Abstract
Objective
Given the well-accepted health benefits, it is important to identify scalable ways to support people with long-term conditions (LTCs) to remain physically active. This systematic review aimed to evaluate the effect of digital tools on the maintenance of physical activity (PA) amongst this population.
Methods
Electronic databases were searched for randomised controlled trials investigating the effect of digital tools on PA maintenance at least three months post-intervention compared with a non-digital control in participants with long-term conditions. Meta-analyses were conducted at post-intervention and longest maintenance timepoint; subgroup analyses explored the effect of the type of control. Prospero registration: CRD42022299967.
Results
Twenty-three studies (n = 5350 participants) were included, with the majority at unclear/high risk of bias. Web-based tools were commonly used (18 studies), 10 monitored PA with a sensor, and digital interventions were often combined with supervised exercise/walks. The overall meta-analysis showed no significant difference in PA between digital interventions compared with any type of control (device-based outcomes: SMD = −0.07, 95% CI [−0.35, 0.21]; self-reported outcomes: SMD = 0.11, 95% CI [−0.01, 0.24]). However, this was moderated by type of control group with subgroup analyses suggesting that the effect of digital interventions on PA surpassed that of an inactive/minimal control at post-intervention (SMD = 0.29) and maintenance timepoints (SMD = 0.24).
Conclusions
This review provides preliminary evidence that interventions incorporating digital tools may be more effective than minimal controls at supporting people with LTCs to maintain PA. Given the quality of the evidence, confidence in these findings is very low and may change with future research.
Keywords
Background
Physical activity (PA) is an important part of maintaining both physical and mental health for people with one or more long-term conditions (LTCs).1,2 Previous systematic reviews and guidelines have reported the benefits of PA for managing people with LTCs and reducing risks of future comorbidity.3,4,5,6 People with LTCs are often less active than the general population3,7,8 and those with more than one LTC, defined as multimorbidity, 9 are even less active. 10 Evidence suggests that PA levels are inversely proportionate to the number of conditions an individual has. 11 In many countries, it is a priority to support this population to be more active, 12 and it is important to identify ways to maintain this PA, given that the clinical and economic benefits depend strongly on how long the effects can be maintained.13,14
Clinician-delivered exercise programmes are recommended for people with LTCs to start to be more active. 15 However, it has been shown that people with diabetes, 16 cardiac disease, 17 chronic obstructive pulmonary disease (COPD) 18 and arthritis19,20 reduce their PA levels after completion of such programmes. One reason for this decline is that interventions designed to support the initiation of PA, such as supervised exercise programmes, typically cease without planned follow-up activities and contact. These interventions are rarely underpinned by behaviour change theory, 21 and therefore, lack the necessary behaviour change components for PA maintenance, such as becoming autonomous and creating new habits. 22 Given that PA benefits accrue over time as people move from initiating changes in PA behaviour to the maintenance of this behaviour change, 23 it is vital that we find effective, sustainable and scalable solutions, that account for the increasing complexity of providing PA for people with multiple LTCs.24,25
Digital technologies have been advocated as a means to provide patient-centred, tailored programmes and deliver evidenced-based behaviour change techniques. 26 These usually require minimal healthcare professional guidance, 27 and may be particularly suitable for PA maintenance as people can continue to harness behaviour change features once a main intervention, such as an exercise programme, ends. 28 Although there is no consensus on how to define PA maintenance,14,23,29,30 both stage-based 30 and time-based definitions have been used.31,32 While the duration varies for time-based definitions, we previously included studies with a follow-up timepoint of at least three months after intervention ends in our scoping review. 7
The aim of the scoping review was to identify and describe the literature on digital tools used for PA maintenance across 18 LTCs. 7 Using a wider lens of knowledge synthesis, the scoping review identified 34 studies that used a range of digital tools.7,33 Systematic review evidence for both PA maintenance5,18,32,34 and digital interventions35–39 for people with LTCs is growing. To date, systematic reviews of digital tools for people with LTCs have often focused on single conditions.34–37 Two reviews including a range of LTCs did not include PA outcomes, 38 or did not focus specifically on the maintenance of PA. 39 This indicated the value of developing a more focused research question using systematic review methodology 40 where we could make judgements on methodological quality of these digital interventions, and combine data across these studies in order to determine the effectiveness of digital tools to support PA maintenance.
Therefore, the aim of this review was to synthesise the evidence from randomised and quasi-randomised controlled trials (RCT) on the effectiveness of digital interventions compared with non-digital or inactive (for example, usual care, waitlist) controls in supporting the maintenance of PA among adults with one or more LTC and explore which intervention design elements are associated with effectiveness.
Objectives:
To investigate the effects of digital interventions in people with ≥1 health condition on objectively and subjectively-measured PA at post-intervention and its maintenance at least three months after the end of the intervention compared with controls. To explore intervention design elements considered key for PA maintenance in effective interventions.
Methods
The protocol for this review was developed a priori according to the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines 41 and registered on Prospero (CRD42022299967).
