Abstract
This prospective study examines gender-specific psychological risk factors of myocardial infarction. Out of 41,248 participants free of coronary heart disease at baseline, 822 cases of myocardial infarction were identified in the Nord-Trøndelag Health Study or the mortality register. The participants completed the Hospital Anxiety and Depression Scale. Cholesterol, blood pressure, and waist–hip ratio were measured by medical staff. Smoking, diabetes, non-fatal myocardial infarction, and history of depressive episode were self-reported. Anhedonic depression (Hospital Anxiety and Depression Scale-D ≥8) was a significant predictor of myocardial infarction in women but not in men. Gender difference in risk estimate based on Hospital Anxiety and Depression Scale-D was significant (p < .01). History of depressive episode was a significant predictor of myocardial infarction in men. Symptoms of anxiety (Hospital Anxiety and Depression Scale-A ≥8) reduced the risk of having a myocardial infarction.
Women have been marginalized in the field of cardiac research, and studies focusing specifically on women’s psychological risk profiles are lacking (Naqvi et al., 2005; Wenger, 2003). Coronary heart disease (CHD) in women does not present or manifest the same way as in men; symptoms of myocardial infarction (MI) are more atypical and cardiac events more likely to occur asymptomatically. Women are less likely to experience chest pain before an MI than men, but significantly more likely to experience neck pain, nausea, right-arm pain, dizziness, jaw pain and fatigue (Coventry et al., 2011). Furthermore, women typically experience a heart attack later in life than men and hence have more comorbid conditions, and they are also less likely to be diagnosed and treated efficiently (Low et al., 2010; Möller-Leimkühler, 2007). The European guidelines on cardiovascular disease (CVD) prevention in clinical practice (Perk et al., 2012) emphasize the inclusion of psychosocial factors in CHD risk assessment. However, the guidelines offer no gender-specific risk profile in regard to anxiety and depression.
Several reviews about the relationship between depression and CHD (e.g. Nicholson et al., 2006; Stampfer et al., 2012; Van der Kooy et al., 2007; Wulsin and Singal, 2003) and prospective studies (e.g. Gustad et al., 2013; Hawkins et al., 2013) strongly support that there is an association between depression and CHD. Among women, there has been an increased prevalence of CHD, and the role of depression as a gender-specific factor is still unclear (Naqvi et al., 2005). In a Finnish study, symptoms of depression predicted cardiovascular mortality only in women (Haukkala et al., 2009). In another study, depressive symptoms were an independent risk factor for CHD in women but not in men (Mendes de Leon et al., 1998), and depressive symptomatology has been found to be more important in predicting CVD in women than in men (Lloyd et al., 1996). In contrary, a French population-based study found that depressive mood predicted cardiovascular mortality for men only (Lemogne et al., 2012). These findings emphasize the importance of both screening for depression in general and a focus on the gender-specific role of depression in preventive heart interventions.
Women and men represent different populations, and therefore, the practice of using men as references should be reconsidered (Wenger et al., 2008). Prevalence, incidence, and morbidity risk of depressive disorders are higher in females than in males (Piccinelli and Wilkinson, 2000). Different measures of depression tend to display differentiated gender differences (Salokangas et al., 2002), and in some self-report instruments, the differences are reversed, that is, men have a higher score on depression (Nortvedt et al., 2006; Stordal et al., 2001). At the symptom level, different gender subtypes can be identified (Alexandrino-Silva et al., 2013), and this might have implications for understanding the depression–CHD relationship. Gender differences in depression differ from gender differences in CHD, suggesting that the association between depression and CHD is gender specific (Faravelli et al., 2013; Low et al., 2010).
