Abstract
Background:
Kidney transplantation is the most effective treatment for patients with end-stage kidney disease. Modern immunosuppressive treatment has prolonged graft survival and reduced transplant rejection. However, these immunosuppressive regimens are associated with multiple side effects, including increased infections and cancer risk, nephrotoxicity, and metabolic complications. Attaining transplant tolerance with minimal reliance on immunosuppressive drugs is considered the ultimate goal in transplantation. Regulatory T cells (Tregs) adoptive immunotherapy has been proposed as a strategy to achieve transplant tolerance. However, this approach is labor-intensive and expensive, limiting its large-scale applicability. There is, therefore, a need to develop methods that promote Treg-mediated tolerance in vivo. Treg function and numbers are influenced by dietary components, and dietary interventions could provide a new therapeutic opportunity. Recently, our group demonstrated that sucralose supplementation, a commonly used sugar substitute (sweetener), reduces proinflammatory T-cell function and unpublished data indicating enhanced Treg frequencies. Building on this discovery, we propose to investigate the impact of sucralose on modulating T-cell populations and function in humans.
Objective:
To determine whether sucralose supplementation increases circulating Treg frequencies and alters T-cell function and populations in healthy adults.
Design:
This is a randomized, double-blind, placebo-controlled, crossover pilot trial.
Setting:
The study will be monocentric, at the Maisonneuve-Rosemont Hospital.
Patients:
Twelve healthy adult volunteers (>18 years of age), of both sexes, with no prior history of autoimmune disease or current treatments of immunomodulatory drugs. In addition, pregnant women will also be excluded from this study.
Measurements:
The primary outcome will be an alteration in circulating Treg frequency after sucralose supplementation. The secondary outcomes include modulation in the frequency of CD45+ cells, the frequency of CD4+ T-cell subsets, the differentiation of both CD4+ and CD8+ T cells, and T-cell function after antigen-specific and alloreactive challenges. Feasibility will also be evaluated, including adherence to visits, blood draw, ease of recruitment, percentage of study completion, and adherence to supplements. Exploratory outcomes in response to sucralose supplementation include changes in circulating metabolites and gut microbiome composition.
Methods:
Participants will be randomly assigned to receive either a placebo or sucralose (5–7 mg/kg/day) for four weeks, separated by a two-week washout period. This will be followed by a crossover phase, where patients receiving sucralose will receive the placebo and vice versa for an additional four weeks. Capsules will be prepared and blinded by a licensed pharmacy. Blood will be collected at baseline, after four weeks of treatment or placebo, after washout and after four weeks of crossover for Peripheral Blood Mononuclear Cell (PBMC) isolation, flow cytometry, functional assays, and metabolomics. Stool samples will be collected for microbiome sequencing.
Limitations:
This will be a small, single-center, short-duration trial in healthy volunteers. Findings might not apply to transplant recipients.
Conclusion:
The SWEET trial will provide first-in-human data on sucralose as a potential, inexpensive oral immunomodulator to promote Tregs.
Trial Registration:
NCT06997133 (2025-06-05)
Introduction
Chronic kidney disease (CKD) is a condition that results in progressive loss of kidney function. 1 It affects one in ten adults globally and is associated with high mortality. 2 In patients with the most severe form, kidney transplantation is the preferred treatment as it increases both survival and quality of life. 3 However, patients need to take life-long immunosuppressive drugs, which are associated with several side effects, including an increased risk of infection and cancer. 4 In Canada, the ten-year graft survival rate is 58% for recipients of deceased donor transplants and 77% for those who receive transplants from living donors. 5 Infection and cardiovascular disease are the most common reasons for death after a kidney transplant. 6 Graft loss is also a very relevant problem and challenge. The primary causes of functional graft failure are infections, cardiovascular diseases, and chronic allograft dysfunction (a progressive decline in renal function) that can result from over- or under-immunosuppression. 7 Therefore, there is a critical need to improve transplant tolerance while limiting the side effects of immunosuppressive drugs to protect patients from infection.
