Abstract
Objectives
The present study reports the multilocus genotyping of Giardia duodenalis isolates from cats maintained in breeding catteries in Japan and discusses their potential for zoonotic transmission.
Methods
A total of 41 faecal samples positive for Giardia-specific antigen were procured from cats maintained in five breeding catteries and subjected to PCR to amplify four gene loci, namely small subunit ribosomal RNA (SSU rRNA), glutamate dehydrogenase (gdh), beta-giardin (bg) and triose phosphate isomerase (tpi ). The PCR-amplified DNA fragments were sequenced to determine the G duodenalis genotypes (synonym for assemblages).
Results
The most commonly occurring single assemblage was assemblage F (68.3%; n = 28/41), followed by assemblage A (12.2%; n = 5/41) and assemblage C (2.4%; n = 1/41). The mixed assemblages were identified as follows: assemblages F and A (9.8%; n = 4/41), assemblages F and C (4.9%; n = 2/41) and assemblages C and D (2.4%; n = 1/41). Additional sub-genotyping of assemblage A isolates based on three of the sequenced loci (gdh, bg and tpi ) revealed that all eight isolates were identified as sub-assemblage AI and/or AII.
Conclusions and relevance
The present study is the first to report the detection of dog-adapted assemblages C and D in feline isolates from Japan. In addition, zoonotic sub-assemblage AI and human-adapted sub-assemblage AII were also identified. Thus, we concluded that the risk of transmission of G duodenalis from breeding cattery cats to humans is considerable and cannot be ignored.
Introduction
The protozoan Giardia duodenalis is an intestinal pathogenic parasite that is frequently detected in several mammals and can cause gastrointestinal obstruction in its hosts.1,2 Based on its molecular biological characteristics, G duodenalis has been recognised as a multispecies complex comprising eight different genotypes, namely assemblages A–H, which possess different host ranges.3,4 Assemblages A and B are zoonotic genotypes, whereas the remaining assemblages (C–H) are host-adapted genotypes.4–6 Although cats are commonly infected with assemblage F, a cat-adapted genotype, assemblages A and B have also been detected in cats.7–9 Recent studies have demonstrated that multilocus genotyping provides comprehensive information for the identification of G duodenalis and mixed-assemblage-mediated infections.8,10 Therefore, multilocus genotyping is highly recommended.
Breeding catteries are the major source of kittens for many private owners, via pet shops or direct purchase. A high prevalence of G duodenalis has been reported in breeding cattery cats. 11 However, the genetic characteristics of G duodenalis isolates from breeding cattery cats are not sufficiently understood. The present study reports the multilocus genotyping of G duodenalis isolates from breeding cattery cats in Japan using four loci (small subunit ribosomal RNA [SSU rRNA], glutamate dehydrogenase [gdh], beta-giardin [bg], triose phosphate isomerase [tpi]) and discusses their potential for zoonotic transmission.
Materials and methods
We previously reported that 64 faecal samples were positive for Giardia-specific antigen using ELISA (SNAP Giardia; IDEXX Laboratories) in 342 fresh stools from breeding cattery cats in Japan. 11 In the present study, a total of 41 fresh faeces sufficient for DNA extraction from 64 positive samples for Giardia-specific antigen were obtained from cats maintained in five breeding catteries (Nagano-I, n = 4; Nagano-II, n = 26; Aichi, n = 3; Gifu, n = 6; Miyagi, n = 2) in Japan (Figure 1). Giardia species cysts were microscopically identified in 19/41 samples using the formalin–ethyl acetate sedimentation technique. The conditions of the 41 analysed faecal samples were normal (n = 32), soft (n = 8) and diarrhoea (n = 1).

Location of five breeding catteries in Japan
PCR and sequencing analysis were performed for four gene loci – namely SSU rRNA, gdh, bg and tpi – to determine G duodenalis assemblages. Fragments of these four genes were individually amplified according to previously described protocols.12–15 All PCR amplicons were sequenced at FASMAC (Atsugi, Kanagawa, Japan). Sequence alignment and compilation were performed using MEGA 6.06 software (www.megasoftware.net). The deduced DNA sequences were compared with the GenBank sequences of G duodenalis using BLAST (http://www.ncbi.nlm.nih.gov/) to determine the assemblages. In addition, the sub-assemblages of assemblage A were also determined based on three loci (gdh, bg and tpi).
