Abstract
Introduction
Continuous monitoring is the hallmark of managing chronic disease. Multiple sclerosis (MS), in particular, requires patients to visit their treating neurologists typically twice a year, at least. In that respect, the COVID-19 pandemic made us rethink our communication strategies. This study determined satisfaction with remote visits for people with MS (pwMS) by comparing non-inferiority to conventional visits.
Methods
TELE MS was a randomized controlled trial that was open to any person with MS. We randomized a volunteer sample of 45 patients. We compared satisfaction with remote visits (via phone or via videochat) with conventional outpatient visits. The primary endpoint was patient satisfaction determined by the Telemedicine Perception Questionnaire (TMPQ, min: 17 and max: 85 points) with the hypothesis of non-inferiority of televisits to conventional visits. Physician satisfaction measured on the PPSM score (Patient and Physician Satisfaction with Monitoring, min: 5 and max: 25 points) was the secondary endpoint.
Results
The trial met both endpoints. Mean (SD) TMPQ scores in the individual groups were 58 (6.7) points for conventional visits, 65 (7.5) points for phone visits, and 62 (5.5) points for video visits. Physician satisfaction over the whole cohort was similarly high. Median (range) PPSM scores were 23 (16–25) for the whole cohort, 19 (16–25) for conventional visits, 25 (17–25) for phone visits, and 25 (16–25) for video visits.
Conclusions
Televisits in multiple sclerosis yield a high level of satisfaction for both patients and treating physicians. This concept for remote patient monitoring adopted during the current pandemic may be communicable to other chronic diseases as well.
Introduction
Multiple sclerosis (MS) is a chronic disease that requires regular care. It affects about 2.5 million people worldwide and is the most frequent cause of disability in young adults, aside from trauma. 1 . 2 Monitoring the health status of people with MS (pwMS) encompasses periodic visits to their treating neurologists in order to review the most recent patient history, receive a clinical assessment and discuss the latest paraclinical exams. 3 In light of the current pandemic, we have faced an interruption to face-to-face care. Beyond doubt, this exemplifies an imperative need to develop, offer and implement strategies to maintain consistent monitoring and create an opportunity for the exchange of health information. 4
The hallmark of telehealth is the provision of remote healthcare as it can sustain disease management without the physical co-presence of the patient and physician.5–7 The majority of recent telehealth applications in the field of MS centered on telerehabilitation and biosensors. These studies illustrated, among others, attenuated adherence and improved quality of life as palpable benefits.8–11 In contrast, only a handful of investigations focused on remote visits per se. One randomized crossover trial reported feasibility of remote monitoring demonstrated by the percentage of completed televisits compared to conventional outpatient appointments. 12 Another study concluded that pwMS highly appreciate scheduled teleconsultations, which may even enhance access to specialized care. 13 However, these investigations lack an association between mere acceptance of telehealth on one side and MS-related patient reported outcomes or standardized clinical outcome measures on the other (such as the Expanded Disability Status Scale (EDSS) 14 or quality of life). Furthermore, these observations did not account for physician satisfaction. It remains unknown how satisfaction with remote visits compares to regular visits in the view of the patient and physician.
Therefore, in this randomized controlled trial, we aimed to demonstrate non-inferiority of remote visits in pwMS based on satisfaction expressed by both patients and physicians. We used standardized and validated outcome measures to test our hypotheses. Additionally, we performed ancillary analyses to identify patients particularly pleased with this alternative form of healthcare delivery.
Methods
Ethics review, consent and trial registration
The ethics review board at the Medical University of Vienna approved this study (EK2157/2020). We obtained informed written consent from all study participants and followed guidelines set by the Declaration of Helsinki. We adhered to CONSORT guidelines when writing this report. 15 This study is registered with ClinicalTrials.gov (identifier: NCT04838990).