Study eligibility
This review included full-text articles of randomised controlled trials (RCTs) and quasi-RCTs published in English that included a digital intervention for PA maintenance among people with at least one LTC using the inclusion/exclusion criteria defined in Appendix 1 – eligibility criteria. While the previous scoping review included all study designs to meet its aim of descriptively mapping the evidence, non-randomised study designs were excluded from this systematic review so its conclusions on the effectiveness of the interventions could be based on the best available evidence. 42
Types of participants
This review includes studies with adults ≥18 years of age with at least one LTC, from a pre-defined list of LTCs based on The Quality and Outcomes Framework 43 and the National Institute for Health and Care Excellence PA pathways. 44
Types of interventions
Eligible studies included any digital intervention aimed at increasing or maintaining PA. In line with the World Health Organisation definition of digital health interventions, 33 eligible interventions included a digital element that was accessed via a smartphone, computer or other handheld device. Interventions where the digital tool was not a major component were excluded, judged by team consensus, for example when the participants received more in-person than digital interaction. There were no limits on duration of intervention. There were no limits on setting, geographical location or language of delivery.
Comparisons
Studies were eligible for inclusion if the digital intervention was compared with either an inactive (for example, usual care or waitlist) or non-digital control group (which included minimal controls, such as education only, or active controls, such as supervised exercise). Initially, there were no restrictions placed on comparative control group; however, a change from the protocol, to exclude trials comparing different types of digital interventions, was implemented to focus on understanding how digital interventions compared with non-digital interventions.
Outcome measures
The primary outcome of interest for this review was level of PA, either self-reported (subjective) or device-based (objective), at least three months after the end of the intervention. Measurement of adherence to the intervention or changes in sedentary behaviour were not sufficient to be included. The intention was to include studies where the intervention stopped at least three months prior to the measurement of the PA outcomes; however, some studies were agreed as eligible for inclusion where a lesser version of the intervention continued to the maintenance measurement time point.
Data sources and search methods
Studies identified from our previous scoping review 7 were screened for inclusion in this review. This was supplemented by an updated search of the same databases (Cumulative Index to Nursing and Allied Health Literature (CINAHL), Medline, OVID EMBASE, IEEE Xplore, PsycINFO and Scopus) and using the same search strategy, which was made up of keywords (e.g. digital, PA, maintenance and the list of included LTCs) as well as synonyms of these terms, which were connected using Boolean operators, see Appendix 2. The search was conducted from January 2020 (date of the scoping review search) to August 2023. Reference lists of eligible studies were hand searched.
Data collection and analysis
Study selection process
Search results were imported into Covidence systematic review software 45 for screening. Duplicates were removed. Titles and abstracts were screened by two independent reviewers, and conflicts were reviewed and resolved by the review team. The same process was conducted for full text screening of potentially eligible studies. The reasons for exclusion for all records excluded after full-text assessment were recorded.
Data extraction and management
Study details and data were extracted from each included study independently by two reviewers using a customised form piloted prior to use. The form was used to capture relevant information including study-, participant- and intervention-characteristics, outcomes and results (means and standard deviations). Intervention characteristics of interest included: the theoretical underpinning used to inform intervention development which was extracted based on the 83 theories of behaviour change from the ABC of behaviour change theories 46 ; digital device characteristics, including hardware used and use of sensors to monitor PA. Delivery of PA details included: participant support to embed PA in their daily routines, supervised or autonomous PA, PA delivery via the digital system or non-digitally. Information on human support or healthcare professional communication was extracted, to capture whether health coaching and/or motivational interviewing approaches were delivered via the digital system or non-digitally. Finally, the form captured other non-digital intervention components, behaviour change strategies, such as self-monitoring, setting goals and problem solving; and, maintenance support elements, including follow-up booster sessions, support with becoming autonomous and creating new habits.
Assessment of risk of bias in included studies
Two reviewers independently assessed each included study using the Cochrane risk of bias tool version 1. 47 Blinding of participants was not assessed as this was considered difficult to be obtained due to the active nature of the interventions. For ‘other sources of bias’, studies were assessed for sample size (considered high risk of bias if not powered based on a sample size calculation, or <50 participants per group if a sample size calculation not available) and baseline differences (considered high risk of bias if statistically reported or observed in baseline data). Conflicts were discussed to reach consensus on judgement by the review team. A study was considered at high risk of bias overall if one domain was at high risk, or there were some concerns for multiple domains in a way that substantially lowers confidence in the result. 48
Measures of treatment effect
For calculation of treatment effects, PA outcome data at post-intervention and maintenance timepoints were entered into Cochrane Collaboration Review Manager (RevMan version 5.4). Continuous outcomes were reported as means and standard deviations, with effect sizes presented as standardised mean difference (SMD) and 95% confidence intervals (CI) as PA outcomes were reported across different scales.
Unit of analysis issues
To avoid double counting in studies with multiple intervention arms, the digital intervention was compared only to the minimal intervention control. Where studies reported multiple PA outcomes, both objective and subjective outcomes were extracted for separate meta-analyses. However, if a study reported multiple outcomes for either objective or subjective measures, the measure most representative of overall PA was agreed and extracted. For example, this was usually preferentially total PA (for example, total steps/minutes/metabolic equivalents (METS)), followed by minutes of activity at moderate to vigorous intensity of PA (MVPA).
Dealing with missing data
Where data were not presented in a form that enabled quantitative pooling, estimates were calculated from the published data, as per the method suggested by the Cochrane Collaboration. 49 Where tabulated results were not presented, an attempt was made to extract data from graphs. Where it was not possible to obtain missing data, the study was excluded from the meta-analysis.