Results
PCR-amplified fragments from the 41 samples were observed to contain at least one of the four loci (Table 1). The samples contained isolates from all five breeding catteries (Nagano-I, n = 4; Nagano-II, n = 26; Aichi, n = 3; Gifu, n = 6; Miyagi, n = 2). Sequencing analysis revealed the genotypes of the isolates based on the loci that were amplified (80.5% [n = 33/41] SSU rRNA; 70.7% [n = 29/41] gdh; 46.3% [n = 19/41] bg; 26.8% [n = 11/41] tpi). Overall, the results of genotyping using one or more loci revealed that single assemblages were identified in 82.9% (n = 34/41) of the samples, whereas the remaining 17.1% (n = 7/41) of the samples showed mixed assemblages. The most commonly detected single assemblage was assemblage F (68.3%; n = 28/41), followed by assemblage A (12.2%; n = 5/41) and assemblage C (2.4%; n = 1/41). The percentage of assemblage F was significantly higher than the percentages of assemblage A and assemblage C (P <0.001) using Fisher’s exact probability test. The mixed assemblages were identified as follows: assemblages F and A (9.8%; n = 4/41), assemblages F and C (4.9%; n = 2/41) and assemblages C and D (2.4%; n = 1/41). Assemblage F was detected in samples from all five breeding catteries, assemblage C from three facilities (Nagano-I, Nagano-II and Miyagi), assemblage A from two breeding catteries (Nagano-II and Gifu) and assemblage D from one breeding cattery (Nagano-I).
Multilocus genotyping of Giardia duodenalis in breeding cattery cats
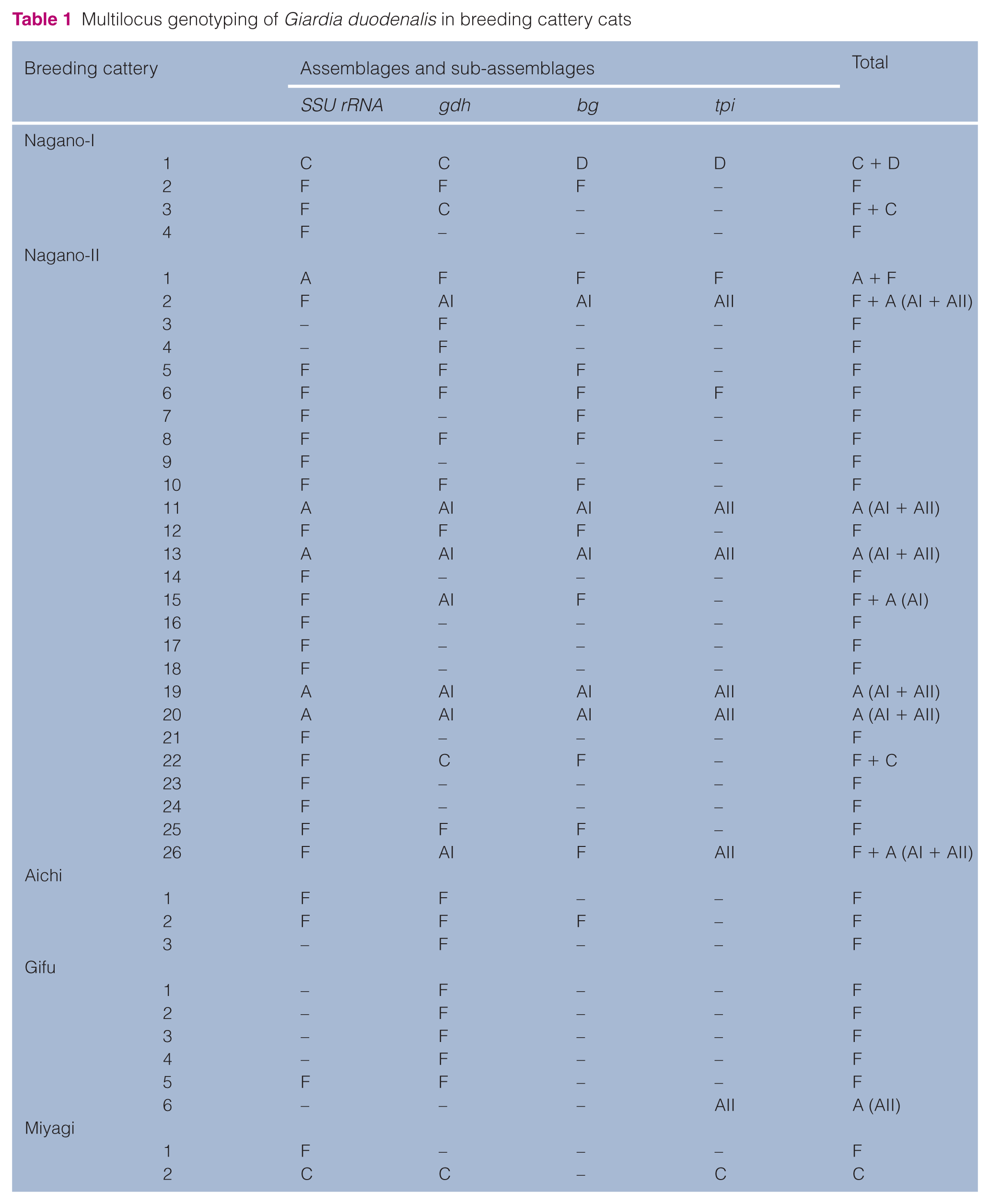
Assemblages of G duodenalis isolates detected with differential loci are summarised in Table 2, and all isolates showed 99–100% similarity to corresponding sequences in GenBank. Of the 33 isolates successfully sequenced at the SSU rRNA locus, 26 were identified as assemblage F and another five and two isolates were identified as assemblages A and C, respectively. In the 29 isolates amplified at the gdh locus, 18 were identified as assemblage F and seven isolates were assemblage A (sub-assemblage AI). The remaining four isolates belonged to assemblage C. Of the 19 isolates successfully sequenced at the bg locus, 13 isolates belonged to assemblage F and five isolates were assemblage A (sub-assemblage AI). Only one isolate belonged to assemblage D. Of the 11 isolates successfully sequenced at the tpi locus, seven belonged to assemblage A (sub-assemblage AII) and two were assemblage F. In the remaining two isolates, one was identified as assemblage C, whereas the other one belonged to assemblage D.
Assemblages of Giardia duodenalis isolates detected at differential loci

Discussion
