Abstract
Background
Mental health problems in patients with chronic obstructive pulmonary disease (COPD) are common and frequently neglected. Digital psychological interventions may reduce mental health problems, but their effectiveness has not been evaluated in the Chinese COPD population. In this study, we will develop an integrated digital psychological intervention (EmoEase) and evaluate its effectiveness and cost-effectiveness in enhancing the mental wellbeing of patients with COPD in China.
Methods
This study is a multicenter, two-arm, randomized controlled trial (RCT) with a parallel-group design to enroll at least 420 patients with COPD with age over 35 years. Participants will be assigned to receive either usual care (control group) or usual care + EmoEase (intervention group). Assessments will take place at baseline (T0) and 4 weeks (T1), 8 weeks (T2), and 16 weeks (T3) after baseline, and participants will be asked to complete questionnaires and physical measurements. The primary outcome measure will assess mental wellbeing using the Warwick Edinburgh Mental Wellbeing Scale (WEMWBS). Secondary outcome measures will assess mental health, physical health, COPD symptoms, health risk behaviors, socioeconomic indicators, and healthcare utilization and expenditure. Analyses will utilize an intention-to-treat approach.
Discussion
This is the first RCT to examine the value of EmoEase, a novel digital psychological intervention for patients with COPD. If this intervention is effective and cost-effective, it could be rapidly scaled up to provide mental healthcare for patients with COPD in China.
Trial registration
ClinicalTrials.gov Identifier: NCT06026709. Date of first submission: 30 August 2023. https://clinicaltrials.gov/study/NCT06026709
Background
Many patients with chronic obstructive pulmonary disease (COPD) also have mental health problems. A recent study found that rates of depression and anxiety in people with COPD were 14% and 11%, respectively, which are significantly higher than those observed in patients without COPD. 1 However, current COPD management standards do not attend to patients’ mental health needs. 2 For instance, routine screening for depression and anxiety among patients with COPD is absent from existing clinical guidelines, leading to the neglect of mental health problems in many patients. Overall, less than one-third of patients with COPD with mental health problems receive appropriate psychological treatment, partly due to a lack of standardized management of mental health illnesses in patients with COPD. 2 Studies have also found low awareness of existing mental health issues among patients with COPD. For example, the prevalence of self-reported depression was only 0.09% among 29,201 patients with COPD enrolled in the Enjoying Breathing Program in China, 3 whereas active screening data indicated a depression prevalence among patients with COPD in China of 14%. 1
Several factors suggest that digital psychological interventions may be effective in alleviating mental health issues among patients with COPD and more accessible and cost-effective than traditional medical help. First, digital interventions have been validated as an effective means of addressing various mental health-related problems such as depression, anxiety, stress, and insomnia.4,5 Second, by leveraging online platforms, digital interventions enable patients to access psychological support in a relatively anonymous environment, avoiding the social pressure and stigma that may accompany face-to-face counseling. In the case of patients with advanced respiratory system diseases, some individuals refuse referrals to psychologists or psychiatrists due to the stigma associated with mental health conditions. 6 Third, from a healthcare policy perspective, although China's health insurance covers pharmaceutical treatment for mental health, psychotherapy, preventive care, and rehabilitation services are typically not included. 7 As China seeks to expand the scope of services offered, digital psychological interventions may be a more cost-effective option than traditional psychotherapy. Such an approach has been taken in other countries. For example, Germany has already integrated digital health applications, including digital psychological interventions, into its healthcare insurance system. 8
Although the prevalence of COPD is higher among individuals over 40 years old, those with lower socioeconomic status, and residents in rural areas with poorer internet coverage, 9 smartphone usage in China is highly prevalent. As of December 2023, China had 1.091 billion mobile internet users, with 99.9% accessing the internet via smartphones, a national internet penetration rate of 77.5%, and 66.5% in rural areas, with users aged 40 and above comprising half of all internet users. 10 Additionally, 87.5% of internet users have at least one digital literacy skill, such as using smartphones for searching, downloading, installing software, expressing opinions online, and communicating with others. 10 This suggests that implementing digital psychological interventions among the COPD population in China is relatively feasible.
There has been limited implementation of digital psychological interventions at scale for patients with COPD worldwide. To assess whether digital psychological interventions represent an effective and cost-effective approach to improving the mental wellbeing of patients with COPD in China, we aim to develop EmoEase, a digitally integrated WeChat Mini Program and validate its impact through a large-scale randomized controlled trial (RCT).
