Abstract
Objective
Digital therapeutics (DTx) are promising technologies. However, current assessment and access frameworks, when they exist, are heterogeneous and fragmented. We analysed and compared health technology assessment (HTA) criteria for DTx across European countries that had assessed the same DTx products. This allowed us to conduct a direct comparison of these countries’ DTx assessment frameworks.
Methods
A scoping review identified HTA DTx documents from European HTA bodies with specific DTx frameworks in place. The HTAs of the same DTx products assessed across multiple countries were then extracted and analysed.
Results
Deprexis and Velibra were the only DTx products assessed by multiple countries: Deprexis, indicated for depression, was assessed by Germany, the United Kingdom (UK) and France; and Velibra, indicated for anxiety, was assessed by Germany and the UK. There are notable differences among those frameworks, but what they share is an emphasis on the context-specific positioning of products within the disease landscape, choice of comparators and usage and usability data. Safety data are not a major focus in DTx assessments in these countries, but a request is often made for the addition and evaluation of risk-flagging systems.
Conclusion
The majority of European countries do not have a specific framework in place for the assessment of DTx, and major differences exist among recently implemented frameworks. The current policy landscape at the European Union level offers an opportunity to establish a harmonized framework for the assessment of DTx and possibly faster access and integration of such promising technologies.
Keywords
Introduction
The past five years have seen a substantial change in healthcare towards a more digitized approach. This digitization of the healthcare system has occurred across multiple levels, including interoperability, electronic health records and digital health technologies (DHTs), allowing for improved efficiency, reduction in errors and lower costs. However, the current rate of this digital transformation of the healthcare system is slow, and great potential in this area remains unachieved. DHTs include systems that employ computing platforms, connectivity, software and sensors and can be standalone medical products or used in addition to other medical products (devices, drugs, biologics). 1 Digital therapeutics (DTx) are a subtype of DHTs; while there is no universally agreed-upon definition of DTx, the International Organization for Standardization has defined DTx as products ‘intended to treat or alleviate a disease, disorder, or injury by generating and delivering a medical intervention that has a demonstrable positive therapeutic impact on a patient's health’. 1
Although DTx have been available for years, the rise of personalized healthcare and the overall digital transformation of healthcare have accelerated their uptake and led to a change in how DTx are perceived and used. DTx are now considered not only a potential option, but a highly necessary and beneficial solution to the treatment of many diseases. As a result, many European countries have invested in digital health and launched initiatives to encourage the development and use of DTx, as has the European Union (EU). 2
Despite this, there generally remains a lot of ambiguity around the definition, regulation and assessment of DTx products, creating barriers for developers to launch their products and for patients to access them. 3
Background
DTx regulatory framework in Europe
Before a patient can access a DTx product, the product must receive regulatory market approval. In the EU, DTx are considered medical devices, falling under the Medical Device Regulation (MDR), and must receive Conformité Européenne (CE) marking. 4 DTx can also be assessed for market approval at a national level, though this rarely occurs. In the United Kingdom (UK), DTx with a CE marking are currently still accepted; alternatively, manufacturers can seek a UK Conformity Assessed mark, allowing their DTx products to be placed on the UK market.
According to Article 51 of the MDR, medical devices are divided into four classes: I, IIa, IIb and III based on the intended purpose of the device and its inherent risks. DHTs classified as Class IIb or III devices require a great degree of evidence to be developed and submitted for market approval to be received. 4
Current market access and reimbursement landscape
Following market approval, manufacturers can subsequently undergo DTx health technology assessments (HTAs) to secure market access and reimbursement. HTAs are a systematic evaluation of existing evidence for medical products to determine the clinical and cost-effectiveness impact of those products. Thus far, only a few countries in Europe (including Germany, the UK, France, Belgium, Finland and Sweden) have introduced detailed policies specific to the assessment of DTx. Among them, Germany and the UK were the first to pass legislation and issue guidelines specific to digital health products.5,6 As part of these policies, Germany and the UK were also the first countries to introduce specific assessment criteria for DHTs, namely the Digitale Gesundheitsanwendungen-Verordnung (DiGAV; focused exclusively on DTx) and the Evidence Standards Framework for Digital Health Technologies (ESF), respectively.7,8 At the time of this manuscript's composition, a separate framework for digital care applications had been established in Germany, but no apps had yet completed the approval process. 9
Based largely on German's DiGA framework, France has now developed its own DTx assessment framework, known as Prise en charge anticipée des dispositifs médicaux numériques (PECAN), introduced on 30 March 2023. While many countries are now in the process of developing DTx assessment frameworks, a number of challenges remain in the process of gaining market access and reimbursement for these products. An overview of the DTx assessment and reimbursement frameworks of the first movers, namely Germany, the UK and France, is presented in Table 1.
