Abstract
Background
Internet of Things (IoT) innovations such as wearables and sensors promise improved health outcomes and service efficiencies. Yet, most applications remain experimental with little routine use in health and care settings. We sought to examine the multiple interacting influences on IoT implementation, spread and scale-up, including the role of regional innovation ‘ecosystems’ and the impact of the COVID-19 context.
Methods
Qualitative study involving 20 participants with clinical, entrepreneurial and broader innovation experience in 18 in-depth interviews, focusing primarily on heart monitoring and assistive technology applications. Data analysis was informed by the NASSS (non-adoption, abandonment, scale-up, spread, sustainability) framework.
Results
Interviewees discussed multiple tensions and trade-offs, including lack of organisational capacity for routine IoT use, limited ability to receive and interpret data, complex procurement and governance processes, and risk of health disparities and inequalities without system support and funding. Although the pandemic highlighted opportunities for IoT use, it was unclear whether these would be sustained, with framings of innovation as ‘disruption’ coming at odds with immediate needs in healthcare settings. Even in an ‘ecosystem’ with strong presence of academic and research institutions, support was viewed as limited, with impressions of siloed working, conflicting agendas, fragmentation and lack of collaboration opportunities.
Conclusions
IoT development, implementation and roll-out require support from multiple ecosystem actors to be able to articulate a value proposition beyond experimental or small-scale applications. In contexts where clinical, academic and commercial worlds collide, sustained effort is needed to align needs, priorities and motives, and to strengthen potential for good value IoT innovation.
Keywords
Background
Digital health innovations such as smartphone apps and wearables are commonly presented as having the potential to improve care, streamline an increasing clinical workload and extend independent living.1–3 Despite research studies suggesting reduced hospital admissions, 4 improvement in service,5,6 and patient outcomes,6–8 when such innovations are implemented routinely in healthcare settings they face a high failure rate,9–11 especially when the change effort is large, ambitious and complex.12–16 Little attention is paid to underlying complexities of embedding new technologies in existing infrastructures (technical, human and organisational) so that innovations work to the extent and scale intended.17,18
In recent years increasing emphasis has been placed on the ‘Internet of Things’ (IoT), which refers to wireless interconnectivity between digital devices (or objects) enabling them to collect, send, store and receive data over a network.19,20 Common applications in health and care include wearables, sensors and other connected medical devices for remote monitoring, management or diagnosis of chronic conditions such as heart failure, 8 asthma, 21 chronic obstructive pulmonary disease, 22 dementia 23 and diabetes. 24 Health and care IoT innovations are often promoted as a means of addressing capacity and staff shortages, 20 supporting inexpensive, efficient and reliable diagnoses,25,26 reducing the need for repeat hospital monitoring,3,27 improving patient safety in the community,28,29 bringing clinical monitoring and care closer to home 30 and reducing caregiver burden. 31
However, IoT implementation can also be problematic with concerns raised over usability, data accuracy, data interpretation and responsibility for acting on alerts.12,32–34 Previous qualitative research on the use of wearable devices highlights privacy, confidentiality and data ownership challenges, as well as issues around data robustness, overload, consistency and reliability to support clinical decision making. 35 For patients, data from wearable devices can both create and alleviate fear and anxiety. 36 There are also tensions between IoT integrity and reliability versus whether IoT innovations align with financial, clinical, and wider social interests and motives of clinicians and patients. 35 It is the technical development of IoT innovations that tends to attract the focus of researchers, alongside financial and service outcomes, 37 with emphasis placed on IoT devices as technical ‘tools’ decoupled from their sociotechnical context. 38 Most research on IoT is carried out in controlled and experimental settings, rather than addressing the challenges of ‘real-world’ widespread implementation over time in clinical and personal contexts.39,40
Given the complexity of development, implementation and roll-out, health and care IoT innovations cannot be understood in isolation, away from the context in which they are embedded, but rather as part of an ‘emerging IoT ecosystem’. 35 Innovation ecosystems refer to networks of actors, activities, objects, organisations at all levels, and relationships, that are important for innovation. 41 Supportive and effective digital health ecosystems consist of members that are interested in, and value, innovation in the health system, and engage with different communities, working together for common goals. 37 Strong innovation ecosystems offer potential for exchange and collaboration, with a view to overcoming challenges in digital health adoption and spread. Such exchange is often facilitated by organisations enabling sharing and networking between academic, industry and clinical stakeholders (e.g. the non-profit Network of Digital Evidence in Health in the United States). 42
While health and care IoT innovations are favoured at policy level, 1 there is little research into how regional innovation ecosystems can better support good value IoT development and roll-out, especially in areas where these ecosystems involve strong academic, rather than purely commercial, actors. In England, this is further complicated by the complexity and largely devolved nature of the health and care system, which means high-level strategic priorities on innovation become translated differently and implemented variably depending on needs at local level.11,43 Innovators navigate a shifting landscape of funding disparities, lack of clarity around procurement processes, and a multiplicity of organisational National Health Service (NHS) ‘customers’.43,44 There also appears to be imbalanced distribution of government investment in digital health innovation, in areas already seen as ‘digitally mature’ or ‘digitally forward’ receiving investment in the form of, for example, Test Beds promising to pilot innovations in ‘real-world’ settings, 45 among other approaches. As a result, some areas appear to be moving forward at pace, and attract further investment and attention from those in the innovation ecosystems, while others lag behind. 43 However, there are national efforts to reduce disparities in innovation infrastructures, as, for example, with levelling up electronic patient record systems across England. 46
Objective
In this qualitative study, we identified opportunities and challenges clinical and industry innovators face in the implementation, spread and scale-up of IoT innovations in health and care, including the COVID-19 context. We considered how local innovation ‘ecosystems’ support or hinder health IoT roll-out and examined how such ‘ecosystems’ can be strengthened to maximise the potential of good value IoT.
Methods
Study design
This qualitative study took place in the context of a large mixed methods project, which began with an exploratory survey and desk research to: (a) map health and care IoT innovations developed and/or deployed in a region in South East England, (b) describe the regional ecosystem supporting innovation and (c) assess key challenges for IoT, including the COVID-19 context. Guided by early findings, we developed an interview agenda and recruitment strategy for the qualitative work, which took place in October 2020 to February 2021.