Assessment of heterogeneity
Heterogeneity was qualitatively assessed in terms of intervention, participant demographics, outcome measures and follow-up, followed by assessment of statistical heterogeneity using the I2 statistic in RevMan. Given the qualitatively assessed and statistical (>50%) heterogeneity, studies were pooled using a random effects model.
Data synthesis
Data synthesis followed the process recommended by the Cochrane Collaboration.
49
After summarisation of the characteristics of each study, similarity of studies was determined for grouping in two ways:
by control group: inactive/minimal control or an active control by inclusion of intervention design elements that were considered key to support PA maintenance i.e. theoretically informed30,50,51 + building a habit through practice of PA skills in daily life50,51,52 + multiple digital elements that included a wearable to support goal setting, self-monitoring and feedback51,53 + health care professional (HCP) support to build autonomy and self-efficacy through communication approaches54–56 +follow up and support to maintain PA behaviour.
52
Where appropriate, data were pooled for the primary outcome. Meta-analyses were conducted using RevMan; a random effects model was applied to incorporate heterogeneity.
57
Separate meta-analyses were completed for subjective and objective PA outcome data at two time points; immediately post intervention and at the longest maintenance time point (which varied across studies). As the continuous outcomes reported in the included studies were pooled on different scales and units, SMDs and 95% CIs were calculated. For comparisons of the results, pooled effect sizes were categorised according to Cohen's classification; SMD: 0.2-<0.3 as small, 0.3–<0.8 as moderate, >0.8 as large.
58
P-values of <.05 and CIs that excluded null values were considered statistically significant.
Subgroup analysis and investigation of heterogeneity
Subgroup analyses were completed to compare the findings from studies with active controls groups versus those with minimal/ inactive control groups.
Sensitivity analysis
Two sensitivity analyses were planned. The first was to explore the influence of study risk of bias by excluding studies which were judged as having high risk of bias in ≥1 domain. The second was to explore the impact of intervention design elements identified as key to supporting PA maintenance (listed above, in Data synthesis).
Results
The PRISMA Flow diagram (Figure 1) summarises the study selection process. Twenty-three studies were eligible for inclusion in this review,59–81 including n = 18 from the scoping review. 7 Reasons for exclusion of original scoping review studies were: protocol or abstract with no available results paper (n = 9); PA not reported at 3 months post-intervention (n = 4); not an RCT (n = 1); digital control group (n = 2).

PRISMA Flow diagram of study selection process.
Study characteristics
See Table 1 for study characteristics of the 23 studies including 5350 participants (range: 54 to 761 participants per study),59–81 with 49% male and mean age 59.8 years. Studies included participants with the following LTCs: diabetes mellitus (n = 5 RCTs); cardiovascular disease (n = 5 RCTs); osteoarthritis (n = 4 RCTs); COPD (n = 3 RCTs) and obesity (n = 2 RCTs). Four RCTs were not limited to one condition, while in one RCT eligible participants had both knee osteoarthritis and hypertension. Additionally, six RCTs reported additional co-morbidities experienced by included participants. The longest maintenance timepoint/length of follow-up ranged from 3 months to 12 months, with only one study with a 4-year follow-up timepoint (Figure 2). PA was measured using devices in n = 19 studies: Actigraph accelerometer (n = 9), pedometer (n = 5), Sensewear armband (n = 3), Axivity (n = 1) and PA Monitor (n = 1); and measured using self-report in n = 10 studies (paper-based measures, n = 9; semi-structured interview, n = 1).

Intervention duration (months), maintenance elements and follow up. This figure shows the duration of each intervention (red bar), whether a reduced version of the intervention continued into the maintenance period and its duration (pink bar), and the timing of the longest maintenance time point (black bar) for each individual study.
Study characteristics.

Abbreviations: IG, Intervention group; CG, Control group; COPD, Chronic obstructive pulmonary disease; PASE, Physical Activity Scale for the Elderly (PASE); SQUASH, Short QUestionnaire to ASsess Health-enhancing physical activity; IPAQ, International Physical Activity Questionnaire.
Intervention characteristics
Intervention characteristics are summarised in Table 2, with colour coding to identify similarities across studies according to the groupings defined in Data synthesis: by control group; and, by inclusion of intervention design elements that were considered key to support PA maintenance. Interventions frequently supported participants to complete PA independently in their daily lives (n = 11,59–64,67,70,71,75,76), while three interventions included PA delivered via the system asynchronously via exercise videos (n = 2,68,69) or remotely tailored by therapist via the system (n = 1, 80 ). Seven of the interventions comprised in-person exercise delivered either alongside a digital component (n = 5,71,73,74,78,81) or participants completed an in-person exercise intervention prior to continuing with a digital intervention (n = 3,67,72,79). One intervention was exergaming only 77 and another was online education only. 65 Nine studies did not include a supervised exercise programme.59,60,62–64,66,70,75,76
Digital interventions frequently included a website (18/23 studies), while six studies used a custom smartphone application.66,67,71,74,80,81 To assist in self-monitoring, some form of sensing device was used in 43% (10/23) of studies, with three of these67,71,81 utilising the onboard accelerometer within the user's smartphone. Seven interventions used a wearable sensor to objectively monitor PA,66,72,73,76,78–80 with devices ranging from a consumer wearable such as a smartwatch, to a less high-tech connected pedometer, and two systems utilised heart rate data as part of the intervention.79,80 Other methods of self-monitoring and feedback were provided via websites in six studies59–61,63,64,68 but relied on manual entry of the data by participants.
