Abstract
Objective
This review examined the effectiveness of mobile-health (m-health) app-supported interventions in improving patient health outcomes. It also sought to describe the behavior change techniques (BCTs) used in these interventions and identify effective BCTs or combinations of BCTs to facilitate glycemic control.
Method
MEDLINE, EMBASE, and Web of Science databases were searched for relevant studies published until November 2024. Forty-three randomized controlled trials (RCTs) examining the effects of m-health app on glycemic control were identified and included in the analysis. The BCTs utilized in each intervention were coded based on a widely used BCT taxonomy. Health outcomes (e.g. change in hemoglobin A1c [HbA1c]) were meta-analyzed using random effect models. Meta-regression models were used to examine associations between the use of BCTs and HbA1c improvements. Cochrane Collaboration's tool was used for the evaluation of the risk of bias.
Results
Using m-health app-supported interventions significantly reduced HbA1c, fasting blood glucose, systolic and diastolic blood pressure, low-density lipoprotein cholesterol, triglycerides, weight, and waist circumference. The examined interventions utilized 5.77 BCTs on average. The most frequently used BCTs included “credible source,” followed by “social support (general),” “self-monitoring of outcome(s) of behavior,” “biofeedback,” “self-monitoring of behavior,” and “instruction on how to perform a behavior (skills training).” Interventions that utilized “problem solving” and “reward and threat” were associated with greater HbA1c improvement than those did not. Using BCTs in groupings of “feedback and monitoring,” “social support,” and “comparison of outcomes” formed the simplest and most effective combination.
Conclusion
Our study provides evidence about effective BCTs and combinations for better glycemic control. However, optimal BCT combinations warrant further examination. Future RCTs that clearly report the BCTs used are recommended. Experimental designs such as a multiphase optimization strategy should be used to examine the effects of single BCTs and their interactions.
Introduction
Approximately 537 million adults are living with diabetes, 1 and 90% of them have type 2 diabetes. 2 Diabetes can cause serious complications (e.g. cardiovascular disease, kidney failure, and retinopathy), and it is a leading cause of death and disability worldwide.3,4 To improve glycemic control, patients with type 2 diabetes must perform diligent self-management, such as taking prescribed medications, eating a healthy diet, conducting regular self-monitoring, and exercising. 5 However, patient compliance is often suboptimal, and many patients struggle to adhere to treatment regimens consistently.5–7 Nonadherence to self-management regimens is associated with health consequences.8,9
Recently, with the ongoing and dramatic spread of mobile technologies, mobile health (m-health) has emerged as a promising tool to encourage behavior changes and support disease management among patients with diabetes who need lifelong self-care.10–12 Health apps are among the most common delivery modalities for m-health interventions. Specifically, m-health app-supported interventions capitalize on various features (e.g. self-monitoring of blood glucose, education, reminders, and patient-care provider communication) to address the constraints of conventional self-management processes.10,12,13 For instance, Or et al. 11 developed a tablet-based self-management app for patients with comorbid type 2 diabetes and hypertension that enables patients to record and retrieve their blood glucose and blood pressure levels. Agarwal et al. 14 designed an m-health app-assisted intervention to support the self-monitoring of health behaviors and outcomes and deliver tailored, real-time feedback messages to facilitate patient behavior and motivation.
Systematic reviews of randomized controlled trials (RCTs) consistently illustrate the positive effects of m-health app-supported interventions.10,15,16 However, very few studies have examined the underlying theoretical basis of the intervention.17,18 Notably, m-health apps developed for diabetes self-management are often complex and include various behavior change techniques (BCTs),19,20 which are “an observable, replicable, and irreducible component of an intervention designed to alter or redirect causal processes that regulate behavior.” 21 Individual BCTs may have additive, synergistic, or antagonistic interactions with others.22,23 Identifying the most effective BCT components and combinations is crucial to investigating potential causal relationships between BCT components and glycemic control, subsequently guiding the optimization of future interventions. 24 Although a few reviews have addressed this issue,17,18,24 their findings are inconsistent.