Population sample
We conducted 18 semi-structured qualitative interviews with 20 participants. We used a maximum variation sampling approach to reach a wide set of professional groups with experience in health and care IoT and combined this with snowball sampling. Participants included those involved in the development, testing, implementation or spread of health IoT solutions in a region in South East England, UK (see Table 1). Interviews included clinicians and allied health professionals using or piloting health and care IoT, representatives from established or start-up IoT companies, and senior innovation leads from the local authority and other organisations functioning as innovation ‘catalysts’. Drawing on early findings, we focused on two areas where IoT use appeared to be increasing: (a) monitoring and management of heart-related physiological signs and conditions and (b) monitoring and supporting older people at home. Due to COVID-19 restrictions most interviews took place online on Microsoft Teams, while some were conducted face-to-face. Interviews lasted between 25 and 80 min. Following either digitally recorded or written, informed consent, participants took part in semi-structured interviews. Interviews were audio-recorded and then either transcribed verbatim (n = 12) by a professional transcriber, or detailed notes were taken (n = 6). Formal interviews were supplemented with additional informal discussions and email exchanges with IoT innovators and other ecosystem actors.
Interviewee roles.
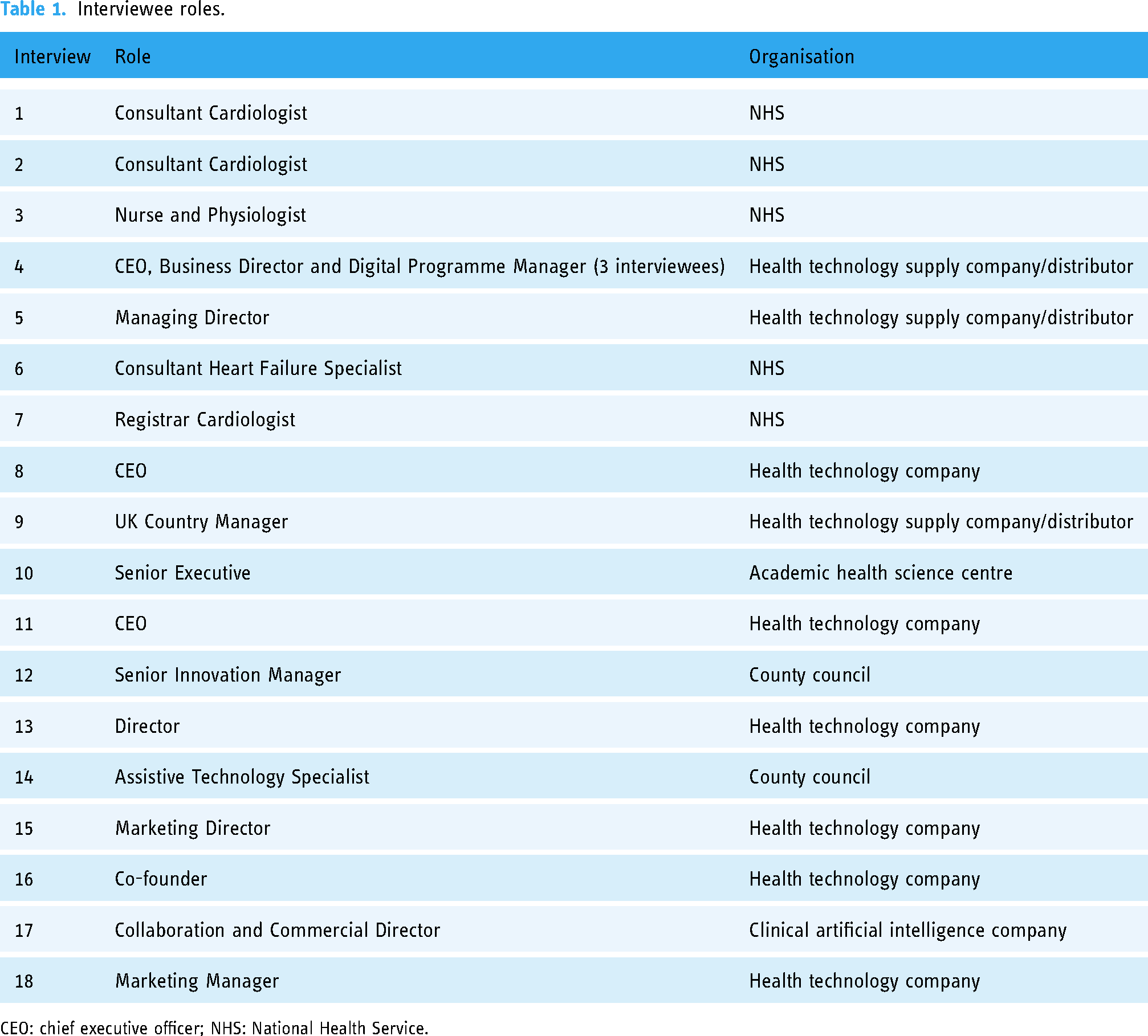
CEO: chief executive officer; NHS: National Health Service.
Ethics
The project received approval from the Medical Sciences Interdivisional Research Ethics Committee (IDREC) at the University of Oxford (R68653/RE001). Interviewees received a participant information sheet ahead of the interview and signed a consent form confirming their agreement to participate in the study.
Data analysis
Analysis was informed by the non-adoption, abandonment and challenges to scale-up, spread and sustainability (NASSS) framework (Figure 1), an evidence-based framework to guide thinking on implementation, roll-out and embedding of technology-supported innovations in health and care.13,47–49 NASSS allowed us to surface and explain the multiple forms and manifestations of complexity which affect uptake and use of health IoT innovations.

Non-adoption, abandonment and challenges to scale-up, spread and sustainability (NASSS) framework.