While kidney transplantation provides the best long-term solution for end-stage renal disease, it still faces considerable challenges, including the disparity between donor and recipient major histocompatibility antigens, which activate the recipient’s immune system and cause rejection of the donor kidney. In case of limited compatibility between donor and recipient, the allograft will activate a pool of the recipient’s naïve T cells, leading to the generation of pathogenic and allograft-specific effector T cells. CD4+ and CD8+ T cells play a key role in transplant rejection, leading to T-cell-mediated rejection (TCMR), which can manifest as acute TCMR or progress to chronic TCMR, ultimately resulting in fibrosis and allograft loss. The activation of CD4+ and CD8+ T cells also leads to the formation of memory T cells, which are continuously reactivated by the allograft, contributing to organ rejection. 8 Identifying treatments to prevent and/or treat TCMR may improve the longevity and function of kidney transplants.
The body is inherently designed to sustain tolerance, as exemplified by processes like wound healing and pregnancy. 9 Therefore, promoting tolerance mechanisms could be therapeutically beneficial in patients. Immune tolerance is achieved via a subpopulation of T cells known as regulatory T cells (Tregs). 10 Tregs elicit their suppressive function by cytokine-mediated suppression (such as IL-10 and TGF-β), IL-2 deprivation, inhibition of dendritic cell maturation, cytolysis (Granzyme B and Perforins) of inflammatory cells, and the production of immunosuppressive metabolites.11,12 Therefore, modulating Treg numbers and suppressive function in the context of allograft transplantation is a holistic approach to transplant survival. 10 The expansion and adoptive transfer of autologous Tregs during kidney transplantation have demonstrated safety in Phase I/II trials. 13 However, such procedures are inaccessible to most transplant recipients because of the cost of a designated and certified manufacturing facility, time, and scalability. Therefore, identifying new and accessible strategies that promote Tregs while dampening proinflammatory and host anti-graft (alloreactive) T cells will profoundly benefit patient long-term survival.
Environmental factors, such as diet, modulate CD4+ and CD8+ T-cell priming, differentiation, and function.14-16 Both dietary composition and calorie intake regulate T effector, memory, and regulatory populations.9,17,18 Commercial food often contains additives besides nutrients to preserve or increase food palatability, such as artificial sweeteners, which enhance sweetness while providing negligible calorie intake. 19 While all artificial sweeteners have undergone extensive toxicity testing and have been approved by the health and food authorities for human consumption, recent studies have shown that artificial sweeteners can affect biological responses.20-23
Sucralose is a chlorinated sucrose derivative approximately 600 times sweeter than sugar and is approved for human consumption by health authorities worldwide. The acceptable daily intake (ADI) set by Health Canada is 9 mg/kg/day. While generally considered metabolically inert, emerging research indicates that sucralose can modulate immune cell function. In a recent study by our group, 24 sucralose consumption in mice at human-equivalent doses reduced T-cell proliferation and dampened proinflammatory CD4+ T-helper 1 and effector CD8+ T-cell differentiation. Importantly, sucralose favors anti-inflammatory Tregs in both cell culture differentiation and multiple murine models, including autoimmune colitis and an MHC-mismatched tumor rejection model (unpublished data). However, the therapeutic potential of sucralose supplementation as a tool to increase Tregs population in humans remains unexplored.
The Sucralose as a Way to Enhance Regulatory T cells (SWEET) trial is a randomized, double-blind, placebo-controlled, crossover pilot study designed to assess the effects of sucralose supplementation on circulating Treg frequencies and T-cell function in healthy adult volunteers. By using a controlled dosing regimen close to, but below, the Health Canada-approved ADI and incorporating detailed immunophenotyping, metabolomics, and microbiome analyses, this study will provide the first human data on the immunologic impact of sucralose.
If sucralose is found to reproducibly increase Tregs and reduce proinflammatory T-cell responses in humans, it could be rapidly translated into clinical trials in kidney transplant recipients as an adjunct to conventional immunosuppression, with the potential to improve graft survival while minimizing toxicity.
Methods
Study Design and Patient Characteristics
The Sucralose as a Way to Enhance Regulatory T cells (SWEET) trial is a single-center, randomized, double-blind, placebo-controlled, cross-over pilot study that will be conducted at the Maisonneuve-Rosemont Hospital (HMR) Research Center in Montreal, Canada. The study is designed to determine whether short-term sucralose supplementation in healthy adults alters circulating Treg frequencies and T-cell function. The study protocol has received Research Ethics Board (REB) approval (#2026-3912) and is registered at ClinicalTrials.gov (NCT06997133).