DHT assessment and reimbursement policy frameworks in Germany, the UK and France.

For the UK, a number of the products identified fell under the Improving Access to Psychological Therapies Programme (IAPT), which was implemented to promote mental health-related innovations.
It is notable that for France the search covered products assessed by the old access framework rather than by the new framework currently under development.
Economic evaluation in France is done only if the intervention would have a substantial financial impact on the health system.
DTx access frameworks in Europe
Germany, the UK and France all have established or are establishing fast-track schemes to speed access to innovative DTx while additional evidence is being generated.8,10 Only Germany and the UK have DTx-specific assessment frameworks in place and running and have developed guidelines for evidence requirements specific to DHTs.7,8
All three countries also favour high-level clinical evidence from randomized controlled trials (RCTs) and meta-analyses.7,8,11 In Germany, the DiGA framework focuses exclusively on DTx products, for which clinical benefits and/or patient-relevant improvement of structure and processes are assessed. In the UK and France, the focus is broader than on DTx products alone. Indeed, both countries assess and reimburse DHTs that address organizational aspects (e.g. telemonitoring devices) and are not, per se, DTx products.8,10
Only the UK has structured economic assessments for DTx, with the most expensive ones requiring the submission of a cost-effectiveness analysis on top of a budget impact analysis. In France, economic analysis is requested only for DTx that will have a substantial economic impact on the healthcare system. In Germany, no economic analysis is requested or performed as part of the current assessment process.
Finally, only Germany and France have centralized market entry frameworks in place for DTx. To be reimbursed, products need to be added to the DiGA repository for Germany and the LPP (
Current market access and reimbursement challenges
In order for manufacturers to gain market access, they must navigate a series of hurdles. Firstly, there are a limited number of clear access and reimbursement frameworks, as well as a lack of guidance on the evidence requirements for assessment. 12 In addition, there is a high level of heterogeneity in market access landscapes between countries, with a variety of stakeholders involved across healthcare systems, leading to confusion and overlapping of responsibilities for decision-making and reimbursement across stakeholders. 13 As the number of countries developing DTx assessment frameworks increases, there is a greater need for alignment across countries. Many stakeholders also have a lack of knowledge of DTx, particularly regarding the clinical and economic impact of DTx. 13 This is further worsened by the lack of consensus across stakeholders such as HTA agencies, payers and physicians on assessment of value/benefit. 13 Furthermore, there are also country-specific challenges that reflect the heterogeneity of HTA and reimbursement frameworks across countries. 3
While other papers have described the market access and reimbursement policies for DTx in Europe and globally, none have directly compared HTA assessment criteria and requirements between countries by identifying DTx products assessed by multiple HTA bodies.3,14
This study aims to provide a detailed analysis and comparison of HTA assessment criteria for DTx across European countries that have introduced DTx-specific frameworks by comparing assessments of the same products. More specifically, the study aims to:
Identify published HTAs of DTx products assessed by multiple European countries with a DTx assessment framework. Analyse HTAs of DTx products evaluated by more than one country, to allow for a direct comparison of DTx assessment frameworks.
The intended audience of this manuscript includes DHT and HTA policymakers, as well as DTx developers. We believe these groups are the most relevant because the collaboration of multiple stakeholders is essential to building on the progress made so far. Recognizing past efforts and understanding where synergies can be created will help align policies, streamline decision-making and ensure that innovations in digital health are effectively integrated into healthcare systems.
