Abstract
Case summary
A 15-year-old, spayed female domestic shorthair cat was evaluated for chronic progressive stertor, mucopurulent nasal discharge and intermittent apnoeic episodes. CT revealed a soft tissue mass occupying the nasopharynx and partially obstructing the choanae. Nasopharyngoscopy confirmed a firm, pale, multilobulated lesion. Partial endoscopic debulking of the mass was performed, although further sampling was limited by post-procedural haemorrhage. Histopathology demonstrated dense eosinophilic inflammation with marked sclerosing fibroplasia, consistent with feline eosinophilic sclerosing fibroplasia (FESF); no histological features suggestive of lymphoma, carcinoma, fungal disease or inflammatory polyps were identified. Prednisolone was initially administered to minimise the risk of post-procedural inflammatory nasopharyngeal stenosis and subsequently continued for 31 days after histological confirmation of FESF. Two months later, CT and nasopharyngoscopy demonstrated complete anatomical resolution with full remission of clinical signs.
Relevance and novel information
Nasopharyngeal involvement of FESF has not previously been reported; the condition is typically described in young to middle-aged cats with gastrointestinal disease. More recent reports describe its possible lymph node localisation in sites beyond the abdominal cavity. This case documents a geriatric-onset, extra-gastrointestinal presentation mimicking neoplasia or granulomatous disease. Diagnosis was achieved on biopsies obtained during partial debulking, and follow-up investigations confirmed complete anatomical resolution. This case expands the recognised anatomical and age spectrum of FESF and supports consideration of this entity in the differential diagnoses for nasopharyngeal masses in cats.
Keywords
Introduction
Feline gastrointestinal eosinophilic sclerosing fibroplasia (FGESF) is an inflammatory fibroproliferative condition initially recognised as a mass-forming gastrointestinal disease characterised by dense collagen trabeculae, activated fibroblasts and marked eosinophilic infiltrates. 1 It primarily affects the stomach, proximal small intestine and mesenteric lymph nodes, often mimicking neoplasia on imaging. 2 Lesions may develop insidiously and resemble metastatic disease, including in extraintestinal presentations. 3 Intralesional bacteria, typically Gram-positive cocci, were once considered central to pathogenesis, but later evidence showed their inconsistent presence, suggesting a non-essential role.2 –4
More recently, the term eosinophilic sclerosing fibroplasia has been extended to extraintestinal sites such as lymph nodes, subcutaneous tissues and the mediastinum, indicating a shared eosinophilic fibroinflammatory pathway irrespective of location.5,6
Despite this broader anatomical distribution, reported cases remain confined to abdominal and thoracic structures and predominantly involve young to middle-aged cats. Nasopharyngeal involvement has never been documented, and geriatric cases are rare.
This report describes the first known case of nasopharyngeal feline eosinophilic sclerosing fibroplasia (FESF) in a 15-year-old cat, expanding the recognised anatomical and age spectrum and emphasising the need to consider FESF among the differential diagnoses for nasopharyngeal masses in elderly cats, especially when lesions are firm or show eosinophil-rich fibroinflammatory features.
Case description
A 15-year-old, spayed female domestic shorthair cat, living strictly indoors, was presented for investigation of chronic progressive stertor, mucopurulent nasal discharge and intermittent apnoeic episodes that had gradually worsened over several months. The cat had no significant previous medical history, had tested negative for feline immunodeficiency virus and feline leukaemia virus, and remained bright with preserved appetite and stable body weight. Physical examination revealed marked stertor at rest, reduced nasal airflow bilaterally and intermittent open-mouth breathing, without facial deformities or oral masses. Systemic or concurrent disease was excluded based on normal history, physical examination, clinicopathological testing and CT, with no evidence of stomatitis or gastrointestinal disease.
Pre-anaesthetic evaluation, including haematology, serum biochemistry, echocardiography and coagulation profile, revealed no abnormalities. Given the chronicity and progression of upper airway noise, cross-sectional imaging followed by endoscopic evaluation was recommended under a single general anaesthetic episode.
A CT scan of the skull was performed first. The study revealed a soft tissue attenuating mass occupying the nasopharynx (Figures 1a and 2a), partially obstructing the choanae and causing dorsal displacement of adjacent soft tissues. CT showed non-turbinate destruction, bone lysis or sinus involvement, supporting FESF as a differential for nasopharyngeal masses. The mass exhibited markedly heterogeneous contrast enhancement, with intralesional hypo-enhancing, fluid-attenuating regions (Figure 1b). Immediately after CT, and with the patient still under the same anaesthetic, a retroflexed nasopharyngoscopy was performed with the cat positioned in sternal recumbency. Endoscopic examination confirmed the presence of a firm, pale, smoothly contoured, multilobulated mass with a glistening surface, occupying the majority of the nasopharyngeal lumen and resulting in marked luminal narrowing compatible with a subocclusive lesion. A moderate amount of adherent mucous secretion was present on and around the lesion, contributing to luminal obstruction (Figure 3a).

