming to outline current trends and future directions.
Methods
A comprehensive search was conducted across Medline, EBSCO, and Web of Science, covering records up to February 2025. Peer-reviewed articles were included without restrictions on study design. The focus was on pediatric and adolescent populations with neurodevelopmental or neuromuscular disorders undergoing telerehabilitation or remote rehabilitative interventions.
Results
The findings support the potential efficacy of tele-neurorehabilitation in improving motor outcomes, with promising results for children with cerebral palsy and hemiplegia. Interventions were shown to enhance motor function (SMD = 0.48; 95% CI: 0.23 to 0.72), range of motion (SMD = 0.31; 95% CI: 0.11 to 0.51), and walking ability (SMD = 0.25; 95% CI: 0.03 to 0.46), suggesting that remote rehabilitation can be an effective complement or alternative to traditional in-person therapy in pediatric care. Moreover, beyond functional improvements, several studies reported positive effects on children's quality of life, highlighting the broader psychosocial benefits of remote interventions.
Conclusions
These results underscore the holistic value of telerehabilitation, reinforcing its role as a comprehensive and accessible strategy in pediatric rehabilitation services. Multidimensional assessments are essential to capture the broader benefits of telerehabilitation and to guide the development of more holistic, child- and family-centered care models.
Keywords
Introduction
The advancement and widespread availability of Information and Communication Technologies (ICTs) have created new opportunities for delivering healthcare services remotely, both during hospitalization and after discharge. 1
According to the World Health Organization (WHO), telehealth refers to the provision of healthcare services when patients and providers are separated by distance, using ICTs to support communication for diagnosis, treatment, research, evaluation, and continuing professional development. 2 Originally adopted to bridge geographic barriers and improve access in underserved areas telehealth has since evolved into a critical component of modern healthcare systems.3,4 Its relevance surged dramatically during the COVID-19 pandemic, when social distancing and overwhelmed healthcare facilities forced a shift toward virtual care solutions.
Telerehabilitation is an essential and expanding area within the broader field of telehealth.5–8 It represents a clinical rehabilitation service that leverages telehealth technologies to enhance accessibility, improve care quality, and maximize patient functionality. 9
In recent years, telerehabilitation has attracted growing attention as an innovative strategy for managing musculoskeletal conditions and motor impairments and has been proposed as an effective care model also for pediatric populations.10–13 The WHO has acknowledged the effectiveness of telerehabilitation as a viable service delivery model for rehabilitation professionals.2,14 Similarly, the American Occupational Therapy Association and the American Physical Therapy Association support the use of telerehabilitation in clinical settings, recognizing its potential to improve patient outcomes and its flexibility in addressing the specific needs of patients and their families.15,16
Despite its recognized potential, the use of remote rehabilitation services in pediatric care remained limited before the COVID-19 pandemic. This was primarily due to cultural factors, limited access to technology, and challenges related to regulatory frameworks and reimbursement policies.17,18
More recently, childhood motor rehabilitation has gained increasing recognition as a critical component in supporting neuromotor development, enhancing coordination, and fostering independence in children with congenital or acquired motor disabilities. Within this framework, telerehabilitation has emerged as a flexible and effective modality for delivering individualized therapeutic interventions without requiring physical presence. Its advantages include the enhancement of both gross and fine motor skills, 19 improved self-perception of physical competence, 20 reduced access barriers, remote treatment personalization and monitoring, and greater opportunities for active family involvement in the rehabilitation process. Additionally, telerehabilitation encourages collaboration between therapists and caregivers, who become active participants in the therapeutic process and gain practical tools to support the child's motor development in daily life. 20
In this systematic review, we analyzed the use of home-based telerehabilitation for pediatric neurological motor disorders, starting from the neonatal age, with the aim of presenting current trends and future perspectives in this field of telemedicine.
Material and methods
Study design
The review was conducted in accordance with the Cochrane Handbook for Systematic Reviews guidelines, as well as other relevant methodological standards for systematic reviews and meta-analyses. 21 The reporting of findings followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, reported in Table S1. 22 The PRISMA flow diagram summarizing the selection process is presented in Figure 1. The Population-Concept-Context (PCC) framework 23 was used to create search strings, as detailed in Table 1.

