Abstract
Background
Helping people recover from aphasia is among the top 10 research priorities relating to life after stroke.
Objective
We aimed to evaluate the feasibility of dubbing techniques (using newly developed software) for post-stroke aphasia therapy and explore its potential efficacy.
Methods
Randomised, crossover, interventional, feasibility trial that included patients with chronic post-stroke non-fluent aphasia. The intervention consisted of an individualised programme (16 sessions; 8 weeks) based on dubbing words and sentences progressively adapted to the severity of the aphasia. Patients were allocated to groups that underwent therapy within the first 3 months, or between 3 and 6 months from inclusion, each group serving as the control during the non-therapy periods. Outcomes were the pre-post differences in the Communicative Activity Log, the Boston Diagnostic Aphasia Examination, the General Health Questionnaire-12, the Stroke Aphasia Quality of Life Scale, and the Western Aphasia Battery Revised, administered by psychologists blinded to the patients’ allocation.
Results
Recruitment was limited due to COVID-19 and prematurely stopped because of funding coming to an end. A total of 23 patients were randomised, 20 of whom completed the study (1 withdrew consent, and 2 dropped out). The adherence rate to the allocated group was 95.3%. No statistically significant differences were found in any of the outcomes; however, 17 (85%) patients reported subjective improvements in communication skills.
Conclusions
This trial shows the feasibility of dubbing therapy (using dedicated software) for patients with post-stroke non-fluent aphasia. Although it lacks statistical power, certain effects on language and communication cannot be ignored.
Introduction
Stroke is a highly prevalent and devastating disease. According to the World Stroke Organization, stroke is the second-leading cause of death, and the third-leading cause of death and disability combined worldwide. 1 It is estimated that over 12.2 million new strokes occur each year and that, globally, 1 in 4 people over 25 years of age will experience a stroke in their lifetime. 1
Aphasia is one of the most devastating sequelae after stroke occurring in 15%-42% of stroke survivors, 2 affecting all or certain language modalities, including the production and understanding of speech, reading, and writing. Although the definition of aphasia is still debated, a recently published consensus underscored its relevance as a communication disorder beyond language impairment. 3 Aphasia is a communication disability that affects daily functioning, social inclusion, and quality of life (QoL),2,3 so recovering functional communication is a strong predictor of social re-integration. 4
A consensus among survivors, caregivers, and health professionals concluded that helping people recover from aphasia was one of the top 10 research priorities related to life after stroke. 5 Current international guidelines recommend speech and language therapy (SLT) for stroke survivors; however, there are no universal standards for its regimen (intensity, dosage, duration), delivery model (group, one-to-one, volunteer, computer-facilitated), approach, or outcome measurements in patients with post-stroke aphasia.6,7 High dropout rates (up to 30%) pose challenges, possibly due to the use of child-like materials in many SLTs. 7 Thus, adult stroke survivors might become frustrated with feeling that their needs and preferences are not being covered. 8 Computer-based technologies (e.g., mobile apps, video games, virtual reality) show promise in engaging therapy and increased spontaneous self-expression.8–10 Factors underlying language recovery, such as visual cue integration, self-performance assessment, motor coordination, intonation, emotion, and motivation, are supported by the latest discoveries in neuroscience as separate components.11–14 The DUbbing Language-therapy CINEma-based in Aphasia post-Stroke (DULCINEA) project aims to contribute to increasing patients’ quality of life by developing a computer-based therapy consisting of dubbing scenes from popular TV series. Dubbing is fun and stimulating and comprises key components of speech. Specifically, dubbing scenes that are familiar to the patient could capitalise on the effect of repetition and familiarity on word recall.15,16 Moreover, when dubbing, actors articulate words within a given emotional context by paying attention to an audio-visual input. Therefore, dubbing sentences from daily situations could improve the patient's functional communication. We aimed to evaluate the feasibility of dubbing therapy and explore its potential efficacy in a clinical trial of patients with post-stroke non-fluent aphasia.
Methods
Study design
The DULCINEA trial was a randomised, crossover, interventional feasibility study. The study followed the CONSORT guidelines for feasibility of randomised studies 17 and the Standard Protocol Items Recommendations for Interventional Trials 2013 declaration. 18 The clinical trial protocol was registered in Clinicaltrials.gov (NCT04289493).
Full design details of this clinical trial are available elsewhere. 19 Briefly, the primary inclusion criteria were as follows: a) non-fluent aphasia due to ischaemic stroke in the left hemisphere without neuroimaging evidence of lesions in the right hemisphere; b) previously completed standard programme of conventional SLT; c) severely restricted language, defined as poor repetition even for single words and moderately preserved language comprehension (i.e., under the 70th and 15th percentiles in Repetition and Listening Comprehension, respectively, as an average score in word comprehension, command, and complex ideational material subscales) on the Boston Diagnostic Aphasia Examination (BDAE) 20 ; and d) signed informed consent. Beyond the standard informed consent, an aphasia-friendly information sheet was provided to ensure the patient's understanding of the study procedures. The local ethics committee approved both documents. Exclusion criteria were a) any clinical condition or other characteristic precluding appropriate follow-up and b) simultaneous participation in any therapeutic trial assessing post-stroke recovery.