Intervention components summarised per the groupings defined in

Note: The intervention design elements that were considered key to promote PA maintenance are provided in the top row. Within the corresponding columns, the use of
Intervention duration ranged from 6 weeks to 12 months, with 35% (8/23) of studies offering some form of reduced intervention following the initial period, this was either in the form of continued access to the web portal without the supervision of clinicians, or the ability to continue to self-monitor with the use of the sensing device (see Figure 2). For one included study, 65 the digital intervention was intended to be the control group, where participants received only online education via a web platform instead of a comparatively more intensive in-person exercise programme.
Digital interventions were compared with an active non-digital comparator control in 10 studies, while 13 studies had an inactive control, such as usual care or a waitlist, or a minimal control, such as education only.
PA intervention approaches
All of the studies aimed to embed PA into daily life but varied in their approach (Appendix 3 – PA details). Eleven of the 23 studies in this review integrated some behaviour change strategies (e.g. action planning, problem solving, goal setting and self-monitoring with diaries or devices) into an exercise programme.61,65,69,71,73,74,77,81 The exercise programmes ranged from existing community-based exercise referral schemes,73,78 in-person supervised cardiac rehabilitation, 79 supervised walking classes,65,71,81 physiotherapy-led programmes69,74 or unsupervised remote exercise.68,69,74,77,80 Some studies65,81 tapered off the supervised element (e.g. supervised walking) in order to support people to start building a habit of independent walking over the course of the intervention, and others supported people to build new habits into their daily life by using wearable devices and setting goals. Nine studies did not include a supervised exercise programme59,60,62–64,66,70,75,76 but did include a self-management programme60,66 or education elements59,62,63,70,75,76 with advice and practical tools to support people to build PA into their everyday life. Three studies explored the benefits of adding on PA following exit from a supervised exercise programme, either Phase II cardiac rehabilitation61,72 or pulmonary rehabilitation 67 in order to maintain the effects of the initial exercise programme.
PA maintenance approaches
Studies were included if it was initially judged that they had a follow-up at least three months following the end of the main intervention. Of all, 35% (8/23) of the studies included continuing PA elements past the end of the main intervention period60,61,67,68,73,79–81 such as peer-supported email discussion groups, 60 recorded daily PA,61,67,68,73,79,81 feedback and or follow-up via the digital system or from the research team.61,68,79
Communication with a health care professional
The utilisation of telehealth approaches was evident in 10 studies either through asynchronous (7/10) or synchronous communication (6/10) between the participant and the healthcare professional. For the latter, the healthcare professional gave feedback whilst people exercised in the system.64,65,72,73,77–81 Health coaching, defined in this review as communication approaches aimed at changing health behaviour, was evident in very few studies (13% or 3/2366,79,81). When present, this was described as motivational interviewing with only two focusing specifically on PA coaching,66,79 and all were non-digital elements of the intervention.
Theory
Where interventions had multiple components, theoretical underpinnings are presented for the whole study intervention as it was not possible to isolate digital/non-digital components (Table 2 – intervention components table). Seven studies (30%) clearly specified the use of behaviour change theory/taxonomy in the development of the intervention. Five studies (22%) implied that theory or taxonomy was used, but it was unclear which exact theory/taxonomy was used, how it was used, or if it was based on another intervention that suggested use of a theory/taxonomy. One intervention clearly stated that theory/taxonomy was used in intervention development without clarification of the exact theories used. The use of the behaviour change techniques 82 was specified in two interventions.64,78 In the remaining 11 studies (48%), there was either no underpinning theory/taxonomy reported or limited evidence to suggest their use in development of the digital intervention.
Risk of bias
Risk of bias of included studies is summarised in Figures 3 and 4. Two studies77,78 were assessed as having low risk of bias across all domains. Five studies were assessed as having unclear risk of bias in one or more domain. The remaining sixteen studies were assessed as having unclear and/or high risk of bias in one or more domain.

Risk of bias graph: review authors’ judgements about each risk of bias item presented as percentages across all included studies.