Firstly, we analysed the data iteratively across the seven domains of the NASSS framework: the clinical condition, the technology, the value proposition, the adopter system, the organisation, the wider system and temporal change. By going through each of the transcripts in detail, we mapped contents to the different domains and developed seven coding reports, as well as an overview document to keep track of the coding process. Then, we delved into each of the coding reports (i.e. domains) to compare and contrast our findings with the original formulation of the NASSS framework, and iteratively refined the analysis. To inform and extend our analysis, we also presented findings (at different stages in the analysis) to our patient and public involvement contributors (n = 6) and to a group of consultant cardiologists and general physicians with extended roles in cardiology (n = 14) interested in the research.
Results
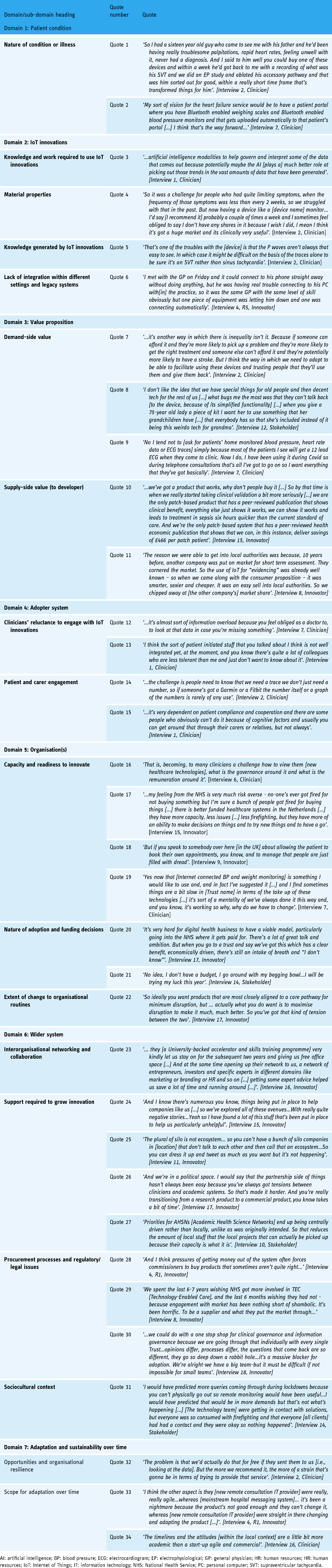
Figure 2 provides an overview of key findings mapped across each of the NASSS framework domains; (1) condition/situation, (2) technology, (3) value proposition, (4) adopter system, (5) organisation(s), (6) wider system and (7) challenges to adaptation and sustainability over time. The figure demonstrates how each of the domains overlap and relate to one another, and the opportunities and challenges faced by study participants in the implementation, spread and scale-up of IoT innovations in health and care. Key points are presented to illustrate pertinent findings from each domain, rather than providing an exhaustive list. Illustrative quotes for each of the domains, and sub-domains have been included as an Appendix (Appendix 1).
Domain 1: Patient condition
Nature of condition or illness
Clinician interviewees focused on the use of IoT for the diagnosis and management of conditions such as arrhythmias (particularly the ubiquitous and increasingly common atrial fibrillation), hypertension and heart failure. These conditions were described as more amenable to IoT-based diagnosis and management than conditions like ischaemic heart disease. For example, paroxysmal arrhythmias giving rise to symptoms, but occurring with relative infrequency, were considered well suited to patient-initiated self-monitoring with a number of innovative and commercially available technologies. The sporadic nature of the condition meant traditional diagnostics were often unable to capture the event, whereas self-monitoring devices could be used at the moment symptoms occurred. Clinicians gave examples of where such devices supported prompt diagnosis and treatment (see Appendix 1, Quote 1).
One of the clinical interviewees described a vision for IoT-based monitoring of selected patients with chronic heart failure, offering the possibility of accelerating the process of medicines optimisation and early identification of those whose condition was deteriorating, potentially pre-empting unplanned hospital admissions (see Quote 2).
Other conditions cited by interviewees as likely to benefit from remote monitoring IoT-based innovations were frailty and isolation (particularly in the elderly), neurological conditions such as Parkinson's disease, and patients developing sepsis.
Domain 2: IoT innovations
Participants specifically discussed 16 IoT innovations piloted or used across different health and care settings. These included sensors recording activities of daily living for short-term social care assessment; home blood pressure monitors uploading data to the cloud; wearable devices providing activity tracking and heart rates with or without accompanying single lead electrocardiograms (ECGs); a wearable device to track Parkinson's disease progression and help with gait freezing; ‘one button computers’ designed to tackle loneliness for older people; disposable patches monitoring vital signs; a small freestanding electrode blue-toothed to smart phones providing a simplified, single lead ECG akin to a rhythm strip on a conventional 12 lead ECG, and with algorithmic interpretation; and an app-linked stethoscope using audio and electrical signals to characterise heart murmurs indicative of heart valve dysfunction.
Knowledge and work required to use IoT innovations
Devices were not necessarily useable without user knowledge of specific technical requirements, as in the example of a digital stethoscope where clinicians had to remember to use headphones to listen to the recording rather than rely on the quality of their phone speakers, according to an industry representative. Another recurring theme in the data was the gap between IoT devices collecting data and the analysis required to understand if these data pointed to meaningful and useful patterns. The anticipatory role of artificial intelligence (AI) was often mentioned, with the expectation that routine use of IoT would only be viable if large scale, automated analysis could be performed in the background (see Quote 3).
Material properties
The material properties of different technologies made them more or less suitable for different contexts; such as handheld devices for symptoms occurring at rest, and smartwatches for identifying exercise-induced arrhythmias. A small commercially available IoT device used to identify and monitor heart arrhythmias seemed to fulfil a need for some cardiology clinicians interviewed. Patients could record a simplified ECG by placing their fingers on the device (and left ankle or leg in the more advanced version) without having to attend a hospital appointment. This meant they did not have to wear bulky and uncomfortable hospital equipment (e.g. body-worn Holter monitors) which could only be lent for short periods (24 hours to 10 days), making it difficult to capture infrequent cardiac events (see Quote 4).
Knowledge generated by IoT innovations
Activity and fitness trackers were frequently acknowledged by clinicians as carrying risks, mainly associated with limited interpretability of heart rate and rhythm on which their notification systems were based. Even with simplified ECG at-home devices that some consultant cardiologists favoured, there were still limitations in that the knowledge gained was sometimes insufficient to reach a conclusive diagnosis (see Quote 5).