Methodology
Study design
The study will consist of two main components: (1) design prototyping and pilot study to test and modify the study design; (2) formal RCT, adopting a multicenter, two-arm, individually randomized controlled clinical trial design to examine the effectiveness and cost-effectiveness of the EmoEase program in China (Figure 1).

CONSORT flowchart of the study procedure.
Study setting and participants
The target population will comprise patients with COPD recruited through the hospitals covered by the China County Respiratory Committee, a branch of the China Association for Health Promotion and Education. The China County Respiratory Committee is responsible for building a health management model for chronic respiratory diseases at the grassroots level and promoting early diagnosis and intervention of common chronic diseases. The Committee involves nearly 100 healthcare organizations at all levels across 31 provinces. We will select 10 of these hospitals at different levels and in different provinces—and with willingness and ability to participate—for inclusion in the study.
The inclusion criteria for participants are defined as follows: (1) age 35 years or older; (2) post-bronchodilator ratio of forced expiratory volume in one second to forced vital capacity (FEV1/FVC) < 0.70 or confirmed patients with COPD; (3) literate and able to type; (4) have a smartphone; (5) proficient in using the WeChat Mini Program; (6) willingness to participate in digital psychological intervention sessions for 8 weeks, with 10 to 15 minutes of practice per session for a total of roughly 1 hour per week, and at a flexible time and place; (7) willingness to receive three follow-up visits within 16 weeks (two in-person and one online) and complete a physical exam and online questionnaire; and (8) willingness to provide the hospital with their usual cell phone number. Exclusion criteria are defined as follows: (1) physician diagnosis of asthma or asthma-COPD overlap syndrome, (2) hospitalized for COPD within the previous year, (3) severe cognitive dysfunction and unable to communicate, and (4) having been listed on the lung transplant waiting list. The anticipated demographics of potential participants will align with the characteristics of patients with COPD in China, primarily focusing on individuals over 40 years old, those with lower socioeconomic status, and residents in rural areas with an internet penetration rate close to 70%.9,10 Most of these participants are expected to have at least one digital literacy skill. 10
Design prototyping and pilot study
In the design prototyping phase of the study, we will use the Rapid Iterative Testing and Evaluation (RITE) method to optimize EmoEase.11,12 (Plans for the initial intervention design are described below.) We will conduct approximately five rounds of face-to-face interviews with physicians and patients in secondary and tertiary hospitals. Each interview round will include physicians and patients from respiratory, psychosomatic, and other relevant departments who meet the criteria. During each testing round, participants will receive in-person guidance on utilizing EmoEase. Following that, we will collect on-site feedback focusing on EmoEase's feature usability, page design (including elements like color scheme, layout, font style and size, and images), and other factors contributing to the seamless use of the mini-program. Alongside on-site feedback, we will incorporate concise questions at the end of each training session to gather online feedback within the mini-program. Subsequently, we will enhance EmoEase based on this feedback before advancing to the next testing round. The number of iterations will be determined by the modifications made and user feedback received. Follow-up phone calls will be conducted 2 and 4 weeks after intervention to gather feedback on the prototype and complete brief questionnaire surveys (System Usability Scale).13,14
The study will include a 2-month internal pilot RCT, for which roughly 42 patients with COPD (10% of the total sample size) 15 will be recruited from several hospitals in Beijing. The aim of this pilot is to assess the success of participant recruitment and retention, as well as the utilization of EmoEase. Participant usage will be determined by the activation, frequency of access, and duration of use of the online training or any components in EmoEase. The research design for the formal RCT will be improved based on participant recruitment, retention, and EmoEase usage during the pilot.
Procedure
Recruitment and consent
The second phase of the study will consist of the formal RCT. Recruitment for the RCT will begin with a brief introduction to the trial by a trained site staff member (usually a clinician) who will screen participants for eligibility and assess inclusion and exclusion criteria. Participants who do not pass screening (e.g. those without smartphone skills or equipment) will receive a paper-based COPD health brochure to help them receive health guidance. Eligible participants will receive written study information and will sign a written informed consent form.
