Abstract
Background:
Metabolic acidosis is a common complication of kidney failure that is treated with bicarbonate supplementation. The addition of bicarbonate to the dialysis solution and oral bicarbonate supplementation are used to treat metabolic acidosis in patients receiving dialysis, but the treatment approach that is best for patient health remains unknown.
Objective:
The purpose of this study was to determine whether the concentration of dialysate bicarbonate or the use of oral bicarbonate supplementation alters the risk of mortality, hospitalizations, cardiovascular and nutritional outcomes, and laboratory measurements in patients treated with maintenance dialysis.
Design:
Systematic review and meta-analysis.
Setting:
Any country of origin.
Patients:
Adult patients (≥18 years) receiving maintenance dialysis.
Measurements:
Extracted data included demographic characteristics and outcomes such as mortality, hospitalizations, cardiovascular events, surrogate markers of nutrition, and pre-dialysis and post-dialysis levels of serum bicarbonate, pH, calcium, potassium, and parathyroid hormone.
Methods:
We searched MEDLINE, Embase, CENTRAL, and Google Scholar through October 7, 2024 for studies examining dialysate bicarbonate concentration and/or oral bicarbonate supplementation in adults undergoing maintenance dialysis. Meta-analysis was performed for pre-dialysis serum bicarbonate and for pre-dialysis and post-dialysis calcium and potassium.
Results:
We identified 37 studies (n = 24,782 patients) with patients treated with hemodialysis (13 randomized trials, 10 non-randomized interventional studies, 14 observational studies) and 4 studies (n = 347 patients) with patients receiving peritoneal dialysis (3 randomized trials, 1 non-randomized interventional study). No randomized trials reported mortality or hospitalizations in hemodialysis patients. Studies reporting cardiovascular outcomes (n = 20) were small with inconsistent results. Most studies reporting nutritional outcomes (n = 21) reported no significant differences with dialysate bicarbonate concentration or oral bicarbonate supplementation but were small in sample size (largest study n = 200). Meta-analysis of parallel-group randomized trials comparing dialysate bicarbonate >35 mmol/L with ≤35 mmol/L found a mean difference of 3.5 mmol/L (95% confidence interval [CI] −0.6 to 7.7) in pre-dialysis serum bicarbonate.
Limitations:
Non-English and gray literature were excluded. Most studies were small or observational in nature, and heterogeneity further limited the ability to perform meta-analysis of outcomes such as mortality, hospitalizations, and cardiovascular outcomes.
Conclusions:
The evidence for the effect of higher vs lower dialysate bicarbonate concentration and oral bicarbonate supplementation on clinical outcomes in dialysis patients is very uncertain. There is a need for large, high-quality randomized controlled trials in this area.
Introduction
Kidney failure leads to metabolic acidosis, which is associated with adverse patient outcomes, including malnutrition, bone disease, hospitalizations, and increased mortality. Metabolic acidosis contributes to protein-energy wasting, which is common among patients receiving maintenance dialysis, and is associated with increased morbidity and mortality. 1 For patients receiving hemodialysis (HD) or peritoneal dialysis (PD), metabolic acidosis is treated primarily by the addition of bicarbonate to the dialysate; however, daily oral sodium bicarbonate supplementation may also be used. Guidelines recommend a serum bicarbonate concentration ≥22 mmol/L, measured pre-dialysis in patients treated with HD, which is based on low quality evidence that suggests an improvement in measures of nutrition and bone metabolism with correction of metabolic acidosis.1,2 However, observational data from the international Dialysis Outcomes and Practice Patterns Study (DOPPS) showed that a higher (vs lower) concentration of dialysate bicarbonate concentration was associated with a higher risk of hospitalizations and death. 3 This association may be causal, potentially explained by an increased risk of significant intra-dialysis and post-dialysis metabolic alkalosis with high dialysate bicarbonate concentrations. However, this result may be confounded (i.e., higher observed mortality with higher dialysate bicarbonate concentration may be driven by differences in age, comorbidities, and central venous catheter use). 3
The optimal concentration of dialysate bicarbonate and whether oral bicarbonate supplementation should be used remain uncertain. The purpose of this systematic review and meta-analysis was to determine whether the concentration of dialysate bicarbonate or the use of oral bicarbonate supplementation alters the risk of mortality, hospitalizations, cardiovascular and nutritional outcomes, and laboratory measurements in patients treated with maintenance dialysis.
Methods
This systematic review is reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) criteria 4 (Supplemental Appendix A), with the exception of prospective protocol registration. We support protocol registration in our future research to prevent unintended duplication and to increase transparency.