Trial design and participants
TELE MS was a single center parallel assignment unblinded randomized controlled trial designed to demonstrate non-inferiority of remote visits (video visits or phone visits) compared to conventional visits (control group). We enrolled 45 patients and randomized them 1:1:1 into three study groups (one control group and two intervention groups). The three groups differed in their type of follow up visits. Patients treated at the MS outpatient clinic at the Department of Neurology at the Medical University of Vienna, Austria, and fulfilling current McDonald diagnostic criteria for MS were eligible to participate in this study. 16 Study inclusion was irrespective of parameters such as age, gender, disease phenotype, disease duration or current treatment. Exclusion criteria were obvious language barriers or technical obstacles (no smart device that would enable video chatting, no phone or no internet connection). There was no change in protocol or re-allocation of study participants once the first patient was enrolled. Treating neurologists documented, in a structured format, clinical characteristics of participating patients including age and gender, disease duration in years, disease phenotype (relapsing MS, progressive MS, 17 ) neurological disability on the EDSS, 14 number of relapses over the past 12 months, and disease-modifying treatment (DMT, categorized as moderately effective [dimethyl fumarate, glatiramer acetate, interferons, teriflunomide], highly effective treatment [cladribine, fingolimod, natalizumab, ocrelizumab, rituximab]) or no treatment.
For interpreting our data, it is important to discuss our national COVID-related restrictions concerning patient care. As stated later, our study was open from November 2020 through April 2021, a time when Austria experienced a nationwide lockdown. For our tertiary care center, we were encouraged to limit patient contact to a tolerable and safe minimum as to not expose our patients to a risk of contracting COVID-19. These measures, however, had no influence on patient recruitment as our study participants were recruited on site during a regular visit and the subsequent study visits were randomized.
Interventions and outcomes
Figure 1 illustrates the overview of the trial design. We informed patients about the study protocol, which consisted of five steps. First, the regular (baseline) visit at the MS outpatient clinic at which study participation was offered and baseline disease characteristics were assessed (age, gender, disease phenotype, disease duration, number of relapses in the past year, EDSS, and Multiple Sclerosis Impact Scale [MSIS]. 18 Second, a randomized allocation to one of the three intervention groups determining the type of follow-up visit (a conventional physical visit to our outpatient department as a control group or a visit and clinical exam over the phone or over a videochat application). The time of the study visit was determined consensually between the patient and treating physician at baseline and followed the usual frequency of appointments. Third, a self-assessment filled out 3–7 days prior to the subsequent study visit by means of an online tool, in which participants could enter a self-report on their health status in their own words and fill out an MSIS questionnaire. The treating physician would read these reports before the visit and document them in the patient record along with the MSIS scores. Fourth, the actual study visit for which the participating patient and treating neurologist would meet at the scheduled appointment. Over the course of this visit, doctor and patient could discuss the patients’ current condition and other relevant information (e.g. laboratory reports, radiological reports, information on adherence to medication, etc.). Furthermore, physicians assessed follow up scores on the EDSS either using the conventional physical method (control group) or the phone EDSS (phone or video group) which is a comparable and validated tool to assess EDSS scores remotely. 19 In addition, participating neurologists filled out the PPSM (Patient and Physician Satisfaction with Monitoring) questionnaires, a five-question validated survey to rate the respective study visit. This score has been validated in German and each question counts up to five points, leading to a maximum of 25 points with higher points indicating greater satisfaction. Fifth, the day after the study visit, participating patients received an invitation for an online tool where they could fill out the TMPQ (Telemedicine Perception Questionnaire) and they were offered the opportunity to give open comments. The TMPQ is a validated 17-item survey, through which patients can rate their experience with a televisit 20 . 21 Similar to the PPSM, each question is worth up to five points and higher scores demonstrate greater satisfaction.

Study overview of the TELE MS RCT. MSIS-29 – Multiple Sclerosis Impact Scale, MS – Multiple sclerosis, MSologist – Neurologist treating persons with MS, PPSM – Patient and Physician Satisfaction with Monitoring, RCT – Randomized Controlled Trial, TMPQ – Telemedicine Perception Questionnaire.