Assessment
After signing the written informed consent form, all participants will be invited to complete a baseline assessment (T0). This will encompass completion of the baseline questionnaire and a physical examination (including a pulmonary function test and a routine physical examination) under supervision and with assistance of the site staff.
Follow-up assessments will be conducted at week 4 (T1; mid-intervention), week 8 (T2; postintervention), and week 16 (T3; 8 weeks postintervention) after the baseline assessment. T1 will be conducted by a trained staff member who will contact the participants by phone to complete a short questionnaire interview to ascertain their baseline psychological wellbeing at the mid-intervention stage. T2 and T3 will follow a similar procedure as the baseline assessment; patients will be required to visit their designated hospital to receive relevant physical measurements and complete a questionnaire. Participants will be given a transportation allowance of 200 CNY for each of the two in-person follow-up visits.
Randomization and blinding
Participants will be randomized after completing the baseline assessment. Patients are randomly assigned (1:1) to usual care (control group) or usual care + EmoEase (intervention group). For participants assigned to the intervention group, on-site staff will not only provide training on using EmoEase during the baseline assessment but also offer a detailed user guide to help them better receive and utilize the intervention. Random assignment will be conducted using an electronic data capture system with a block size of four and stratified by study center location and degree of mental health status (WEMWBS scores ≤45 or >45 16 ). Due to the design of the intervention, participants, and field workers will not be blinded to intervention allocation, but they will be reminded to keep their groupings confidential.
Intervention
Brief introduction
EmoEase, the digital intervention, will be delivered through a WeChat Mini Program. It will integrate two main pieces of training for anxiety and depression. We will develop the training mainly based on CBT. Each training will include approximately 20 sessions, and each session will center around a specific theme (e.g. The Role of Emotions, Acceptance, Concentration and Engagement, Positive Activities, Value of Life, Examining Our Thoughts, Interpersonal Skills, and Social Support) and can be completed in 10 to 15 minutes. The sessions will be structured as simulated conversations in which EmoEase describes and demonstrates concepts, knowledge, and skills relevant to the theme, directs the user in practical exercises, and prompts users to participate by choosing a range of response options. The intervention will span 8 weeks. Participants will be recommended to engage in two or three sessions per week. To ensure sequential progression through the intervention, only the first module will initially be accessible to the user, and later modules will be locked until the preceding module has been completed. Additionally, users can freely access information and tasks from sessions they have already completed, allowing them to deepen their understanding of these sessions. Participants will receive periodic automated Short Message Service (SMS) notifications appraising their engagement with EmoEase. The scheduling of these SMS notifications can be tailored to participants’ preferences. The prototyping phase will be conducted among patients with COPD, psychologists, and some colleagues in the research team. Feedback will be collected to inform the improvement of EmoEase to meet the demands of the target population. During the development of EmoEase, mental health professionals from Heidelberg University, Southeast University, Beijing Hospital, China-Japan Friendship Hospital, and Lanzhou Institute of Technology will provide continuous guidance. These experts will contribute significantly to the intervention development by conducting multiple interviews, designing and developing platform content, and reviewing and providing feedback on the intervention components.
Rationale, functions, and components
EmoEase will integrate various evidence-based therapeutic approaches for treating depression and anxiety. For example, CBT techniques, including cognitive restructuring and behavioral activation, have demonstrated effectiveness in the treatment of depression and anxiety.17–19 As a complementary technique, acceptance and commitment therapy (ACT), which emphasizes mindfulness and acceptance to develop psychological flexibility, 20 helps individuals openly face challenges and adjust their behavior to participate in valued activities rather than avoiding uncomfortable experiences, emotions, and thoughts.20–22
EmoEase will be primarily based on CBT, with ACT as a third-generation CBT therapy. 20 CBT techniques, including cognitive restructuring and behavioral activation, have demonstrated effectiveness in treating depression and anxiety.17–19 As a complementary technique, ACT emphasizes mindfulness and acceptance to develop psychological flexibility. It helps individuals openly face challenges and adjust their behavior to participate in valued activities rather than avoiding uncomfortable experiences, emotions, and thoughts.20–22 Multicomponent CBT programs often include therapeutic elements such as sleep management, problem-solving, and stress management, demonstrating effectiveness across various symptoms.23–25 These therapeutic approaches are incorporated mainly in the form of psychoeducation, addressing issues related to anxiety and depression without overwhelming participants. Depression and anxiety often coexist and interact with sleep problems, stress, and interpersonal issues.26,27 Different components can have synergistic effects. For instance, improvements in sleep can enhance mood and reduce anxiety. 28 Addressing them comprehensively reflects the real-world complexity of these issues and can lead to more comprehensive improvements in mental wellbeing.