In medical practice as well as in the research field addressing depression and CHD, a variety of operationalizations of depression exist (Davidson, 2012; Davidson et al., 2005; Stampfer et al., 2012). The relationship between affective disorders and CHD seems to vary in strength by subtype (Baune et al., 2012). The treatment of depression as a unidimensional construct in the research field has left several questions unanswered regarding symptom clusters (Hawkins et al., 2013) and whether some aspects or subtypes of depression are more cardiotoxic than others (Doyle et al., 2010). Furthermore, different symptomatic profiles of depression have been identified in men and women (Alexandrino-Silva et al., 2013), so including gender-specific analysis is important in understanding the role of depression as a risk factor of CHD. It is also possible that gender-specific strategies could improve the predictive power of a psychological risk profile, but prospective studies are needed to identify gender-specific risk profiles.
Shaffer et al. (2012) address the importance of research on different depression phenotypes and emphasize the role of anhedonic depression in the association between depression and CHD. Recent research on MI has emphasized the role of anhedonia (reduced positive affect and loss of interest and pleasure and lack of reactivity to usually pleasurable stimuli) as independently associated with MI (Davidson et al., 2010; Denollet et al., 2007). Anhedonia, which refers to the reduced or lost capacity to experience pleasure, is a feature of major depressive disorder (Di Giannantonio and Martinotti, 2012). An emphasis on the distinction between depressive syndromes and symptoms and the examination of the predictive ability of different operationalization has also been requested (Davidson et al., 2005).
Although the study of psychological risk factors of CHD has mainly focused on depression, there is a growing amount of literature on the link between anxiety and the development of CHD (Batelaan et al., 2014; Roest et al., 2010; Tully et al., 2013). However, the heterogeneity in assessment of anxiety imposes major limitations in the investigation of the impact of anxiety (Batelaan et al., 2014). Generalized anxiety disorder (GAD), but not symptoms of anxious arousal, has been found to predict mortality in CHD patients (Tully et al., 2011). In another study, symptoms of anxiety predicted mortality in CHD patients independent of symptoms of depression (Watkins et al., 2013). In a study of middle-aged women, a three-item measure of symptoms of anxiety predicted fatal cardiovascular incidents (Denollet et al., 2009). In contrast, the study by Mykletun et al. (2007) showed that symptoms of anxiety were negatively associated with cardiovascular mortality. Similarly, others have documented that higher anxiety scores are associated with reduced mortality (Meyer et al., 2010). Furthermore, GAD has been found to predict superior outcome after an acute coronary syndrome (Parker et al., 2011). Other studies have found that symptoms of anxiety are not associated with CHD events when controlling for depression (Versteeg et al., 2013). Furthermore, gender differences in anxiety are highly prevalent, and comorbidity between the disorders is higher among women (McLean et al., 2011). It is therefore important to include measures of anxiety in the search for a gender-specific psychological risk profile.
The European guidelines on CVD prevention (Perk et al., 2012) do not specify how depression should be measured and offer no gender-specific recommendations. This study addresses the value of using simple psychological screening tools in order to identify men and women at risk of having an MI based on their psychological profile.
Although highly comorbid in psychiatric clinical samples, the comorbidity rates between anxiety and depression in CHD patients are substantially lower (Tully and Cosh, 2013). This is also the conclusion made by Hek et al. (2011): Comorbid depression and anxiety are less prevalent in older adults, while current anxiety disorders are substantially related to past depression. In this prospective, population-based study, a unique linkage between a large health survey, clinical assessment, and the national mortality register is utilized. This enables the investigation of the roles of anhedonic depression, depressive episode, and anxiety as gender-specific risk factors of MI.
Method
Study design and participation
The Nord-Trøndelag Health Study (The HUNT Study) is collaboration between the HUNT Research Centre (Faculty of Medicine, Norwegian University of Science and Technology NTNU), the Nord-Trøndelag County Council, the Central Norway Health Authority, and the Norwegian Institute of Public Health. HUNT is one of the largest health studies ever conducted and is described in Holmen et al. (2003). It is a large database of personal and family medical histories collected during three intensive studies over two decades. HUNT1 was carried out from 1984 through 1986 to establish the health history of 75,000 people. The succeeding HUNT2 was carried out from 1995 through 1997 and focused on the evolution of the health history of 65,049 people. HUNT3 was completed in June 2008, and 48,289 people participated (52% participation rate). Data were collected by means of questionnaires, interviews, clinical examinations, and collection of blood and urine samples.