To fill the aforementioned research gap, this systematic review addressed two objectives: (1) to investigate the effects of m-health app supported interventions on health outcomes (e.g. hemoglobin A1c [HbA1c], fasting blood glucose [FBG], and body mass index [BMI]) among patients with type 2 diabetes, and (2) to identify which BCTs and BCT combinations were effective when delivered via m-health apps to patients with type 2 diabetes.
Methods
This systematic review and meta-analysis were conducted based on the Cochrane Handbook for Systematic Reviews of Interventions 25 and reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement 26 (see Appendix 1). This review was not pre-registered, and a research protocol was not prepared. Two researchers independently performed study selection, data extraction, and risk of bias assessment. Any discrepancies were addressed and resolved through discussion.
Search strategy
We searched the MEDLINE, EMBASE, and Web of Science databases to identify relevant studies published between January 2007 and November 2024. The search was initially conducted in December 2022 and updated in December 2024. We chose 2007 as the starting year because most m-health apps were released after 2007.10,27 The search strategy included four sets of terms for type 2 diabetes (type 2 diabetes or type 2 diabetic or T2DM), technological medium (phone* or tablet*), disease management (monitor* or manag* or care or control), and effect examination (examin* or evaluat* or assess* or compar*). The detailed search strategy and limits employed across databases were presented in Appendix 2.
Eligibility criteria
The eligibility criteria were: (a) RCTs, (b) studied type 2 diabetic patients, (c) assessed the effectiveness of m-health app-supported interventions on changes in HbA1c and other patient outcomes relative to the effectiveness of usual care, and (d) were published in English-language, peer-reviewed journals.
Study selection
Two researchers initially assessed the titles and abstracts of the sources retrieved via the literature search. They then determined the eligibility of potentially relevant papers by reading the full text. The researchers also scrutinized the reference lists of the selected papers and relevant review articles to identify any publications that had been missed by the search.
Data extraction
Data extraction was guided by a preconstructed coding scheme. Extracted information included study characteristics (e.g. study location, sample size, HbA1c eligibility, participants’ mean age, male proportion) and the quantitative data on changes in HbA1c and other patient outcomes. Specifically, means and SDs of outcome measures at baseline and last time-point follow-up were extracted for both the intervention and control groups. If SD was missing, the authors estimated it using standard error (SE) and 95% confidence interval (CI) (if reported), according to the Cochrane Handbook. 25 For RCTs with multiple intervention groups related to using m-health apps, the control group was divided into two or more groups to facilitate multiple pairwise comparisons, as the Cochrane Collaboration Guideline suggests. 25 In addition, the BCTs used in each intervention were coded according to Michie's BCT taxonomy. 21 A BCT was coded as “present” if it was used in the intervention group but not the control group.17,23
Outcome measures
Primary outcome was changes in HbA1c at the end of each trial. Secondary outcomes included changes in other clinical outcomes, for example, FBG, systolic/diastolic blood pressure (SBP/DBP), and low-density/high-density lipoprotein (LDL/HDL) cholesterol.
Risk of bias assessment
Researchers evaluated the risk of bias for each trial using the Cochrane Collaboration's tool. 25 Risk of bias was evaluated in terms of: (a) selection bias, (b) performance bias, (c) detection bias, (d) attrition bias, and (e) reporting bias.
Data analysis
The level of agreement between the two researchers during the study selection process was examined with Kappa statistics. All of the study outcomes were meta-analyzed. The standardized mean difference (SMD) for each outcome was calculated with a random-effect model to represent the effect size. I2 statistic was calculated to examine the between-trial heterogeneity, with 50% and 75% indicating moderate and high levels of heterogeneity, respectively. 28 Egger's test was used to examine the publication bias of each outcome. 25 Forest plot was used to represent the meta-analysis result of intervention versus control regarding HbA1c improvement. Quality of evidence for each outcome was evaluated using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) system. 29 Quality of evidence was evaluated in terms of risk of bias, imprecision, indirectness, inconsistency, and publication bias. A univariate random-effect meta-regression model was planned using the BCT number and the presence or absence of each BCT grouping and individual BCTs as independent variables. Only BCTs and BCT groupings coded as present for at least two trials were included in the analysis. The adjusted R2 was calculated as a measure of how much of the outcome heterogeneity the covariate accounted for. Subgroup analyses were performed to examine the effects of certain combinations of BCTs on glycemic control. Comprehensive meta-analysis statistical software was used for all analyses.