Preferred reporting items for systematic reviews and meta-analyses (PRISMA) flow diagram.
PCC elements.

Protocol registration
The protocol for systematic review and meta-analysis was registered in the International Prospective Register of Systematic Reviews (PROSPERO) under registration number CRD420251036180. 24
Search strategy
A comprehensive literature search was performed across three major biomedical databases: Medline, Elton B. Stephens Company (EBSCOhost), and Web of Science, encompassing records from database inception to February 2025. Search strategy combined controlled vocabulary (e.g., Medical Subject Headings [MeSH]) and relevant free-text terms, customized according to the thesaurus, and indexing structure of each database. No limitations were applied concerning study design or publication type; eligible sources included peer-reviewed articles. The search targeted the pediatric and adolescent population (including neonates, infants, children, and adolescents) diagnosed with neurodevelopmental or neuromuscular disorders and undergoing telerehabilitation or remotely delivered rehabilitative interventions.
Study selection process
Tailored terms such as “Children,” “Telerehabilitation,” and “Neurodevelopmental disorders” and their synonyms were combined to create search strings (detailed in Table S2). Boolean operators (AND, OR) were used to refine search. Two independent reviewers carried out the study selection process in accordance with the predefined inclusion and exclusion criteria. Initially, duplicate records were removed. Next, the titles and abstracts of all retrieved references were screened to exclude studies that were clearly irrelevant. Full-text articles of potentially eligible studies were then obtained and assessed in detail to determine their suitability for inclusion. Any discrepancies between reviewers were resolved through discussion, and if consensus could not be reached, a third reviewer was consulted to make the final decision. Additionally, the reference lists of all included studies were manually screened to identify any further relevant publications.
Eligibility criteria
Studies were considered eligible for inclusion if they met the following criteria: (i) population comprised pediatric patients (0–18 years), (ii) participants presented with neurological motor disorders or motor impairments, including but not limited to neurodevelopmental disorders, cerebral palsy, muscular atrophy, neuropathies, myopathies, hemiplegia, or other neuromuscular conditions; (iii) the intervention involved home-based telerehabilitation or remote rehabilitation modalities, such as digital therapy, mobile health and telemedicine; and (iv) articles were published in English. Moreover, studies were excluded if they met any of the following criteria: (i) involved adult populations aged more than 18 years; (ii) addressed non-neurological or non-motor disorders; (iii) focused exclusively on in-person rehabilitation without any component of remote or digital intervention; (iv) were structured as observational studies, qualitative studies and reviews.
Data selection and extraction
To streamline the study selection process, all references retrieved from the database searches were imported into Rayyan®, an online platform tailored for systematic review management. 25 Duplicate entries were carefully removed through a combination of automated tools and manual verification. Following this, two reviewers (PP and LG) independently conducted a two-stage screening process. The initial phase involved evaluating titles and abstracts to eliminate studies that were clearly ineligible. Articles deemed potentially relevant then proceeded to a full-text review, conducted independently and without influence from the other reviewer's decisions. Any disagreements that arose during the selection process were discussed to reach a mutually agreed-upon resolution. In cases where consensus could not be achieved, a third reviewer (VR) was brought in to provide an objective decision. This approach was designed to uphold consistency and reduce bias throughout the selection process.
Each article that advanced to full-text screening was reviewed in-depth to assess its methodological soundness and relevance to the research question. Only those studies that met the inclusion criteria and demonstrated sufficient methodological quality were included in the final synthesis. For every study included, a standardized set of information was extracted, including: author(s), year of publication, study purpose, design, sample size, participants’ age, setting and intervention and main outcomes. The extracted information is reported in Table 2.
Descriptive characteristics of the studies included in the systematic review.