Study Selection
Eligible populations were adults ≥18 years undergoing maintenance dialysis (any modality) for the treatment of kidney failure. Studies with at least 2 comparison groups evaluating dialysate bicarbonate concentration or oral sodium bicarbonate supplementation were included. Outcomes included the following: mortality, hospitalizations, cardiovascular outcomes (e.g., myocardial infarction, stroke, blood pressure (BP), intradialytic hypotension), surrogate markers of nutrition (e.g., serum albumin, body mass index (BMI)), pre-dialysis and post-dialysis serum bicarbonate, pH, calcium, potassium, and parathyroid hormone (PTH). Additional eligibility criteria were English language and articles that were publicly available or could be ordered through our library system. Publications in the form of consensus statements, protocols, abstracts, conference proceedings, letters, or editorials were excluded. Case studies, case series, studies with less than 20 patients, studies comparing acetate dialysate versus bicarbonate dialysate and studies performed on animals or conducted in vitro were also excluded.
Searching and Screening
MEDLINE, Embase, CENTRAL, and Google Scholar were searched from inception to October 7, 2024 inclusive. The search strategy was developed with the assistance of a librarian and included Medical Subject Headings and keywords that reflect terms commonly used in the literature to refer to dialysis and bicarbonate treatment in patients with kidney failure (see Supplemental Appendix B). Using Covidence software (Covidence, Australia), AOM and AMA screened articles retrieved from the search, first by titles and abstracts, and subsequently by full text to confirm eligibility for inclusion in the review. Disagreements were resolved through discussion between the two reviewers in accordance with the eligibility criteria.
Data Extraction and Synthesis
Data extraction was performed in duplicate by AMA and ZKF and verified by AOM using a standardized data extraction form developed and piloted on Covidence software (see Supplemental Appendix C for data abstraction form). When data were available only in figures and not numerically reported, WebPlotDigitizer software (Automeris, US), which has demonstrated high reliability and validity, was used.5,6 Where appropriate, data were pooled using meta-analysis; otherwise, results were summarized narratively.
Assessment of Study Quality
Study quality was assessed independently and in duplicate by AMA and ZKF and verified by AOM. The risk of bias for randomized controlled trials (RCTs) was assessed using the Cochrane Risk of Bias 2 tool for parallel or crossover trials; 7 non-randomized studies of interventions were assessed using the ROBINS-I tool; 8 prospective or retrospective cohort studies were evaluated using the Newcastle-Ottawa Scale; 9 and cross-sectional studies by the Joanna Briggs Institute (JBI) checklist. 10 Risk-of-bias figures were created using the Risk-of-bias VISualization (robvis) tool (University of Bristol, UK).
Statistical Analysis
Meta-analysis was conducted using a random-effects model on RevMan 5.4 software (Cochrane, UK) and performed on data extracted from studies that were deemed to be reasonably homogenous in study population, intervention, methodology, and outcome measurement. A priori, we planned to perform meta-analysis on randomized, non-randomized interventional and observational studies separately and HD and PD studies separately. Final decisions regarding studies included in meta-analysis and comparator groups were data-driven; data from parallel-group RCTs or crossover RCTs that included patients on HD were separately pooled when deemed appropriate, whereas results from non-randomized studies and PD studies were described narratively. Dialysate bicarbonate concentration categories used for the meta-analysis comparisons were >35 mmol/L versus ≤35 mmol/L (parallel-group RCTs; outcomes of pre-dialysis serum bicarbonate, total calcium, and potassium) and ≥32 mmol/L versus ≤29 mmol/L (crossover RCTs; outcomes of pre-dialysis and post-dialysis ionized calcium and potassium). All outcomes were continuous, and the effect estimates were mean differences (MD). Where applicable, standard error was converted to standard deviation (SD) prior to meta-analysis.
Assessment of the Certainty of the Evidence
Certainty of the evidence for each outcome included in the meta-analysis was evaluated using the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) framework. 11 RCTs started as high certainty and were rated down based on concerns in any of the five domains: risk of bias, inconsistency, indirectness, imprecision, or publication bias. Funnel plots were not used to assess publication bias because none of the comparisons had 10 or more studies. With fewer studies, funnel plot asymmetry may arise by chance, 12 leading to potentially misleading assessments of publication bias. Therefore, no formal assessment of publication bias was conducted. The overall grading of the evidence related to each outcome was generated using GRADEPro GDT software (McMaster University and Evidence Prime, Canada).