An ambitious vision for IoT was common in the accounts of our interviewees. One of the developers described their wearable IoT innovation (still at development stage) as ‘very simple’, aiming to support patients to ‘own’ their condition and to ‘unlock’ treatment optimisation (Interview 11, Innovator). Such vision was appealing, however, in reality the majority of IoT devices discussed by our participants were only used as prompts for further investigations through standard pathways or complementing existing support systems, rather than having the ability to lead to diagnosis or treatment/management decisions on their own.
Lack of integration within different settings and legacy systems
Background technological infrastructure in healthcare organisations was complex, often obsolete, and configured to protect technological infrastructure from third-party devices (rather than to easily integrate them), which meant clinicians could not always rely on legacy equipment within the practice to support routine use of external IoT devices (see Quote 6).
In social care, despite the modernist vision of shiny, new IoT devices, the equipment assistive technology teams had access to described as ‘very old and very clunky’ and often sensors did not work in old buildings with thick stone walls. Instead, the social care teams were seeking: ‘real simplicity in how you operate it and set it up […] I keep wishing for interoperability of devices, that we could just add things on so that they could just connect’ [Interview 14, Stakeholder].
Domain 3: Value proposition
Demand-side value
Many of the devices examined were still at development stage and their financial and non-financial value proposition (to developers and adopters) remained conditional. For example, in heart monitoring, clinicians suggested that specialist IoT devices would provide value by reliably monitoring heart rhythm at a distance, preventing unplanned hospitalisations and giving patients a sense of control. However, IoT innovations were in use by a minority of patients rather than being embedded in routine practice. To the contrary, commercially available, generic IoT devices (such as smartwatches) were deemed a source of inappropriate referrals, or even patient anxiety, when used without adequate clinical support to clarify or reassure where needed.
Establishing value proposition around IoT innovations was not straightforward, especially when they were not available through the NHS but required purchase by patients, which meant there was potential for expediting treatment for some but generating inequalities for others, if patients with greater digital literacy, or higher incomes were encouraged to buy devices that would enable them to reach faster diagnoses (see Quote 7).
One of the innovation managers interviewed made a similar point on social inclusion. Their role in a local authority involved delivering innovation projects and piloting early stage applications of good value technologies. They had rejected a custom-made IoT innovation for older patients that offered limited, simplistic functionality, and instead prioritised not just practical or financial considerations, but symbolic value (see Quote 8).
In the COVID-19 context, the value proposition of IoT innovations increased as remote monitoring was the only option to acquire data that supported clinical-decision making (see Quote 9).
Supply-side value (to developer)
Different IoT innovators attempted to develop and demonstrate value propositions in different ways, primarily focusing on short-term benefits, as they deemed the NHS (the main payer) to be geared less towards long-term value proposition. Value was contingent on clinical validation, peer-reviewed publications showing clinical benefit and health economics analysis (see Quote 10).
Creating value proposition was not just a matter of a single company or product proving their worth, but about multiple providers building and establishing a case for IoT innovation over time, as was the case with IoT use by local authorities in the region where our study was conducted (see Quote 11).
There were also discussions around conflicting priorities, for example where value assessments of those purchasing IoT innovations may conflict with innovators/academics and clinicians’ attempts to address distinct difficulties for particular patient groups.
Domain 4: Adopter system
Intended adopters of IoT innovations described in our dataset included patients, carers, clinicians and social care professionals. A gait monitor for Parkinson's required patients to fasten a device onto each lower leg and use a handheld controller to track activity and potentially prevent gait freezing, as well as falls and inactivity. Using algorithms, the device would buzz when a freezing event was about to occur, allowing the patient to either be distracted, or think about something else for a short time, potentially preventing an occurrence of frozen gait (although the device was not yet fully developed). In another IoT application for patient assessment, clinicians used a digital stethoscope or digital attachment to enhance sound options (such as noise cancellation, filtering and amplification) but also to live-stream, share with other clinicians and save recordings (with the option of AI-enabled disease detection). Another application involved clinicians placing disposable patches on the patient's chest to automatically monitor and score vital signs (such as heart rate, respiration rate and axillary temperature), and receiving notifications via an app (as a treatment prompt rather than diagnostic tool). The aim was to reduce the need for physical observations by clinicians, increase capacity and provide information around vital signs more frequently.
Clinicians’ reluctance to engage with IoT innovations
Not all clinicians were prepared to engage with remote monitoring devices; there was reluctance to support further adoption and spread of IoT heart monitoring devices compared to business as usual for several reasons including: information overload and inability to process the volume of incoming data in the context of time pressures and increased workload; difficulties managing sometimes contradictory data coming from clinically validated (e.g. permanent pacemakers) versus unvalidated devices (e.g. fitness and activity trackers); concerns around clinical governance, quality and responsibility over patient care (see Quote 12). The majority of clinicians seemed to prefer to follow standard pathways, rather than integrate patient-generated data in their decision making (see Quote 13).
Interviewees also noted inequalities in digital literacy between clinicians and insufficient training, preventing engagement with IoT innovations.
Patient and carer engagement
Clinicians mentioned that it could become challenging to explain to patients which data might be useful for clinical decision making and which are less useful (see Quote 14). They expressed the view that older patients (a key population with heart conditions) may have limited digital literacy, with a lack of IT skills and/or confidence, or may be limited by cognitive factors. However, this view shifted during the pandemic as they saw older people using and extending their IT skills to be able to communicate with family, challenging previous assumptions. Carers or relatives were also considered as playing an important role when devices had to be used at a distance. Device recommendations seemed to be done on an informal basis, relying on case-by-case judgments and the patient–doctor relationship (see Quote 15).
Domain 5: Organisation(s)
Capacity and readiness to innovate
There seemed to be little organisational support for the additional work needed to embed IoT-based monitoring in care processes (especially considering the extent of false positive alerts), the funding models required, as well as reduced appetite to account for and cover potential medico-legal risks and insurance (see Quote 16).