Results
Study selection
The study selection process (Figure 1) identified 36 eligible articles (29 from databases and 7 from manual search).11,14,30–63 Of them, five RCTs31,35,45,55,63 had multiple relevant intervention groups; therefore, the control groups in these studies were split into more groups accordingly. Forty-three trials were included in our data analysis. Kappa statistics indicated a good level of agreement during the abstract screening phase (kappa = 0.83) and the full-text analysis (kappa = 0.91).
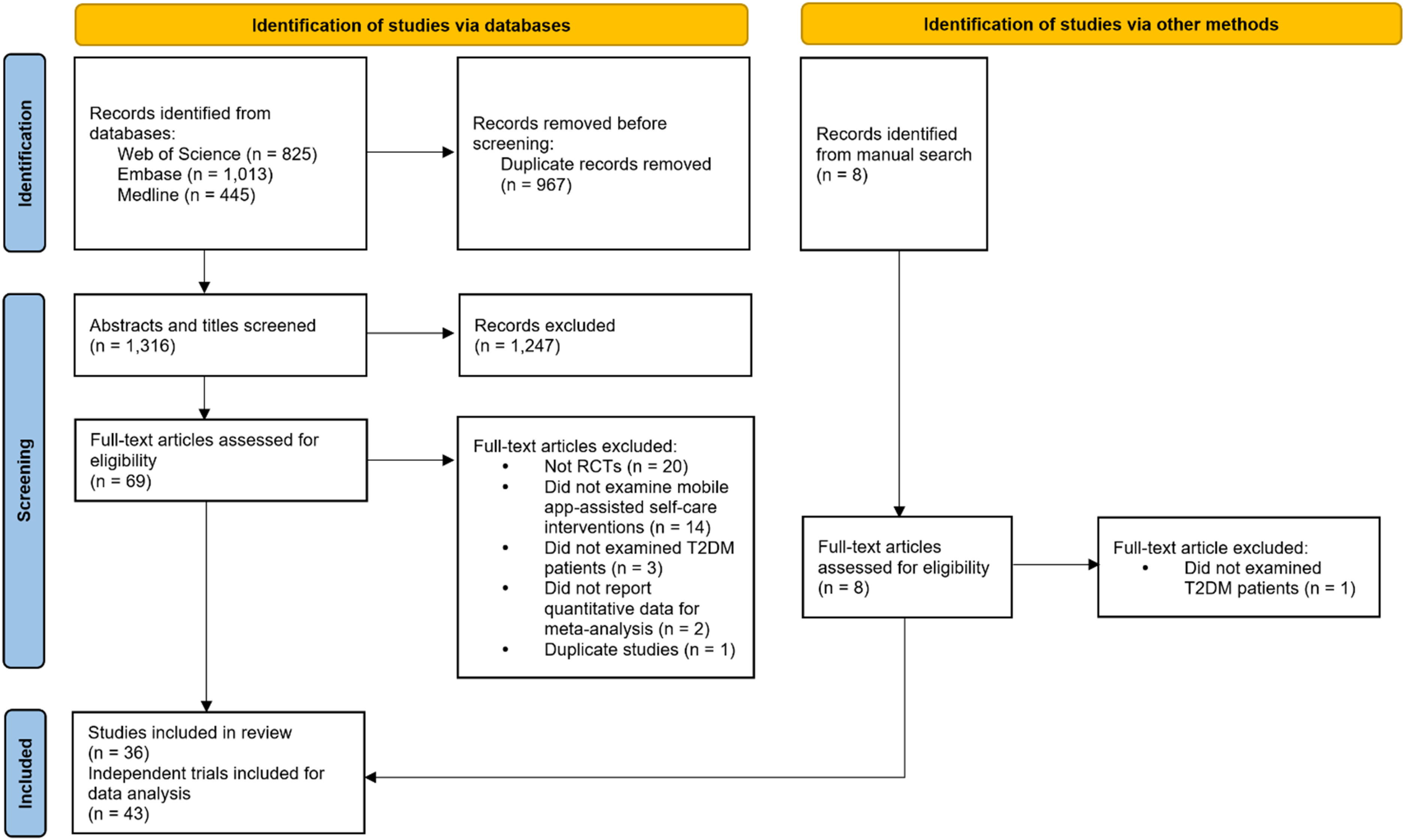
Flow diagram of study selection process.