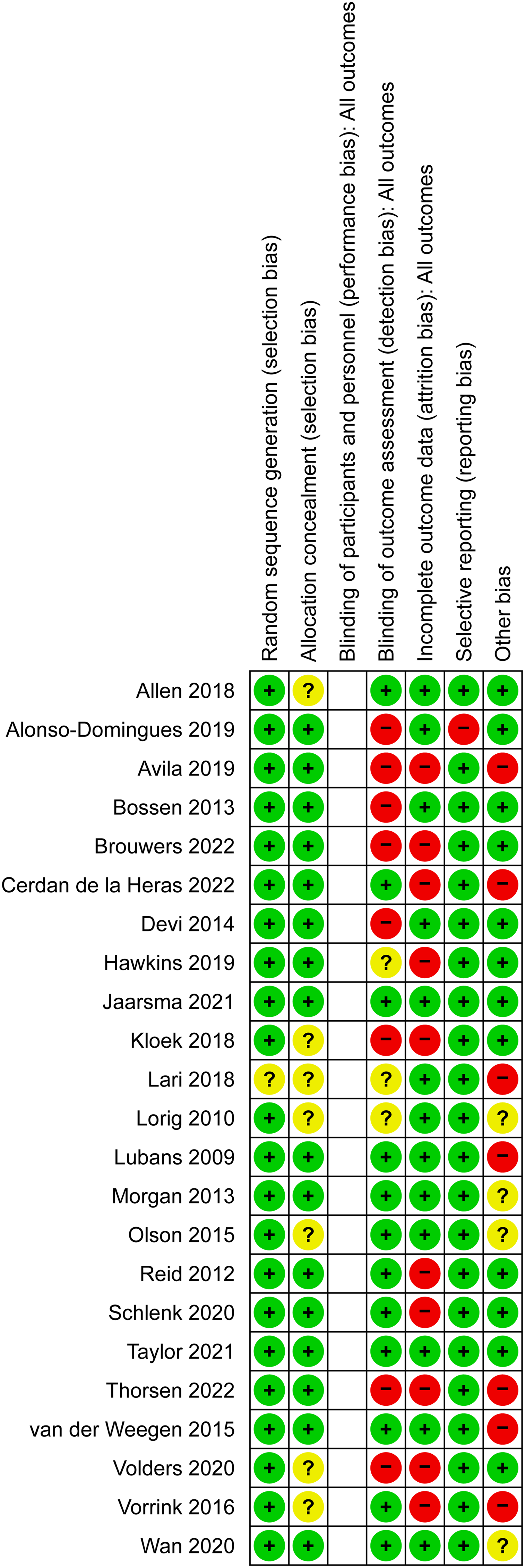
Risk of bias summary: review authors’ judgements about each risk of bias item for each included study.
Effects of interventions
Data were presented in a format available for pooling in 14 studies at post-intervention and 13 studies at the maintenance timepoint. Meta-analyses for the included outcomes are shown in Figures 5–12.

Forest plot of objective PA outcomes at post-intervention.

Forest plot of objective PA outcomes at post-intervention excluding Olson 2015.

Forest plot of subjective PA outcomes at post-intervention.

Forest plot of objective PA outcomes at the maintenance timepoint.

Forest plot of objective PA outcomes at the maintenance timepoint excluding Olson 2015.
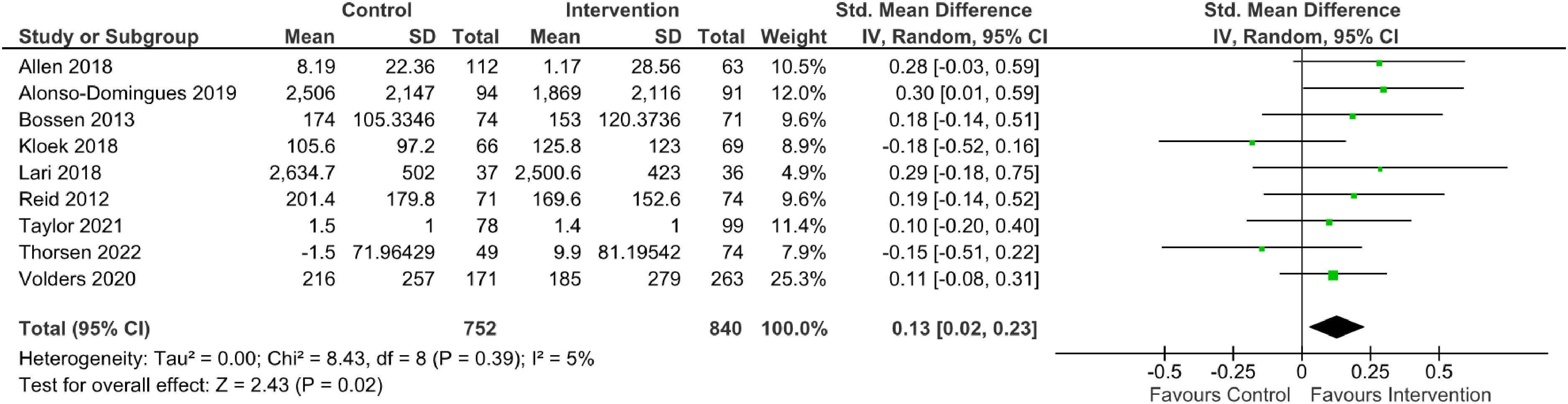
Forest plot of subjective PA outcomes at the maintenance timepoint.

Subgroup analysis by control group at the post-intervention timepoint.

Subgroup analysis by control group at the maintenance timepoint.
Effect of digital health interventions on PA at post-intervention timepoint
Thirteen studies (n = 1904 participants) measuring objective PA were pooled and showed no significant effect post-intervention (SMD = −0.29, 95% CI [−0.68, 0.09], I2 = 189.20, p = .14), Figure 5. Excluding the study with a minimal digital intervention as the intended control comparator, 65 did change the direction of the effect but did not show a significant effect (12 studies, n = 1801 participants, SMD = 0.09, 95% CI [−0.06, 0.23], I2 = 22.77, p = .23), Figure 6. Six studies (n = 1475 participants) measuring subjective PA were pooled and showed no significant effect post-intervention (SMD = 0.10, 95% CI [−0.03, 0.24], I2 = 7.86, p = .13), Figure 7.