This study had a crossover design with 2 treatment sequences (treatment-washout/waitlist-treatment) and 2 phases (Phase 1 and Phase 2). Using a computerised random number generator provided by an independent statistician, patients were randomly allocated (1:1) to one of the following groups:
Group 1: therapy to be started within the first 3 months of the patients’ inclusion in the study, followed by a subsequent period of 3 months without therapy (washout period), thus serving as controls for the study's second phase. Group 2: therapy to be started between 3 and 6 months after the patients’ inclusion in the study. They did not receive any SLT during the first 3 months, thus serving as controls for the study's first phase (waitlist controls) and as the active intervention group in the second phase.
The cross-over design allows this experimental therapy to be offered to all the patients who participate in the trial, therefore reducing the possible rate of refusal to be enrolled in the control group and allows to explore the stability of the effects.
Intervention
The dubbing programme included a baseline session for training and sixteen 40-min sessions of therapy, distributed to twice a week.
The DULCINEA software:
We used ad hoc-designed software. The DULCINEA software is an experimental dubbing program that explores the utility of dubbing techniques as a therapeutic tool to integrate essential features of language production and functional communication. It allows the introduction of scenes reflecting everyday situations, which in turn contain each of the words intended to be dubbed by the patient. This software is registered at https://www.safecreative.org/work/2312216447769-codigo-software-dulcinea-?0 and the user manual (Spanish language) is available at https://www.safecreative.org/work/2312216447752-manual-de-uso-proyecto-dulcinea-?1
In brief, for each target word or sentence, approximately 3 to 4 clips lasting 5 to 15 s each were employed, following the principle of incremental learning (least complex first). Once the scene has been prepared, and the necessary devices (headphones and microphone) have been set up, the work session begins. Initially, the scene is previewed, while the dubbing actor puts the patient in context and narrates what is happening in the scene, so that the patient correctly understands the emotion that is intended to be transmitted when intoning the target word. The speech therapist, in parallel, offers the necessary aids to the patient to articulate the word correctly, in addition to using different tools offered by the software, such as lip zoom and speed of movement, so that the patient can better capture the orolingual movements produced in the emission. In addition, this software offers auditory and visual feedback with a bar that marks the time the clip lasts, a bold outline of the bar segment that identifies the specific moment in which the target word is found/emitted (Figure 1). All these aids are ultimately intended to ensure or increase the amount of feedback the patient receives both before and after giving a response, assuming that feedback from audio-vocal integration and motor control may be impaired in people with aphasia. 21 Thus, we guarantee the anticipation of the expression of the target word to be articulated by means of visual or auditory aids, and finally we provide feedback so that the patient can check how he/she has performed the emission and can carry out the different subsequent corrections. Once we consider that the patient is ready, we proceed with the recording. At this point, the aids related to lip zoom and speed of movement disappear. As in rehearsals, the word is framed when it is to be uttered, to facilitate synchrony. In addition, and only at the time of recording, a triple beep, which becomes more intense as the time when the patient should pronounce the target word approaches, also appears. Once recorded, the exercise can be listened to by the patient, and together with the therapist, to decide whether to discard or retry the exercise because it may not have been performed correctly, or whether to send it finally for further evaluation. After confirming that the exercise has been recorded correctly, it is sent to the speech therapist, who will be able to carry out the evaluation of the exercise, assigning the following characteristics for each of the words in each exercise: (motor coordination-dysarthria (severe/moderate/mild); intonation (no intonation/intonation); fragmentation (split/joint); synchrony (desynchronized/synchronized) and repetition (repeats/repeats with difficulty/does not repeat). As far as possible, we aim for the patient to score correctly on all the evaluated variables; however, for a target word to be considered acquired, it does not necessarily need to score positively in all of them. Nevertheless, to consider the word acquired, the patient must produce it in a single utterance (not fragmented), articulate it correctly, meaning fully intelligible and without any phonetic distortion, as well as with the appropriate intonation, with synchrony being less critical.

Screenshot of the tools included in the DULCINEA software. Footnote: 1.
The software allows this data to be recorded and the evolution and progress of each patient to be displayed, as well as the words worked on by each patient and the order of presentation.
The DULCINEA software also allows the patient to perform reinforcement exercises at home without the assistance of the speech therapist or the voice actor. A family member facilitates correct access to the program, thereby ensuring optimal delivery of the exercises.
Target words
The words included in the study were selected carefully. Initially, a survey was conducted with aphasia patients and caregivers asking them about everyday words and short phrases that would be useful for their communication. These words were classified according to the level of difficulty, considering the length of the word (monosyllabic, disyllabic, more than two syllables and phrases) and according to their semantic category. Based on the survey responses, where 216 words were recorded, we finally compiled a list of 183 words or short sentences, from which the patients and their relatives jointly selected a limited number of words to include in their treatment. The speech therapist prioritized those most suitable for progressive introduction based on the individual's ability to produce certain phonemes, the simplicity of the word (e.g., mono- vs. disyllabic), and the frequency of use of the word. As a means of facilitating participant habituation to the study procedures, the speech therapist selected words of relatively low articulatory difficulty and high frequency of use as initial targets. As the intervention progressed, the complexity of the target words was gradually increased to improve phonetic repertoire and articulatory accuracy accordingly. When a patient could articulate individual words correctly, the word combinations, formulaic phrases (e.g., “thank you very much”), and more complex words or phrases were introduced.