Results
Patient and Study Characteristics
There were 41 studies that met inclusion criteria (n = 37 HD or hemodiafiltration [HDF]), n = 4 PD) (Supplemental Figure 1).3,13 -52 Eligible HD or HDF studies were published from 1989 to 2024. All studies were single-center, except one international cohort study that used DOPPS data, 3 and the number of included patients ranged from 2036,37 to 17,031. 3 Mean age (SD) of included patients varied from 40 (11) to 74 (13) years. HD or HDF study designs were parallel-group RCTs (n = 7), crossover RCTs (n = 6), non-randomized interventional (n = 10), cohort (n = 11), and cross-sectional (n = 3) studies. Interventions were variable across studies (Supplemental Table 1). Eligible PD studies were published from 1997 to 2017, and sample sizes ranged from 40 to 200 patients. There were 3 randomized parallel-group RCTs42,43,48 and 1 non-randomized interventional study 41 (Supplemental Table 2). The largest HD and PD parallel-group RCT included 93 and 200 patients, respectively.17,48
Study Quality and Risk of Bias
Most HD/HDF RCTs had some concerns (n = 7)16,18 -21,24,52 or high risk of bias (n = 4)17,22,25,33 (Figure 1), as did most of the non-randomized interventional HD and HDF studies (Figure 2). The quality of HD/HDF observational studies is summarized in Supplemental Tables 3 and 4. Cohort studies varied in quality with 5 out of 11 studies rated as good quality,3,44,45,49,51 while all cross-sectional studies (n = 3) were of moderate quality.14,30,39 PD RCTs had low risk of bias42,43 or some concerns 48 (Figure 1), while the non-randomized interventional study had a moderate risk of bias 41 (Figure 2).

Risk of bias of randomized controlled trials.a

Risk of bias of non-randomized studies of interventions.a
Mortality and Hospitalizations
No HD/HDF RCTs examined mortality or hospitalizations. A prospective cohort study using DOPPS data found that a higher dialysate bicarbonate concentration (per 4 mmol/L higher) was associated with a higher risk of mortality (adjusted hazard ratio [HR] = 1.1, 95% confidence interval [CI] 1.0-1.2) and hospitalizations (adjusted HR = 1.1, 95% CI 1.0-1.1). 3 A retrospective cohort study found a higher all-cause mortality risk (HR = 3.3, 95% CI 1.5-7.4) associated with a dialysate bicarbonate concentration ≥33.6 mmol/L versus 31.3-32.3 mmol/L, but no significant association with the outcome of first hospitalization (Table 1). 44 An RCT conducted in patients treated with PD (n = 200) found the hospitalization rate and number of days spent in hospital per year to be significantly lower in the group treated with high alkali dialysate and oral sodium bicarbonate than those in the low alkali dialysate group. 48 Another RCT of patients treated with PD (n = 60) found no difference in all-cause mortality or the number of hospitalizations with oral bicarbonate supplementation versus placebo (Table 2). 43
Mortality and Hospitalization Outcomes in HD/HDF Studies.

Note. Conversion factor: Bicarbonate conventional unit (mEq/L) to SI unit (mmol)/L = 1. Values rounded to one decimal place or one significant figure. HR = hazard ratio.
Mortality and Hospitalizations in PD Studies.

Note. Values rounded to one decimal place or one significant figure. Conversion factors: Lactate conventional unit (mg/dL) to SI unit (mmol/L) = 0.111.
Cardiovascular Outcomes
There were 17 HD/HDF studies that reported cardiovascular outcomes (Table 3).3,15,17,20 -24,26,27,30,33,35,38,39,49,52 No studies reported stroke or myocardial infarction. The most commonly reported outcomes were systolic (SBP) and/or diastolic blood pressure (DBP) (9 studies),15,22 -24,26,35,38,39,52 and intradialytic hypotension (4 studies),3,17,24,38 and results were mixed. One crossover RCT reported significantly lower mean intradialytic SBP and DBP with a higher dialysate bicarbonate concentration (32 vs 26 mmol/L), but no significant difference in lowest intradialytic DBP. 24 Two parallel-group RCTs found no significant differences in pre-dialysis SBP and DBP with oral bicarbonate supplementation (compared to placebo).15,52 Two crossover RCTs comparing higher vs lower dialysate bicarbonate concentrations found no significant differences in intradialytic SBP or DBP.22,23 One non-randomized interventional study examining an increase in dialysate bicarbonate depending on the pre-dialysis serum bicarbonate concentration found no difference in SBP from baseline to end of follow-up. 26 Two other non-randomized interventional studies found no significant difference in pre-dialysis SBP or DBP with oral bicarbonate supplementation,35,39 whereas one study reported increased maximum mean pre-dialysis SBP and DBP following treatment with oral bicarbonate, but no impact on post-dialysis SBP or DBP. 38
Cardiovascular Outcomes in HD/HDF Studies.