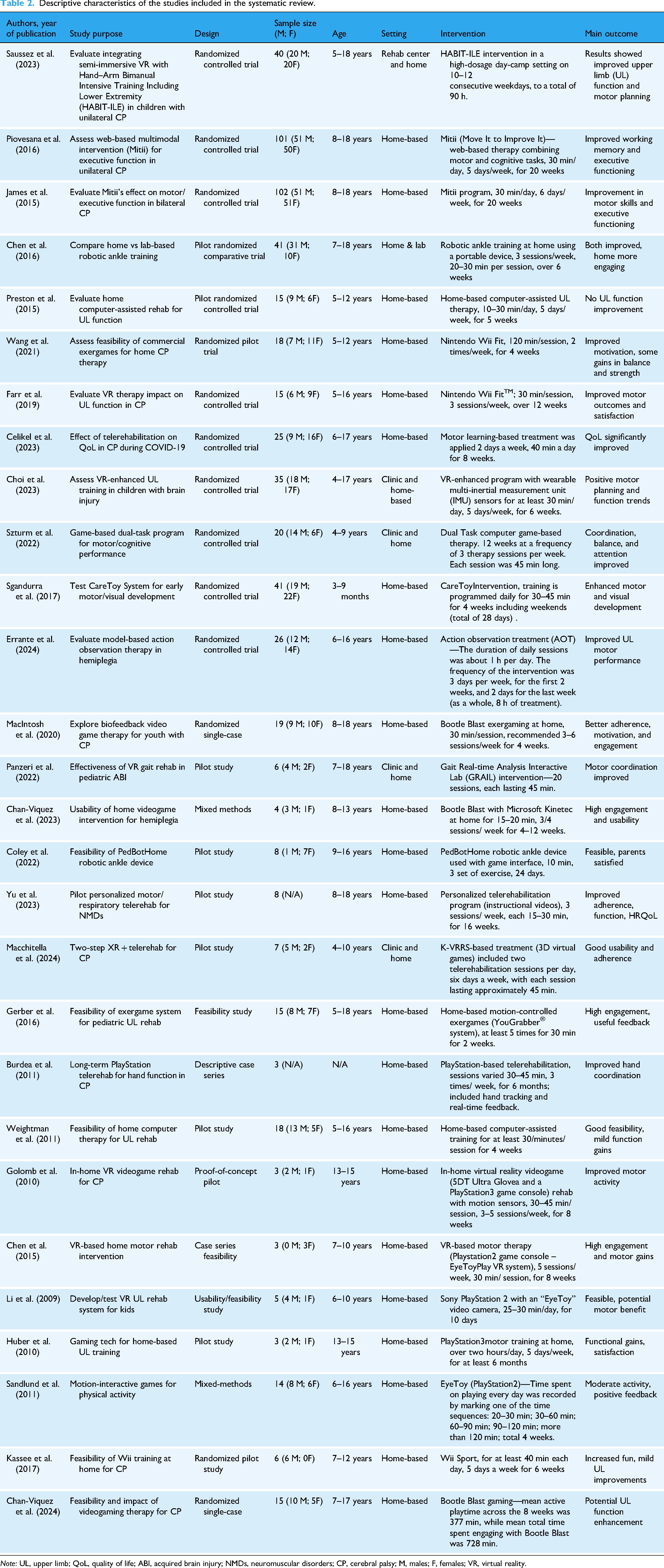
Note: UL, upper limb; QoL, quality of life; ABI, acquired brain injury; NMDs, neuromuscular disorders; CP, cerebral palsy; M, males; F, females; VR, virtual reality.
Outcome measures
In this systematic review, we examined a range of outcome measures used to assess the efficacy of tele-neurorehabilitation in the pediatric population with motor-related conditions. Across the included studies, key outcomes evaluated included gross motor function, range of movement, general strength, walking function and quality of life. These measures provided a comprehensive overview of the potential benefits of remote rehabilitation interventions for children with motor impairments. Quality of life was considered as an important outcome in the review, although it was not consistently reported across studies. For the purposes of the meta-analysis, only outcome measures that were consistently reported and suitable for quantitative synthesis were included. Specifically, data related to motor function and/or performance, range of movement, and general and or specific strength were analyzed. These outcomes enabled a pooled analysis to assess the overall efficacy of tele-neurorehabilitation in improving motor performance. Although quality of life was included in the systematic review, it could not be incorporated into the meta-analysis due to limited data availability, as only two studies reported on this outcome.