Of the 65,049 people who participated in HUNT2, those with a history of MI or reported current or previous incidents of angina were excluded from the analyses. Participation in the HUNT3 study or identification in the mortality register was used as the inclusion criteria, resulting in 41,248 candidates for this study. A total of 573 participants in the HUNT3 study (2.5%) reported that they currently or prior to the examination had a non-fatal MI. According to the mortality register, 249 were classified with mortality due to MI, that is, having a fatal MI. The Regional Committee for Ethics in Medical Research approved the protocols for HUNT2, HUNT3, and for this specific study, including the linkage to the mortality register. All participants in this study gave their written consent.
Measurement of depression and anxiety
The Hospital Anxiety and Depression Scale (HADS; Zigmond and Snaith, 1983) includes 14 items with a 4-point scale indicating how the respondent has felt over a 2-week period prior to measurement. HADS has been found superior to other depression scales when measuring cardiotoxic symptoms, as HADS focuses mainly on anhedonia (Doyle et al., 2012). However, HADS does not include somatic symptoms, a symptom cluster identified as important in relation to CHD (Hawkins et al., 2013).
The HADS anxiety scale contains items that cover worrying, restlessness, and panic attacks. It has been proven to have good clinical value and case-finding abilities (Olssøn et al., 2005). In the depression subscale, the dominant focus is on anhedonia (Mykletun et al., 2001). In this study, a Norwegian translation of HADS was used (Mykletun et al., 2001). A score on HADS-A and HADS-D ≥8 on the respective scales was used as an indicator of case-level anxiety and depression (Bjelland et al., 2002).
Depressive episode
In an extended part of the HUNT2, the participants were instructed to indicate whether, during their life, there had been periods of two consecutive weeks or more when they “felt depressed, sad, and down,” “had appetite problems or ate too little,” “felt weak (adynamic) or lacked energy,” “really reproached yourself and felt worthless,” or “had problems concentrating or had difficulty making decisions.” Participants were classified as having experienced a depressive episode if they answered yes to the question, “During your life, have there been periods of two consecutive weeks or more when you had at least three of the above-mentioned problems simultaneously?”
Hypertension, cholesterol, waist–hip ratio
Participants’ systolic blood pressure (SBP) was measured by specifically trained nurses using a cuff adjusted for arm circumference in HUNT2. SBP was measured three times, and a mean was calculated from the second and third observations to ensure reliable measurements. Total serum cholesterol was measured applying an enzymatic colorimetric cholesterol esterase method (Holmen et al., 2003). Waist and hip circumferences were measured with a steel band to the nearest 1.0 cm with the participant standing and with the arms hanging relaxed. The waist circumference was measured horizontally at the height of the umbilicus, and the hip circumference was measured at the thickest part of the hip (Holmen et al., 2003). The International Diabetes Federation (IDF) has also provided recommendations for cut-offs for waist circumference and waist–hip ratio. The recommendations are gender, population, and geography specific. For Europeans, the recommended cutoff scores are .80 for women and .94 for men to identify central obesity (Alberti et al., 2006).
Diabetes and smoking
Smoking was self-reported and operationalized as numbers of years of daily smoking. Diabetes was also self-assessed by answering yes or no.
End point: fatal and non-fatal MI
The mortality data were obtained from the National Mortality Register by combining the mortality database and the HUNT data by means of an 11-digit personal identity number. In the mortality registry, diagnoses are encoded according to International Classification of Diseases, 10th Revision (ICD-10). MI mortality in this study encompasses ICD-10 codes I21-9, unspecified acute MI, and I22-9 subsequent MI with unspecified location, with the majority of cases being I21-9. Non-fatal MI was indicated by self-report in the HUNT3 study as a “yes” or “no” response to the question, “Have you ever had or do you have myocardial infarction?”