Trial characteristics
Among the 43 trials, most of the trials were published after 2017 and in Asia and North American countries (Table 1). The mean sample size of the trials was 86, and the mean trial length was 6 months. Detailed characteristics of the included studies are presented in Appendix 3.
Trial characteristics.

Risk of bias assessment
Of all trials, 81%, 53%, 0%, 14%, 70%, 100%, and 26% were assessed as low risk in terms of the seven aspects, respectively (see Figures 2 and 3).

Risk of bias of the 43 trials.

Risk of bias for each trial.
Effects of m-health apps on study outcomes
Eleven outcomes were meta-analyzed (Table 2). M-health app-supported interventions significantly lowered HbA1c levels (SMD = −0.42, 95% CI, −0.56 to −0.27, p < 0.001, corresponding to an absolute mean difference [MD] = −0.42%, 95% CI, −0.57 to −0.27, p < 0.001). Figure 4 presents the forest plots for HbA1c change. For secondary outcomes, m-health apps also yielded more improvements in SBP (SMD see Table 2, MD = −2.69 mmHg, 95% CI, −4.72 to −0.67, p = 0.009), DBP (MD = −1.66 mmHg, 95% CI, −2.78 to −0.53, p = 0.004), LDL cholesterol (MD = −0.06 mmol/L, 95% CI, −0.12 to 0.001, p = 0.052), triglycerides (MD = −0.12 mmol/L, 95% CI, −0.21 to −0.03, p = 0.01), FBG (MD = −0.92 mmol/L, 95% CI, −1.07 to −0.76, p < 0.001), weight (MD = −1.01 kg, 95% CI, −1.87 to −0.16, p = 0.02), and waist circumference (MD = −2.01 cm, 95% CI, −3.55 to −0.47, p = 0.01). There was no significant effect of using m-health apps on HDL cholesterol, BMI, and total cholesterol. The GRADE assessment indicated that the quality of evidence for HbA1c was low due to a moderate-to-high level of inconsistency and risk of publication bias.

The forest plot for HbA1c change.
Meta-analysis and GRADE assessment results for the outcome measures.

a SMD: standardized mean difference.
b HbA1c: hemoglobin A1c.
c Downgraded by one level for inconsistency (I2 > 50%).
d Downgraded by one level for risk of publication.
e SBP: systolic blood pressure.
f DBP: diastolic blood pressure.
g LDL: low-density lipoprotein.
h HDL: high-density lipoprotein.
i FBG: fasting blood glucose.
*p < 0.05, **p < 0.01, ***p < 0.001.
Use of BCTs
Among the BCT taxonomy proposed by Michie et al., 21 23 BCTs (of 93 BCTs, 24.73%) from 11 groupings (of 16 groupings, 68.75%) were used. Interventions utilized an average of 4.02 (SD = 1.35, range 1–7, median = 4) BCT groupings. The most frequently used BCT groupings were “feedback and monitoring,” “comparison of outcomes,” “social support,” and “shaping knowledge” (Table 3, BCT coding details see Appendix 3). Other BCT groupings were utilized in less than 50% of the trials. Interventions utilized 5.77 BCTs (SD = 1.85, range 2–9, median = 6) on average. The most frequently used BCTs were “credible source,” followed by “social support (general),” “self-monitoring of outcome(s) of behavior,” “biofeedback,” “self-monitoring of behavior,” and “instruction on how to perform a behavior (skills training).” Other BCTs were utilized in less than 30% of the trials.