From their perspective, innovators described the NHS and social care as risk averse and limited in their capacity to engage with innovations. There were common comparisons with healthcare systems in other countries, seen as superior for decision-making and experimentation capacity, whereas the NHS was seen as tied down by an effort to avoid liability (see Quote 17). There were also other cultural concerns at play; as IoT innovations shifted some of the responsibility and power for generating data to patients, this was an uncomfortable balance for a service with more paternalistic traditions, as it required a different way of engaging with patients and their role in the service (see Quote 18).
Beyond clinical reluctance, organisational limitations in terms of readiness for innovation seemed to hinder IoT adoption and spread. Clinicians spoke about organisational inertia and lack of incentives to adopt new ways of monitoring (see Quote 19).
One of the social care IoT providers (working across several local authorities) suggested that in areas with a dedicated ‘tech for care’ team there was better capacity for successful adoption. However, financial as well as reputational concerns generally complicated adoption processes in social care, especially when innovations were still at early stages of development.
Nature of adoption and funding decisions
Innovators also discussed extensive limitations of current funding models for IoT spread and sustainability, including the disconnect between the policy vision for innovation and the reality; slow cycles of fixed-term grant funding, availability of innovation funding without the capacity for healthcare organisations to take advantage of it, and lack of focus from the payer on long-term/system benefits yet the somewhat disparate requirement for robust evidence (see Quote 20).
Other innovators commented on the difficult-to-reconcile tensions between ‘move fast and break things’ approaches to innovation and the robust evidence base needed for implementation in healthcare compared to other industries.
Social care and local authority teams also grappled with the complexity of innovation funding, in terms of resource scarcity, add-on costs for data-intensive technologies, and the additional formality and complexity of social care and local authority tendering and contracting processes. Such processes are less amenable to dynamic technology companies (see Quote 21).
Extent of change to organisational routines
Technologies which required fewer changes in organisational routines seemed to integrate more easily into existing pathways, but from an innovator's perspective this appeared to be limiting as they saw more value in providing solutions that apparently ‘disrupted’ established processes rather than sustaining usual practice (see Quote 22).
Domain 6: Wider system
Interorganisational networking and collaboration
The local system supporting innovation consisted of a complex configuration of academic and research institutions, a major university-owned technology transfer and consultancy company, a large health and social care innovation team based with the local authority, as well as different types of innovation incubators and accelerators providing training, coaching, networking opportunities, workspace, access to funding and general business support (based on a mapping exercise we conducted as part of this study). Collaborative partnerships across institutions had been established to support exchange, especially between the NHS and industry, and to support implementation of government-backed innovation programmes.
Some of the support provided by this ecosystem was identified as helpful, in terms of opening up important networks, bringing people together, linking ideas, even providing workspace (see Quote 23). However, with academic and research institutions playing such a large, anchoring role within this ecosystem, some industry interviewees highlighted difficulties when holding an ‘outsider’ role, and challenges in creating strong supportive networks and aligning with priorities of other stakeholders.
Support required to grow innovation
The majority of interviewees from innovation companies were critical of the support provided in the local ecosystem, challenging the extent to which accelerator programmes met their intended aims and suggesting that structured, one-size-fits-all support, no matter how well-meaning, could in fact become distracting for innovators, especially at later stage product development. Instead, they valued ad hoc, informal support structured around their specific needs, and increased exchange and collaboration between innovation companies to strengthen the ecosystem as a whole (see Quote 24 and Quote 25).
Development and implementation of IoT solutions took place in what was described as ‘a political space’ where clinical, academic and commercial worlds collide, and different motives needed to be reconciled and accommodated (e.g. furthering academic knowledge versus building solutions that sell). Interviewees suggested that the ecosystem became blind-sighted by the need to prioritise academic achievements (such as high-impact journal articles) and placed less emphasis on developing successful commercial technological ventures, with capacity taken up by centrally mandated solutions rather than innovations developed locally (see Quote 26 and Quote 27).
Procurement processes and regulatory/legal issues
Aside from local ecosystem support, commissioning and payment processes were generally seen as time-consuming and challenging, or when funds were available they often had to be spent fast without due consideration for the relevance of solutions purchased or their sustainability in care pathways (see Quote 28).
One provider seeking to promote an entirely new service and redesign a pathway around their technology described how the NHS procurement process ‘is set up for devices, not for change or innovation as they are always looking to compare at least three options. They need a comparator in their mind’. [Interview 18, Innovator].
Broader challenges were also identified with government procurement and purchasing processes, especially during the COVID-19 pandemic (see Quote 29). An even bigger challenge reported by this relatively large industry provider was around meeting clinical and information governance requirements that differed between Hospital Trusts (see Quote 30).
Sociocultural context
Interviewees agreed there were difficulties progressing innovation projects in the context of the COVID-19 pandemic (e.g. having to stop clinical trials, or being unable to meet in person), but physical distancing requirements had other positive impacts in raising ‘the profile of digital health’ and accelerating adoption. Some companies suggested they saw ‘a peak of sales’ to Hospital Trusts during this period (e.g. heart rhythm monitoring equipment) but in other settings, such as social care, firefighting took precedence and there was little capacity (or funding) to implement new remote monitoring solutions (see Quote 31).
Domain 7: Adaptation and sustainability over time
Opportunities and organisational resilience
Remote monitoring had become more attractive since the pandemic as it enabled some care continuity without the need for face-to-face consultations. Interviewees described the nationwide shift to remote downloads and battery checks for well-established implantable cardiac devices such as permanent pacemakers and implantable cardiac defibrillators, avoiding the need for clinic attendance. On a smaller scale, newer technologies enabling patient-initiated self-monitoring were also deployed to facilitate remote delivery of care. If the trend of increased patient-initiated self-monitoring became more mainstream, it would test the robustness of the service and its capacity to safely deal with larger-scale remote monitoring. Little attention had been paid to developing the conditions that would allow remote monitoring to become routine and, as one of the clinicians admitted, increased use of patient-owned devices providing ECG traces would not be sustainable in terms of workload (see Quote 32).
Scope for adaptation over time
Adaptation and sustainability was discussed at organisational and system levels. Innovators were largely critical of large hospital systems that did not have the agility to respond to emerging needs compared to smaller and more responsive competitors in the COVID-19 context (see Quote 33). At system level, the local area was seen as geared more towards academic needs and timelines, which did not always match the adaptability and fast turnaround times start-ups and established companies needed (see Quote 34).