The intervention will have the following functions (see Figure 2 for screenshots of the first-generation EmoEase application in the WeChat Mini Program, with iterative updates to follow):
Online training:
(a) Depression: Sessions for depression will be based on a combination of treatments recommended by clinical guidelines for treating depression.29,30 They will include self-guided education and awareness training focused on depressive symptoms, relaxation and physical exercise, increasing positive activity, ACT techniques (acceptance, mindfulness, and value exploration), CBT techniques (behavioral activation and cognitive restructuring).30–36 We also include components such as sleep management, problem-solving skills, pressure management techniques, and interpersonal skills to comprehensively address sleeping problems, stress, and social support related to depressive symptoms, thereby enhancing the overall effectiveness of the intervention.37–38 As the depression training is delivered through a WeChat mini-program, it offers participants the flexibility to customize their learning schedule, allowing them to choose the most suitable times and locations for practice. (b) Anxiety: We develop this training based on clinical guidelines for the treatment of anxiety.39–42 Meanwhile, while designing sessions for anxiety, we also refer to previous published intervention studies using CBT to alleviate anxiety symptoms.43–47 It will be a series of online self-guided sessions containing eight modules in easily understandable language. The sessions will progress as follows: a basic introduction to anxiety, relaxation skills, ACT techniques (e.g. cognitive defusion, acceptance, and value exploration), CBT techniques (cognitive restructuring, behavioral activation, and self-management approach), graduated exposure to address anxiety, and seeking social support. The EmoEase application will use psychological scales to assist participants in assessing their level of anxiety, and participants can set personalized anxiety relief goals based on this assessment. EmoEase will also guide participants in acquiring effective anxiety relief skills through a combination of theoretical learning and practice, alleviating anxiety symptoms, and improving overall mental wellbeing. Finally, this mini-program will aid participants in establishing harmonious social relationships and mobilizing external forces to alleviate anxiety symptoms. Support from experts (Mood Tree Hole): This is a safe space that provides an outlet for participants to release their emotions and thoughts. Posts made by participants in Mood Tree Hole may receive responses or inspiration from mental health professionals. The privacy and the potential for support encourage users to focus more on self-expression and emotional release rather than relying solely on external feedback. It also offers a low-pressure option for those who need support but are hesitant to seek direct counseling. Mood recording: Participants will be actively prompted to observe and document their mood. They can record the state and intensity of their emotions, along with the situation (when the relevant event took place, where they were, who was involved, and what happened) and triggers contributing to their mood. Problem-solving: We will actively encourage participants to document the challenges they face and explore realistic coping strategies. This process will involve the development of action plans to mitigate psychological distress. Positive activity: Positive activities are simple, intentional, and regular practices associated with naturally happy people.
48
They encompass activities that are enjoyable, rewarding, or personally meaningful, such as listening to music, reading, and hiking. Participants can select and plan positive activities to improve their happiness. Mindfulness: Participants will have access to guided mindfulness audio covering various topics, such as relaxation, mood management, self-acceptance, anchoring, concentration, sleep management, presleep body scanning, and pressure management. Quick self-support toolbox: Participants can save and access favorite techniques from the depression and anxiety training sessions in this toolbox. Health promotion: Participants will have access to various self-assessment scales and learning materials pertaining to depression, anxiety, and general mental health to enhance their knowledge and awareness. Incentives: Participants can accumulate credits during practice sessions, which can be subsequently redeemed for digital stamps within EmoEase. For instance, earning 50 credits qualifies for a silver stamp, and achieving 100 credits allows participants to obtain a golden one. Notifications: To motivate engagement, participants will receive text message reminders two to three times per week. These notifications will include encouragement, as well as information on health benefits and incentives for completing practice sessions.

Screenshots of intervention modules in EmoEase.