The primary endpoint was patient satisfaction measured by the TMPQ to determine non-inferiority of televisits compared to a conventional visit. We considered a difference of 10% (≥9 points) to be clinically meaningful. The secondary endpoint was physician satisfaction (with the hypothesis of non-inferiority) measured by the PPSM. We considered a difference of 10% (≥3 points) as relevant. Exploratory outcomes were determined with respect to identifying patients particularly satisfied with their remote visits translating as high scores on the TMPQ.
Sample size, randomization and implementation
Sample size was determined based on the hypothesis of non-inferiority defined as a <10 point difference in the primary endpoint at 80% power and an alpha-error of 5%. We performed randomization by preparing 45 concealed envelopes that would determine the study group (15 for each of the three groups). Upon enrollment, patients would randomly draw an envelope that would irrevocably allocate them to their respective study group. After this process, the envelope was discarded.
All participating neurologists were board certified in neurology and were trained and examined EDSS raters for both the conventional EDSS and phone EDSS. 19
Considerations of data protection and privacy
Our study protocol adhered to current and strict data protection guidelines set by both the country of Austria and the Medical University of Vienna as of December 1st, 2020. Patients were educated on these guidelines on the informed consent form, which was approved by our local ethics review board. Patients consented to having their relevant study information implemented into their regular patient record (i.e. MSIS scores plus a copy of their self-description). The provision of questionnaires through an online tool (self-description and satisfaction) was enabled via an academic platform widely used in German speaking countries (http://www.soscisurvey.de). Only one of the authors involved in this study had access to the data on that platform. All information on the survey portal was stored in a pseudo-anonymized manner through a study identification number (study ID) which we assigned to participants upon enrollment. For the video visit group we used an end-to-end encrypted video chat system that would operate on personal computers, smartphones or tablets all the same (Webex by Cisco, Milpitas, CA, USA).
Statistical analyses
Statistical analysis was performed using SPSS 26.0 (SPSS Inc, Chicago, IL, USA). We report categorical variables as frequencies and percentages, continuous variables as mean and standard deviation or median and interquartile range or range as appropriate. Continuous variables for normal distribution were tested by the Kolmogorov-Smirnov test with Lilliefors correction. Group comparisons were calculated by independent analyses of variance (ANOVA), Kruskal-Wallis-Test or by chi-square test A value of p <0.05 was considered statistically significant. All multiple analyses were corrected using Bonferroni method. We performed our final analysis in a per-protocol manner.
Results
Patient characteristics
Figure 2 demonstrates the patient flow. We screened 49 patients for eligibility. Four patients declined participation upon informed consent stating the following reasons: (i) no internet connection at home (n = 1), (ii) not wanting to be allocated to the remote visit group (n = 1), (iii) not being interested without giving a reason (n = 2). Three participants dropped out over the course of the observational period due to (i) scheduling conflicts (n = 1) and (ii) incomplete or missing satisfaction questionnaires (n = 2). Recruitment was open from November 2020 (first patient in) and lasted until April 2021 (last patient in). Study visits took place from April 2021 (first patient out) through September 2021 (last patient out). The trial ended with data analysis in October 2021. Table 1 lists clinical information and patient reported outcome measures from 42 patients who completed this study and were included in the final analysis. The mean age in our cohort was 34 (SD 8) years, gender distribution was 2:1 (f:m) and 37 patients had relapsing MS (88%). Patient characteristics were distributed evenly between all three intervention groups. Two patients experienced a disease relapse over the course of the observation period (one on moderate and one on highly active treatment).

CONSORT flow diagram for the TELE MS trial. CONSORT 15 flow diagram showing participant flow through each stage of the TELE MS randomized open controlled trial (enrollment, intervention allocation, follow-up and final analysis).
Demographic and clinical characteristics for the whole cohort and each intervention group.

EDSS: Expanded Disability Status Scale, MS: multiple sclerosis, MSIS: Multiple Sclerosis Impact Scale, N/A: not applicable or not relevant, n.s.: statistically not significant, Δ: change in value.