Methods
A scoping review was conducted to identify European countries with specific DTx frameworks in place at the national level and the respective assessor. These included Bundesinstitut für Arzneimittel und Medizinprodukte (BfArM) (Germany), 15 the National Institute for Health and Care Excellence (NICE) (UK), 16 Haute Autorité de Santé (HAS) (France), 17 Finnish Coordinating Center for Health Technology Assessment (FinCCHTA) (Finland), 18 Tandvårds-och läkemedelsförmånsverket (TLV) (Sweden) 19 and National Institute for Health and Disability Insurance (NIHDI) (Belgium).20,21 This was followed by a search across these European HTA bodies’ databases to identify HTAs of DTx products. The search strategy for this is presented in Table 2. The initial search was undertaken on 10 October 2022, with a first update search conducted on 19 June 2023 and a second update search conducted on 7 February 2024. The population, intervention, comparator, outcomes and study design (PICOS) eligibility criteria used to identify relevant HTAs in the systematic search are presented in Table 3. This review was not registered with any prospective review database (e.g. PROSPERO).
Search strategy.

Inclusion/exclusion criteria.

In line with the objectives of this study, once the relevant HTAs of DTx products were identified, an overview of the HTAs identified was conducted. Secondly, the HTAs of the same DTx products assessed across multiple countries were then extracted and analysed.
Results
Health technology assessments of DTx
HTAs identified
A search of DTx HTAs across relevant European countries revealed that the only countries with comparable DTx assessments of the same DTx products were Germany (with the Digitale-Gesundheitsanwendungen (DiGA)), 15 the UK (with the Digital Technology Assessment Criteria (DTAC) and the ESF) 16 and France (with the broader assessment principles established by the Medical Device and Health Technology Evaluation Committee (CNEDiMTS) to determine the reimbursement eligibility of medical devices for individual use). 11
A total of 70 HTAs of DTx products were identified across these three countries, as presented in Figure 1. In all three countries, the majority of DTx products that were assessed target mental health: 21 out of 52 in Germany, 16 out of 17 in the UK and one out of one in France. This disease area also saw the disease increase in HTA assessments over the last two years, with 10 assessments having been conducted for mental health products since October 2022. In the case of the UK, the focus on mental health DTx products is in part due to the implementation of the NHS Talking Therapies, for Anxiety and Depression programme (formerly known as Improving Access to Psychological Therapies, IAPT), developed to promote mental health-related innovations. 22 Endocrine/metabolic-related and nervous system-related DTx were the next most commonly assessed products, accounting for 11 out of 52 assessments in Germany and one out of 17 in the UK, respectively.

Types of DTx assessed in Germany, the UK and France (
HTAs analysed
Only two products were assessed across multiple countries, Deprexis and Velibra, the former developed for the treatment of depression and the latter for the treatment of anxiety. Both these DTx products are patient-facing mobile applications.
Deprexis was assessed across all three countries, first in the UK in 2018 23 (then reassessed in 2023 24 at the end of the IAPT program), then in Germany in 2021, 25 and finally in France in 2021 26 –2022. 27 Velibra was assessed only in the UK (in 2019)28–30 and in Germany (in 2020). 31 An overview of the evidence submitted can be found in the Appendix (for Deprexis, this refers to the 2018 assessment only; in the 2023 assessment, it is not possible to distinguish the evidence specific to Deprexis).
While Deprexis was ultimately recommended, or considered eligible, for reimbursement across all three countries, Velibra was only approved in Germany and not recommended in the UK. The assessments of both products focused on positioning and comparators, efficacy and safety, usability, unmet need and – in the UK – economic impact. It is also important to consider that in Germany, DHT assessments are not publicly available; however, certain assumptions can be made based on the final decision and the requirements that need to be fulfilled, for example, in the implementation of risk-flagging tools (e.g. alerts to practitioners or therapists of potential concerns for a user). The primary endpoints considered in the assessment of Deprexis and Velibra, as well as intervention and comparator details, are presented in Table 4. An overview of the assessment of these products is presented in Tables 5 and 6 for Deprexis and Velibra, respectively.
Overview of primary endpoint, intervention details and comparator details across HTAs.

