Abstract
Case summary
A 2-year-old female intact domestic shorthair cat was referred to the neurology service at the Foster Hospital for Small Animals as a result of lifelong weakness, seizure-like episodes after excitement, muscle spasms, stiffness of the limbs and shortness of breath. A diagnosis of myotonia congenita (MC) was made based on compatible history, clinical signs and electromyography findings, and excluding other causes of muscle weakness using muscle biopsy, baseline bloodwork and echocardiography. Initial treatment with procainamide had overall low efficacy. The cat was then switched to carbamazepine, which provided long-term improvement in clinical signs at a dosage of 7.4 mg/kg PO q12h with no observed clinical side effects.
Relevance and novel information
To date, there is only a single case report describing the successful treatment of MC in cats, and no report describing the use of carbamazepine to treat this condition in this species. This anti-convulsant medication was shown to be anecdotally effective in the case reported. Further research is needed to categorize the metabolism, pharmacokinetics, tolerance, dose range and efficacy of carbamazepine in cats with and without MC.
Introduction
Myotonia refers to a disorder of skeletal muscle characterized by prolonged muscle contraction in response to mechanical, electrical or voluntary stimulation. 1 Myotonia can be divided into dystrophic and non-dystrophic forms. 1 Non-dystrophic myotonia includes abnormalities of chloride channels (myotonia congenita [MC]) and sodium channels (paramyotonia congenita). 1 MC occurs secondarily to a mutation in the CLCN1 gene encoding for voltage-gated chloride channels at the level of the sarcolemma in cats.1,2
The mutation in the chloride channels reduces inhibitory signals, hindering muscle relaxation. Sodium influx through acetylcholine ligand receptors and voltage-gated sodium channels triggers muscle contraction. 3 Repolarization occurs via potassium efflux through ATP-dependent potassium channels, leading to muscle relaxation. 3 CLCN1 voltage-gated chloride channels facilitate chloride influx and efflux, contributing to 80% of inhibitory membrane current. 3 Without CLCN1 channels, there is reduced chloride permeability and increased membrane resistance, leading to decreased inhibitory currents. This results in the prolonged muscle contraction observed in MC, causing muscle stiffness and hypertrophy characterized by runs of action potentials.
Clinical signs of MC manifest when affected individuals begin walking and may include muscular hypertrophy, dysphonia, blepharospasm, a protruding tongue, a stiff jaw, hyperextension of the limbs, falling over and a stiff gait that improves with exercise.1,4 Signs may worsen with excitement 1 and/or cold temperatures. 4 Diagnosis involves clinical signs, electrodiagnostics, imaging, muscle biopsy and genetic analysis. Neurological examination may reveal muscle dimpling upon percussion.1,5 Bloodwork, including muscle enzyme activities, is typically normal. MRI and muscle biopsies may show mild non-specific changes, while electromyography shows spontaneous myotonic discharges characterized by waxing and waning amplitude and frequency. 1
MC has been reported in Chow Chows 6 and Miniature Schnauzers, 7 linked to an inherited autosomal recessive trait. In cats, cases of MC have been described only sporadically. These reports discuss clinical signs,4,5 diagnosis and genetic findings,1,2 with only one report discussing the use of phenytoin 1 as a potential treatment.
Case description
A 2-year-old female intact domestic shorthair cat presented to the neurology service at the Foster Small Animal Hospital for a second opinion of a lifelong history of pelvic limb weakness, 20 s seizure-like episodes consisting of acute collapse, intermittent hypersalivation as well as protrusion and curling of the tongue. The cat had persistent blepharospasm in the right eye and intermittent dragging of the left pelvic limb. The littermates were unaffected.
Physical examination revealed a grade I/VI systolic heart murmur, muscular hypertrophy of the limbs and head, slight tongue hypertrophy and increased respiratory effort associated with excitement. Neurological examination revealed a stiff gait and a kyphotic posture when stressed. A delayed palpebral reflex in both eyes and increased generalized muscle tone and muscle mass were also noted. The remaining neurological examination was unremarkable.