Frequency of BCTs used in the trials.

Effectiveness of BCTs
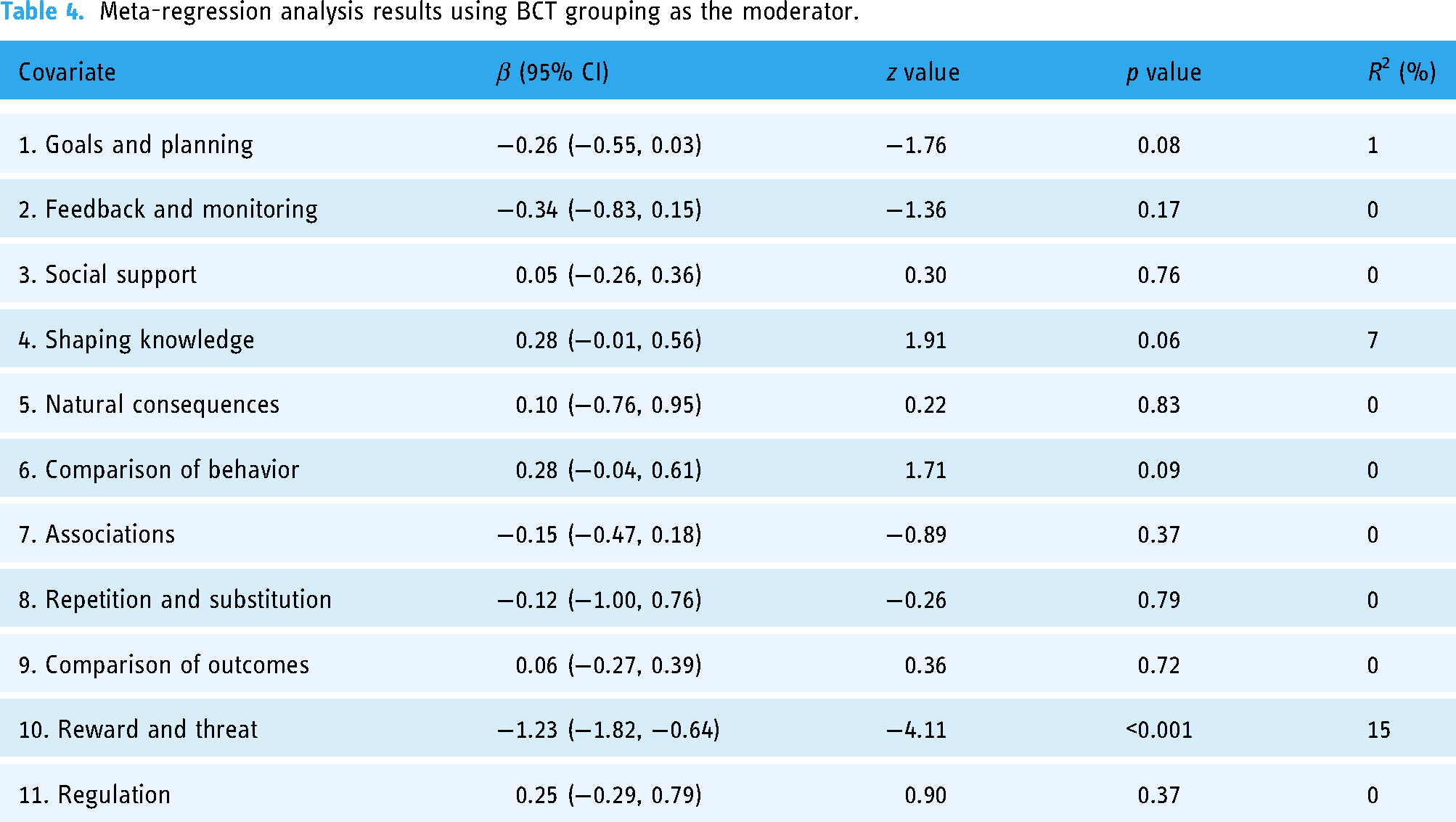
Meta-regression analysis showed that the number of BCTs used in the intervention was not found to be associated with more HbA1c improvement (β = −0.01, p = 0.80). At the grouping level, utilizing BCTs in grouping “reward and threat” yielded significantly greater improvement in HbA1c (β = −1.23, p < 0.001, Table 4). At the individual BCT level, utilizing “problem solving (coping planning)” (β = −0.70, p = 0.003) was found to be associated with greater improvements in HbA1c (Table 5). Using “feedback on behavior” yielded a marginally significant effect on HbA1c change (β = −0.31, p = 0.05).