It refers to the 2018 assessment only; in the 2023 assessment it is not possible to distinguish the evidence specific to Deprexis.
Submitted as supplementary evidence
HTA assessment criteria for Deprexis.

No published evidence submitted.
 Requirements fulfilled.
Requirements fulfilled.
 Requirements partially fulfilled.
Requirements partially fulfilled.
 Unclear if assessed or if evidence was submitted.
Unclear if assessed or if evidence was submitted.
 Requirements not fulfilled or evidence missed.
Requirements not fulfilled or evidence missed.
HTA assessment criteria for Velibra.
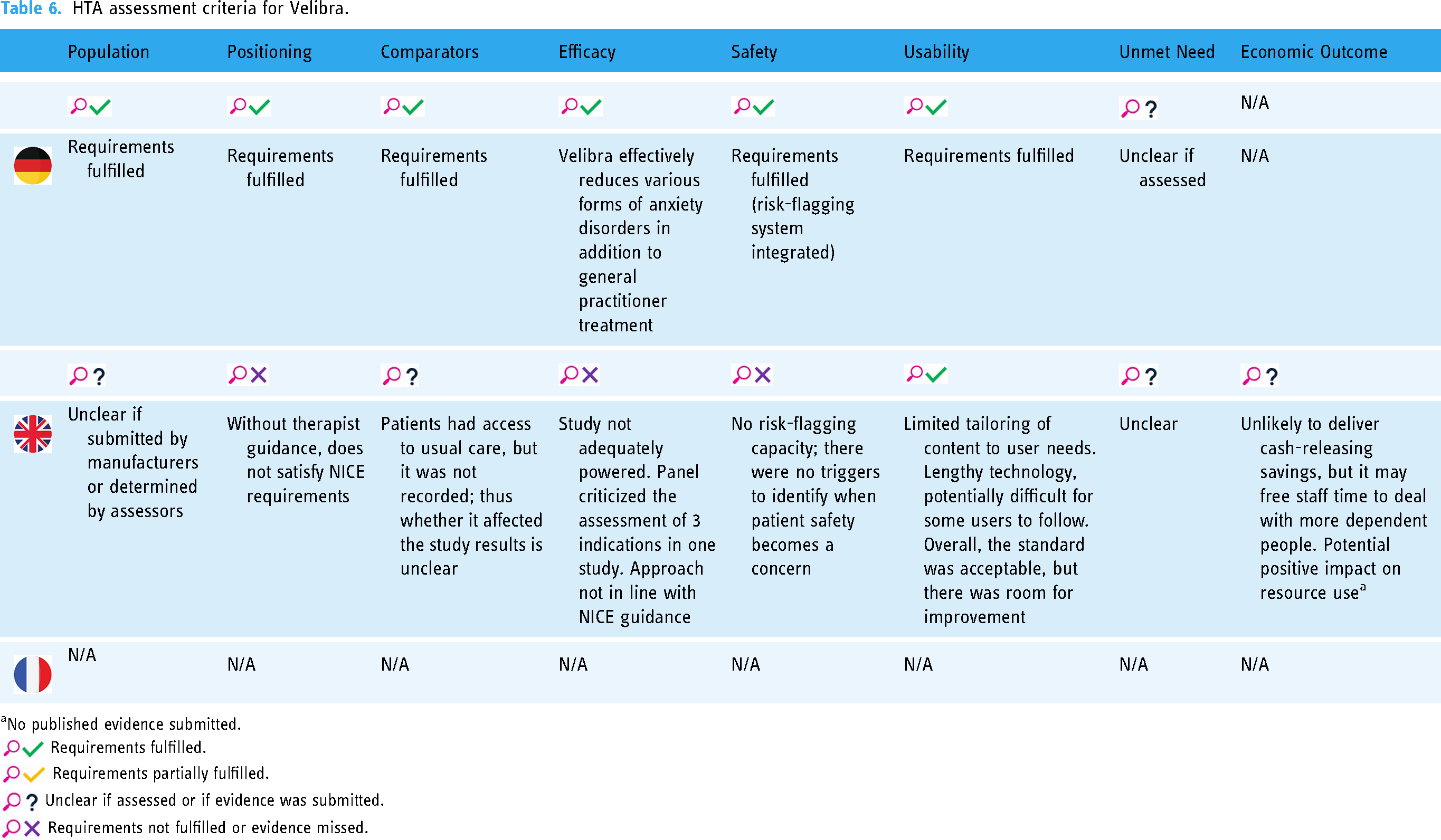
No published evidence submitted.