Outcomes
Primary outcomes were the scores in the Communicative Activity Log (CAL) questionnaire, 22 a validated measure for evaluating functional communication that includes 2 measurements (amount and quality of communication), with higher scores reflecting better performance. Secondary outcomes were the BDAE 20 and additional tests following the Research Outcome Measurement in Aphasia consensus statement 23 for aphasia treatment research: the General Health Questionnaire (GHQ)-12,24,25 the Stroke and Aphasia Quality of Life Scale-39 (SAQOL-39) 26 and the Western Aphasia Battery Revised (WAB-R), in particular the aphasia quotient (AQ), as the summary score that indicates overall severity of language impairment. In addition, we included the Stroke Aphasic Depression Questionnaire (SADQ)-10 27 to detect depressed mood. The GHQ-12 is a screening instrument designed to evaluate psychological well-being, already validated for use in Spain, with higher scores indicating poorer well-being, and with a maximum score of 12. 24 The SAQOL-39 evaluates QoL divided into 4 domains: physical, psychosocial, communication, and energy. SAQOL-39 scores range from 1 to 5, with higher scores indicating better QoL. The SADQ-10 27 has a maximum score of 30, with higher scores indicating more severe symptoms of depression. All the tests and questionnaires referenced above were purchased legally or downloaded from published articles. We used versions validated in Spanish samples except for two: CAL 22 (the original sample was in German) and the (SADQ)-10 27 which was validated in the UK. For this latter questionnaire we used a Spanish translation of developed by Investen/ISCIII Centro Español para los cuidados en salud basados en la evidencia, with permission from the authors of the original questionnaire. We considered that both questionnaires were validated in samples with a similar culture to the Spanish; however, the interpretation of their results should be taken cautiously in case there were significant cultural differences.
Psychologists, blinded to allocation, assessed all tests. Due to COVID-19 restrictions, tests were conducted online in 2 sessions to prevent fatigue. To aid participants with severe expressive aphasia, questions and answers were displayed and pointing was permitted. Family members could assist in connecting and understanding questions. Additionally, a Likert scale survey was administered to relatives at the study's end, assessing the frequency of trained word usage in patients’ spontaneous speech: very often, frequently, sometimes, rarely, or hardly ever. The specific question was: Has the patient used any of the trained words in his/her daily life?
Data management and monitoring body
The data were prospectively included in a study-specific database developed with REDcap software (REDCap 8.2.0, 2020 Vanderbilt University). All data management followed the principles of the European regulations for biomedical research ensuring confidentiality. Monitoring was conducted by dedicated personnel at the Clinical Trials Unit at La Paz University Hospital.
Sample size estimates
According to a previous clinical feasibility trial on a different SLT in patients with non-fluent post-stroke aphasia, 28 we estimated a sample size of 54 patients for 80% power and a 0.05 2-sided significance level to detect a difference in the CAL evaluation. 22
Statistical analyses
The statistical analyses were conducted by the biostatistics platform at the Hospital La Paz Research Institute (IdiPAZ), using R software. 29 The data are shown as absolute and relative frequencies for categorical variables or median and interquartile ranges (IQR) for numeric variables. Data were first compared using a chi-squared test, Student's t-test, or the Mann-Whitney U test, as appropriate. To evaluate the benefit of DULCINEA therapy through the CAL and BDAE questionnaires, we used mixed effects linear regression models, a statistical model particularly useful for longitudinal studies with repeated measures. Due to their advantage in dealing with missing values, mixed effects models are usually preferred over other approaches, allowing for an adjustment of the treatment effects by the baseline values and the period effect in crossover trials. Given that this study has a crossover design with 2 treatment sequences (treatment-washout/waitlist-treatment) and 2 phases (Phase 1 and Phase 2) with a baseline evaluation of all participants, we considered treatment and phase as principal fixed effects, the treatment × phase as an interaction effect, and the patient nested in the treatment sequence as a random effect. For pairwise post hoc comparisons, we used the Bonferroni test. Lastly, for the calculation of the sample size needed for a definitive trial, we employed nQuery Advisor software (Statistical Solutions Ltd, Cork, Ireland). We used a per-protocol analysis for the analysis of the principal outcome variables and an intention-to-treat analysis for secondary outcomes.
Availability of data and materials
Raw data will be deposited in a public repository (https://repositoriosaludmadrid.es/).
Results
Recruitment began in January 2021 and continued until February 2023. A total of 44 patients signed informed consents; however, 21 of these were excluded at the screening visit. Figure 2 shows the study flow diagram according to CONSORT guidelines.17,30 Table 1 presents the demographic and clinical data of the 23 patients with post-stroke aphasia ultimately included in the trial. Eight (34.8%) were women, with a median age of 65 years. Only 1 patient (allocated to group 2) was illiterate. A total of 12 (52%) patients reported never using computers, 11 (47%) never using electronic tablets, and 5 (21%) never using smartphones. The primary family member/accompanying person assisting the patient in the completion of the tests was a spouse in 9 (39%) cases, offspring in 6 (26%), a sibling in 1 (4%), and an informal caregiver in another (4%). Twelve (52%) patients reported gestural help for daily-life communication, and only 1 patient (allocated to group 2) used electronic devices to support communication. No significant differences in baseline BDAE scores were found between the groups (Table 1).

Flow diagram.
Baseline characteristics (intention to treat population).

BDAE, Boston Diagnostic Aphasia Examination; SLT, speech and language therapy.
*Daily need for help with any of the following activities: toilet use, feeding, transfers, walking, dressing or bathing.
Primary outcomes
Figure 3 shows the distribution of CAL scores at baseline and at follow-up visits. No significant differences were found at the end of phase 1 (3-month follow-up) between group 1 (at the end of the DULCINEA therapy) and group 2 (waiting-list controls) (Table 2). The before-and-after therapy analysis showed no significant differences in either of the groups: group 1, median 93.5 (IQR 77.5; 96.5) at baseline and median 92 (IQR 86.5; 124.75) at the end of therapy (p = .185); group 2, 91.5 (79.5; 105.5) at baseline and 106 (84.5, 113.25) at the end of therapy (p = .092). A trend to higher CAL scores after treatment was observed in group 2 (Figure 4).