Participants
Adults aged 18 years or older are eligible to participate. All participants must be able and willing to restrict their additional dietary sucralose intake during the trial and provide written informed consent. Exclusion criteria include a history of autoimmune disease, use of chronic immunomodulatory medication, pregnancy or breastfeeding, and habitual high sucralose consumption estimated at more than 1.5 mg/kg/day. No more than eight of the 12 participants will be of the same sex to limit sex-related immunologic variability.
Recruitment and Screening
Recruitment will be carried out through word-of-mouth within the research network at Maisonneuve-Rosemont Hospital. At screening, participants will complete an eligibility assessment and a dietary questionnaire to estimate baseline sucralose intake. They will meet with a registered nutritionist for education on identifying and avoiding sucralose-containing foods, beverages, and pharmaceuticals, such as splenda packets, diet sodas, flavored water, protein bars, protein powder, and low-fat dairy products.
Randomization and Blinding
Participants will be randomized in a 1:1 ratio to receive either sucralose or placebo in the first phase. Randomization will be performed by the hospital pharmacy using the NIH Clinical Trial Randomization Tool. The pharmacy will conceal allocation and is responsible for preparing, coding, and dispensing the study capsules. A licensed compounding pharmacy will manufacture sucralose and placebo (cellulose) capsules. Capsules will be identical in size, color, and packaging. Blinded labels will include a removable section for pharmacy use and a concealed section to maintain allocation concealment. Randomization codes will be held by the research pharmacy and will not be accessible to study staff until the end of the trial. Outcome assessors and investigators performing statistical analyses will also remain blinded to treatment allocation throughout the study to minimize bias.
Intervention
Participants will receive sucralose at a dose of 5–7 mg/kg/day, divided into two doses taken morning and evening, for four weeks. Combined with unconscious dietary intake (approximately 1.5 mg/kg/day), 25 total exposure will remain below the Health Canada acceptable daily intake of 9 mg/kg/day. The placebo will consist of cellulose capsules matched for appearance and weight. After completing the first phase, participants will undergo a two-week washout period before crossing over to the alternate treatment arm for another four weeks (Figure 1).
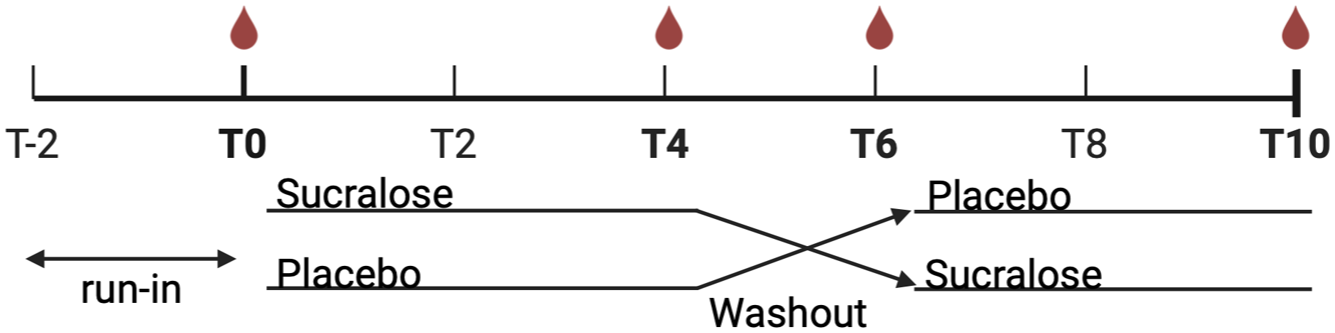
SWEET pilot study design.
Study Visits and Sample Collection
The trial will begin with a two-week run-in phase during which participants will abstain from dietary sucralose. The baseline visit will include anthropometric measurements and the collection of blood and stool samples. Subsequent visits take place at the end of each intervention phase and after the washout period. At each visit, blood will be collected in lithium-heparin tubes for isolation of peripheral blood mononuclear cells (PBMCs), and stool samples will be provided in anaerobic gas packs. Adherence, dietary changes, and adverse events will be recorded at every visit.