 Requirements fulfilled.
Requirements fulfilled.
 Requirements partially fulfilled.
Requirements partially fulfilled.
 Unclear if assessed or if evidence was submitted.
Unclear if assessed or if evidence was submitted.
 Requirements not fulfilled or evidence missed.
Requirements not fulfilled or evidence missed.
Positioning and comparators
The HTA analysis revealed an extensive focus from HTA authorities on a context-specific positioning of the product and appropriate choice of comparators based on national frameworks and policies. HTA authorities stressed how essential it is to contextualize the product to country-specific care models and, particularly in France, with context-specific real-world data. HTA authorities in France and the UK criticized the positioning of Deprexis in their respective markets and therefore the comparators chosen. In the 2018 assessment, NICE said that Deprexis together with psychotherapy was not representative of the NHS care model and that ideally Deprexis would be compared with face-to-face cognitive behavioural therapy in a therapist-guided care model. In the updated NICE assessment of Deprexis, it was concluded that Deprexis should be integrated into a system alongside face-to-face treatment by therapists and practitioners, rather than serve as a standalone treatment. Similarly, HAS in France stressed that Deprexis needed to be positioned as a complement to conventional treatment and would not replace existing therapies. Positioning and comparators were not discussed in the German assessments, but both Deprexis and Velibra were indicated in addition to usual care.
Efficacy and safety
The submitted evidence overlapped across Germany, the UK and France, as can be seen in Table S1 in the Appendix, and the efficacy outcomes were generally considered sufficient for Deprexis across all three countries. Efficacy data for Velibra were considered sufficient in Germany but not in the UK; specifically, the UK HTA found the efficacy data study to be insufficiently powered and criticized the assessment of three different indications in the same study. HTA authorities in France and the UK asked for additional efficacy data across all assessments. In the UK, after the assessment of Deprexis, assessors sought supplementary data concerning the rates and causes of treatment discontinuation, additional treatment needs and insights into the patient experience. Another common element across both the UK and France was the need for country-specific efficacy outcome data. In the UK, it was suggested to generate evidence within NHS Talking Therapies with appropriate comparators for Deprexis. Similarly, French studies with real-world data were requested for re-assessment in one year's time by HAS. Regarding safety, no major adverse events related to the use of the technologies were investigated and reported. Often HTA agencies asked for additional safety data to be collected, but no major concerns were raised by any assessor. HTA authorities in Germany and the UK emphasized that digital tools such as Deprexis and Velibra should be used for obtaining real-time safety alerts and data through a structured risk-flagging system embedded in the software. In contrast, France focussed more traditionally on the assessment of adverse events associated with the technology. However, this may change with the new French DTx legislation, known as PECAN, introduced on 30 March 2023. 10 This new fast-track framework is based largely on Germany's DiGA framework.
Usability and usage
HTA analysis also revealed an important focus on usability and usage evidence. Usage refers to data on the amount of product use, while usability refers to the state or condition of use of a product (ease of use). Usability evidence was extensively analysed from NICE in the UK, where reviewers criticized the limited tailoring of content to user needs, the comprehensibility of that content and the lack of patient experience data. For example, it was highlighted that digitally enabled therapies may not be suitable for patients with limited access to technologies or an internet connection, or who may have visual or hearing impairment. In both Germany and the UK, users’ involvement is required across the entire development phase of the product. Usability was also an important element for approval by BfArM in Germany; however, no details on this are publicly available. Usability needs to be assessed as part of the technical documentation.32,33 Usage data and the generation of real-world evidence, which provide better insight into actual usage and effectiveness, are gaining increasing relevance and may play an important role in future price negotiations. 32 In France, HAS seems to focus more on usage data than usability, particularly on metrics concerning user engagement, such as duration of use, frequency of use, number of connections and time between dispensing and first connection.