CAL scores. Footnote: Dark grey, group 1 (start of treatment after baseline visit; washout period between the 3- and 6-month visits); light grey, group 2 (start of treatment after the 3-month visit, serving as waiting list during the first 3 months). Footnote: Mixed effects linear regression model for crossover trial analysis p = .092.
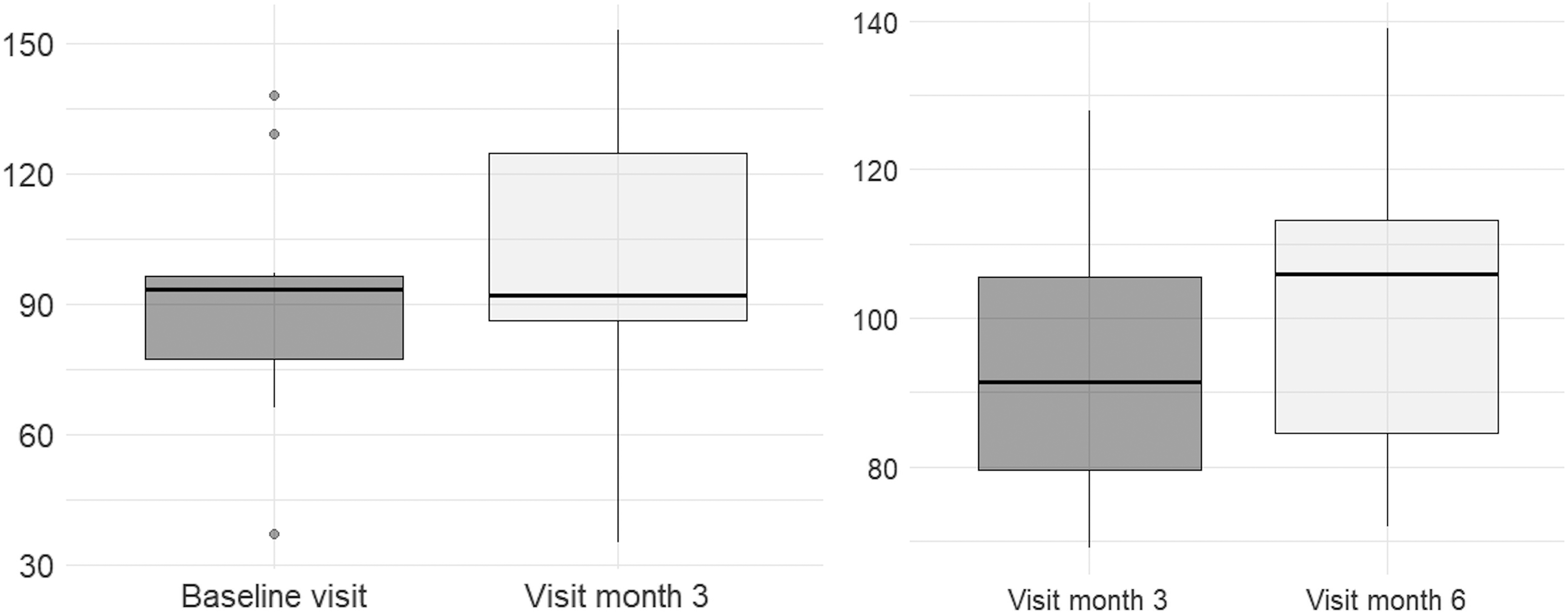
Before and after analysis of CAL scores
Outcome analysis at the 3-month evaluation (group 1 end of active treatment; group 2 waiting-list).