Primary endpoint: Patient satisfaction
Figure 3 demonstrates patient satisfaction with the study visits determined on the TMPQ and displayed for the whole cohort and the three study groups. For the whole cohort, the mean satisfaction measured on the TMPQ was 62 (SD 7) points. Mean scores in the individual groups were: 58 (SD 6.7) points for conventional visits, 65 (SD 7.5) points for phone visits, and 62 (SD 5.5) points for video visits. Mean scores for the phone and video visits were slightly higher than in the control group. However, as the lower border of the 95% confidence interval in both the phone and video visit groups were less than 10 points below the mean score in the conventional group, our results fall within the predetermined threshold for non-inferiority (respective differences were 2.6 points [conventional vs. phone] and 0.1 points [conventional vs. video]). We also asked patients to rate their visit experience on a Likert scale. Percentages of answers are presented in Table 2.

Patient satisfaction with the study visit. This box and whiskers plot shows scores on the TMPQ scale (y-axis, higher scores indicate greater satisfaction) for the three study groups and the whole cohort (x-axis). Boxes indicate the interquartile range and whiskers min-max. TMPQ – Telemedicine perception questionnaire.
Patients rating their TELE MS study visit.

Pooled results for favorable (“very good” or “good”), neutral, and unfavorable ratings (“very poor” or “poor”) on a Likert-scale.
Secondary endpoint: Physician satisfaction
Physician satisfaction over the whole cohort was high (Figure 4). Median PPSM scores were 23 (range: 16–25) for the whole cohort, 19 (range: 16–25) for conventional visits, 25 (range: 17–25) for phone visits, and 25 (range: 16–25) for video visits. Physician satisfaction did not differ statistically significant between the three groups (p = 0.175). The lower border of the 95% confidence interval in the remote visit groups were less than 3 points below the mean score in the conventional group, thus, the secondary endpoint of assumed non-inferiority was met (respective differences were 0.6 points [conventional vs. phone] and 0.4 points [conventional vs. video]). Moreover, we asked physicians participating in remote visits (n = 28) to state, whether they believe that individual visit had resulted in a loss of information. 25 (89%) gave a favorable rating (Likert scale, pooled for “strongly disagree” and “disagree”) and 3 (11%) an unfavorable rating (pooled for “strongly agree” and “agree”).