Complete blood count and serum biochemistry analyses showed mild hyperkalemia (5.5 mmol/l, reference interval [RI] 3.6–5.4) and hyperproteinemia (8.5 g/dl, RI 6.0–8.4). On urinalysis, urine specific gravity was 1.050, pH was 5.0 and findings were otherwise unremarkable. Fasting total serum bile acids (5.0 µmol/l, RI >20), adrenocorticotropic hormone stimulation test (pre-ACTH 4.8 µg/dl, RI 0.5–5.4; post-ACTH 10.2 µg/dl, RI 4.7–16.3) and total thyroxine were within normal limits. Thoracic radiographs revealed a subjectively enlarged cardiac silhouette and subjective hypertrophy of the body musculature. On abdominal ultrasonography, a thickened diaphragm was observed. An abbreviated electrocardiogram showed low-voltage QRS complexes; however, the study was discontinued owing to stress-induced cyanosis. An echocardiogram showed speckled hyperechoic regions of the papillary muscle, slight dilation of the left ventricle, trivial regurgitation of the tricuspid valve and occasional premature ventricular beats. Electromyography was then performed under general anesthesia, showing spontaneous waxing and waning myotonic discharges consistent with a myotonic myopathy (Figure 1). Motor nerve conduction velocity measured from the peroneal nerve was within normal limits. A biopsy from the left cranial tibial muscle was obtained under the same general anesthesia (Figure 2). Except for a subjectively increased variability in myofiber size, no specific abnormalities were identified in the cranial tibial muscle biopsy. Immunostaining of muscle cryosection for dystrophin protein sarcolemmal localization was normal (not shown), ruling out X-linked (hypertrophic) muscular dystrophy. Therefore, a diagnosis of MC was made.

(a) The patient’s electromyogram (EMG) showed myotonic discharges. The insertion of a monopolar needle into the cranial tibial muscle produced an EMG characterized by continuous waxing and waning action potentials. These findings strongly suggest myotonia. (b) The patient’s nerve conduction activity of the peroneal nerve showed a normal conduction velocity and a normal amplitude of the recorded compound muscle action potentials, making neuropathy very unlikely
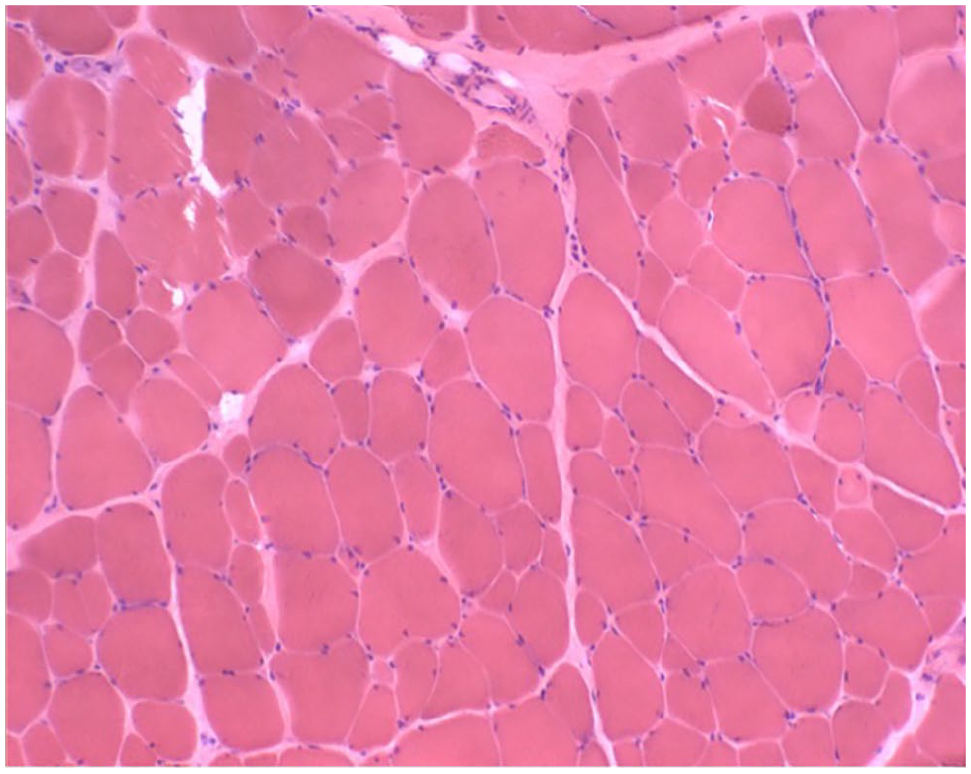
Biopsy of the left cranial tibial muscle. A hematoxlyin and eosin-stained cryosection from the cranial tibial muscle showed an excessive variability in myofiber size without other specific pathological changes. Magnification = ×20
The cat initially responded to procainamide (35 mg/kg PO q8h), showing fewer episodes of stiffness and collapse for 2 months. When re-evaluated owing to worsening clinical signs, serum levels of procainamide revealed significant fluctuations, with peak levels approximately 2 h after administration (Figure 3). Consequently, the dosage was switched to extended-release procainamide at 37 mg/kg q6h. Procainamide was stopped after 2 years because of inadequate improvement and carbamazepine treatment was initiated. The dose was slowly titrated based on the observed clinical response and side effects, including the degree of sedation and either spastic or flaccid limbs as described below.