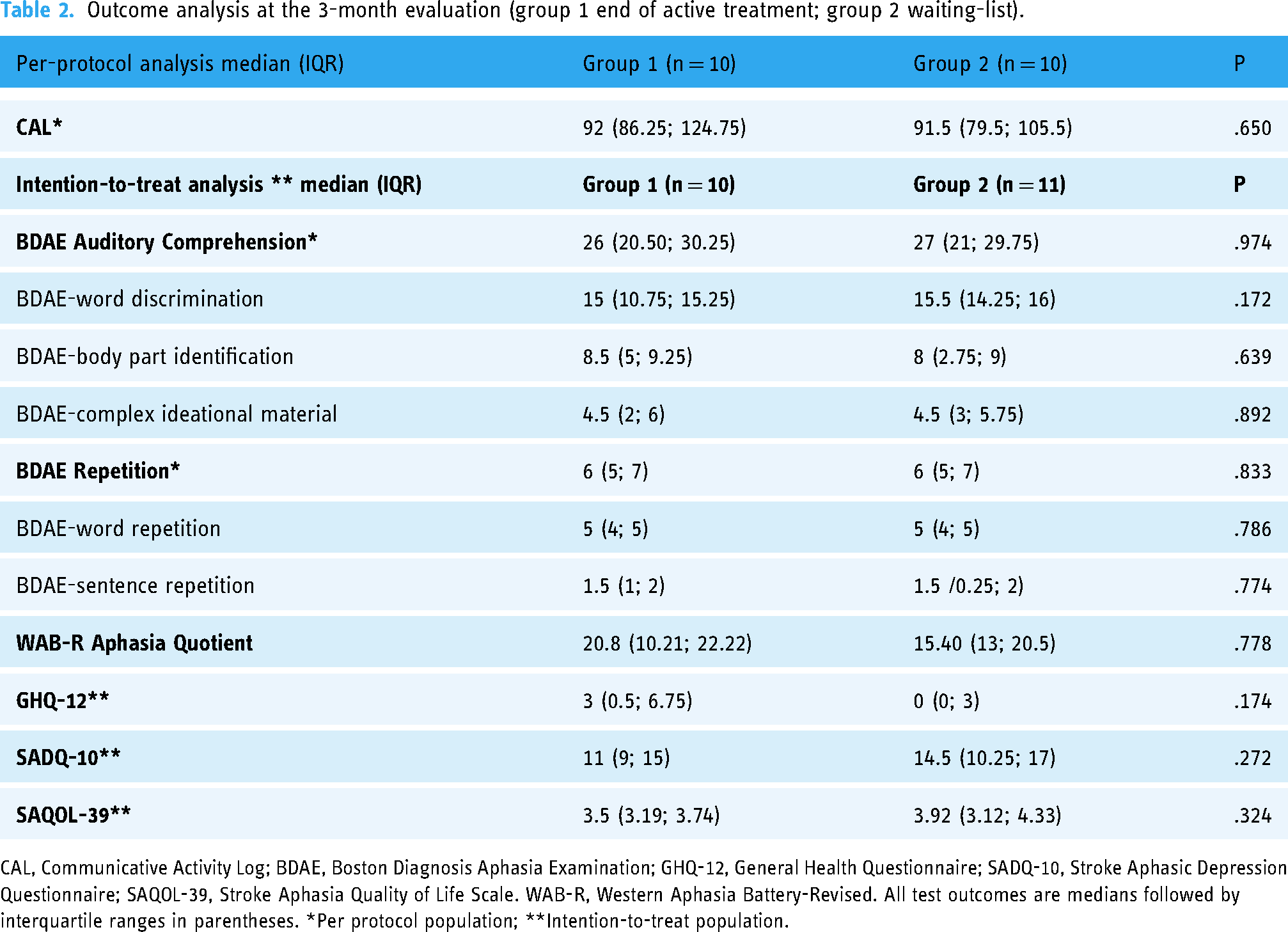
CAL, Communicative Activity Log; BDAE, Boston Diagnosis Aphasia Examination; GHQ-12, General Health Questionnaire; SADQ-10, Stroke Aphasic Depression Questionnaire; SAQOL-39, Stroke Aphasia Quality of Life Scale. WAB-R, Western Aphasia Battery-Revised. All test outcomes are medians followed by interquartile ranges in parentheses. *Per protocol population; **Intention-to-treat population.
Secondary endpoints
Similarly, no statistically significant differences were found in the analysis of total scores for the BDAE Auditory Comprehension, either in BDAE repetition features or in any of their components (word discrimination, body part identification, complex ideational material, word repetition, or sentence repetition) at the end of phase 1 in group 1 (end of the DULCINEA therapy) and group 2 (waiting list controls) (Table 2). Again, there were no statistically significant differences in the scores of the GHQ-12, SAQD-10, SAQOL-39, AQ or WAB-R (Table 2 and Figure 5).
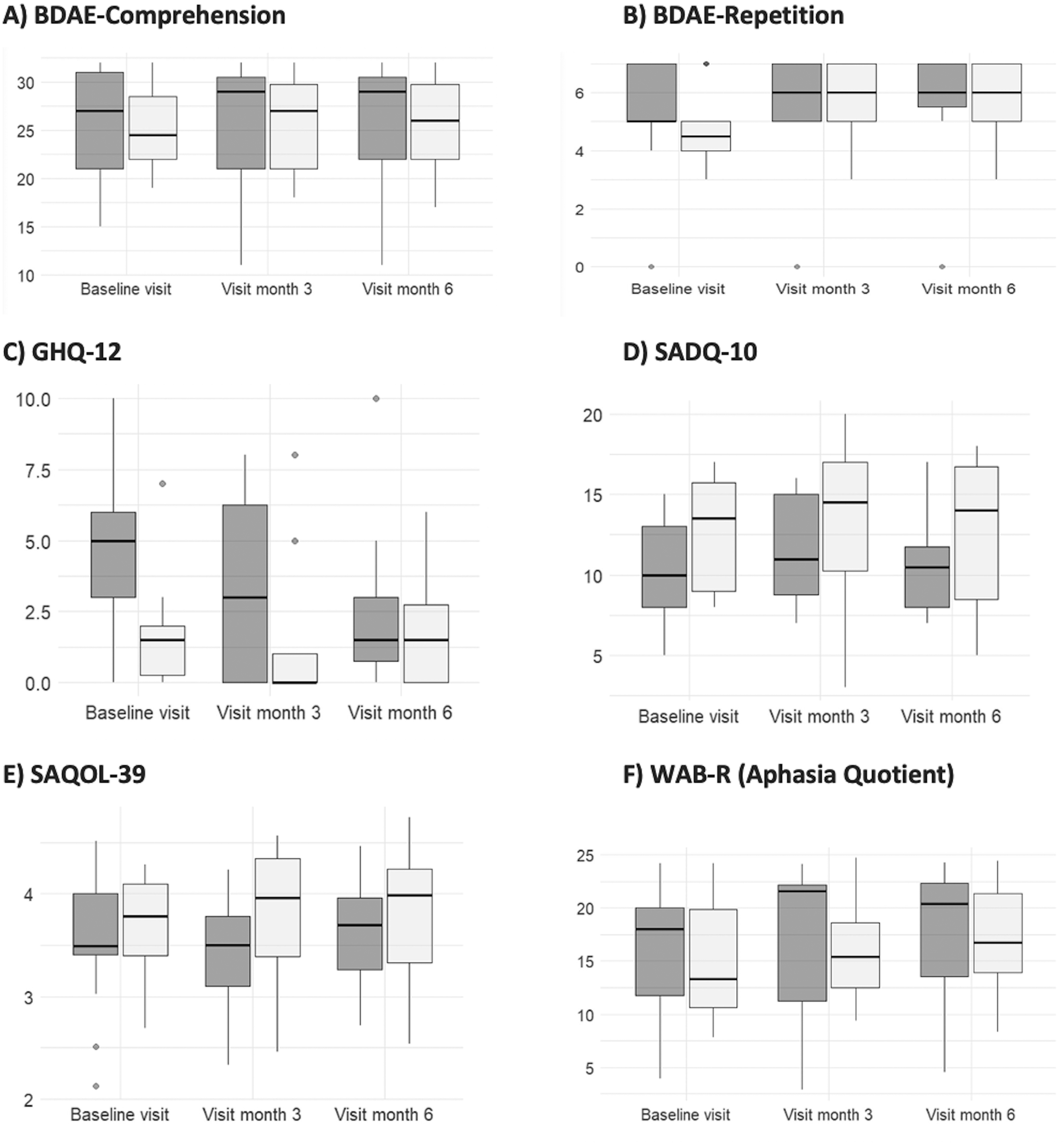
Secondary outcomes
Dropouts and adherence to the study visits
Two patients dropped out of the trial, one after completing 11 of the 17 scheduled sessions, the withdrawal from the study being related to transportation issues (he lived 334 km away). The second dropout was due to the onset of an unrelated disease after completing 7 study sessions.
The adherence rate to the allocated group was 95.3%, with only 1 patient crossing from group 1 to group 2 due to the development of severe COVID-19-related pneumonia, with 2 hospital admissions during the 3 months in which he was supposed to start the DULCINEA sessions. This period coincided with a temporal interruption of the study sessions due to the absence of the speech therapist, also due to SARS-CoV-2 infection. Therefore, the decision to cross this patient from group 1 to group 2 was offered to the patient by the study team.
Patients’ acceptability and other reported qualitative outcomes
At the end of the study visit, the relatives were surveyed through a Likert scale regarding the frequency with which the patients introduced the trained words into their spontaneous speech: very often (2; 9.5%), frequently (4; 19%), sometimes (11; 52.3%), rarely (2; 9.5%), or hardly ever (9.5%). In addition, a total of 9 patients/relatives provided spontaneous comments regarding their overall experience during the clinical trial. Seven of them reported subjective improvements in communication skills mainly related to losing the fear of trying to speak. Here is a sample of the comments: “She used to say the words made in the sessions quite a few times right after leaving the session and a little the next day. She uses them from time to time. It is true that she uses more words”; “She is very happy with having participated, and we would say that she has sometimes incorporated the words worked on”; “We have observed an improvement in communication”; “The DULCINEA project has been a very positive experience for my brother and me. I have learned to communicate with my brother much better and to understand things about aphasia that I did not understand or know”; “He presents a little more spontaneous language, he has overcome the fear of trying to communicate, although he only uses the specific words he trained sometimes”; “She recovered 4-5 words, but