The present study is the first to evaluate the zoonotic potential of a large number of feline G duodenalis isolates from multiple breeding catteries, using multilocus genotyping analysis. Previously, a study based on multilocus genotyping reported that all 16 feline G duodenalis isolates from one breeding cattery belonged to assemblage F. 16 These results are similar to those in the present study, wherein the most dominant genotype, including mixed assemblage infections, was assemblage F because it was present in the highest percentage of genotyped isolates in all breeding cattery samples. The predominance of assemblage F in breeding cattery cats is understandable because this assemblage is a cat-adapted genotype.4–6
Other assemblages, such as A, C and D, were also identified, including in the mixed assemblage infections. The identification of assemblage A was not surprising, as it is recognised as a zoonotic genotype and has been often detected in cats.7–9 Although sub-assemblage AI has been reported in feline isolates in Japan, 17 the present study is the first to report the occurrence of sub-assemblage AII. Sub-assemblage AI has been commonly observed as a zoonotic sub-genotype in several mammals.2,3 In contrast, sub-assemblage AII was mostly isolated from humans.5,18 Thus, sub-assemblage AII was presumed to be a human-adapted genotype. However, recent multilocus genotyping analyses have routinely identified sub-assemblage AII in animals other than humans.6,8,9,19 Therefore, it is suggested that sub-assemblage AII is also potentially zoonotic. The results of the present study suggest that the risk of transmission of these G duodenalis isolates from breeding cattery cats to humans is considerable and cannot be ignored. In addition, although assemblages C and D were recognised as dog-adapted genotypes,1,7 the present study provides the first evidence for the occurrence of these assemblages in cats in Japan. Previous studies have demonstrated the isolation of assemblages C, D and E from cat faeces.8–10 However, breeding cattery details were unavailable. We suspect that those facilities had also bred dogs and that the environment was potentially contaminated with assemblages from both cats (assemblage F) and dogs (assemblages C and D). In addition, mixed assemblage infections were observed in the samples from all but one of the breeding catteries. Recent studies have revealed that mixed assemblage infections with assemblage F were often observed in feline stool samples.2,3,8 Therefore, cats might be simultaneously infected with not only dominant assemblage F and zoonotic assemblage A, but also non-cat-adapted assemblages.
Conclusions
The present study is the first report of the detection of dog-adapted assemblages C and D from feline isolates in Japan. In addition, zoonotic sub-assemblage AI and human-adapted sub-assemblage AII were also identified. Thus, we concluded that the risk of transmission of G duodenalis from breeding cattery cats to humans is considerable and cannot be ignored.
Footnotes
Conflict of interest
The authors declare that there is no conflict of interest regarding the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