Discussion
Principal findings
This qualitative study examined opportunities and challenges around IoT implementation, spread and scale-up, with a specific focus on the role of innovation ‘ecosystems’ as facilitators of good-value technology-supported health and care. Interviewees with clinical, entrepreneurial and broader experience with innovation support, discussed the tensions and trade-offs they faced (including during the early stages of the COVID-19 pandemic) drawing on a range of IoT development and adoption efforts (see Figure 2 for an overview of findings).

Overview of findings mapped across the NASSS domains. NASSS: non-{PI}adoption, abandonment, scale-{PI}up, spread, sustainability.
Interviewee accounts centred around tensions in establishing the value proposition (financial and non-financial), which in most cases remained conditional given IoT innovations were primarily used in ad hoc, experimental ways and at small scale (if at all). Perceived value of IoT innovations was largely dependent on the aims different stakeholder groups were seeking to achieve and the costs they were prepared to incur, for example, in piloting different solutions for reliable monitoring of patients at a distance to prevent unplanned hospital admissions. There were also concerns that widespread use of IoT innovations in the health service would create disparities and inequalities if not centrally funded, as well as contributing to digital and social exclusion. Health and care professionals stressed difficulties with IoT integration in legacy infrastructures and uncertainty about interpretation of data generated by some IoT innovations (e.g. especially those without regulatory clearance), including the additional workload data interpretation and communication would entail. Even when interviewees recognised potential for improved care in specific cases, this was still hindered by lack of organisational capacity and readiness for routine use of innovations, resource scarcity and liability concerns.
Several tensions and challenges were also identified from the perspective of innovators. For example, common framings of innovation as ‘disruption’ came at odds with the reality of health and social care where short-term needs were prioritised and specific types of evidence were valued. In some cases there was a fundamental divergence between healthcare service providers’ aspiration to adjust and improve existing care delivery mechanisms versus some innovators vision to break the mould with approaches that entailed disruption of the status quo. This led industry stakeholders to characterise the service as resistant to change and risk averse, with innovation attempts compounded by the difficulties navigating multiple organisational boundaries with different procurement and governance processes. Some gains in IoT adoption were made during the pandemic, but without attention to underlying infrastructures, it was unclear whether these would be sustained.
Although various ecosystem actors were tasked with facilitating innovation in the local area, the priorities of academic and research institutions played such an anchoring role, that innovators found it difficult to navigate support networks, and to align between academic and commercial timelines and competing priorities in what was described as a ‘political space’. Partnerships between the NHS and industry were identified as helpful for those at start-up phase, in terms of opening up important networks and providing resources. Yet tailored support for those further down the innovation journey appeared to be missing and there was a general impression of siloed working and fragmentation. Other systems, such as in the United States and Europe, were considered more responsive to innovation. In the local ecosystem innovators also described distinct concerns about limited options for translational research, and lack of opportunities to network and connect with key players.
Strengths and limitations
Although our sample is relatively small, it represents a wide range of clinical, industry and other stakeholder perspectives on innovation development and deployment in health and care systems, as well as interviewees with dual academic roles and experience. Further interviews with academic stakeholders and with patients would broaden our understanding of opportunities and challenges, although this perspective has been covered elsewhere.23,29,36 Using NASSS as our analytical framework enabled us to systematically surface different aspects of complexity in the accounts of our interviewees, with a view to understanding how regional innovation ecosystems can be strengthened. Negative views about the role of regional ecosystems may be overrepresented as actors who have had worse experiences may have been more likely to agree to participate in the study, although we did also interview stakeholders from established and successful companies.
Comparison with prior work
Previous work examining digital health ecosystems has considered their role in the delivery of national technology programmes at scale.13,50 Our study focused instead on a local digital health ecosystem in a specific region in the United Kingdom, to develop a detailed understanding of opportunities and challenges with growing, supporting and adopting innovations locally. We highlighted the dual role of academic actors that both enabled and hindered innovation development, by providing opportunities and exposure, while at the same time making it more difficult to address competing timelines, resource constraints and priorities. Siloed, fragmented working was common in this ecosystem and despite an abundance of training opportunities in an environment heavily oriented towards education and capacity-building, tailored support for innovators did not appear as readily available as expected. Our study fills a gap identified by previous survey research which called for more qualitative studies on tri-sectoral collaboration between academia, healthcare and industry. 51
As we carried out this research during the early stages of the COVID-19 pandemic in the United Kingdom, we have also been able to observe the influence of the pandemic and infection control measures on IoT development and roll-out. We found that although the value proposition for IoT innovations became more prominent in some sectors, there was less demand than expected during national lockdowns in other sectors such as social care. Other research pertaining to the COVID-19 pandemic also found that there was a dramatic uptake of IoT innovations in health settings during the COVID-19 pandemic, 52 for example, wearable devices for monitoring of vital signs in hospital settings. However, Kaihlanen et al. 53 found that while IoT was more prominent during the COVID-19 pandemic, IoT devices were not always preferred by patients, and there were significant developmental needs to improve equal access. Further, while there appears to be less focus on the lower uptake of IoT in social care, Anderson et al. 54 contended that the COVID-19 pandemic has emphasised the ‘chronic underfunding of social care’, and requirements for investment and improvements in resource management.
Our study further confirms findings from previous IoT research highlighting the importance of digital literacy, infrastructure, professional responsibility and capacity, including the time required for clinicians to interpret IoT-generated data. 13 We elaborate on similar issues in a separate publication that focuses on the usefulness of patient-generated IoT data for heart monitoring. 55 Clinical training and guidelines may further support responsible use of higher value IoT innovations and support roll-out. Such guidelines and training may consider Smuck et al.'s 56 key features of successfully integrated IoT innovations: determining a clearly defined problem or need, providing technology support, personalised technology (for the identified need), aligned payment/reimbursement models, and clinician champions/support.