Risk of bias and quality assessments
The methodological quality of the included studies was assessed using validated tools tailored to the specific study designs. For randomized controlled trials (RCTs), the Revised Cochrane Risk-of-Bias Tool (ROB2) was applied. This tool evaluates five domains: the randomization process, deviations from intended interventions, missing outcome data, measurement of outcomes, and selection of the reported results. Two reviewers (VR and AG) independently assessed each domain, rating studies as having “low,” “some concerns,” or “high” risk of bias. Discrepancies were resolved through discussion, with the involvement of a third reviewer (PP) when necessary.
For non-randomized studies, the Risk Of Bias In Non-randomized Studies of Interventions (ROBINS-I) tool was used. This framework addresses seven domains of potential bias: confounding, selection of participants, classification of interventions, deviations from intended interventions, missing data, measurement of outcomes, and selection of reported results. As with the RCTs, two reviewers independently conducted the assessments (LG and VR), resolving any differences by consensus or consultation with a third reviewer (AG).
Importantly, studies were not excluded based solely on their quality assessment. Instead, the results of these appraisals were used to guide the interpretation, weighting, and contextualization of the evidence during synthesis.
Statistical analysis
The effectiveness of telerehabilitation interventions was assessed using a post-pre analysis approach. Standardized mean differences (SMDs) were calculated based on the change from baseline to post-intervention within each study. To ensure comparability across studies, effect sizes were standardized using the pooled baseline standard deviation. A random-effects meta-analysis model was employed, with restricted maximum likelihood (REML) estimation used to account for between-study variance. Each study's contribution to the pooled estimate was weighted using the inverse variance method. Heterogeneity across studies was assessed using the I² statistic and Cochran's Q test, with I² values interpreted according to established guidelines (e.g., 0–40%: low; 30–60%: moderate; 50–90%: substantial; 75–100%: considerable). To assess possible sources of heterogeneity, mixed-effects meta-regression models were used to explore the impact of study-level moderators, including intervention type, age group, and, where applicable, intervention duration. The proportion of heterogeneity explained by moderators was reported using R² values, and the overall contribution of moderators was assessed with the QM statistic. Residual heterogeneity was examined to determine whether the included variables accounted for variation across studies. In addition, prediction intervals were calculated using the method described by Riley, providing an estimate of the likely range of effects in future studies. All analyses were conducted using R (version 4.4) with the “metafor” package.
Results
As shown in Table 2, most of the participants included in the studies were diagnosed with cerebral palsy or hemiplegia, reflecting the primary populations targeted by pediatric motor tele-rehabilitation programs. Furthermore, most interventions were specifically focused on upper limb rehabilitation, which may limit the generalizability of the findings to other motor domains, such as lower limb function or global mobility.
The structured process for identifying and selecting studies is presented in the PRISMA flowchart (Figure 1). After removing duplicate entries, a total of 1008 titles and abstracts were screened. From these, 94 full-text articles were evaluated in more detail. Studies were excluded at this stage for several reasons: (i) the intervention was not performed using any type of technology in the hospital or clinics; (ii) the intervention was performed at home without using the support of any technology. Finally, 28 studies met all inclusion criteria and were included into this systematic review. However, only 21 studies were found eligible to be included in the meta-analysis.
Risk-of-bias assessment for every study is shown in Figure 2. Overall, the risk of bias has risen “some concerns,” with only several pilot studies considered as “high.” Most of the concerns were related to problems in the allocation concealments and in the statistical analyses performed.

Summary plot of the risk of bias for the included studies, assessed using the Risk Of Bias In Non-randomized Studies of Interventions (ROBINS-I; on the left) and the Risk of Bias tool 2 (RoB 2; on the right).