Meta-regression analysis results using BCT grouping as the moderator.
BCT combinations
There were 29 different combinations of BCT groupings. Twenty-six (60.5%) trials simultaneously included BCTs in groupings “feedback and monitoring,” “social support,” and “comparison of outcomes.” Among them, six interventions only used BCTs from the three groupings, while others added BCTs from “goals and planning,” “shaping knowledge,” “comparison of behavior,” “association,” “reward and threat,” and “regulation,” or their combinations. Meta-analysis indicated that interventions using BCTs from groupings 2, 3, and 9 (SMD = −0.48, p < 0.001), grouping 2, 3, 9, and 4 (SMD = −0.56, p = 0.03), and groupings 2, 3, 9, 1, and 4 (SMD = −0.27, p = 0.03) were favorable in reducing HbA1c (Table 6). However, other combinations did not show greater improvement in HbA1c.
Meta-regression analysis results using individual BCTs as the moderator.

Meta-analysis results for different BCT combinations.

a SMD: standardized mean difference.
b Feedback and monitoring.
c Social support.
d Comparison of outcomes.
e Goals and planning.
f Shaping knowledge.
g Comparison of behavior.
h Regulation.
Discussion
Principal findings
Our systematic review investigated the effects of m-health app-supported interventions on type 2 diabetes patients’ health outcomes. We identified 43 trials to analyze with a literature search. Our meta-analyses indicated that, compared with conventional care, m-health app-supported interventions achieved greater improvements in HbA1c (SMD = −0.42, corresponding to an absolute MD of −0.42%) than usual care. This effect size was similar to those reported in other systematic reviews (SMD range −0.31 to −0.44, MD range −0.40% to −0.49%).10,15,16,18 It was also considered clinically meaningful because a decrease in HbA1c by 0.3% was associated with a lower risk of diabetic complications.18,64 Among the secondary outcomes, significant improvements in SBP, DBP, LDL cholesterol, triglycerides, FBG, weight, and waist circumference were found. These significant improvements may reflect that most m-health app-supported interventions involve lifestyle management features, such as diet and exercise management. However, m-health app-supported interventions did not improve HDL cholesterol, BMI, or total cholesterol. The reasons for this need to be further examined.
Of the 93 BCTs in Michie's taxonomy, 21 only 23 were used in the selected trials, illustrating that most BCTs in the taxonomy are not currently used in m-health apps for diabetes management. An average of 5.77 BCTs were used in the studied m-health apps. This number was similar to those reported in similar existing reviews (average number of BCTs: 4.5 to 9.07).17,18,24,65 Descriptive analyses indicated that “feedback and monitoring,” “comparison of outcomes,” “social support,” and “shaping knowledge” were identified in more than 50% of the trials. To utilize these types of BCTs, interventions usually included features such as self-monitoring of blood glucose, exercise, and diet; educational materials; and patient–healthcare provider communication.10,13,27
The univariate meta-regression analyses revealed that the number of BCTs was not associated with improved HbA1c. This finding diverged from some previous reviews.17,24 For example, Rhoon et al. 24 found that effective digital interventions for type 2 diabetes used more BCTs than ineffective interventions. El-Gayar et al. 17 indicated that using more BCTs may lead to slightly more HbA1c improvement. However, our results are consistent with other studies18,65 indicating that BCT number was not associated with better health outcomes. This finding affirms that using effective BCTs or BCT combinations is more important than using more BCTs. More research is needed to elucidate the relationship between BCT quantity and health outcomes.