Note. CI = confidence interval, HD = hemodialysis, HR = hazard ratio, SBP = systolic blood pressure, DBP = diastolic blood pressure, IRR = incidence rate ration, aIRR = adjusted incidence rate ratio. Conversion factors: Bicarbonate conventional unit (mEq/L) to SI unit (mmol/L) = 1, Calcium conventional unit (mg/dL) to SI unit (mEq/L) = 0.25, potassium conventional unit (mEq/L) to SI unit (mmol)/L = 1, sodium conventional unit (mEq/L) to SI unit (mmol)/L = 1. Values rounded to one decimal place or one significant figure.
There was a positive association between dialysate bicarbonate concentration and the incidence of intradialytic hypotension in 2 of 3 studies comparing higher vs lower dialysate bicarbonate,3,24 while the incidence of intradialytic hypotension was found to be lower following oral bicarbonate supplementation in one pre-post intervention study. 38 The largest HD parallel-group RCT found no significant difference in the rate of intradialytic hypotension between dialysate bicarbonate groups of 34 vs 30 mmol/L, 17 whereas the largest observational study found a positive association between dialysate bicarbonate concentration and the incidence of intradialytic hypotension (HR per 4 mmol/L higher bicarbonate concentration, 1.1, 95% CI 1.0-1.3). 3
QTc interval was reported in two crossover RCTs; one trial comparing dialysate bicarbonate concentrations of 24 vs 28 mmol/L found no difference between groups in QTc interval pre-dialysis or post-dialysis, 20 and one trial found a significantly larger increase in QTc during dialysis with higher vs lower dialysate bicarbonate concentration (40 ms ± 10 vs 2 ms ± 2 with dialysate bicarbonate 34 vs 30 mmol/L respectively). 21
An RCT in 200 patients treated with PD found no between-group difference in mean SBP or DBP (130 ± 2/78 ± 1 mm Hg vs 129 ± 3/76 ± 1 mm Hg, high vs low alkali) at 1 year. 48 The other two PD RCTs also did not find any differences in SBP or DBP (Supplemental Table 5).42,43
Nutritional Outcomes
There were 18 HD/HDF studies that reported nutritional outcomes (Supplemental Table 6).14 -16,18,25 -28,32,34 -37,39,45,50 -52 RCTs (n = 3) comparing different dialysate bicarbonate concentrations found no difference in serum albumin.16,18,25 An RCT comparing daily oral sodium bicarbonate supplementation to no supplementation found higher albumin in the intervention group than the control group at 3 months (4.2 ± 0.5 g/L vs 3.8 ± 0.5 g/L, P = .01). 52 Most non-randomized interventional studies (n = 4 of 6) found no difference in serum albumin with higher dialysate bicarbonate concentration or with oral bicarbonate supplementation.27,35 -37 Most HD/HDF studies that examined normalized protein catabolic rate (nPCR) (n = 6 of 8) found no difference with higher, compared with lower, dialysate bicarbonate concentrations18,25,27 or with daily oral bicarbonate supplementation.15,36,39 No HD/HDF studies found oral or dialysate bicarbonate interventions to have an impact on Subjective Global Assessment (SGA) (n = 2).32,36
Three PD studies reported nutritional outcomes (serum albumin, lean body mass, protein nitrogen appearance [PNA], and SGA).41,43,48 A parallel-group RCT reported significant increases in normalized PNA and SGA following 12-months of oral bicarbonate supplementation (compared to placebo), but no differences in lean body mass or serum albumin between-groups after adjusting for the Charlson Comorbidity Index. 43 Another RCT found no within-group differences in serum albumin at 1 year or PCR at 1 month in patients treated with high alkali dialysate and oral bicarbonate supplementation vs low alkali dialysate. 48 A non-randomized interventional study showed no difference in PNA with an increase in dialysate bicarbonate concentration from 34 to 39 mmol/L (Supplemental Table 7). 41
Serum Bicarbonate, pH, Calcium, Potassium, and PTH
Results for laboratory measurement outcomes are summarized in Table 4 for HD/HDF studies and Supplemental Table 8 for PD studies. There were 22 of 27 HD/HDF studies that showed significantly higher pre-dialysis serum bicarbonate with higher dialysate bicarbonate concentration3,13,17 -19,27,28,31,32,44,45,51 or oral bicarbonate supplementation.15,33 -40,52 A significantly higher post-dialysis serum bicarbonate concentration was observed with higher dialysate bicarbonate concentration in 11 of 15 HD/HDF studies,13,14,17,19,21,24,28,31,40,44,45 There were 9 of 16 HD/HDF studies that found higher pre-dialysis pH with higher dialysate bicarbonate concentration or oral bicarbonate supplementation.19,22,25,27,31,34,35,37,39 Post-dialysis pH was found to be significantly higher with higher dialysate bicarbonate concentration in 5 of 9 HD/HDF studies.21 -24,46
Bicarbonate, pH, Calcium, Potassium, and PTH in HD/HDF Studies.