Procainamide blood levels. Limited information is available on the metabolic rate of procainamide in cats. To better understand treatment with procainamide, serum levels were measured after an oral dose of 35 mg/kg at multiple time points. Results showed a fast increase of the serum levels after administration to a peak within the therapeutic range reported for humans of 4–14 µg/ml. However, the short half-life of 3 h would have required multiple treatments per day, which was difficult to achieve in this cat
Carbamazepine was started at 8 mg (1.6 mg/kg) PO q12h and increased by 4 mg every 2 weeks. Once 45 mg (9.8 mg/kg) q12h was reached, a recurrence of muscle spasms and stiffness of the limbs was observed. The dose was then decreased to 40 mg (8.7 mg/kg) q12h, with good improvement of clinical signs and minimal side effects. During the cat’s annual recheck examination, recent worsening in the frequency of spasticity was described. On the basis of increased clearance in humans with chronic carbamazepine use, 8 the dosage was adjusted to 50 mg (10.4 mg/kg) every 12 h. However, the cat experienced a sudden onset of shortness of breath, limb stiffness or previously unobserved flaccidity occurring within 1 h of drug administration and lasting approximately 3 h. The dosage was lowered to 35 mg (7.4 mg/kg) q12h, leading to clinical improvement without apparent side effects.
During the 12-month recheck examinations, the cat showed continued improvement of signs. There was a decrease in muscle mass and increased mobility, flexibility and athleticism (as seen by jumping on furniture and using stairs). Episodes of muscle spasms and collapse were minimal (1–2 episodes a week); however, the mild stiff gait and kyphosis remained. The spastic palpebral reflex continued to improve with carbamazepine treatment until resolution was achieved with occasional blepharospasm. Two years after initiating carbamazepine, the cat’s neurological examination remained stable and continued to do so throughout the course of treatment. The cat was maintained on carbamazepine lifelong at 7.4 mg/kg q12h with no apparent side effects observed during yearly recheck examinations.
Twelve years later, the cat died at the age of 17 years of unrelated conditions. Necropsy showed moderate, multifocal cerebellar abiotrophy and masses in the central nervous system consistent with meningioma (Figure 4) and meningeal proliferative lesions. The iris of the right eye contained a mass with typical characteristics of lymphoma. A mass infiltrating the muscles of the right forelimb was most consistent with a plasma cell tumor. The heart showed mild mitral valvular endocardiosis and moderate enlargement of the atria.