Regarding overall motor function, the meta-analysis revealed a moderate and statistically significant improvement (SMD = 0.48; 95% CI: 0.23 to 0.72), Figure 3. However, heterogeneity was substantial (I² = 74.2%, P < .001), likely due to the use of heterogeneous assessment tools across studies. Commonly used instruments included the Gross Motor Function Measure (GMFM-66 and GMFM-88), Bruininks–Oseretsky Test of Motor Proficiency, Second Edition (BOT-2), and other functional motor assessments. These tools differ in sensitivity and applicability, which may have contributed to the variability in the estimated and observed effects. In line with the substantial heterogeneity detected, the prediction for motor function had an estimate of 0.48 (95% CI: −0.25 to 1.29), indicating a moderate treatment effect in future studies with a moderate range variability. To examine sources of heterogeneity, a mixed-effects meta-regression was conducted (k = 11) with intervention type as a moderator. The model explained most of the variance (R² = 95.93%, I² = 9.07%, QM (4) = 25.65, P < .001). The estimated effect for computer-based interventions in children and adolescents was significant (SMD = 0.48; 95% CI: 0.29 to 0.67). Compared to this group, the CareToy system showed significantly larger effects (β = 0.85, P < .001), while no significant differences were found for Nintendo Wii-based, VR-based, or PlayStation 2-based interventions.

Forest plot of standardized mean differences within groups (post–pre estimates) for motor function. A negative value indicates a reduction in outcome after the intervention, whereas a positive value indicates an improvement in outcome after the intervention. Abbreviations: CI, confidence interval; SMD, standardized mean difference.
In the evaluation of muscular strength, a pooled SMD of 0.67 (95% CI: −0.05 to 1.39) was found, indicating a moderate effect size, Figure 4. However, the analysis revealed a not statistically significant effect of telerehabilitation programs. A high degree of heterogeneity was detected (I² = 81.8%, P < .001), likely reflecting both the diversity of strength assessment methods and differences in the specific muscle groups targeted across studies. In line with the substantial heterogeneity detected, the prediction for muscular strength had an estimate of 0.67 (95% CI: −0.93 to 2.27), indicating a wide range of possible treatment effects in future studies. For muscular strength, a mixed-effects meta-regression was conducted (k = 5) with intervention type as a moderator. The model was significant (QM (2) = 10.44, P = .0054), explaining a substantial proportion of between-study variance (R² = 87.33%, I² = 40.80%). The estimated effect size for the reference group (computer-based interventions) was not statistically significant (SMD = 0.33; 95% CI: −0.11 to 0.78; P = .14). Compared to this group, Nintendo Wii-based interventions showed a significantly larger effect on muscular strength (β = 1.83; 95% CI: 0.72 to 2.93; P = .001), while PlayStation 3-based interventions did not differ significantly (β = 0.30; P = .593).

Forest plot of standardized mean differences within groups (post–pre estimates) for muscular strength. A negative value indicates a reduction in outcome after the intervention, whereas a positive value indicates an improvement in outcome after the intervention. Abbreviations: CI, confidence interval; SMD, standardized mean difference.
For range of motion (ROM), the meta-analysis showed a small but statistically significant improvement (SMD = 0.31; 95% CI: 0.11 to 0.51), with moderate heterogeneity (I² = 39.8%, P = .09), suggesting a fair level of consistency in the observed effects across studies, Figure 5. In line with the moderate heterogeneity detected, the prediction for ROM had an estimate of 0.31 (95% CI; −0.12 to 0.74), showing that future studies might observe small positive changes in ROM.

Forest plot of standardized mean differences within groups (post–pre estimates) for range of motion (ROM). A negative value indicates a reduction in outcome after the intervention, whereas a positive value indicates an improvement in outcome after the intervention. Abbreviations: CI, confidence interval; SMD, standardized mean difference.
In terms of walking function, a small but statistically significant effect was observed (SMD = 0.25; 95% CI: 0.03 to 0.46), with low heterogeneity (I² = 17.6%, P = .41), indicating a high degree of consistency among studies, Figure 6. Although walking performance was assessed using various functional tests, including the 6-Minute Walk Test (6MWT), the 1-Minute Walk Test (1MWT), and other similar protocols, this variability in outcome measures did not appear to substantially influence the magnitude or direction of the pooled effect. In line with the low heterogeneity detected, the prediction for muscular strength had an estimate of 0.25 (95% CI; −0.05 to 0.54), showing that future studies might observe small positive changes in walking function.

Forest plot of standardized mean differences within groups (post–pre estimates) for walking function. A negative value indicates a reduction in outcome after the intervention, whereas a positive value indicates an improvement in outcome after the intervention. Abbreviations: CI, confidence interval; SMD, standardized mean difference.