Physician satisfaction with the study visit. This box and whiskers plot shows scores on the PPMS scale (y-axis, higher scores indicate greater satisfaction) for the three study groups and the whole cohort (x-axis). Boxes indicate the interquartile range and whiskers min-max. PPSM – Physician and Patient Satisfaction with Monitoring.
Ancillary analyses
We investigated whether there was a certain group of patients inclined to be more satisfied with remote visits (Figure 5(a)-(f)). A detailed description is included in the supplement.
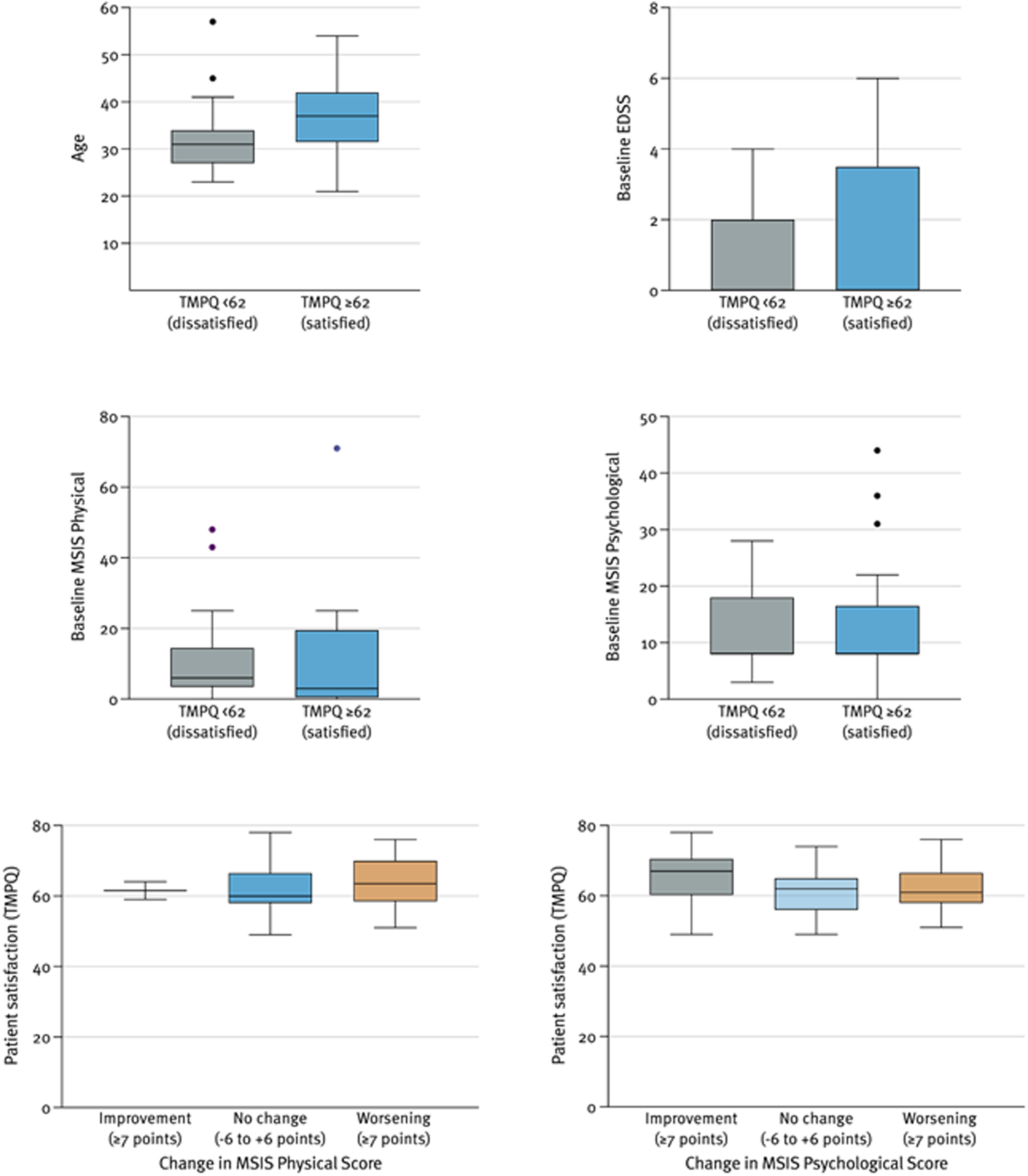
Patient characteristics in groups with satisfaction below and above this study's median TMPQ score. This box and whiskers plots illustrates patient characteristics (y-axis) along scores on the TMPQ scale (x-axis). In case there were no ourliers, boxes indicate the interquartile range and whiskers the min-max. Outliers, if existent, are indicated by black dots and, in that case, whiskers indicate values ranging from 1.5 times the IQR from the first and third quartile. TMPQ – Telemedicine perception questionnaire. Figure 5e-f Satisfaction score (TMPQ) in relation to change in the MSIS score. This box and whiskers plots illustrates patient characteristics (y-axis) along scores on the TMPQ scale (x-axis). Boxes indicate the interquartile range and whiskers min-max. TMPQ – Telemedicine perception questionnaire, MSIS – Multiple Sclerosis Impact Scale.
Open comments from patients
Table 3 lists all open statements that participating patients made available. These statements were collaborately translated by two authors without further interpretation.
Open statements from patients participating in the TELE MS trial.

Harms or unintended effects
We defined harm beforehand as patients reporting significant clinical worsening in their self-report, resulting in them expressing their wish to decline a remote visit in order to have a conventional visit instead (“group switch”). There were no such reports in this study, thus, we could not detect any harms or unintended effects of our interventions.