This study identified several effective BCT groupings and individual BCTs through meta-regression analysis. At the group level, “reward and threat,” although used infrequently, effectively facilitated glycemic control. For example, Kamat et al. 53 provided financial incentives to patients if they maintained 80% medication adherence. This finding is consistent with previous studies indicating that rewards and incentives effectively encourage behavior changes for diet management, 66 tobacco cessation, 67 and medication-taking. 68 However, providing incentives may reduce patients’ intrinsic motivation to improve their behavior. 69 In addition, many studies report that incentives tend to facilitate initial behavior change, but the effects may diminish over time.69–71 As only two trials used this BCT, further research is needed to examine how different incentive types influence behavior change among chronically ill patients.
At the single BCT level, “problem solving (coping planning),” in which patients “analyze factors influencing the behavior and generate/select strategies that include overcoming barriers and/or increasing facilitators,” 21 was useful in reducing HbA1c level. Other digital behavior change intervention literature has also reported its effectiveness.23,72 This BCT was implemented via features that prompt patients to identify and document reasons or barriers causing non-adherence to specific self-management behaviors. 53 Patients were also trained to challenge negative thinking and improve their interpersonal and problem-solving skills.55,59 This approach can foster patients’ reflection on their behavior and facilitate the identification of challenges to adherence, enabling targeted improvements. Moreover, “feedback on behavior,” in which apps “monitor and provide informative or evaluative feedback on performance of the behavior,” 21 yielded marginally significant effects on HbA1c change. This BCT was enacted by sending individualized feedback about patients’ diet, exercise, and medication-taking behaviors. The results indicate that self-monitoring alone may be insufficient and ineffective without feedback. Providing real-time feedback can help patients feel supported and foster sustained engagement.38,73 The effects of these BCTs require further examination because of the limited number of trials that have assessed them.
We also analyzed common combinations of BCTs and performed subgroup meta-analyses of the effectiveness of certain combinations. Most interventions simultaneously included BCTs from feedback and monitoring (group 2), social support (group 3), and comparison of outcomes (group 9). Interventions that only used BCTs from groups 2, 3, and 9 worked to reduce HbA1c. Adding BCTs from group 4 (shaping knowledge) or from groups 4 and 1 (goals and planning) can also be effective. However, the number of trials in such subgroups was limited due to the significant differences in the BCT combinations used. Effective BCT combinations merit further study in the future.
Implications for research
First, using m-health app-supported interventions to assist self-management positively affected glycemic control among patients with type 2 diabetes. However, the mechanisms by which various BCTs affect patients’ health behaviors and outcomes remain unclear. Future research should investigate the factors driving behavior change and screen for effective BCTs and/or their combinations to improve intervention effectiveness. Second, investigating how different BCTs interact with each other is important. While RCTs can provide information about the overall effectiveness of an intervention, they cannot provide evidence about the effects of individual BCTs and their interactions. Therefore, future studies could utilize other experimental designs, such as a multiphase optimization strategy,74,75 to identify effective BCTs and/or their combinations. Third, the implementation time of most trials was six months or less, which is relatively short for chronic disease management studies. The long-term influence of BCTs warrants further examination in trials with longer study periods. Fourth, m-health app-supported interventions for diabetes self-management include multiple target behaviors. Further studies are needed to identify effective BCTs and BCT combinations for different behaviors. Fifth, the most frequently used BCTs were not the most effective ones. 65 Some effective BCTs were used infrequently. Researchers could explore other available BCTs, integrate them into diabetes management apps, and verify their effectiveness through empirical studies. Finally, many studies failed to clearly report the theoretical basis of their interventions or the specific BCTs utilized in the interventions. These vague descriptions posed significant challenges in coding BCTs, 65 making comparisons of the results across different review studies difficult. Therefore, we urge researchers to report on the theoretical basis and BCTs employed in their interventions.