Outcomes
The primary outcome is the change in frequency of circulating Tregs, defined as CD4+FOXP3+ or CD4+CD25HICD127low cells, measured by flow cytometry as a proportion of total CD4+ T cells. Secondary outcomes include changes in (i) the frequency in immune cell composition of CD45+ cells; (ii) the frequency of CD4+ T-cell subsets; (iii) frequencies of naïve, effector memory and central memory CD4+ and CD8+ T cells; (iv) T-cell activation after an antigen-specific challenge (activation marker and cytokine secretion); (v) T-cell activation after an allogenic challenge (proliferation and cytokine secretion). Exploratory analysis will include plasma metabolic profiles and gut microbiome composition. Feasibility will also be evaluated, including adherence to visits, blood draw, ease of recruitment, percentage of study completion and adherence to supplements.
Laboratory Analyses
PBMCs will be extracted by Ficoll isolation, then cells will be stained for flow cytometry, to detect Tregs, CD4+ and CD8+ T cells, as well as subtypes (naive, memory, and effector) and proinflammatory CD4+ T cell effector lineage, NK cells, B cells, and CD11b myeloid cells.
Antigen-specific memory T-cell function will be evaluated by exposing PBMCs to a peptide pool of a common seasonal virus peptide, hexon from Adenovirus (Ad). Cell activation will be assessed by examining activation markers using flow cytometry. Cytokine secretion will be assessed by intracellular cytokine staining or alternatively by an ELISPOT assay. Alloreactivity will be evaluated in a mixed lymphocyte reaction. To do so, we will establish a co-culture of the participant PBMCs with irradiated allogenic PBMCs. Proliferation will be assessed using a cell proliferation dye, and cytokine production will be measured via ELISAs.
Metabolites will be extracted from the plasma via methanol-based extraction and analyzed by liquid chromatography-mass spectrometry (LC-MS). We will first assess the levels of sucralose per volunteer prior to commencing the study, as each individual will have unique starting levels of sucralose due to its widespread use in food and pharmaceutical products (e.g. toothpaste). 25 Second, we will evaluate global metabolite changes by untargeted metabolomics, a comprehensive approach to assessing the full range of metabolites. In addition, using targeted metabolomics, analysis of a predefined metabolite pool, we will analyze sugars (glucose, fructose, mannose, etc.), amino acids, nucleotides, and nucleosides, as well as metabolic intermediates, such as the Krebs cycle, to evaluate their variability in response to sucralose exposure.
Sucralose has been found in some, but not all, studies to modulate the microbiome, which could therefore impact the host.24,26-28 Consequently, microbiome composition will be analyzed from stool DNA extracts by 16S rRNA gene sequencing.
Statistical Analysis
Analyses will be performed using GraphPad Prism (version 10) and R (4.5.1). Paired statistical tests will be used for within-participant comparisons between sucralose and placebo phases. Mixed-effects models will be applied to account for sequence and period effects in the cross-over design. Based on a correlation coefficient of 0.3 and a 2-sided alpha of 0.05, a sample size of ten participants provides 80% power to detect a 20% absolute change in Treg frequency. To account for potential loss to follow-up and unforeseen issues with sample quality, we increased the planned enrollment to 12 participants. Finally, multivariate analyses will be used for metabolomic and microbiome data.
Discussion
The SWEET trial will be the first human study to investigate whether short-term sucralose supplementation can modulate immune cell populations in vivo, with a particular focus on enhancing regulatory T cells (Tregs). This work is motivated by robust preclinical evidence demonstrating that sucralose, at doses equivalent to those approved for human consumption, alters T-cell function, reduces effector responses, and increases Treg frequencies. 24 If similar effects are observed in humans, sucralose could represent a safe, novel, low-cost, and widely accessible immunomodulatory strategy in transplantation.