Discussion
The design of the neurological reflex hammer in the late nineteenth century arguably hailed the advent of the neurological exam 8 . 22 Although this neurological exam, along with radiological investigations, remains the mainstay of disease monitoring in pwMS, advancing patient care to a new era through the use of telemedicine may certainly be warranted 23 . 24 The COVID-19 pandemic has undoubtedly set a new tone in terms of communication strategies. In current literature, the availability of telehealth strategies has demonstrated a substantial benefit in terms of retaining an affinity with patient outcomes in MS.8–13 Studies have shown how telerehabilitation can improve physical functioning and enhance quality of life. 10 , 11 , 25 This development is facilitated by the availability of validated tools that can remotely monitor MS-related disability, for instance via a structured virtual neurological exam, the assessment of health-related quality of life or cognitive functions 26 . 27 There is emerging evidence that pwMS appreciate attempts in remote monitoring as they foster adherence and save time. Our present investigation emphasizes the importance of promoting telehealth concepts in pwMS as we provide evidence from a randomized controlled trial illustrating non-inferior satisfaction with remote visits over the phone or via videochat compared to conventional visits for both pwMS and their treating neurologists.
The majority of patients rated their study visit as satisfying (90%) with a trend towards greater satisfaction with phone visits. It was somewhat surprising that patients seemed to prefer to stay “invisible” during their consultation. While this observation could certainly be random, it definitely warrants further exploration as our study was not designed to determine reasons for divergent satisfaction. In follow-up visits outside the scope of this study, some patients mentioned that they would prefer phone visits to videochat, as they would not want their treating physician to judge their home situation. Moreover, they would feel less restricted in terms of where the remote visit would take place. Physicians conducting the study visits predominantly stated that remote visits did not result in a poor exchange of health information (in 89% of cases) and they did not per se favor conventional appointments over virtual ones. We would have expected to gain deeper insights into our patients’ preferences by performing subgroup analyses. Surprisingly, there were no significant differences identifying a specific population of patients more satisfied with their corresponding visit type. However, our study was not conceived to detect such nuances. We anticipated patients with more advanced or progressive disease to favor remote visits. There was statistical significance with respect to patients with progressive disease being more frequently satisfied with their remote visit. However, this hypothesis cannot be fully supported, as this group of patients was fairly underrepresented in our study (only 5 patients in the remote visit group had progressive MS). This is a common observation in studies with unselected recruiting of pwMS with disregard of their disease phenotype.
Our results should be discussed with regard to strengths and limitations. This randomized controlled trial depicts a real-life cohort with patient satisfaction as the primary endpoint. Patient satisfaction with an intervention is rarely a main endpoint in a clinical trial, yet we believe it is paramount when addressing a particular patient need 28 . 29 We decided to choose a non-inferiority study design as we did not aim to undermine the relevance of a conventional physical appointment. We selected both patient and physician satisfaction as the core of this trial as we wanted to convey the context of an equal doctor-patient relationship. Our study was sufficiently powered with respect to our primary endpoint, yet our sample size was small and, thus, limiting a potential benefit from subgroup analyses. Even so, the results from our investigation may be useful when designing similar studies. Additionally, patients hoping for a personal benefit from a remote visit were potentially more inclined to participate in this study, which could have introduced selection bias and, thus, possibly inflate satisfaction. However, only 4 out of 49 patients asked to participate declined participation, rendering a selection bias unlikely. The fact that we excluded persons with obvious language barriers may be considered a bias. We chose this mainly for safety and feasibility reasons. The main tool to determine our study endpoints (satisfaction questionnaires, MSIS, self-description of health status) required a certain level of ability to communicate in written language. We did not want to risk missing important information beneath this virtual setting. Furthermore, our study design did not include a qualitative survey regarding individual preferences of participants. Nonetheless, we used validated outcome measures throughout this study.