Depression and anxiety trainings will involve multiple components: (1) Single choice, multiple choice, or judgment questions; (2) Text box: Participants can enter their thoughts, ideas, and emotions; (3) Action list: Participants select multiple predefined options or define options by themselves to generate a list of planned actions (e.g. positive activities); (4) Audio: a mindfulness exercise or meditation; (5) Test and scale: some tasks contain a scale or psychological test to assess participants’ urrent emotional state; and (6) Pictures: each session incorporates visual illustrations to enhance participants’ comprehension of the concepts and skills.
Usual care
We will distribute paper-based informational pamphlets to participants in both the intervention and control groups. These pamphlets are convenient for patients with COPD to carry and reference, offering regular health guidance. The pamphlets will include guidance on the following aspects of self-management of COPD
49
:
Concise overview of COPD, encompassing its definition, prevalent risk factors, commonly observed symptoms, and disease progression; Minimizing exposure to high-risk factors, such as tobacco/smoke exposure, air pollutants, household cooking emissions, and respiratory infections; Regularly monitoring respiratory symptoms, including cough, sputum production, and shortness of breath; Adhering to prescribed pharmaceutical treatments under medical guidance; Undergoing an annual pulmonary function assessment; Completing an annual comprehensive physical examination, with attention paid to potential coexisting conditions; Maintaining a well-balanced diet and considering nutritional supplementation if necessary; Engaging in appropriate respiratory rehabilitation activities; Prioritizing mental wellbeing; and Receiving vaccinations for influenza and streptococcus pneumoniae.
Outcomes
Primary outcomes
Patients’ mental wellbeing at baseline and each follow up will be assessed using WEMWBS.50,51 WEMWBS is a self-report measure comprising 14 short statements focusing on positive wellbeing. Each statement is rated on a 5-point Likert scale, resulting in scores ranging from 14 to 70 (a higher score indicates better mental wellbeing). The Chinese version of the scale has good psychometric properties with strong reliability and validity.52–55
Secondary outcomes
The following set of secondary outcomes will be considered. The Chinese versions of all involved scales and instruments have been validated.
Mental health
Mental health outcomes—including depression, anxiety, stress, insomnia, emotion regulation, and self-efficacy—will, respectively, be examined via the following instruments: Patient Health Questionnaire depression module-9 (PHQ-9),56–58 Generalized Anxiety Disorder 7-Item Scale (GAD-7),57,59,60 Perceived Stress Scale-10 (PSS-10),61–63 Insomnia Severity Index (ISI),59,64,65 Emotion Regulation Questionnaire (ERQ),66,67 and general self-efficacy scale (GSE).68–70 In addition, patients will be asked about their daily sleep time.
Physical health
Self-rated health status will be assessed using the EuroQol 5-Dimension 5-Level (EQ-5D-5L) instrument71,72; height, weight, and waist circumference will be recorded by the patient based on their recollection of the most recent measurement.
COPD symptoms and co-morbidities
Self-assessment of symptoms and dyspnea in daily activities will be measured by the COPD assessment test (CAT)3,9 and the modified Medical Research Council dyspnea questionnaire (mMRC),3,9 and pulmonary function tests will be used to assess the severity of airflow limitation. In addition, gastroesophageal reflux disease (GERD) will be assessed through the self-assessment GERD questionnaire (GerdQ). 73
Health risk behaviors
Smoking and alcohol consumption will be assessed by the recall question, the Fagerstrom test for nicotine dependence (FTND),74,75 and the International Physical Activity Questionnaire-Short Form (IPAQ-S). 76
Socioeconomic outcomes
Productivity loss will be measured with the Work Productivity and Activity Impairment Questionnaire: General Health (WPAI:GH).77,78 Social media use will be assessed via recall questions.
Healthcare utilization and health expenditures
Care cascade outcomes for COPD, hypertension, anxiety, and depression will be measured by asking about participant disease awareness and status with respect to prior screening, diagnosis, treatment, and control. Outpatient or emergency care utilization over the past three months will be assessed via recall questions. Healthcare expenditures will include the direct costs and indirect costs associated with mental illness.
Sociodemographic indicators
Patient information to be collected at the baseline assessment will include basic information (e.g. gender, age, education, residence, type of health insurance, and annual patient and family income), disease history (e.g. history of chronic diseases and frequent cough before age 14), and family history (e.g. whether their parents suffered from respiratory disease).
Table 1 lists the primary and secondary outcomes of the trial and shows how each outcome will be measured. These outcomes will be measured at baseline assessment and each follow-up visit. If the current questionnaire does not work well in the prototype phase and pilot study, we will consider replacing any deficient scales or questions as we transition to the full study.