Economic impact
The UK is the only country where economic assessments are required, and they are highly valued in the assessment process. Both Deprexis and Velibra were originally assessed through a budget impact model as unlikely to deliver cash-releasing savings. In the 2023 re-assessment, with new cost-effectiveness analyses available for Deprexis, NICE concluded that it may be a cost-effective option for people with depression, both less and more severe forms. The economic model captured inputs such as licensing costs, clinicians’ time and computer and internet costs, compared with the costs associated with comparators. The major cost drivers were the effectiveness of the treatment and the follow-up treatment for people whose symptoms were not improved with a digitally enabled therapy. Nonetheless, the assessors also stated that additional evidence of both clinical effectiveness and resource use was needed to reduce uncertainty.
Unmet need
In the UK and France, authorities emphasized the importance of evaluating the unmet need and added social value (e.g. increased access, removal of barriers to access and medical need), while for Germany this was not clearly mentioned. Unmet need and added social value relevance in HTA increase with the focus on value-based decision-making in healthcare; this is particularly true in the case of DTx assessments, particularly in the UK and France. In the UK, Deprexis received commendation for its ability to offer flexible access while concurrently alleviating time and resource burdens on mental health professionals. Notably, NICE recognized Deprexis for its role in overcoming access barriers, including travel constraints and time constraints related to work commitments, and for enabling treatment for patients unable to leave their homes. NICE's assessment highlighted Deprexis as addressing a significant clinical gap, thereby offering promise in meeting unmet needs within the healthcare landscape. Similarly in France, despite the criticism of lack of evidence about societal impacts, Deprexis was assessed as responding to a public health need related to problems of access to mental health services (waiting times, distance, etc.).
Discussion
The aim of this study was to identify and compare HTAs for DTx products in major European markets with established assessment procedures for DTx, namely Germany, the UK and France. Across these countries, various similarities and differences were found between the DTx assessment frameworks; all three countries highly value country-specific data and strong efficacy data, while safety data seem to be of lower consideration. In the UK and France, unmet need and social value added are of importance in the assessment criteria, but it is unclear if this is assessed in Germany.
Assessment frameworks and HTAs
Only a handful of countries have established frameworks for evaluating DTx. This, coupled with the absence of a universally agreed-upon definition of DTx and deficiencies in research and development, clinical trial and regulatory standardization, hampers the widespread adoption and implementation of these technologies.34,35 The recently developed PICOTS-ComTec framework helps to address this issue by providing guidelines to define digital intervention characteristics and identify relevant comparators, therefore aiding the design of primary research and the reporting of relevant evidence. 36
Germany, the UK and France seem to be the most extensive and well-established in terms of product assessment and reimbursement frameworks. At the time of this manuscript's composition, a separate framework for digital nursing applications had been established in Germany, known as DiPA, but no apps had yet completed the approval process. 9 In France, the full implementation of the new PECAN framework is still ongoing at the time of this paper's writing. 10 Its evidence requirements seem to fall within the broader guidelines for medical devices. 11
Country-specific positioning and efficacy outcomes
This analysis revealed a focus from HTA authorities on the positioning of products within country-specific disease landscapes and choice of comparators based on national clinical practice. In other words, it is important to select appropriate populations and comparators when designing and implementing RCTs, systematic reviews and meta-analyses. For manufacturers to achieve this, there is a need to engage with key opinion leaders and have a strong understanding of clinical guidelines.