Transverse plane CT images of a case of nasopharyngeal feline eosinophilic sclerosing fibroplasia. There is a mass (white arrowhead) that completely obstructs the nasopharyngeal lumen and displaces the soft palate ventrally in (a) pre-contrast and (b) post-contrast acquisitions. The lesion displays soft tissue attenuation with heterogeneous contrast enhancement, characterised by intralesional hypo-enhancing regions. Window width = 400; window level = 40. L = left; R = right

(a) Post-contrast sagittal plane CT image of a case of nasopharyngeal feline eosinophilic sclerosing fibroplasia. A mass (white arrowhead) completely obstructs the nasopharyngeal lumen and displaces the soft palate ventrally. Fluid-attenuating material is present within the lumen cranial to the lesion. (b) Post-contrast sagittal plane CT image of the same patient after complete resolution. The nasopharyngeal lumen (white asterisk) is completely air-filled and free of expansile lesions. Window width = 400; window level = 40. Cd = caudal; Cr = cranial

Endoscopic views of the nasopharynx with the mass and after its removal. (a) Endoscopic view of the nasopharyngeal mass before debulking, showing near complete luminal obstruction. (b) Endoscopic appearance after approximately 70% debulking, with a markedly enlarged nasopharyngeal lumen. (c) Follow-up endoscopy demonstrating complete resolution of the lesion and full restoration of nasopharyngeal patency
Given the substantial obstruction, a decision was made to pursue mechanical debulking to improve airflow and obtain diagnostic tissue. The patient was repositioned into dorsal recumbency. To allow direct retrograde access to the nasopharynx, a suture thread was passed through the caudal soft palate and retracted cranially, exposing the dorsal nasopharyngeal vault. Through this window, a Heuwieser Antrum Grasping Forceps (3.5 mm, angled down 90°) was introduced. Approximately 70% of the mass was removed through repeated debulking passes. As the procedure advanced, moderate post-bioptic haemorrhage developed, limiting visibility and preventing safe acquisition of additional samples. A final endoscopic inspection confirmed a markedly enlarged nasopharyngeal lumen and improved patency (Figure 3b).
Prednisolone (1 mg/kg q24h) was started preoperatively to limit post-manipulation inflammation and stenosis risk, and buprenorphine (20 µg/kg IM) was administered postoperatively for analgesia. The cat recovered uneventfully from anaesthesia and was discharged with instructions to continue the same dosage while awaiting histopathological evaluation.
The histopathological appearance of the lesion was of an exophytic polypoid, partially ulcerated mass, lined by ciliated respiratory epithelium and expanding into the lamina propria. The mass consisted of dense collagen matrix forming branching and anastomosing trabeculae (Figure 4) associated with reactive fibroblasts and infiltrated by numerous eosinophils, fewer neutrophils and macrophages (Figure 5). Scattered lymphoid aggregates were multifocally visible throughout the sample. No features of atypia were identified. Additional Ziehl-Neelsen, periodic acid Schiff and Gram special stains were negative for bacterial and fungal agents. There was no histological evidence of lymphoma, carcinoma or fungal infection. Special stains excluded fungal organisms, and no bacterial colonies suggestive of primary infection were noted.