Statistical analysis
Independent-samples t-test and χ2 were conducted to compare the groups on the baseline characteristics at HUNT2. Logistic regressions were applied to investigate the effect on MI. The dependent variable was dichotomous (MI/non-MI). Separate analyses were performed for men and women to enable comparison of the risk predictor’s gender-specific magnitude. The psychological variables were entered in Step 1, whereas the traditional risk factors were entered in Step 2 to adjust the model.
Results
In the total sample of 41,248 subjects, 822 of the participants had a fatal (249) or non-fatal (573) MI. Of those with a HADS-D score ≥8 (3621), 59.3 percent (2148) had a HADS-A score ≥8, and 5.7 percent (2148) had a history of depressive episode. Compared to men, women reported higher prevalence of prior depressive episode (6597/22,013 vs 3060/18,413, χ2 = 982.81 (1), Φ = 16, p < .001). Women were overrepresented in the HADS-A ≥8 group as 1907 out of 2203 women had a HADS-A score equal or above 8, whereas the respective numbers for the men were 2291 out of 18,413 (χ2 = 317.71 (1), Φ = .07, p < .001). Men were marginally overrepresented in the HADS-D >8 group; 1714 out of 18,413 men and 1907 out of 22,013 women had a HADS-D score ≥8 (χ2 = 5.12 (1), Φ = .01, p < .03. Analysis between the outcome variable MI versus non-MI group and predictors performed by t-test and χ2 showed significant associations and group differences related to all predictors except depressive episodes. The prevalence of elevated symptoms of anxiety was higher in the non-MI group, although this association was significant only for men. Prevalence of depression measured by HADS-D was significantly different in the MI versus the non-MI group in both men and women, whereas depressive episodes were not. There was a significant association between HADS-D and depressive episode (Φ = .25, p < .001) and a significant association between HADS-D and HADS-A (Φ = .39, p < .001). HADS-A and depressive episode had a significant positive association (χ2 = 4498.32 Φ = .33, p < .001; Table 1).