Early stakeholder engagement is essential to contextualize the product to country-specific care models and efficacy outcome needs. This could be achieved through involvement of users across all steps of clinical research, use of real-world data and consensus or acceptability research. These studies become even more relevant when considering the challenges of more traditional study designs such as RCTs when evaluating DTx. 37 RCTs are challenging for manufacturers to conduct when assessing DTx due to their high cost, the difficulty in finding an appropriate comparator for certain indications and the insufficient account of the complexity of DTx implementation. 37
In addition, early interaction of manufacturers with regulatory bodies is very important prior to manufacturers developing an evidence generation plan. This is the case in Germany, where manufacturers are required to have an initial consultation with the BfArM to discuss their proposed approach. 38
Safety
DTx assessments seem to focus less on safety data related to the use of the product and more on the incorporation of risk-flagging systems within DTx. Therefore, DTx offer an added value to the monitoring of safety and disease progression in patients and, in the future, could be used to provide real-time data related to other health technologies. However, it should be noted that any aspect of a DTx that is used to influence clinical decisions may position the product in a higher regulatory risk class. In light of this, flexibility across regulatory and HTA frameworks becomes essential to ensure adaptability of DTx products to the specific needs of the healthcare system in a timely and cost-efficient manner. Both developers and assessors should consider DTx as integral parts of broader treatment strategies, which include different therapeutic options through a variety of products (pharmaceuticals, medical devices, digital diagnostic tools, etc.). However, at present, multi-product assessment frameworks that address multiple health technologies simultaneously are rare and complicated; therefore, manufacturers often have to navigate multiple regulatory frameworks simultaneously for different product components. 39
Usability and usage
The assessments of DTx focused extensively on usability and usage data; however, the focus on one or the other is different across the countries assessed. The collection and analysis of usability and usage represent an important challenge for both developers and assessors. Usability is a new consideration from an HTA point of view, and it is not clear what type of study designs and outcomes would be the best fit for an appropriate assessment. 40 Due to the nature of DTx, it is essential to show evidence of users’ engagement as proof of a product's efficacy. 41 Moreover, on top of the contextualization of the DTx during development, consensus research could also prove useful here to provide evidence on usability and engagement.
Unmet need and economic impact
Finally, both in the UK and in France, HTA authorities highly valued the impact of DTx on unmet needs and public health policy needs. Indeed, authorities across both countries seem to value the impact of DTx on the social added value (e.g. increased access). DTx are generally seen as technologies that can address health inequities within societies and offer an alternative for disease areas with limited therapeutic options (e.g. mental health). Another area where DTx are thought to prove useful is the financial sustainability of a healthcare system. In the UK, NICE were initially unable to make a conclusion on the cost-effectiveness of Deprexis when considering a budget impact model alone; however, a recent cost-effectiveness model revealed that it may be considered a cost-effective treatment option. 24 Deprexis and Velibra were considered to potentially increase access and free up staff time, respectively. 42 This highlights the importance placed on high-quality economic evidence in the assessment of DTx.
A single European framework for DTx?
Across Europe, national frameworks diverge with regards to assessment criteria and evidence requirements for DTx. This is in line with other studies and publications.3,14 This study's results suggest the need for a more uniform approach internationally and within the EU of assessment criteria and evidence requirements of DTx. This approach might be particularly beneficial if implemented at the EU level. In line with Tarricone et al., we believe that recent regulatory changes such as the European HTA Regulation, the MDR, the European Health Data Space (EHDS) and the revision of pharmaceutical legislation offer an unprecedented window of opportunity. However, none of them addresses this topic specifically and in detail.
The European HTA Regulation refers to the new HTA process coming into force as of 13 January 2025 in the EU, whereby medical products will have to undergo a clinical assessment at the EU level in parallel to the regulatory procedure. As EU members work to develop joint criteria for this new assessment process, joint criteria for the assessment of DTx products should also be considered. At the moment, collaboration between European HTA agency on this topic within the EU HTA regulation framework is exclusively voluntary, and there are no concrete actions foreseen in the near future. 43
The MDR is a new European legal framework that outlines the new regulatory procedures and classification of medical devices in Europe. Currently, this framework does not specifically mention and address DTx products,44,45 including the challenges of changing classes during or after HTA assessment. 44
Similarly, the EHDS is a framework designed to allow for easier and more secure access and sharing of health data between EU members. It could support the evaluation of DTx effectiveness and safety in diverse populations with real-world data. But it can also increase patients' empowerment by giving patients control over the data they generate with DTx products. 46 The potential of this framework is outstanding for DTx products; however, there is no guidance or policy action in place to uncover and utilize the opportunities offered by this regulation for HTA and access.