Long-term kidney graft survival remains limited, with chronic immune-mediated injury as a leading cause of graft loss. 29 Strategies to selectively enhance immune tolerance without increasing the risk of infection or malignancy would therefore have a profound clinical impact. One potentially therapeutic intervention is the adoptive transfer of ex vivo expanded Tregs, which is safe and feasible.13,30 However, Treg expansion remains resource-intensive and inaccessible for widespread use. A dietary intervention that achieves similar Treg-promoting effects could offer an accessible and achievable alternative. This clinical trial will provide insight into the cellular mechanisms of sucralose consumption in humans, thus creating a foundation for subsequent trials in transplant recipients. There is always a potential risk that therapies designed to increase Tregs in kidney transplant recipients could inadvertently elevate susceptibility to infections or malignancies, considering the central role of T cells in immune surveillance. However, results from Phase I/II clinical trials of adoptive Treg immunotherapy demonstrate that the incidence of infections or cancer remains unchanged.13,31 In fact, some studies have shown a reduction in infection rates, likely a reflection of reducing the use of immunosuppressive drugs during Treg immunotherapy.31,32 While this trial is being conducted in healthy volunteers, these considerations will be critical when designing future studies in transplant recipients.
While numerous preclinical studies interrogated how different nutrients and certain diets can modulate immune function,9,17,18 few have assessed how non-nutritive artificial sweeteners influence immune responses. Our group’s preclinical work identified a unique effect of sucralose on dampening proinflammatory T cells and demonstrated protective effects in autoimmune and allograft rejection models. 24 This trial will extend these findings to humans and assess both phenotypic and functional T-cell outcomes.
Strengths and Innovations
This study integrates detailed immune phenotyping, ex vivo functional assays, metabolomics, and microbiome analysis in a controlled, double-blind, randomized crossover design. The within-participant comparison increases statistical power despite the small sample size, and the washout period minimizes carryover effects. The study also controls for confounding dietary intake of sucralose and other artificial sweeteners, an important consideration given their widespread use in processed foods. By including both healthy male and female participants, the trial will allow exploratory assessment of potential sex-related differences in immune modulation.
Limitations
Several limitations must be acknowledged. The small sample size limits the precision of effect-size estimates and precludes definitive conclusions about clinical efficacy. However, this pilot study will be essential to infer the number of patients required for a larger cohort study to perform power calculations. The short duration of exposure may not capture longer-term effects of sucralose on immune function or the microbiome. Restricting the population to healthy volunteers improves internal validity but limits generalizability to patients with CKD or transplant recipients, whose immune systems are altered by chronic disease and immunosuppression. Additionally, seasonal variation in infections and immune responses and antibiotic use could confound results, although these will be monitored and recorded. Finally, while the two-week washout period was chosen based on sucralose pharmacokinetics and prior evidence of Treg normalization within this timeframe with low-dose IL-2, 33 individual variability could result in longer biological effects, which should be considered when interpreting the findings.
Conclusion
The Sucralose as a Way to Enhance Regulatory T cells (SWEET) trial will provide the first in vivo human data on the immunologic effects of sucralose supplementation, with a specific focus on regulatory T cells. This randomized, double-blind, placebo-controlled, crossover study is designed to yield mechanistic insights into how sucralose may shift the immune balance toward tolerance. By integrating immune phenotyping, functional assays, metabolomics, and microbiome analyses, the trial will generate a comprehensive dataset that can guide the development of future studies in kidney transplantation and other clinical contexts in which immune regulation is critical. If the findings mirror preclinical results, sucralose could represent a novel, low-cost and globally accessible way to promote transplant tolerance.
Footnotes
Authors’ Note
This study involves a widely used, commercially available dietary sweetener at doses within the Health Canada acceptable daily intake, minimizing risk to participants. Adverse events will be monitored at each visit, with procedures in place for unblinding and medical evaluation if needed.
Ethical Considerations
This study has been approved by the Research Ethics Board of the Maisonneuve-Rosemont Hospital (#2026-3912). The trial has been registered on ClinicalTrials.gov (NCT06997133). All data will be collected and stored in accordance with Good Clinical Practice guidelines and Canadian privacy regulations. Participants will be identified by coded numbers to protect confidentiality, and only authorized study personnel will have access to identifiable data.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The SWEET trial is funded by the Kidney Foundation of Canada.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