Demographic and baseline risk factors in MI (822) versus non-MI group (40,426) sorted by gender (N = 41,248).
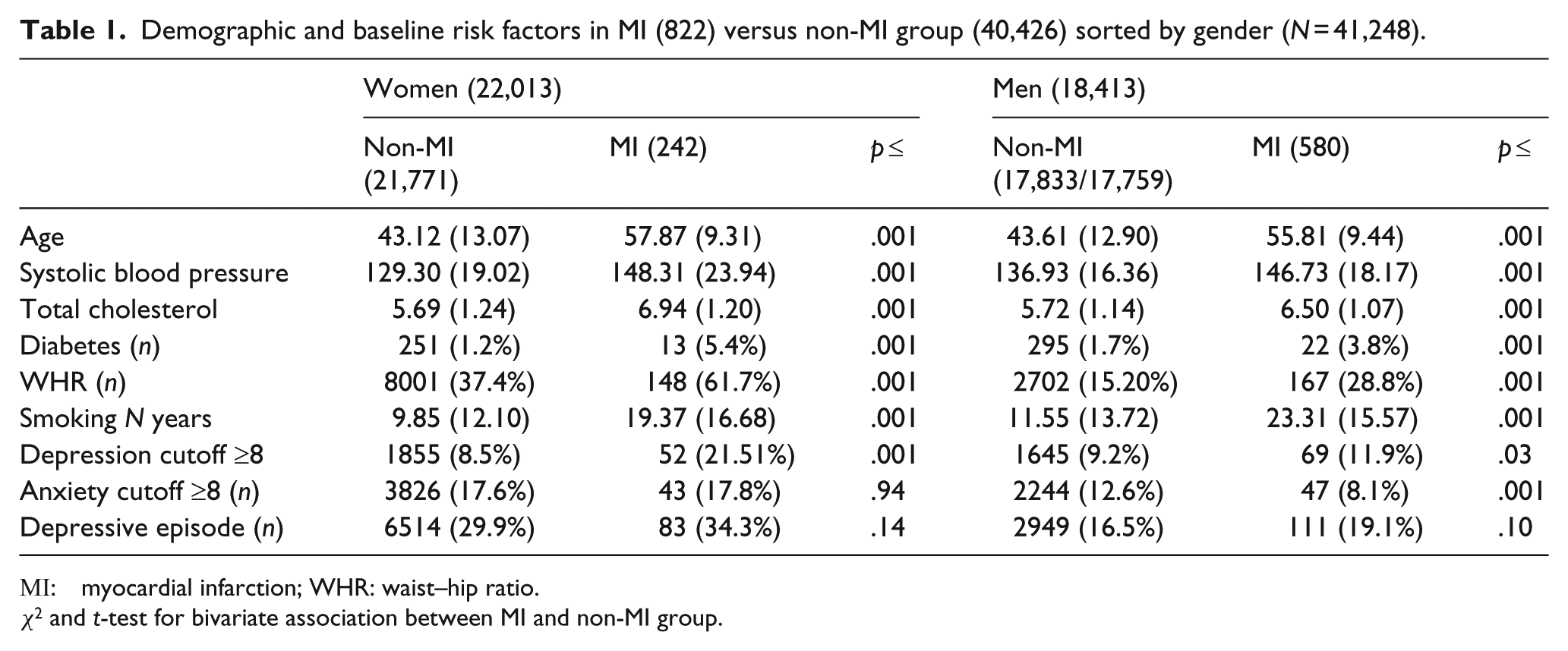
MI : myocardial infarction; WHR: waist–hip ratio.
χ2 and t-test for bivariate association between MI and non-MI group.
Table 2 shows the results of the logistic regression. In the total sample, elevated symptoms of depression (>8) were a significant predictor of MI, whereas history of depression was not. After adjusting for the traditional risk factors (gender, cholesterol, waist–hip ratio, diabetes, smoking, and SBP), history of depressive episode emerged as a significant predictor along with symptoms of depression. A high score on anxiety (HADS-A >8) significantly reduced the risk of having an MI both in the unadjusted and adjusted model. In the separate analysis, history of depression was not a significant predictor of MI in the female sample. Elevated symptoms of depression increased the odds of having an MI, whereas the opposite was the case for anxiety. Higher score on HADS-A represented reduced risk of having an MI in the male sample, and the effect size (odds ratio (OR)) of this predictor was stronger compared to the female sample in both the unadjusted and adjusted model. History of depressive episode was a significant predictor of MI in men, and elevated symptoms of depression were a significant predictor only in the unadjusted model.
Results of logistic regression.

CI: confidence interval; OR: odds ratio; HADS: Hospital Anxiety and Depression Scale.
Post hoc analyses of risk differences
To investigate whether the gender differences in risk were statistically significant, the statistical approach suggested by Paternoster et al. (1998) was applied. In this procedure, a Z-score is calculated, and according to the results, there was a significant difference in risk estimate based on HADS-D; Z = 2.96, p = .002. The difference in risk-based anxiety (Z = .62) and history of depression (Z = 1.57) did not reach statistical significance.