Outcomes and measurements.
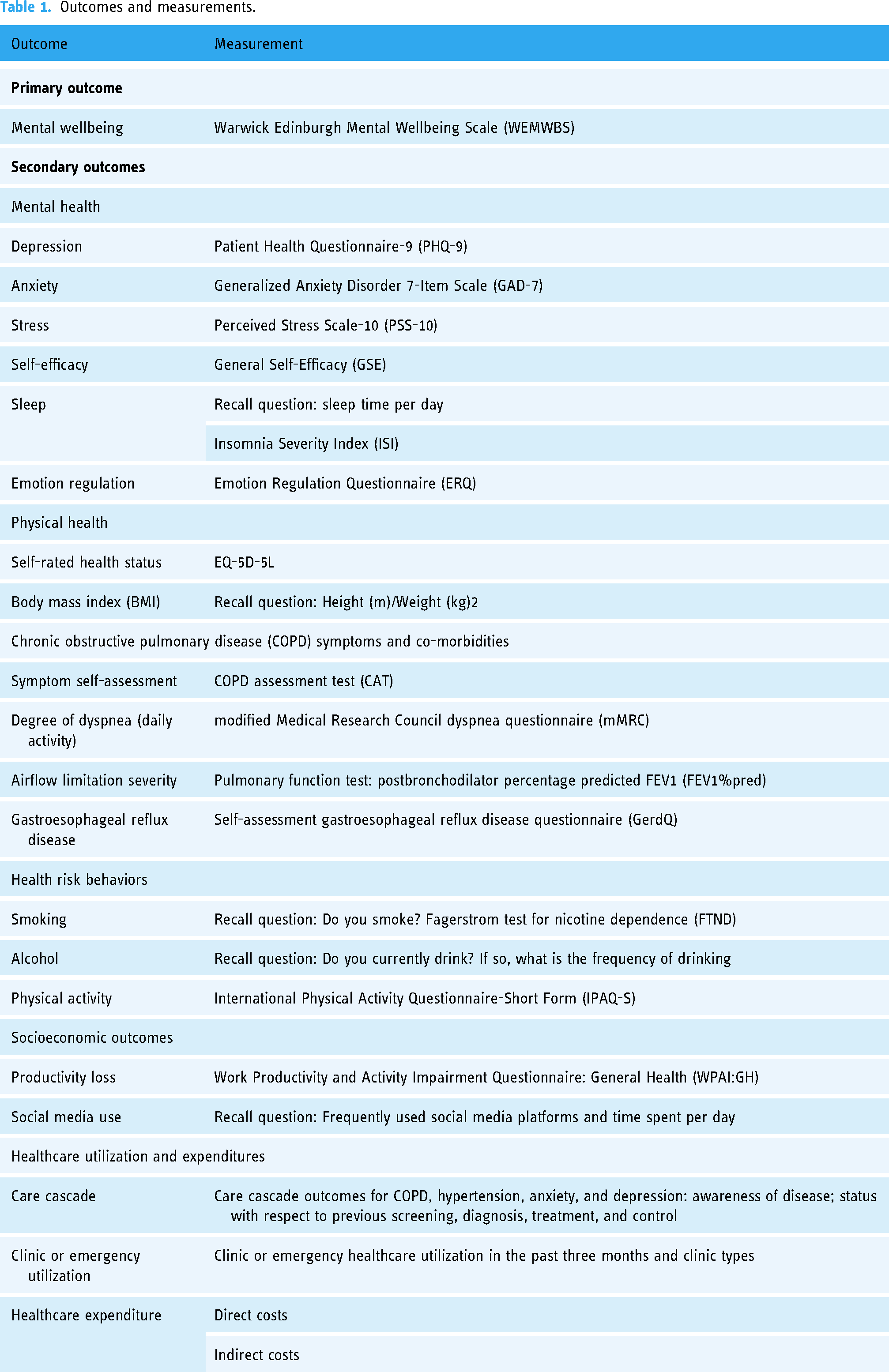
Sample size
To detect a minimally clinically important between-group difference of three points in the WEMWBS score, 79 considering a standard deviation (SD) of 10 as a median value within the variation of SD (ranging from 6 to 14 across different populations),50,80–87 a sample size of 420 participants is necessary. This calculation is based on a significance level (α) of 0.05 (two-tailed) at 80% power, accounting for a 20% dropout rate. The power to detect the between-group difference increases to 90% if the sample size increases to 562. To boost power, we will endeavor to recruit as large of a sample as possible.