after finishing the sessions, it became increasingly difficult for her”; “The research is great, but what I would like would be to continue with the sessions since I notice improvement when speaking.” One relative observed no significant improvement but reported that the patient “attended the sessions with enthusiasm,” reflecting very good acceptability. Five patients could not provide any subjective comments due to aphasia severity and no relatives to ask. Lastly, another relative reported interest in participating again in clinical trials on aphasia.
Sample size calculation for a definitive trial
Using the data on the standard deviation of the main change in the CAL evaluation, we would need a sample size of 287 patients in each arm (for an 80% power and a 0.050 two-sided significance level) for a definitive, randomised, double-blind, parallel clinical trial.
Discussion
This clinical trial was designed to evaluate the feasibility of dubbing therapy using ad hoc-developed software (DULCINEA) for patients with long-term moderate-to-severe post-stroke aphasia who had already completed the standard SLT. Despite being unable to obtain the pre-calculated sample size (due to the COVID-19 pandemic and the end of funding), our study shows the feasibility of dubbing techniques and of the DULCINEA software in treating post-stroke aphasia. Our dropout rate (9.5%) was much lower than the 24.7% reported by the Cochrane systematic review in the comparison of SLT with no SLT. 7 In addition, the adherence to the allocated intervention was excellent (95.3%), which contrasts with the reported 7.25% in the Cochrane systematic review in the comparison of SLT with no-SLT trials. 7
A critical limitation of the current trial was the failure to achieve the predefined sample size. In a similar trial on the efficacy of melodic intonation therapy for patients with aphasia, 28 the estimated simple size was 54 patients for detecting differences in the primary outcome (CAL). Small sample size is a common limitation in clinical trials of post-stroke aphasia. A systematic review that included 62 studies on the use of digital applications developed for new technologies (computers, tablets, and smartphones) alongside classical therapies (randomised controlled trials [RCTs], quasi-experimental studies, case-series, narrative reviews) found that most studies had a sample size of fewer than 50 patients. If we restrict the analysis on the sample size to RCTs, only 1 recruited more than 51 patients. 31
Our study was affected by the COVID-19 pandemic, as were many other clinical trials during 2020 and 2021. 32 We planned to begin recruitment in the fourth quarter of 2020, once the restrictions on hospital access were eliminated after the first wave of the pandemic. At that time, the on-site visits were mainly replaced by telephone consultations whenever possible. Given that, for the sessions conducted face-to-face, use of the DULCINEA software requires support from the speech therapist and the dubbing actor, and that the patients and speech therapist need to work without a mask, we obtained special authorization from the Risk Prevention Department to make some logistical adjustments to the hospital room where the DULCINEA sessions took place. We obtained approval to start face-to-face visits in November 2020, and the first patient was recruited in January 2021.