Discussion
Women have been marginalized in the search for psychological risk factors for CHD, and prospective studies applying separate analyses for men and women have been requested (Low et al., 2010; Naqvi et al., 2005; Wenger et al., 2008). In this study, different psychological risk profiles among men and women were identified and support the assumption that the association between depression and CHD is gender specific (e.g. Faravelli et al., 2013; Low et al., 2010). Depression, especially symptoms of anhedonic depression as measured by HADS, seems to be a strong predictor of MI among women, whereas this specific instrument did not contribute in predicting MI among men after controlling for traditional risk factors. Post hoc analysis of differences in risk estimate showed that this difference was statistically significant. This is consistent with other findings of symptoms of depression being more important in predicting CHD in women than men (Lloyd et al., 1996). History of depressive episodes was more prevalent in women and was a significant risk predictor in men only; however, the gender difference in risk was not significant. Although HADS-D and depressive episodes were correlated (Φ = .25), the strength of the relationship between do not suggest that these differences could be attributed to co-linearity. It rather supports the view that men and women represent different populations when it comes to understanding the link between depression and CHD (Wenger et al., 2008). The higher prevalence of history of depressive episodes in women might be the reason that it fails to differentiate those at risk of MI. The results support that gender differences in depression are important to incorporate in understanding the link between depression and CHD. Elevated symptoms of depression (HADS-D ≥8) were not more prevalent in women, and this is consistent with other studies using HADS (e.g. Nortvedt et al., 2006; Stordal et al., 2001). However, the results in this study are contrary to comparable samples where other measurements of depression have been applied (e.g. Mendes de Leon et al., 1998) and the general literature on gender differences (Piccinelli and Wilkinson, 2000). This suggests that HADS-D represents a distinct symptom cluster of depression that might be especially cardiotoxic for women. HADS does not include somatic symptoms, and HADS-D focuses mainly on anhedonia (Mykletun et al., 2001). Although the somatic cluster has been identified as the most important symptom cluster in predicting CHD (Hawkins et al., 2013), this study found that other symptom clusters of depression are relevant and that positive affect may be cardioprotective, especially for women. Hawkins et al. (2013) identified somatic clusters followed by positive affect as the most important symptom clusters predictive of CHD, but the analyses were not performed separately for men and women, and the role of specific symptom clusters as more cardiotoxic can only be understood by prospective studies including participants free of CHD at baseline. Furthermore, separate data analysis should be performed, not only controlling for gender (Low et al., 2010). The variable “Depressive episode” includes both somatic symptoms and lack of positive affect/depressed affect, and it might be that somatic clusters are more important for men than for women. A lifetime history of depressive episodes was a unique predictor of MI in men and not women in the adjusted models.
HADS does not include somatic symptoms, and this methodological aspect might be the reason why studies using HADS-A (e.g. Versteeg et al., 2013), including this study, do not find a positive association between anxiety and CHD. However, the study by Parker et al. (2011) using diagnostic criteria for GAD did find the same results as in this study, that is, anxiety being cardioprotective. Although distinctive conditions, the relationship between HADS-A and GAD is well documented (Olssøn, et al., 2005)
It is important to differentiate between anxiety and depression as a prognostic factor in CHD groups and risk factor of CHD in healthy populations. While the prognostic studies focus on patients with existing CHD, the etiologic approach focuses on psychological factors as risk factors of CHD, and these represent two different approaches (Nicholson et al., 2006; Stampfer et al., 2012). As suggested by Parker et al. (2011), timing is of great importance, and both anxiety and depression might have quite different roles as etiological factors compared to prognostic factors. However, the findings of this study of anxiety as a protective factor of MI in healthy subjects are consistent with studies of patients with acute coronary syndrome, where anxiety predicted superior 5-year outcomes (Parker et al., 2011). Men are more reluctant to self-refer to primary health care (Jeffries and Grogan, 2012), and this might be why the protective effect of anxiety is stronger for the male sample in this study. Although females were underrepresented, the review of seven etiological studies of initially disease-free individuals points to worry as being associated with fatal and non-fatal CHD (Tully et al., 2013). Limited knowledge about anxiety and CHD has been attributed to the assessment of anxiety, and worry has been found to have the strongest impact on the onset of CVD compared to panic and phobias (Batelaan et al., 2014). More knowledge about the association between depression and anxiety can be achieved through the focus on the differential impact of types of anxiety (Batelaan et al., 2014) informed by the focus on specific symptom cluster/phenotypes of depression (Hawkins et al., 2013; Shaffer et al., 2012). A higher level of specificity might be a promising approach toward a more comprehensive understanding of anxiety as a risk or protective factor of CHD in general and MI in particular for men and women.