Rinopharyngeal mass. Nasopharyngeal polypoid mass lined by pseudostratified ciliated epithelium and composed of dense collagen trabeculae and inflammatory cells. Haematoxylin and eosin, × 4 magnification
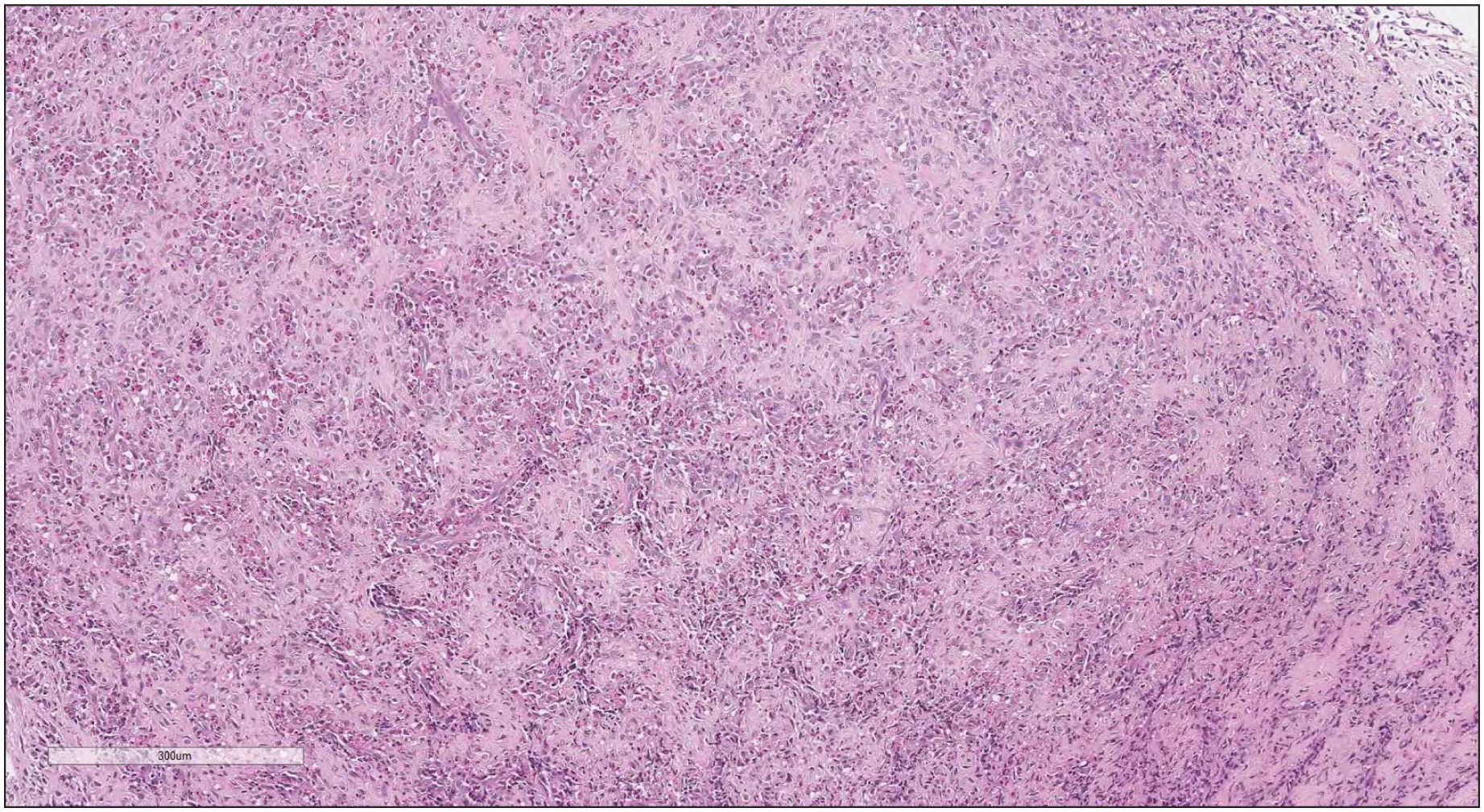
Trabeculae of dense collagen matrix separated by plump reactive spindle-shaped cells and eosinophils. Haematoxylin and eosin, × 10 magnification
After histological diagnosis, prednisolone (1 mg/kg q24h) was continued for a total of 3 weeks. As the cat remained asymptomatic with progressive improvement, the dosage was tapered to 0.5 mg/kg q24h for 5 days, then to 0.25 mg/kg q24h for a further 5 days and subsequently discontinued. During corticosteroid therapy, the owners reported complete resolution of stertor, disappearance of nasal discharge and absence of apnoeic episodes.
One month after cessation of prednisolone, and with no recurrence of clinical signs, repeat CT (Figure 2b) and nasopharyngoscopy (Figure 3c) were performed. Both examinations showed complete anatomical resolution of the previously observed mass, with fully patent choanae and normal nasopharyngeal architecture. No residual tissue, stenosis or structural deformity was present. The cat remained clinically normal at subsequent follow-up.
Discussion
This report documents the first recognised case of FESF arising in the nasopharynx and occurring in the oldest patient described to date. The clinical, endoscopic and histopathological features observed in this cat strongly align with the core characteristics of FESF as originally described in gastrointestinal locations,1,2 –7 yet the anatomical presentation is unprecedented. The case, therefore, extends both the topographical and age-related boundaries of the disease and emphasises the importance of considering FESF among the differentials for nasopharyngeal masses in elderly cats.
The diagnostic process in this patient was particularly informative because it followed a rigorous, methodical sequence that allowed the lesion to be characterised through imaging, endoscopy, mechanical debulking and histopathology. CT revealed a soft tissue mass without turbinate or sinus destruction – an imaging profile consistent with reports describing FESF as a mass-forming but often non-destructive fibroinflammatory process.4,7 –9 Endoscopically, the firm, multilobulated and subocclusive lesion closely resembled the gritty, infiltrative masses reported in abdominal and thoracic forms of the disease,1,2 –6 reaffirming that FESF displays a remarkably consistent macroscopic phenotype regardless of location. This presentation may resemble common feline inflammatory polyps, typically seen in young cats and characterised by prominent lymphoid tissue. However, the lesion in this case lacked lymphoid infiltration and instead showed marked eosinophilic inflammation with sclerosing fibroplasia, features not characteristic of feline nasopharyngeal inflammatory polyps.