Patient recruitment was hindered by the requirement to cease other SLT activity prior to participating in the trial. Given that we considered it unethical to allocate patients still undergoing standard SLT to the control group, we only selected those who had already finished SLT upon giving informed consent. However, we noticed that many patients with chronic aphasia followed privately funded SLT, even after acknowledging lack of improvements.
An additional challenge was the fact that patients in the DULCINEA trial were more severely affected than those in the trial used for the sample size calculation (baseline median CAL of 93 vs. 122). 28 This difference might also contribute to the final low statistical power. All the participants were in a chronic phase (more than 6 months from stroke onset and having completed standard SLT) and had a mean age of 65 years. Enrolment more than 1 month after onset and an age older than 55 years are well-known predictors of poorer recovery in poststroke aphasia. 33 Therefore, the patients included in the DULCINEA trial might have a low pre-trial probability of obtaining benefits from any SLT. Considering that many of the patients were recruited long after the stroke, it is possible that the learned disuse of speech, which appears in chronic patients due to prolonged lack of practice, may have influenced the effectiveness of the therapy. Therefore, we believe that the efficacy of this therapy could be increased if it is administered in earlier phases of stroke, where this disuse of speech is not so established.
It is well known that the treatment response in aphasia also varies in association with the SLT frequency, intensity, and dosage.34,35 All of the participants in the DULCINEA trial were at the lower end (40 min per session, 2 days a week, 16 sessions) compared with other SLTs. A recent systematic review showed that the greatest overall language gains for patients more than 3 months after the onset of aphasia were detected with frequencies of more than 5 days of SLT per week, an intensity greater than 3–4 h per week, and dosage greater than 50 h. 34 Therefore, once we have confirmed the feasibility of the dubbing techniques through the DULCINEA software, we should explore its efficacy as an adjuvant therapy together with standard SLT in a larger clinical trial that includes patients in the early phase and with higher frequency, intensity, and total dosage of the dubbing procedure.
Functionally tailored interventions have been shown to achieve greater gains for overall language ability, naming, and functional communication than untailored SLT. 35 The DULCINEA therapy has this tailored approach, given that the dubbing programme is individualised according to baseline language impairment as well as the patient's preferences regarding words to be dubbed. To reinforce the validity of the words included in our study, we rely on the article by Palmer et al., which includes a record of the words preferentially chosen by 100 participants with aphasia. 36 Thus, we found that 133 of the 216 words recorded in our initial survey also appear in Palmer's study. Similarly, 121 of the 183 words that made up the word list in DULCINEA are also match in Palmer et al.'s study. In addition, we should note that the words were classified according to their semantic category, which was also the case in our study. However, in contrast to DULCINEA, they were not classified according to the level of difficulty.
Future iterations of the DULCINEA procedure could benefit from incorporating recent advances in language reacquisition. Specifically, the incorporation of a procedure based on reinforcing the mechanisms behind oral sensorimotor control in the speech process could improve articulatory outcomes. Indeed, presenting clips that patients can see and hear in the foreground while carrying out the exercises may help patients recruit the articulatory movement that must be performed to pronounce the chosen word. The idea of orolingual movement recruitment is compatible with the motor theory of speech, 37 which explains the ability to produce and understand language through visual and acoustic decoding of the speaker's oral movements, as well as with other theories or approaches in which the interconnection between perception and speech production is a fundamental aspect. 38 It has been proposed that the interaction of mirror neurons helps in this task, 39 since the observation of an action activates the cortical area of the observer's brain, that is, the interaction between vision, proprioception and motor commands promotes the recruitment of mirror neurons, thus enhancing cortical reorganization and functional recovery of patients after stroke. 40 Key to this is the presence of auditory and visual aids, references or feedback provided by the software. These aids allow the feedback received when performing an exercise to be improved or enhanced, thus allowing patients to adjust their response progressively, while being supervised by the speech therapist.
During the DULCINEA sessions, the therapist allows for the gradual development of more complex target utterances through successive approximations, starting, for example, with single sounds, and gradually forming words and short phrases by adapting to the initial phonetic repertoire and the rate of reacquisition of each patient. Moreover, generalisation strategies help support the ecological validity of newly acquired verbal skills by transitioning to more naturalised contexts. For example, practicing in various settings and with multiple partners could facilitate the use of target words and phrases in context, thereby leading to a more functional use of language.41,42
Limitations
Alongside the small sample size, another potential limitation was the online assessment of the language and functional communication performance as well as quality of life, mood and perception of health. We had to adapt to the restrictions imposed by the COVID-19 pandemic and reduce on-site patient evaluations as much as possible. Thus, to ensure homogeneity throughout the trial, we decided to perform all the blinded assessments online. Since interaction via a screen might be a barrier to interpersonal communication, we made major efforts to overcome this by displaying all the questions and potential answers on the screen and allowed the chosen answer to be pointed out. In addition, family members could assist with connecting and understanding questions.