Effect of digital health interventions on PA at maintenance timepoint
Fifteen studies (n = 2304 participants) measuring objective PA were pooled and showed no significant effect at the maintenance timepoint (SMD = −0.07, 95% CI [−0.35, 0.21], I2 = 139.04, p = .64), Figure 8. Excluding the study with a minimal digital intervention as the intended control comparator, did change the direction of the effect but did not show a significant effect (14 studies, n = 2210 participants, SMD = 0.11, 95% CI [−0.01, 0.24], I2 = 24.35, p = .08), Figure 9. Nine studies (n = 1592 participants) measuring subjective PA were pooled and showed a small significant effect at the maintenance timepoint (SMD = 0.13, 95% CI [0.02, 0.23], I2 = 8.43, p = .02), Figure 10.
Subgroup analysis
There was a difference between studies that compared digital interventions to active non-digital controls, versus comparison to minimal/inactive controls. Eight studies (n = 1273 participants) with an inactive or minimal control were pooled and showed a small significant effect in favour of the digital intervention at the post-intervention timepoint (SMD = 0.29, 95% CI [0.08, 0.48], I2 = 62%, p = .003). Whereas six studies (n = 982 participants) with an active control showed no significant effect post-intervention (SMD = −0.08, 95% CI [−0.22, 0.07], I2 = 20%, p = 0.32), Figure 11. At the maintenance timepoint, seven studies (n = 1144 participants) with an inactive or minimal control were pooled and showed a small significant effect in favour of the digital intervention at the post-intervention timepoint (SMD = 0.24, 95% CI [0.05, 0.42], I2 = 56%, p = .01). Whereas six studies (n = 664 participants) with an active control showed no significant effect post-intervention (SMD = −0.06, 95% CI [−0.21, 0.9], I2 = 0%, p = .44), Figure 12.
Sensitivity analysis
It was not possible to conduct a sensitivity analysis exploring the influence of risk of bias on effect size for the post-intervention meta-analyses given that all but one study 78 had high risk of bias. Sensitivity analysis removing all high risk of bias studies from the maintenance timepoint meta-analyses slightly increased the effect sizes observed for both objective (SMD = 0.26, 95% CI [0.17, 0.35], n = 586) and subjective (SMD = 0.22, 95% CI [0.06, 0.39], n = 352) PA outcomes. However, we note that only two studies remained in each sensitivity analysis after excluding high-risk studies, limiting the sensitivity analyses to much smaller sample sizes.
Sensitivity analysis related to intervention design elements was not conducted as none of the studies included all the elements that were considered key to support PA maintenance (see colour coding, Table 2). Some included nearly all the elements; for example, van der Weegen 66 was theoretically informed, included motivational interviewing, and wearable to support goal setting, self-monitoring and feedback (post intervention effect: SMD = 0.39, 0.04 to 0.75; maintenance effect, SMD = 0.30, −0.06 to 0.66 compared to an inactive control), but had no follow-on support once the intervention ended. While Thorsen 81 had follow-on support, used a smart phone accelerometer for self-monitoring, goal setting and feedback, rather than a wearable device which would have been preferable (post intervention, SMD = 0.11, −0.17 to 0.39; maintenance effect, SMD = −0.04, −0.40 to 0.33 compared to an active control).
Discussion
Summary of findings
This review is novel given the focus on digital tools to support people with LTCs to transition from initiating changes in PA behaviour to the maintenance of this behaviour change longer-term. To do this we only included studies with a follow-up at least three months after intervention-end. Data were identified and synthesised from 23 RCTs, and 14 of these studies were included in meta-analyses of device-measured and self-reported PA outcomes immediate post-intervention and the longest maintenance timepoint, which was mostly 12 months or less. We observed only one very small effect when pooling studies overall (on subjective PA at the maintenance timepoint), and this seemed to be moderated by the nature of the control groups. Subgroup analyses suggested digital health interventions support people to transition from initiation to the maintenance of PA compared to an inactive or minimal controls; whereas no differences in effect were observed between digital health interventions and active controls. Confidence in these findings is very low as only two of the included studies were at a low risk of bias.
Given the potential of digital tools to provide tailored support, we investigated whether digital tools were used to support people with multiple LTCs or multimorbidity. In our narrative synthesis, we found only a few studies that included more than one LTC, with the majority designed to support people with single LTCs, with metabolic, musculoskeletal and cardiorespiratory being most commonly included. We identified that a range of digital tools were used; the majority were web-based, with around half including a sensor to monitor PA via a smartphone or a wearable device. It is notable that many were combined with non-digital elements, such as supervised exercise classes, supervised walks or exercise referral schemes, delivered either prior to or simultaneously with the digital intervention. Given the promise that digital can reduce or replace human support, our findings showed that in-person support was a common component either within or outside the digital tool; although in some studies the use of the digital tool did replace some of the human support. Few reported use of communication approaches such as health coaching to support people to build autonomy and self-efficacy, and thus support behaviour change. This discussion will explore a number of factors that may have contributed to the observed effects.