In terms of providing care to pwMS during the current pandemic, strategies for remote patient monitoring acquired greater importance and urgency. Our study provides insight into satisfaction with remote visits, which warrants further investigations in a real-world setting. It is tempting to speculate that, for some patients, recurring remote visits may be attractive. We learned from another study that pwMS appreciate remote visits in terms of them being cost-effective and timesaving. 12 This effect increased the farther participants resided from their clinic or hospital. In the future, it may be useful to conduct a single remote visit and determine, upon bilateral consent, the type of subsequent visit a few weeks before the next appointment. This setting would certainly complement a recent initiative adopted from follow up programs for people with gynecological malignancies: patient-initiated follow-up (PIFU). 30 In short, PIFU provides a framework to individualize patient care with particular interest in holistic needs and optimizing resources. Our present study suggests that perhaps it is not only the pace but also the medium that the patient should manage. That being said, we should appreciate that, currently, we lack guidelines on how to identify patients particularly suitable for remote monitoring. 31 Fortunately, there is an increasing body of evidence supporting the relevance of this topic. To our knowledge, there is no study reporting possible harms resulting from a remote visit. However, reliable follow up data, that would allow detecting potentially adverse effects from a one-time remote visit have not been communicated. Up to now, we do not know how patients with highly active disease (i.e. a high relapse rate) or particular needs (such as affective disorders, poor adherence) would respond to a virtual appointment setting. In our study, the relapse rate prior to entering this study was rather low at 0.5/year and only two patients experienced a relapse over the course of our observation period. In this context, open statements from this trial raised some interesting themes that warrant further exploration. Some comments suggested that satisfaction with remote visits may be context dependent, particularly considering that remote monitoring may not be considered satisfactory if presented as the sole option for care. We believe that no matter what the concept for teleconsultations may be, the patients perception on their health status should factor in these kinds of decisions. Furthermore, we should mention paraclinical exams for pwMS and how to address them in possible teleconsultations. Important paraclinical exams for treatment monitoring include repeated MRI scans and blood tests to monitor drug safety. In Austria, the majority of patients do these tests in outsourced facilities, e.g. specialized facilities for radiology or laboratory medicine. Thus, the running of these tests per se remains unaffected by televisits as their results can be discussed all the same. In addition, we should bear in mind that our study concerned primarily the interaction between patient and neurologist. Multidisciplinary consultations, e.g. with a team of physical therapists, occupational therapists etc., may require a different approach.
At last, we want to make a point to embrace new technology that has the potential to improve individualized patient care. With technology emerging and innovative methods of monitoring (disease) activity in pwMS becoming more and more accessible, we should beware not to inadvertently transform our doctor patient relationship into an exchange of health information between two avatars. Yet, we can use this knowledge to pursue a new narrative when it comes to holistic disease monitoring. The matter of reimbursement of televisits may certainly constitute a barrier to telehealth in general. In Austria, this has been a matter of debate, although there is general consent that televisits are reimbursable. As far as our study is concerned, participating physicians offered their consultations on their own time. It will certainly be interesting to observe how the dynamic imposed on modern patient care will evolve beyond the COVID-19 pandemic.
In summary, this randomized controlled trial demonstrates multifaceted aspects of satisfaction concerning remote visits in a sample of pwMS. Patients and treating physicians reported high levels of satisfaction with their virtual visit comparing favorably with regular physical visits. We provide solid evidence that concepts for remote visits in MS are feasible and worth exploring. The conclusions from our investigation may be communicable to other chronic diseases as well, bearing in mind individual patient needs and preferences.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076221112154 - Supplemental material for Remote visits for people with multiple sclerosis during the COVID-19 pandemic in Austria: The TELE MS randomized controlled trial
Supplemental material, sj-docx-1-dhj-10.1177_20552076221112154 for Remote visits for people with multiple sclerosis during the COVID-19 pandemic in Austria: The TELE MS randomized controlled trial by Patrick Altmann, Fritz Leutmezer, Markus Ponleitner, Dominik Ivkic, Nik Krajnc, Paulus Stefan Rommer, Thomas Berger and Gabriel Bsteh in Digital Health
Footnotes
Author contributions
Availability of data and materials
Data is available from the corresponding author upon reasonable request and after approval from the ethics review board at the Medical University of Vienna.
Competing interests
The authors declare there are no competing financial or non-financial interests relevant to this study.
Conflict of interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The ethics review board at the Medical University of Vienna approved this study (EK2157/2020).
Funding
This study was funded by the Medical University of Vienna.
Guarantor
Patrick Altmann
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