Implications for practice
Our review provides evidence about effective BCTs and combinations for improving glycemic control. Health app designers should include effective BCTs and eliminate ineffective BCTs to make their apps more effective and less burdensome to use. For example, a combination of feedback and monitoring, social support, and outcome comparisons are useful in m-health apps for patients with diabetes. Other BCTs, such as problem-solving and rewards, can also be used to facilitate behavior change. Furthermore, m-health app developers should know that more BCTs do not translate to greater effectiveness. Too many features increase the complexity of health apps, making them difficult for patients (especially older patients) to use. Finally, m-health app designs should be theory-driven. 76 Developers should select an appropriate theoretical basis to help prevent the unnecessary use of ineffective interventions.
Strengths and limitations
This systematic review, which considered 43 individual trials, examined the effectiveness of m-health app-supported interventions in facilitating glycemic control. We employed the BCT taxonomy to systematically investigate the BCTs used across trials and examined the associations between using BCTs from particular groups and HbA1c reduction. However, this study had various limitations that must be acknowledged. First, only a few studies have reported that their interventions were designed according to behavior change theories, and most failed to clearly describe the BCTs used in their interventions.23,77 Therefore, the BCTs were identified based on the authors’ understanding based on the features described in published articles, 22 which may have introduced bias. Second, the targeted behaviors varied among the included trials. Most m-health apps used in the selected studies targeted more than one health behavior. However, the vague descriptions of the interventions in the research articles made clarifying which BCTs applied to each target behavior challenging. Future studies should provide more details about their interventions, including target behaviors, applied BCTs, and the intensity and frequency of each component. Third, relatively few trials were included in this review, which may limit its power to examine the moderating effects of all single BCTs or BCT combinations. Future studies might examine the effects of more moderators when more relevant trials are published. Fourth, only articles published in English were included in this review, which may have introduced language and publication biases. Fifth, we did not search databases such as CENTRAL, although most of its indexed journals are included in other databases we searched. Finally, there is a lack of prior registration of review protocols.
Conclusions
Adhering to self-management activities is necessary but challenging for patients with type 2 diabetes. This review provides compelling evidence that m-health app-supported interventions can facilitate glycemic control. This study identified certain effective BCTs and BCT combinations. BCTs that were used infrequently in our context but were found effective in other behavior change domains should be examined further. Moreover, the interaction effects of various BCTs and optimal BCT combinations warrant further examination. This review provides translational evidence for healthcare practitioners to improve the design and optimization of their interventions to support diabetes care.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076251326126 - Supplemental material for Effective behavioral change techniques in m-health app supported interventions for glycemic control among patients with type 2 diabetes: A meta-analysis and meta-regression analysis of randomized controlled trials
Supplemental material, sj-docx-1-dhj-10.1177_20552076251326126 for Effective behavioral change techniques in m-health app supported interventions for glycemic control among patients with type 2 diabetes: A meta-analysis and meta-regression analysis of randomized controlled trials by Kaifeng Liu and Yuxuan Xia in DIGITAL HEALTH
Footnotes
Contributorship
KL: conceptualization, methodology, formal analysis, writing—original draft, writing—review & editing; YX: formal analysis, writing—original draft.
Data sharing
The data collected for this study will be provided upon request to the corresponding author.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The review was conducted with support from National Natural Science Foundation of China (grant no. 72104176) and the Research Project of the National Intelligent Society and Governance Experimental Base in the Health and Wellness Industry (2023) [grant no. TJHIA-2023-010]. Dr. Liu would like to thank the support of Xiaomi Young Talents Program (Xiaomi Foundation).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