Discussion
The findings of this systematic review and meta-analysis support the potential efficacy of tele-neurorehabilitation in improving motor outcomes in the pediatric population, with particularly promising, and more often assessed, results observed in children with cerebral palsy and hemiplegia. We found that the interventions analyzed were significantly effective in enhancing motor function, range of motion, and walking ability, suggesting that remote rehabilitation strategies can be a reliable complement and/or an alternative to traditional face-to-face occupational therapy in a pediatric setting. Moreover, beyond functional outcomes, several studies also reported improvements in children's quality of life following tele-neurorehabilitation. Although this outcome could not be included in the meta-analysis due to the limited number of studies reporting comparable data, the available evidence indicates that remote interventions may positively impact broader aspects of well-being. This further supports the holistic and comprehensive value of tele-rehabilitation approaches in pediatric care.
Effects of tele-neurorehabilitation on motor function
Our meta-analysis demonstrated a significant improvement in motor function following tele-neurorehabilitation interventions, with a small-to-moderate effect size. While several studies did not reach statistical significance,26–28 most showed a trend toward positive change.29–31 These non-significant findings are likely attributable to small sample sizes, limited statistical power, and variability in intervention protocols. Still, the overall direction of the effect consistently favored telehealth-based approaches. Notably, the observed substantial heterogeneity reflects differences in study design, intervention intensity, outcome measures, and the specific motor domains assessed. This underscores the need for greater methodological consistency in future trials, specifically regarding standardized outcome measures. Improving motor function in children with neurologic conditions is not only clinically relevant, but also critical for long-term functional independence. 32 Even modest gains can translate into increased participation in everyday activities, including school, play, and self-care, which are foundational to healthy development. 33 The variability observed in the prediction intervals likely stems from differences in motor function assessment methods, the heterogeneity of intervention designs, and the specific motor domains addressed across studies. Although the average effects point toward moderate improvements, the moderate range of possible outcomes underscores the need for cautious interpretation.
Effects of tele-neurorehabilitation on range of motion
Our meta-analysis revealed a small but statistically significant effect of tele-neurorehabilitation on improving range of motion (ROM) in pediatric populations. Although the effect size was modest, the consistency of direction across studies and the relatively low heterogeneity strengthen confidence in the reliability of these findings. This suggests that remote interventions can effectively target joint mobility, particularly in children with neuromotor impairments who are at increased risk of developing ROM restrictions over time. 34 Improved ROM is closely linked to functional outcomes, such as reaching, grasping, and ambulation, and is also essential for preventing contractures and maintaining musculoskeletal health. Nonetheless, the gains observed support the integration of tele-rehabilitation into home programs aimed at preserving and enhancing joint flexibility. Regular guided movement sessions, combined with active caregiver participation, appear to contribute to the maintenance of therapeutic routines outside of clinical settings. 35 Future studies should employ standardized and quantitative ROM measures and explore long-term outcomes to better determine the durability and clinical impact of these improvements. The prediction for ROM suggests that future studies may observe small but variable improvements. This range reflects differences in assessment tools and intervention protocols, reinforcing the need for standardized assessment procedures.
Effects of tele-neurorehabilitation on walking function and muscular strength
Our findings suggest that tele-neurorehabilitation may contribute to improved walking function in pediatric populations with neurologic conditions. The meta-analysis revealed a small but statistically significant effect with low heterogeneity, indicating a relatively consistent, though modest, benefit across studies. Improved walking ability is a key functional outcome, often linked to greater independence and participation in daily life activities,36,37 which reinforces the clinical relevance of even small gains in this domain. In contrast, no statistically significant improvement was observed for muscular strength, despite a moderate effect size and several individual studies reporting favorable trends.38,39 The wide confidence interval and substantial heterogeneity highlight inconsistencies in intervention design and outcome measurement, as well as the limited sample sizes across studies. These limitations make it difficult to draw firm conclusions about the effectiveness of remote interventions for strength enhancement. Overall, while tele-neurorehabilitation shows promise in promoting functional mobility, further research is needed to clarify its role in improving muscular strength, ideally through larger trials employing standardized, strength-specific assessment tools.