Our study confirms that the complexity inherent in integrating IoT innovations into care pathways necessitates clinical partnership and understanding. Supportive partnerships are likely to increase the chances of collaboration and sustainability, creating a network for generating consensus over the credibility and legitimacy of technology products, supporting mitigation of potential obstructions, and providing access to responsive and agile financial support. 57 For example, supporting start-ups to access resources and expertise for robust evaluations to meet the needs and priorities of direct and indirect users, and to address regulatory and purchasing requirements.56,58 To overcome fragmentation, innovation hubs could play a significant role in IoT ecosystems if they provide a relevant and ‘enabling space’ to negotiate contrasting motives and processes, bring together expertise and create collaborative connections. 37 Coordinated leadership could further assist in understanding and aligning clinical and academic priorities, as well as opportunities for access to funding and partnerships.59
Conclusions
Although ecosystems with strong academic, commercial and clinical actors appear well placed to support innovation, in many cases co-ordination and alignment costs seem to hinder efforts at meaningful exchange and impact creation. The development, implementation and roll-out of IoT innovations for health and care presents particular challenges as their value proposition (including non-financial) to intended users and organisations is not always clearly articulated outside experimental settings. System support is necessary to enable identification and adoption of useful devices in health and care settings in ways that meet ongoing needs for patients and practitioners, with emphasis on training, infrastructure and equity.
Footnotes
Abbreviations
Acknowledgements
We would like to thank Gemma Hughes and Joe Wherton for contribution to two of the interviews.
Contributorship
CP, AJ and MM-C conceptualised the study and acquired funding. CA, CP, WJ, CR, AR and MM-C carried out data collection and analysis. CP and CR co-authored the manuscript, with input from CA, AJ and MM-C. All authors have approved the final version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The project received approval from the Medical Sciences Interdivisional Research Ethics Committee (IDREC) at the University of Oxford (R68653/RE001).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been funded by the Pitch-In programme, a Research England's Connecting Capability Fund project.
Guarantor
CP.
Appendix: 1: Illustrative quotes
| Domain/sub-domain heading | Quote number | Quote |
|---|---|---|
| Domain 1: Patient condition | ||
|
|
Quote 1 | ‘So I had a sixteen year old guy who came to see me with his father and he’d been having really troublesome palpitations, rapid heart rates, feeling unwell with it, never had a diagnosis. And I said to him well you could buy one of these devices and within a week he’d got back to me with a recording of what was his SVT and we did an EP study and ablated his accessory pathway and that was him sorted out for good, within a really short time frame that's transformed things for him’. [Interview 2, Clinician] |
| Quote 2 | ‘My sort of vision for the heart failure service would be to have a patient portal where you have Bluetooth enabled weighing scales and Bluetooth enabled blood pressure monitors and that gets uploaded automatically to that patient's portal […] I think that's the way forward…’ [Interview 7, Clinician] | |
|
|
||
|
|
Quote 3 | ‘…artificial intelligence modalities to help govern and interpret some of the data that comes out because potentially maybe the AI [plays a] much better role at picking out those trends in the vast amounts of data that have been generated’. [Interview 1, Clinician] |
|
|
Quote 4 | ‘So it was a challenge for people who had quite limiting symptoms, when the frequency of those symptoms was less than every 2 weeks, so we struggled with that in the past. But now having a device like a [device name] monitor… I’d say [I recommend it] probably a couple of times a week and I sometimes feel obliged to say I don't have any shares in it because I wish I did, I mean I think it's got a huge market and its clinically very useful’. [Interview 2, Clinician] |
|
|
Quote 5 | ‘That's one of the troubles with the [device] is that the P waves aren't always that easy to see. In which case it might be difficult on the basis of the traces alone to be sure it's an SVT rather than sinus tachycardia’. [Interview 2, Clinician] |
|
|
Quote 6 | ‘I met with the GP on Friday and it could connect to his phone straight away without doing anything, but he was having real trouble connecting to his PC with[in] the practice, so it was the same GP with the same level of skill obviously but one piece of equipment was letting him down and one was connecting automatically’. [Interview 4, R5, Innovator] |
|
|
||
|
|
Quote 7 | ‘…it's another way in which there is inequality isn't it. Because if someone can afford it and they’re more likely to pick up a problem and they’re more likely to get the right treatment and someone else can't afford it and they’re potentially more likely to have a stroke. But I think the way in which we need to adapt to be able to facilitate using these devices and trusting people that they’ll use them and give them back’. [Interview 2, Clinician] |
| Quote 8 | ‘I don't like the idea that we have special things for old people and then decent tech for the rest of us […] what bugs me the most was that they can't talk back [to the device, because of its simplified functionality] […] when you give a 75-year old lady a piece of kit I want her to use something that her grandchildren have […] that everybody has so that she's included instead of it being this weirdo tech for grandma’. [Interview 12, Stakeholder] | |
| Quote 9 | ‘No I tend not to [ask for patients’ home monitored blood pressure, heart rate data or ECG traces] simply because most of the patients I see will get a 12 lead ECG when they come to clinic. Now I do, I have been using it during Covid so during telephone consultations that's all I’ve got to go on so I want everything that they’ve got basically’. [Interview 7, Clinician] | |
|
|
Quote 10 | ‘…we’ve got a product that works, why don't people buy it […] So by that time is when we really started taking clinical validation a bit more seriously […] we are the only patch-based product that has a peer-reviewed publication that shows clinical benefit, everything else just shows it works, we can show it works and leads to treatment in sepsis six hours quicker than the current standard of care. And we’re the only patch-based system that has a peer-reviewed health economic publication that shows that we can, in this instance, deliver savings of £466 per patch patient’. [Interview 15, Innovator] |
| Quote 11 | ‘The reason we were able to get into local authorities was because, 10 years before, another company was put on market for short term assessment. They cornered the market. So the use of IoT for “evidencing” was already well known – so when we came along with the consumer proposition – it was smarter, sexier and cheaper. It was an easy sell into local authorities. So we chipped away at [the other company's] market share’. [Interview 8, Innovator] | |
|
|
||
|
|