Effect size
Subgroup analysis based on the nature of the control group showed a consistent effect in favour of digital health interventions compared to inactive or minimal control groups, both at the end of the intervention (small to moderate effect, 0.29) and the maintenance timepoints (small effect, 0.24). Our short-term and maintenance effects are similar to those observed in a review of the long-term effects of mHealth PA interventions. 83 In contrast to our review Mönninghoff et al., 83 did not show that control group type moderated the effect size; but did show that effects were larger in people at risk or with health conditions. It is not possible to directly compare our results as we only included people with LTCs. Some of the differences in findings may be explained by our inclusion criteria for digital tools; Mönninghoff et al. 83 excluded trials where only a website was used, whereas these were a common component of interventions in our review, and perhaps are less effective than interventions with mobile digital components that enable people with LTCs to actively self-monitor their PA levels. Wearable devices have been shown to improve PA levels when combined with exercise prescription or advice in the maintenance phase of cardiac rehab. 84 Another similar review, focusing on digital health interventions for PA for people with LTCs, 39 did not exclude studies without follow up of at least three months and searched for RCTs on eight of the 18 LTCs that we included. This review demonstrated similar small effects in favour of digital interventions compared to minimal or usual care controls. However, little detail was given on the usual care controls and so it is difficult to compare to our findings where the control group moderated our meta-analysis results.
Differences dependent on the control group
Forty three percent of included studies compared a digital intervention to an active (either supervised or unsupervised exercise) control group, and in the meta-analysis we observed no difference in effect. Although the variance values are low in these analyses, there was variability in the digital interventions, two studies included digital components only, whereas the other eight studies included a mix of digital and non-digital components such as starting with supervised exercise and then moving to unsupervised exercise supported by digital tools. These findings therefore provide preliminary support that digital tools are comparable to in-person supervised exercise, potentially reducing human resources and making intervention delivery more scalable. The need for in-person support as part of digital interventions was debated in our scoping review. 7 Similar recent reviews have not reported whether trials included this key information. 39 Other authors have argued that scalable digital interventions are those that leverage automated components without any of these human-to-human interactions; and showed no difference in positive effects between scalable and non-scalable mHealth interventions. 83 We were unable to make any judgements on this in this review, however, it may be important for people with health conditions to receive in-person support, to give them the skills and confidence to then continue to be physically active independently. Communication approaches such as motivational interviewing have been associated with the maintenance of behaviour change by facilitating self-regulation and positive motivation,50,85 and Ref. 66 in our review showed that a website used alongside health coaching was more effective than either alone. Although not a common feature in this review (n = 3 studies), it would be interesting in future studies to explore the possibility of integrating this type of communication within the digital tool.
This review did not consider the impact of participant characteristics on response to minimal controls. Previous PA maintenance reviews have found that motivated, well-educated, younger and white participants respond to minimal controls, 32 suggesting that intervention dose could be matched to participant characteristics and titrated depending on their initial response to minimal interventions.
Intervention design elements
While this systematic review aimed to evaluate the effectiveness of digital interventions in supporting the maintenance of PA behaviour change, the studies identified were mainly designed to support participants to initiate PA and had a long-term follow-up measurement of PA, rather than being specifically designed to support long-term maintenance of PA. Only 35% of the included interventions had continuing PA elements past the end of the full intervention period, despite NICE behaviour change guidelines 52 recommending that people receive feedback and monitoring up to one year after the end of an intervention to make sure people can get help to address relapses in behaviour. Despite the opportunity of digital interventions to continue indefinitely, very few of the studies included or reported on these continuing intervention components. A range of strategies have been reported in other reviews such as phone calls, devices, home visits, logbooks, web-based instructions, written material and visual instructions. 5 However, even with such behaviour change support their effectiveness regresses to previous levels once the intervention is completed. 28 This means very little is known about how to support people to maintain PA behaviour long-term. Existing literature in this area demonstrates a lack of reporting of maintenance outcomes, small improvements at follow-up that often diminish over time, or no significant differences in PA maintenance outcomes.28,56,83
Just over a third of studies clearly identified theory with even fewer explicitly naming specific behaviour change techniques, 82 and although 50% listed some behaviour change strategies, for example, action planning, goal setting etc., these were often linked to generic self-management programmes rather than embedded within the exercise classes or linked with the use of the digital tool. In addition to the limited use of theory in behaviour change, it is notable that no single digital intervention utilised all the features described in Table 2. The use of approaches including biofeedback, remote supervision, goal setting, self-monitoring, education, reminders, gamification and social support are regularly incorporated into the design of digital health interventions which offer new opportunities to promote behaviour change. 86 When examining PA in particular, design features such as reminders in the form of text messaging and personalisation have shown increased effectiveness in improving PA in healthy adults, 87 with gamification also shown to have an effect in encouraging maintenance of PA levels during pandemic-related lockdowns. 88 In the clinical cohort, digital interventions which incorporate a variety of passive and interactive features have been shown to be more effective for behaviour change in the management of diabetes, although the effectiveness of more interactive features diminishes over time. 89 Whilst it is unclear what the optimal design features are from a long-term PA maintenance perspective, the opportunity to implement PA interventions on a long-term basis with minimal resource burden, and the effect of this, is one that should be explored further.