Statistical analysis
Effectiveness
Our primary analysis for all endpoints will be intent-to-treat (ITT). To investigate the factors that affect the endpoints, we will use generalized linear mixed models. We will use ordinary least squares regression for our continuous endpoints (e.g. body mass index (BMI)), we will use modified Poisson regression for our binary endpoints (e.g. whether the participant quit smoking), and we will use negative binomial regression for our count endpoints (e.g. WEMWBS score).
The reasons for choosing modified Poisson regression are as follows: first, compared to the log-binomial regression model, it avoids convergence problems and exhibits greater robustness in the face of model misspecification.88–90 Second, the risk ratios generated by modified Poisson regression are considered to be more interpretable than effect size measures produced by alternative methods, such as odds ratios or marginal effects.91–94 In addition to the ITT analysis, we will carry out a complier average causal effect analysis—also referred to as compliance-adjusted ITT 95 —to quantify the effects among those complying with the trial assignment. 96 This effect size can provide a benchmark for policymakers to understand the maximum impact that could be achieved if all individuals complied with digital psychological intervention. To this end, we will carry out an instrumental variable analysis using the ITT assignment as the instrumental variable and receiving the digital psychological intervention (vs. not) as the treatment variable. In addition, multiple imputation will be used for missing data and sensitivity analysis to determine the impact of missing data on study findings. We will also consider hospital-level fixed effects to filter out unobserved factors at the hospital level that may affect the outcome variables. Standard errors will be adjusted for clustering at the level of the primary sampling unit, that is, the hospital level. We will use R statistical software to perform the planned analysis.
Cost-effectiveness analysis
We will conduct a within-trial cost-effectiveness analysis from a broad societal perspective, accounting for both direct (medical and nonmedical) and indirect costs associated with mental illness. The direct medical costs will be obtained from hospital information systems and patient-completed questionnaires. Questionnaires will be administered to patients at baseline and at the follow ups during the trial period. Indirect costs will be analyzed using the human capital approach, 97 which quantifies productivity losses from absenteeism or reduced work productivity caused by work disability. A detailed description of the types of costs, sources, and breakdowns can be found in Supplemental Appendix Table S1. Health outcomes are measured in quality-adjusted life years derived from the EQ-5D-5L.71,72 Both cost and health outcomes will be discounted at an annual rate of 5%, following recommendations for low- and middle-income countries.98,99
Ethics and data safety
The study protocol has been reviewed and approved by the Ethics Committee of Peking Union Medical College (approval number: CAMS&PUMC-IEC-2023-018). Participants can withdraw from the study at any time without any adverse consequence.
All data sets will be securely protected with passwords, and all project members will be required to sign confidentiality agreements.
Discussion
This article describes the study protocol for an RCT designed to evaluate the effectiveness and cost-effectiveness of an integrated digital psychological intervention (EmoEase) in improving COPD patients’ mental health and wellbeing in China. Given the high prevalence and adverse effects of mental health problems in patients with COPD, it is critical to identify suitable remedies. To our knowledge, this study is the first to examine the effectiveness of a digital psychological intervention in patients with COPD.
This study has several strengths. First, design prototyping will use the RITE method, which will optimize the trial design and save resources. Second, this is a nationally conducted multicenter RCT with a substantial sample size, elevating external validity and rendering the findings more universally applicable, thereby fortifying the study's reliability and generalizability. Third, we will take several proactive measures to address the common challenge in digital psychological interventions of low adherence and high dropout rates. 100 Literature shows that guidance can enhance adherence and increase effectiveness compared to unguided digital interventions.100,101 To improve treatment adherence, we will therefore adopt the following measures: (1) a trained physician will instruct patients to use EmoEase; (2) all patients will receive a COPD health promotion pamphlet, and patients in the intervention group will also receive EmoEase registration and use guidelines; and (3) EmoEase will be designed with relevant modules to answer participant questions. Other measures to improve participant adherence include short follow-up periods, relatively few follow-up visits, informational reminders for intervention use, and a transportation allowance.