Cerebellar mass. The histopathological analysis of the cerebellar mass showed a prominent whorling of elongated spindle-like cells (arrow) and few psammoma bodies (star). These findings are most compatible with a psammomatous meningioma, which is a common histological subtype in cats
Discussion
Based on clinical signs, electrodiagnostic testing and the absence of specific myopathic changes on examination of the muscle biopsy, a presumptive diagnosis of MC was made in the cat in this report. Limited information is available on treatment options for cats with MC or on long-term outcome. The current report focuses on the description of the response to treatment with procainamide and carbamazepine in a young cat with MC.
Class 1 antiarrhythmic drugs are the preferred treatment in people with MC. 1 In dogs with MC, procainamide9,10 and mexiletine 10 have shown some efficacy. Woelfel et al 1 reported on the use of phenytoin in one cat with MC, with a partial response. The cat in our case report had a simple incomplete response to procainamide. After 2 months of treatment and initial improvement at home, a partial ‘honeymoon effect’ was described and a worsening in the cat’s signs was observed.
Because of the overall unsatisfactory improvement with procainamide, the cat was started on carbamazepine, as described above, and maintained on 7.4 mg/kg q12h as the long-term dose, with no side effects observed and improved clinical signs.
Carbamazepine, a dibenzazepine anticonvulsant, enhances sodium channel inactivation, acts on post-synaptic serotonin transmission 8 and binds to other voltage-gated channels, such as calcium channels. 11 Its efficacy in treating MC has been reported in human cases. For instance, a 14-year-old boy with MC due to a CLCN1 gene mutation showed significant improvement in stiffness and transient weakness 1 year after starting carbamazepine treatment (initially 100 mg q12h, later increased to 900 mg/day). 12 In a cohort of nine individuals with MC, including one with a CLCN1 mutation, carbamazepine effectively reduced clinical signs with minimal side effects. 13 Another case report described a 10-year-old girl with MC who achieved total symptom relief after 1 month of carbamazepine (15 mg/kg per day), with complete resolution at the 7-month recheck and no observed side effects. 14
Carbamazepine induces its own metabolism in humans, necessitating dosage increases over time. 8 In this case, plasma concentrations were not available, but a dosage increase was not needed based solely on clinical signs and observed side effects at higher doses. This suggests potential metabolic differences between cats and humans. The optimal dosage and plasma level for controlling clinical signs in cats remain uncertain. Hence, it is important to evaluate improvement with both clinical signs and side effects alongside serum levels. Monitoring serum levels can help maintain consistent drug levels within the desired effective range.
Despite cerebellar abiotrophy observed on necropsy, clinical signs were never reported at home or during routine examinations. Currently, it is unclear whether MC contributes to heart anatomical changes or arrhythmias in humans. Low CLCN1 levels are found in various organs, including the heart, and some case reports suggest a possible relationship in humans. 15 Lack of genetic testing and identification of a specific CLCN1 variant limits our ability to correlate drug effectiveness to a specific mutation. However, feline genetic testing was less advanced and cost-effective at the time of diagnosis. Furthermore, there are no available data on carbamazepine pharmacokinetics, dose range or tolerance in cats for conditions such as MC.
Conclusions
Carbamazepine is an antiepileptic drug that should be considered for the treatment of MC in cats, as described in this case. Our case demonstrates that it can be used safely and effectively, can decrease the signs of MC and contributed to a long-term survival of 12 years post diagnosis in this cat. Studies evaluating pharmacokinetics and possible toxicity are needed to best guide the use of this drug in veterinary species. Efficacy compared with other drugs in cats and dogs remains unknown and further research is needed. Carbamazepine has been implicated in positive responses when used to treat dystonia and dyskinesia in humans. 16 This might indicate efficacy in animals with similar signs. Treatment of other conditions affecting muscle tone and excitability, such as familial reflex myoclonus and Scottie cramps, with carbamazepine should be considered.
Footnotes
Acknowledgements
The authors would like to thank Dr Cynthia R Leveille-Webster for her editorial contribution to this report, Dr M Sawkat Anwer for his pharmacological contribution and the radiology team at the Foster Hospital for Small Animals for their contributions in imaging interpretation.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