Multimorbidity management
Participants with metabolic, musculoskeletal and cardiorespiratory LTCs were most commonly included, and the majority of studies focused on these single conditions, despite the fact that the demographic profiles in some studies indicated multimorbidity. We know that experiencing multiple health conditions is common, with increasing numbers of conditions associated with lower PA levels in individuals. 11 We also know that exercise and remaining active is recommended for people across different health conditions.1,90 This indicates that targeting these interventions for people with multiple health conditions might be particularly feasible and effective, as PA interventions are able to improve health across multiple different LTCs. 11 Two recent reviews have concluded that being active is safe for people with multiple health conditions.39,91 Both reviews recognised that future studies should focus on interventions tailored to individuals’ goals and preferences in order to improve effect estimates and adherence. From a clinical perspective, it may be necessary to tailor interventions to an individual as the presence of other conditions may lower their baseline levels of physical function and require adaptions, such as lower PA intensity for people with cardiac conditions or more time and support to overcome higher levels of pain, fear of pain and disability. 11 In our previous work we have measured people's PA over seven days and used tools such as a six-minute walk test, and a 10-min self-efficacy walk to establish baseline functional ability and confidence levels in people with low back pain or COPD92–94 which allowed us to tailor our initial PA intervention goals. This worked well for people with LTC who were supported by the clinician to set and progress their PA goals, particularly in those with other health conditions, very low levels of baseline PA, physical function and confidence to be active. 94 Harnessing digital tools to automate these processes may be valuable in the future.
Strengths
A strength of this systematic review is the inclusion of 18 different LTCs and the focus on trials with outcomes at least three months after the main intervention ended. This allowed us to identify a significant body of evidence in order to understand the impact of digital tools across this wide range of conditions, and to explore whether the digital tools were designed to manage PA maintenance in people with multiple health conditions. Other strengths were the use of two authors to screen studies, extract data and complete the risk of bias assessments.
Limitations
In the title/abstract screening stage we excluded studies that did not have follow-up at least three months after the end of the main intervention. It is therefore possible that we excluded well designed theoretically-informed PA maintenance feasibility or pilot trials for this reason. Any updates to our search dates will identify these studies once they report on future definitive trials with maintenance time point outcomes measures. In terms of interpretation of our findings, a limitation is the heterogeneity of the interventions across the studies. This is indicated in the wide CIs around our effect estimates and the high I2 values in the overall comparisons, and in the subgroup analyses versus an inactive/minimal control. Given that few studies included a digital-only group, any positive results can only be attributed to combined effects of digital and non-digital interventions and we do not know the individual contribution of the digital element to these results. Finally, the quality of evidence was low, suggesting that the true effect of the interventions may differ from the ones reported in this systematic review. We were limited in our exploration of the factors that could have explained our meta-analysis findings. However, as more studies are published a wider number of factors could be explored. For example, we did not explore the impact of intervention design elements as none of the studies included all the elements that were considered key to support PA maintenance. We could consider a meta-regression in a future review, like that planned in a systematic review of PA referral scheme components. 95 A meta-regression could be conducted if there are sufficient studies available for each component, to test their impact on effect estimates. We did not extract adherence with the digital tools and therefore could not explore this impact on our findings. A previous review has demonstrated a weak but consistent positive association between engagement with a PA digital health intervention and PA outcomes, 96 and we would recommend this is included in future reviews in this area.
Conclusion
This review provides some preliminary evidence that interventions incorporating digital tools may be more effective than minimal controls at supporting people with LTCs to increase and maintain PA levels up to 12 months after the main intervention ends. Notably, digital tools were often used within multicomponent interventions, either simultaneously with or following in-person exercise interventions. Future research should explore effective intervention components and consider the interplay of the need for human support versus the scalability of digital interventions. Confidence in the findings of this review is very low given the quality of the evidence and we expect that future research will change these findings.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076241299864 - Supplemental material for The effectiveness of digital tools to maintain physical activity among people with a long-term condition(s): A systematic review and meta-analysis
Supplemental material, sj-docx-1-dhj-10.1177_20552076241299864 for The effectiveness of digital tools to maintain physical activity among people with a long-term condition(s): A systematic review and meta-analysis by Sarah Howes, Aoife Stephenson, Chloe Grimmett, Rob Argent, Paul Clarkson, Ameera Khan, Emily Lait, Leah Rose McDonough, Giorgia Tanner and Suzanne M McDonough in DIGITAL HEALTH
Footnotes
Acknowledgements
We would like to thank RCSI librarians, Paul Murphy, Andrew Simpson and Killian Walsh, for their assistance in the conduct of the searches.
Contributorship
SMD and PC researched literature and conceived the study. All authors were involved in data screening and extraction. SMD and SH conducted data analysis and wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and consent
As this was a systematic review, ethical approval and consent were not required.
Funding
AS's time this was funded by the Health Research Board and ARTHRITIS IRELAND under the HRCI-HRB Joint Funding Scheme HRCI-HRB-2022-010. PC's time was supported by the National Institute for Health Research Applied Research Collaboration Wessex, United Kingdom
Guarantor
SMcD.
Supplemental Material
All supplemental material mentioned in the text is available in the online version of the journal.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