Quote 12 | ‘…it's almost sort of information overload because you feel obliged as a doctor to, to look at that data in case you’re missing something’. [Interview 7, Clinician] |
| Quote 13 | ‘I think the sort of patient initiated stuff that you talked about I think is not well integrated yet, at the moment, and you know there's quite a lot of colleagues who are less tolerant than me and just don't want to know about it’. [Interview 1, Clinician] | |
|
|
Quote 14 | ‘…the challenge is people need to know that we need a trace we don't just need a number, so if someone's got a Garmin or a Fitbit the number itself or a graph of the numbers is rarely of any use’. [Interview 2, Clinician] |
| Quote 15 | ‘…it's very dependent on patient compliance and cooperation and there are some people who obviously can't do it because of cognitive factors and usually you can get around that through their carers or relatives, but not always’. [Interview 1, Clinician] | |
|
|
||
|
|
Quote 16 | ‘That is, becoming, to many clinicians a challenge how to view them [new healthcare technologies], what is the governance around it and what is the remuneration around it’. [Interview 6, Clinician] |
| Quote 17 | ‘…my feeling from the NHS is very much risk averse - no-one's ever got fired for not buying something but I’m sure a bunch of people got fired for buying things […] there is better funded healthcare systems in the Netherlands […] they have more capacity, less issues […] less firefighting, but they have more of an ability to make decisions on things and to try new things and to have a go’. [Interview 15, Innovator] | |
| Quote 18 | ‘But if you speak to somebody over here [in the UK] about allowing the patient to book their own appointments, you know, and to manage that people are just filled with dread’. [Interview 9, Innovator] | |
| Quote 19 | ‘Yes now that [Internet connected BP and weight monitoring] is something I would like to use and, and in fact I’ve suggested it […] and I find sometimes things are a bit slow in [Trust name] in terms of the take up of these technologies […] it's sort of a mentality of we’ve always done it this way and, and you know, it's working so why, why do we have to change’. [Interview 7, Clinician] | |
|
|
Quote 20 | ‘It's very hard for digital health business to have a viable model, particularly going into the NHS where it gets paid for. There's a lot of great talk and ambition. But when you go to a trust and say we’ve got this which has a clear benefit, economically driven, there's still an intake of breath and “I don't know”’. [Interview 17, Innovator] |
| Quote 21 | ‘No idea, I don't have a budget, I go around with my begging bowl…I will be trying my luck this year’. [Interview 14, Stakeholder] | |
|
|
Quote 22 | ‘So ideally you want products that are most closely aligned to a care pathway for minimum disruption, but … actually what you do want is to maximise disruption to make it much, much better. So you’ve got that kind of tension between the two’. [Interview 17, Innovator] |
|
|
||
|
|
Quote 23 | ‘… they [a University-backed accelerator and skills training programme] very kindly let us stay on for the subsequent two years and giving us free office space […] And at the same time opening up their network to us, a network of entrepreneurs, investors and specific experts in different domains like marketing or branding or HR and so on […] getting some expert advice helped us save a lot of time and running around […]’. [Interview 16, Innovator] |
|
|
Quote 24 | ‘And I know there's numerous you know, things being put in place to help companies like us […] so we’ve explored all of these avenues…With really quite negative stories…Yeah so I have found a lot of this stuff that's been put in place to help us particularly unhelpful’. [Interview 15, Innovator] |
| Quote 25 | ‘The plural of silo is not ecosystem… so you can't have a bunch of silo companies in [location] that don't talk to each other and then call that an ecosystem…So you can dress it up and tweet as much as you want but it's not happening’. [Interview 11, Innovator] | |
| Quote 26 | ‘And we’re in a political space. I would say that the partnership side of things hasn't always been easy because you’ve always got tensions between clinicians and academic systems. So that's made it harder. And you’re really transitioning from a research product to a commercial product, you know takes a bit of time’. [Interview 17, Innovator] | |
| Quote 27 | ‘Priorities for AHSNs [Academic Health Science Networks] end up being centrally driven rather than locally, unlike as was originally intended. So that reduces the amount of local stuff that the local projects that can actually be picked up because their capacity is what it is’. [Interview 10, Stakeholder] | |
|
|
Quote 28 | ‘And I think pressures of getting money out of the system often forces commissioners to buy products that sometimes aren't quite right…’ [Interview 4, R1, Innovator] |
| Quote 29 | ‘We spent the last 6-7 years wishing NHS got more involved in TEC [Technology-Enabled Care], and the last 6 months wishing they had not - because engagement with market has been nothing short of shambolic. It's been horrific. To be a supplier and what they put the market through…’ [Interview 8, Innovator] | |
| Quote 30 | ‘…we could do with a one stop shop for clinical governance and information governance because we are going through that individually with every single Trust…opinions differ, processes differ, the questions that come back are so different, they go so deep down a rabbit hole…it's a massive blocker for adoption. We’re alright-we have a big team-but it must be difficult if not impossible for small teams’. [Interview 18, Innovator] | |
|
|
Quote 31 | ‘I would have predicted more queries coming through during lockdowns because you can't physically go out so remote monitoring would have been useful…I would have predicted that would be in more demands but that's not what's happening […] [The technology team] were getting in contact with solutions, but everyone was so consumed with firefighting and that everyone [all clients] had had a contact and they were okay so nothing happened’. [Interview 14, Stakeholder] |
|
|
||
| Opportunities and organisational resilience | Quote 32 | ‘The problem is that we’d actually do that for free if they sent them to us [i.e., looking at the data]. But the more we recommend it, the more of a strain that's gonna be in terms of trying to provide that service’. [Interview 2, Clinician] |
| Scope for adaptation over time | Quote 33 | ‘I think the other aspect is they [new remote consultation IT provider] were really, really agile…whereas [mainstream hospital messaging system]… it's been a nightmare because the product's not good enough and they can't change it, whereas [new remote consultation IT provider] were straight in there changing and adapting the product […]’. [Interview 4, R1, Innovator] |
| Quote 34 | ‘The timelines and the attitudes [within the local context] are a little bit more academic than a start-up agile and commercial’. [Interview 16, Clinician] | |
AI: artificial intelligence; BP: blood pressure; ECG: electrocardiogram; EP: electrophysiological; GP: general physician; HR: human resources; HR: human resources; IoT: Internet of Things; IT: information technology; NHS: National Health Service; PC: personal computer; SVT: supraventricular tachycardia.