Mechanical debulking was pivotal both diagnostically and therapeutically. Substantial removal of the obstructive tissue improved airflow immediately and permitted the acquisition of deeper samples, reducing the risk of superficial biopsy artefact – a known limitation in FESF, where dense fibrosis and necrotic debris can obscure diagnostic features.2,10,11 The technique used in this case – retrograde nasopharyngeal exposure via soft palate traction – allowed safe, targeted sampling while minimising trauma in a confined anatomical space.
Histologically, the combination of marked eosinophilic infiltration, abundant fibroblastic proliferation and sclerosing collagen deposition (Figures 4 and 5) was consistent with classical histological characteristics of FESF.1,2 –4 Although sharing some histological features with inflammatory polyps, this diagnosis was excluded because of the absence of lymphoid follicles and glandular stromal structures, despite the compatible location. Importantly, there was no evidence of carcinoma, lymphoma or fungal infection – three essential exclusions in elderly cats with nasopharyngeal lesions. Special stains and conventional histology excluded fungal organisms, aligning with reports where infectious triggers may or may not be present.2 –12 No intralesional bacteria were detected, a finding compatible with the increasing recognition that bacterial involvement, although common in some cohorts,2,6,13,14 is not obligatory for diagnosis and may represent a secondary modifier rather than a primary cause. 4
A critical point in interpreting this case is the absence of lymphoid atypia. Although rare, an association between FESF and T-/natural killer-cell lymphoma has been described,15,16 raising the possibility of clonal evolution in chronic eosinophilic fibroinflammatory lesions. Given the absence of morphological atypia, additional tests such as immunohistochemistry or clonality were deemed unnecessary and potentially misleading, as chronic inflammation can yield false-positive monoclonal results. 15 In this patient, however, histopathology showed no evidence of malignant lymphoid infiltration, and complete clinical resolution without relapse during follow-up further supports a non-neoplastic inflammatory origin.
Another distinctive aspect of this case is the cat’s age. Most published cases involve young or middle-aged animals, with median ages in the range of 6–8 years.2 –4 The present 15-year-old patient markedly exceeds this range, demonstrating that FESF can occur even in geriatric individuals. This has practical implications because, in older cats, nasopharyngeal masses are overwhelmingly interpreted as neoplastic, with lymphoma and carcinoma being the most common diagnoses. The current case shows that FESF should remain a differential diagnosis irrespective of age, particularly when imaging or endoscopic features suggest a firm, fibroinflammatory process.
The clinical response to prednisolone was both complete and rapid. Although FESF has traditionally been regarded as a mass-forming condition often necessitating surgical excision,10,17,18 increasing evidence – including the present case – indicates that a substantial subset of patients may achieve full remission with medical management alone. This favourable outcome is consistent with several reports describing marked responsiveness to glucocorticoid therapy.5,11,19 The absence of recurrence 1 month after tapering and discontinuation, combined with complete anatomical resolution on CT and endoscopy, reinforces the concept that FESF is a potentially reversible fibroinflammatory disease when adequately treated. The degree of resolution observed in this case is particularly striking given the lesion’s initial size and the anatomical constraints of the nasopharynx.
The novelty of this localisation raises interesting pathophysiological considerations. Extra-gastrointestinal forms of FESF, including mediastinal,6 –13 intrathoracic,6–14 mesenteric, 17 retroperitoneal 11 and hepatic/lymph node involvement, 20 support the hypothesis that FESF represents a process that appears to be driven by eosinophil-mediated inflammation in response to a poorly defined antigen. In humans, sclerosing diseases are linked to type 2 immune responses involving T helper 2 cells, M2 macrophages and, in IgG4-related disease, B cells and autoantibodies, although the exact triggers remain unclear.21,22 The nasopharynx, with its rich lymphoid tissue, mucosal immune activity and susceptibility to chronic antigenic stimulation, may provide a permissive environment for the same fibroinflammatory cascade characterised in abdominal and thoracic sites. Given the absence of fungal organisms, bacteria or neoplastic transformation in this case, an immune-mediated mechanism remains the most plausible explanation. Bacterial, fungal or neoplastic findings associated with FESF may be secondary rather than primary disease drivers.
Conclusions
This case expands the known anatomical sites and age range of FESF, emphasising its inclusion in the differential diagnosis of nasopharyngeal masses, especially when lesions show eosinophil-rich fibrosis and respond to glucocorticoids. The complete resolution demonstrated through follow-up imaging and endoscopy provides compelling evidence that FESF, even when extensive and obstructive, remains a treatable and reversible condition.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