Conclusion
This trial aims to evaluate the effectiveness and cost-effectiveness of EmoEase, a convenient and low-cost digital psychological intervention, in providing timely mental healthcare for patients with COPD. If proven effective and cost-effective, EmoEase can rapidly deliver mental healthcare to a large number of patients with COPD. The study's results will guide future research, potentially establishing a framework to address barriers to digital psychological interventions and developing more effective strategies for managing mental health risks.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076241277650 - Supplemental material for Effectiveness and cost-effectiveness of an integrated digital psychological intervention (EmoEase) in Chinese chronic obstructive pulmonary disease patients: Study protocol of a randomized controlled trial
Supplemental material, sj-docx-1-dhj-10.1177_20552076241277650 for Effectiveness and cost-effectiveness of an integrated digital psychological intervention (EmoEase) in Chinese chronic obstructive pulmonary disease patients: Study protocol of a randomized controlled trial by Simiao Chen, Wenjin Chen, Yanfei Li, Yiwen Yu, Qiushi Chen, Lirui Jiao, Ke Huang, Xunliang Tong, Pascal Geldsetzer, Aditi Bunker, Xingyuan Fang, Shu Jing, Yuhao Liu, Yanming Li, Liu He, Chao Wang, Weiyu Wang, Zhoutao Zheng, Shiyu Zhang, Jinghan Zhao, Ting Yang, Till Bärnighausen and Chen Wang in DIGITAL HEALTH
Supplemental Material
sj-doc-2-dhj-10.1177_20552076241277650 - Supplemental material for Effectiveness and cost-effectiveness of an integrated digital psychological intervention (EmoEase) in Chinese chronic obstructive pulmonary disease patients: Study protocol of a randomized controlled trial
Supplemental material, sj-doc-2-dhj-10.1177_20552076241277650 for Effectiveness and cost-effectiveness of an integrated digital psychological intervention (EmoEase) in Chinese chronic obstructive pulmonary disease patients: Study protocol of a randomized controlled trial by Simiao Chen, Wenjin Chen, Yanfei Li, Yiwen Yu, Qiushi Chen, Lirui Jiao, Ke Huang, Xunliang Tong, Pascal Geldsetzer, Aditi Bunker, Xingyuan Fang, Shu Jing, Yuhao Liu, Yanming Li, Liu He, Chao Wang, Weiyu Wang, Zhoutao Zheng, Shiyu Zhang, Jinghan Zhao, Ting Yang, Till Bärnighausen and Chen Wang in DIGITAL HEALTH
Footnotes
Acknowledgements
The authors would like to thank the hospitals that were involved for showing their interest and support for this project.
Contributions
Funding acquisition was completed by SC. SC, WC, YL, YY, AB, SJ, WW, TB, and CW contributed to the study concept and design. KH, TY, XT, YL, and CW provided resource support, contributing to the project implementation. SC, WC, and YL were responsible for coordinating resources and led the coordination and implementation. WC, YL, YY, SJ, LJ, YL, LH, CW, ZZ, SZ, and JZ collected the data. WC, QC, PG, and XF conducted the sample size analyses and contributed to the analysis plan. WC, YL, YY, and SJ wrote the first draft of the article. QC, PG, AB, TB, SC, TY, and CW critically revised the manuscript for important intellectual content. WC, YL, YY, SJ, and LJ contributed to the literature review. All authors reviewed and approved the final manuscript before submission.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
This study was approved by the institutional review boards at the Chinese Academy of Medical Sciences & Peking Union Medical College with registration CAMS&PUMC-IEC-2023-018.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this publication was supported by the China Medical Board (CMB) under Award Number Grant#22-469, the Chinese Academy of Medical Sciences & Peking Union Medical College under Project Number 2024-CFT-QT-034, and Horizon Europe (HORIZON-MSCA-2021-SE-01) (Project 101086139 — PoPMeD-SuSDeV). The content is solely the responsibility of the authors and does not necessarily represent the official views of the funding agencies. The study funder had no role in study design; collection, management, analysis, and interpretation of data; writing of the report; nor the decision to submit the report for publication, or authority over any of these activities.
Guarantor
SC, WC, YL, YY, KH, TY and CW.
Data availability statement
The individual-level datasets generated and analyzed during the current study are not publicly available due to avoid risk of participant re-identification but are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