However, these conclusions should be interpreted with caution due to the heterogeneity observed across studies, especially in the domains of motor function and muscular strength. This variability can be attributed to several factors, including the diversity of assessment tools used and the differences both in the intervention protocols and in the targeted motor domains. The variability found also in the prediction estimates likely reflects the diversity of walking function and strength measurement protocols, intervention formats, and specific motor domain targeted across study. While the mean effect suggests a moderate improvement, the range of potential outcomes cautions against overly optimistic generalization.
Clinical implications and future perspectives
Implementing tele-rehabilitation in a pediatric neurorehabilitation can help delivering continuous and individualized care within the child's home environment. This is particularly advantageous for families living in rural or underserved areas, or for those facing logistical and financial barriers to in-person services. Moreover, telerehabilitation helps to achieve a more involving model of care, encouraging both parents and caregivers to be active partners in the therapeutic process. 1 Studies have shown that caregiver engagement not only facilitates the delivery of remote interventions but also improves their effectiveness by strengthening family support and integrating therapy into daily routines.17,34,38 Encouraging caregiver participation is therefore crucial to maximize the outcomes and sustainability of pediatric telerehabilitation programs.
Nonetheless, the successful implementation of telerehabilitation also depends on addressing critical technological and infrastructural barriers. Disparities in access to high-speed internet, appropriate digital devices, and digital literacy represent significant challenges to the equitable delivery of remote rehabilitation services. 8 Without careful consideration, these gaps may limit the inclusivity and effectiveness of digital health interventions. To promote broader accessibility, future telerehabilitation programs should prioritize adaptable solutions that work across diverse socioeconomic contexts, including low-bandwidth environments and widely available devices.40,41 Investing in intuitive, user-friendly platforms is crucial to ease the technological burden on families and enhance engagement. Additionally, initiatives to improve digital literacy and partnerships with community organizations can support equitable adoption on a larger scale. 42 By proactively addressing these barriers, telerehabilitation can fully realize its potential as a high-quality, accessible, and inclusive tool for pediatric care.
As telerehabilitation continues to expand, it is also important to ensure that rehabilitation professionals receive adequate training to deliver care effectively in virtual environments. 43 Telerehabilitation programs require adapted communication strategies, proficiency in virtual assessment tools, and innovative therapeutic approaches adapted to digital platforms. 44 Furthermore, hybrid care models, which combine in-person and remote sessions, could be another promising strategy. Through both in-person assessments and home-based tele-rehabilitation, hybrid models may help sustain therapeutic engagement over time. Particularly for children requiring long-term, intensive rehabilitation, hybrid models may help sustain therapeutic engagement, enhance continuity of care, and optimize clinical outcomes over time. 45
Moreover, nowadays, the integration of emerging technologies holds great potential to further enhance pediatric telerehabilitation. Tools such as motion-tracking systems, gamified therapy applications, and AI-driven adaptive feedback mechanisms can increase patient motivation, support individualized care plans, and enable real-time progress monitoring.40,46 Motion-tracking systems, leveraging accessible hardware like high-definition webcams and smartphones equipped with innovative technologies such as LIDAR, allow for precise tracking of body movements.47,48 Advanced computer vision frameworks facilitate real-time analysis, enabling clinicians to provide immediate corrective feedback and systematically assess progress. Gamified therapeutic applications, through interactive mini-games that reward correct physical performance, can sustain motivation and transform exercises into engaging activities.48,49 Meanwhile, AI-driven adaptive feedback mechanisms offer a personalized rehabilitation experience by analyzing motion data in real time, delivering tailored feedback, and dynamically adjusting task difficulty based on patient performance.50–52
The synergistic application of these technologies promises to create a more dynamic, engaging, and patient-centered telerehabilitation environment, ultimately enhancing the effectiveness of remote interventions and strengthening the therapeutic alliance with families.