We also believe that streamlining an EU-wide approach to the assessment and integration of DTx could stimulate digitalization of health and facilitate access to scientifically validated DTx across the union. Moreover, the move towards a single European framework could avoid duplication of work across agencies, accelerate patient access and allow for universal European health coverage of DTx.12,47 This EU framework implementation would need to occur through the harmonization of:
Nomenclature and classification of DTx based on their scope and assessment criteria Standards for quantity, quality and type of evidence needed for assessing DTx Clinical evidence assessment frameworks of DTx based on national mandates from the relevant authorities and HTAs Guidelines for stakeholder engagement across development and assessment phases
Limitations
One limitation of our research is that it focusses only on markets for which DTx were assessed by multiple countries to allow for direct comparison of frameworks. This resulted in a framework analysis that only captured information for Germany, the UK and France; however, it should be noted that other markets also have frameworks in place for the assessment and reimbursement of DTx. In order to conduct a more in-depth assessment of all existing DTx HTA frameworks, comparisons could have been made between similar DTx within the same indication or disease area across all countries with a HTA framework in place for DTx products. It might also have been beneficial to conduct primary research alongside our searches by engaging with relevant stakeholders in order to gain a more comprehensive understanding of the DTx HTA processes in each country.
Our research was also limited by a lack of publicly available information with regard to HTA processes and decisions. In the case of Germany, there was limited availability of context around certain decisions in the assessment process, and therefore assumptions had to be made, such as whether usability was sufficient as per guidelines. Another limitation of this study is due to the ever-changing nature of DTx assessment frameworks. In both France and the UK, the results presented in this paper are based on the old assessment frameworks, which have now been discontinued, and new frameworks have been developed.6,10 Therefore, this research may require an expansion in scope as well as regular updating to account for changes in assessment frameworks,6,10 including shorter times to patient access, as well as more risk classes covered, and as new frameworks are developed.
Conclusion
Our study aimed to identify and analyse HTA assessments for DTx products, as well as to conduct a direct comparison and analysis of HTA assessments for DTx products assessed by multiple countries. Our study found that currently, mental health is the area with the most DTx being developed and assessed, in line with national strategies and health policies. 22 Additionally, it was revealed that there is a lack of consistency in the type of evidence required and criteria used for DTx assessment across Germany, the UK and France, such as the need for country-specific evidence, the inclusion of risk-flagging systems for safety assessment, the focus on usability or usage evidence and the value of unmet need and cost-effectiveness data. Future research should expand on this to compare evidence requirements and assessment criteria for all existing DTx HTA frameworks and across a broader range of indications.
It is essential that HTA agencies and policy makers consider that, as more countries begin developing frameworks for the assessment and reimbursement of DTx, there is a need for harmonization across markets so that assessment criteria and processes are similar. Policy changes such as the EU HTA, the MD and the EHDS Regulations offer a unique opportunity to move towards a single DTx HTA assessment framework at the EU level. This will streamline the market access and reimbursement process for DTx developers, thus stimulating the digitalization of healthcare systems, easing patient access and encouraging development of a globally leading European DTx industry.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076241308704 - Supplemental material for Comparison of health technology assessments for digital therapeutics in Germany, the United Kingdom and France
Supplemental material, sj-docx-1-dhj-10.1177_20552076241308704 for Comparison of health technology assessments for digital therapeutics in Germany, the United Kingdom and France by Emanuele Arcà, Dorothea Heldt and Martina Smith in DIGITAL HEALTH
Supplemental Material
sj-docx-2-dhj-10.1177_20552076241308704 - Supplemental material for Comparison of health technology assessments for digital therapeutics in Germany, the United Kingdom and France
Supplemental material, sj-docx-2-dhj-10.1177_20552076241308704 for Comparison of health technology assessments for digital therapeutics in Germany, the United Kingdom and France by Emanuele Arcà, Dorothea Heldt and Martina Smith in DIGITAL HEALTH
Footnotes
Acknowledgements
The authors thank Christina DuVernay of OPEN Health for editorial assistance.
Contributorship
All authors contributed equally to the study and to the composition of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and patient consent
As this study is a narrative review that involved no patients or patient data, patient consent was not required, and ethical approval was not sought.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Guarantor
EA.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