Strengths
The current feasibility trial has significant strengths. First, it was an RCT with a blinded outcome assessment. Second, we provided a complete description of the patients’ baseline characteristics included with post-stroke aphasia, as well as the overall assessment of perceived health, QoL, mood, and functional communication, using standard and validated tests, following current recommendations for post-stroke aphasia research.23,43
The DULCINEA project exemplifies citizen science, emphasizing a co-design process involving stakeholders (individuals with communication disorders, caregivers, therapists). This inclusive approach enhances tool effectiveness, balancing clinical content and user experience. 44 In DULCINEA, stakeholder collaboration was extensive, as we addressed different methods already reported 45 : consultation (word selection), involvement (trial design, grant proposal), collaboration (consortium agreement signing), empowerment (patients’ association managing education and dissemination), and information (activities for public understanding of aphasia and clinical research). This comprehensive engagement reflects a commitment to citizen science principles, ensuring relevance, and effectiveness in addressing communication disorders.
Conclusion
This trial shows the feasibility of dubbing techniques using ad hoc-developed DULCINEA software for patients with long-term moderate-to-severe non-fluent aphasia. Patient adherence was excellent. Although the study lacks statistical power, certain positive effects on language and communication cannot be ignored. Larger multicentre RCTs are needed to confirm its feasibility and efficacy as adjuvant therapy to conventional SLT in early stages after stroke.
Footnotes
Acknowledgements
The patients’ association “Afasia Activa” was a partner of the research consortium. They contributed to the design of the research project; provided advice on the call application draft; delivered a list of target words (together with 2 other Spanish stroke patient associations, “Asociación Ictus Madrid” and “Freno al Ictus”) and organised dissemination activities. We acknowledge the collaboration of the speech and language therapists Lidia de la Fuente and Celia Delgado (†), and the psychologists Sofía Blanco, María Muñoz, Marta Provencio, Ana Navarro, and Blanca Aldea. We also thank the public Spanish Radio and Television (RTVE) because they allowed us the right to use and provided us with videoclips of some Spanish series for the dubbing material. We greatly appreciate the support of Morote Traducciones for editing assistance.
Contributorship
BF: Study concept and design, patient recruitment, data analysis and interpretation, drafting the manuscript, and final approval of the version to be published. PJ-P: data acquisition, analysis, and interpretation, critical review of the manuscript, and final approval of the version to be submitted for publication. CS-I: data acquisition, analysis, and interpretation, critical review of the manuscript, and final approval of the version to be submitted for publication. AT-R: Study concept and design, data acquisition, analysis, and interpretation, critical review of the manuscript, and final approval of the version to be submitted for publication. EC-R: patient recruitment, critical review of the manuscript, and final approval of the version to be submitted for publication. MM-A: Study concept, patient recruitment, critical review of the manuscript, and final approval of the version to be submitted for publication. RR: patient recruitment, critical review of the manuscript, and final approval of the version to be submitted for publication. JR-P: patient recruitment, revising the manuscript critically for important intellectual content, and final approval of the version to be published. EA-L: patient recruitment, critical review of the manuscript, and final approval of the version to be submitted for publication. GR-A: patient recruitment, critical review of the manuscript, and final approval of the version to be submitted for publication. MAL: patient recruitment, critical review of the manuscript, and final approval of the version to be submitted for publication. JV-O: Study concept and design, data acquisition, analysis, and interpretation, critical review of the manuscript, and final approval of the version to be submitted for publication. AMB: data analysis and interpretation. MJ-G: data analysis and interpretation. MM-B: data analysis and interpretation. PB: Study concept and design, critical review of the manuscript, and final approval of the version to be submitted for publication. NB: Study concept and design, data acquisition, analysis, and interpretation, critical review of the manuscript, and final approval of the version to be submitted for publication.
Competing interests
The authors declare that they have no competing interests.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
All versions of this protocol (version 1.0, date 15 July 2019 and version 2.0, dated 7 October 2020) were approved by the Hospital Ethics Committee for Clinical Research. The protocol was registered with the ClinicalTrials.gov register: NCT04289493. All patients, or their guardian or legal representative, signed a written informed consent document after a detailed explanation of the nature and purpose of this study and before undergoing any of the procedures related to the clinical trial. An aphasia-friendly information sheet containing large text and simplified language was provided.
Funding
This study was promoted by the Research Foundation of La Paz University Hospital, which hosted a research consortium joined by the Department of Neurology at La Paz University Hospital, the Department of Psychology at Comillas Pontifical University, and the patients’ association, Afasia Activa. This project has received funding from “la Caixa” Banking Foundation under the project code LCF/PR/HR19/52160009. The funder was not involved in any of the following processes: design of the trial, data collection, analysis, or interpretation, or writing the manuscript. BF, EdC-R, RR, GR-A, JR-P, EA and MA-L are members of the Spanish Stroke Research Network RICORS ICTUS (RD21/0006/0012) funded by the Carlos III Institute of Health and the European Union (NextGenerationEU). ‘la Caixa' Foundation, Instituto de Salud Carlos III, (grant number LCF/PR/HR19/52160009, RD21/0006/0012).
Guarantor
BF
Trial registration
NCT04289493, Registered 28 February 2020, https://clinicaltrials.gov/ct2/show/NCT04289493?term = dulcinea&draw = 2&rank = 1