Conclusions
This study demonstrates the effectiveness and practicality of telerehabilitation in supporting motor and functional improvements in children with developmental and physical impairments. The positive outcomes observed underscore the value of remote delivery models in maintaining therapeutic engagement and facilitating access to care, particularly for families facing geographic, logistical, or socioeconomic barriers to traditional in-person services. The results indicate that telerehabilitation is not only a feasible alternative but also a clinically meaningful approach that can be integrated into standard pediatric rehabilitation pathways. Furthermore, the findings of our study highlight the potential of telerehabilitation to enhance continuity of care, promote greater family involvement in therapy, and extend the reach of interventions beyond conventional clinical settings. These implications are particularly relevant in the context of ongoing healthcare challenges that demand more flexible and inclusive service delivery models. By demonstrating improvements in key functional outcomes, this study supports the broader adoption of telerehabilitation as a strategy for achieving child- and family-centered care goals. It also provides critical evidence for clinicians, policymakers, and healthcare systems seeking to implement more adaptable, and responsive rehabilitation services for pediatric populations.
Take home message
Telerehabilitation has a strong potential as an effective and accessible intervention for improving gross motor function, targeted upper and lower limb abilities, and walking performance in children with neurological conditions, particularly those with cerebral palsy and hemiplegia. Despite variability among studies, the overall evidence supports its use as a valuable complement or alternative to traditional in-person therapy. Additionally, telerehabilitation promotes active caregiver participation and enhances access to care, especially for families in underserved areas. To maximize its benefits, future research should prioritize the use of standardized outcome measures, address technological and infrastructural barriers, and use emerging technologies to deliver personalized rehabilitation experiences.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076251357504 - Supplemental material for Home-based telerehabilitation for pediatric neurological motor disorders: Current trends and future perspectives. A systematic review and meta-analysis
Supplemental material, sj-docx-1-dhj-10.1177_20552076251357504 for Home-based telerehabilitation for pediatric neurological motor disorders: Current trends and future perspectives. A systematic review and meta-analysis by Valeria Calcaterra, Luca Marin, Luca Guardamagna, Alessandro Gatti, Virginia Rossi, Pamela Patanè, Matteo Vandoni and Gianvincenzo Zuccotti in DIGITAL HEALTH
Supplemental Material
sj-docx-2-dhj-10.1177_20552076251357504 - Supplemental material for Home-based telerehabilitation for pediatric neurological motor disorders: Current trends and future perspectives. A systematic review and meta-analysis
Supplemental material, sj-docx-2-dhj-10.1177_20552076251357504 for Home-based telerehabilitation for pediatric neurological motor disorders: Current trends and future perspectives. A systematic review and meta-analysis by Valeria Calcaterra, Luca Marin, Luca Guardamagna, Alessandro Gatti, Virginia Rossi, Pamela Patanè, Matteo Vandoni and Gianvincenzo Zuccotti in DIGITAL HEALTH
Supplemental Material
sj-docx-3-dhj-10.1177_20552076251357504 - Supplemental material for Home-based telerehabilitation for pediatric neurological motor disorders: Current trends and future perspectives. A systematic review and meta-analysis
Supplemental material, sj-docx-3-dhj-10.1177_20552076251357504 for Home-based telerehabilitation for pediatric neurological motor disorders: Current trends and future perspectives. A systematic review and meta-analysis by Valeria Calcaterra, Luca Marin, Luca Guardamagna, Alessandro Gatti, Virginia Rossi, Pamela Patanè, Matteo Vandoni and Gianvincenzo Zuccotti in DIGITAL HEALTH
Footnotes
ORCID iDs
Author contributions
Conceptualization: V.C., M.V., L.M., G.Z.; independent collection of the contributions: A.G., A.G., V.R., P.P.; writing—original draft preparation: A.G., A.G., V.R., P.P.; writing—review and editing: V.C., M.V., L.M., G.Z.; supervision: V.C., M.V., L.M., G.Z. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported within the (1) MUSA—Multilayered Urban Sustainability Action—project, funded by the European Union—NextGenerationEU, under the National Recovery and Resilience Plan (NRRP) Mission 4 Component 2 Investment Line 1.5: Strengthening of research structures and creation of R&D “innovation ecosystems,” set up of “territorial leaders in R&D”_CUD G43C22001370007; (2) “PSR_LINEA6_2021/Research Development Plan-University Grand Challenges-Line 6 Disease prevention and sustainability and efficiency of healthcare: a distributed virtual hospital for Italy (e-Care).”
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data sharing is not applicable to this article as no new data were created or analyzed in this study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