Treatment of depression in CHD patients thus far has demonstrated only moderate effects (Baumeister et al., 2011), especially among women (Low et al., 2010). However, treatment of depression before the onset of CHD has been found to reduce the risk of CVD (Stewart et al., 2014). This points to the importance of primary prevention and suggests that addressing depression as a risk factor has more impact than the treatment of depression as a prognostic factor, and hence interventions should aim at screening those at risk. Effective and simple screening for depression in identifying CHD risk in non-cardiac patients could reduce the number of MI deaths among men and women. Screening for depression would have a positive impact regardless of its association with CHD as depression represents a major health problem (Möller-Leimkühler, 2007).
In the research on depression and CHD, depression has been treated as a unidimensional cluster, whereas more recent studies have demonstrated that particular depressive symptoms are stronger predictors (Hawkins et al., 2013), and the role of anhedonia has received particular attention (Davidson et al., 2010; Denollet et al., 2007; Shaffer et al., 2012). The implication of anhedonia as a specific cardiotoxic symptom is that this symptom cluster or depression subtype, or intermediary phenotype, should be more carefully addressed in the preventive strategies (Shaffer et al., 2012). It is also possible that the type of treatment has not been optimal for women (Low et al., 2010; Naqvi et al., 2005). In the research on anhedonia in major depression, agomelatine has shown promising results (Di Giannantonio and Martinotti, 2012). The potential of agomelatine to reduce the risk of CHD in patients with an anhedonic depression subtype should be explored as a possible new intervention in prospective studies applying a gender-specific approach.
Efficient ways of identifying women with increased CHD risk due to psychological factors is necessary in order to develop effective prevention and intervention strategies (Low et al., 2010). This study suggests that HADS-D could be used to identify risk in women, whereas information about lifetime depression/depressive episode is more promising in identifying risk among men. Both represent simple and non-invasive approach that easily could be adopted by health personnel. Anxiety as measured by HADS-A is not a suitable screening tool in identifying risk, according to the results of this study, which is in line with other studies that have used HADS to explore the association between anxiety and CHD (Versteeg et al., 2013). Further research should address whether symptoms of anxiety might be protective of MI in a general population.
It has been argued that it is premature to conclude that depression in general is an independent causal risk factor for CHD (Stampfer et al., 2012). The findings from this study support this notion, as it seems that too little is known about the specific aspects of depression as a CHD risk factor in men and women. A general recommendation to screen for anxiety and depression (Perk et al., 2012) might have little benefit, as specific subtypes of depression differentiate in predictive power, and the role of anxiety is far from conclusive. In research on the link between depression and CHD, more attention should be given to measurement issues and levels of specificity. Gender differences in depression are important to incorporate, as well as a focus on specific aspects of depression symptom clusters and different anxiety diagnoses. The results of this study suggest that it would be useful to develop gender-differentiated screening tools for depression in preventive CHD care and that the role of anxiety as a risk factor needs to be re-evaluated or reversed.
Strengths and limitations
This study contributes to the existing literature in the following ways: (1) prospective study of a large community-based sample free of CHD at baseline; (2) it includes all the major risk factors; (3) separate analysis is applied for men and women (4); two different measures of depression are included as potential screening tools; (5) it includes measures of anxiety; and (6) it includes both fatal and non-fatal MI as end-point measures.
The most obvious limitations in this study are as follows: (1) the reliance on self-reporting measures for several variables, and (2) the measure of depressive episode is unspecific, as it does not ask when it occurred or whether there have been recurrent episodes. Furthermore, a depressive episode as described here does not imply a valid assessment of major depressive episode as diagnosed in the Diagnostic and Statistical Manual of Mental Disorders (5th ed.; DSM-V). Although HADS has been found to be a reliable screening instrument of psychological stress, the factorial structure of HADS has been questioned (Martin et al., 2003).
This study indicates that men and women represent different populations in the investigation of psychological risk factors in the development of MI and that the association between depression, anxiety and CHD is gender specific. This study emphasizes the importance of screening for depression in women with no CHD at baseline. Symptoms of anhedonic depression measured by HADS-D might be a gender-specific risk factor of MI in women.
Footnotes
Acknowledgements
The authors would like to thank the HUNT Research Centre for enabling this research, without whose permission and helpful assistance this study would not be possible.
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