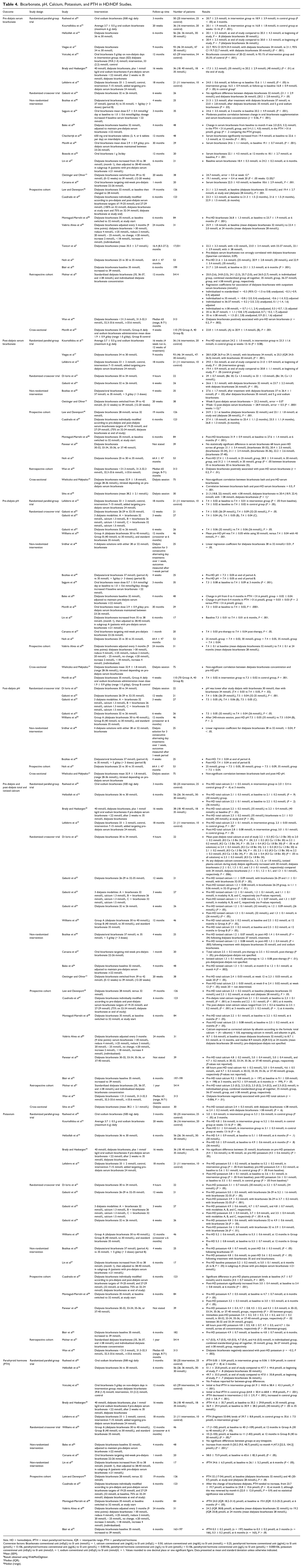
Note. HD = hemodialysis, iPTH = intact parathyroid hormone, IQR = interquartile range, PTH = parathyroid hormone.
Conversion factors: Bicarbonate conventional unit (mEq/L) to SI unit (mmol/L) = 1, calcium conventional unit (mg/dL) to SI unit (mEq/L) = 0.50, calcium conventional unit (mg/dL) to SI unit (mmol/L) = 0.25, parathyroid hormone conventional unit (pg/mL) to SI unit (pmol/L) = 0.106, parathyroid hormone conventional unit (pg/dL) to SI unit (pmol/L) = 0.00106, parathyroid hormone conventional unit (ng/L) to SI unit (pmol/L) = 0.106, parathyroid hormone conventional unit (pg/L) to SI unit (pmol/L) = 0.000106, potassium conventional unit (mEq/L) to SI unit (mmol)/L = 1, sodium conventional unit (mEq/L) to SI unit (mmol)/L = 1. Values rounded to one decimal place or one significant figure. Data presented as mean and standard deviation unless otherwise indicated.
Mean (SEM).
Result obtained using WebPlotDigitizer.
Median (IQR).
Median (range).
Meta-analysis was performed on three HD parallel-group RCTs16,18,19 with 113 total participants that reported pre-dialysis serum bicarbonate, total calcium, and potassium at end of study follow-up and compared dialysate bicarbonate concentrations greater than 35 mmol/L to those ≤35 mmol/L (i.e., 30 vs 36 mmol/L, 35 vs 40 mmol/L, 33 ± 2 mmol/L vs additional 7-15 mmol/L). We found an uncertain effect of higher vs lower dialysate bicarbonate concentration on pre-dialysis serum bicarbonate (MD 3.5 mmol/L, 95% CI −0.6 to 7.7), serum total calcium (MD −0.01 mmol/L, 95% CI −0.07 to 0.6), and serum potassium (MD −0.1 mmol/L, 95% CI −0.4 to 0.1) (Figures 3-5, Supplemental Table 9). The overall certainty of the evidence for all three outcomes is very low due to serious concerns with risk of bias (randomization concerns), inconsistency (substantial statistical heterogeneity for serum bicarbonate, I1 = 94%), and imprecision (wide CIs and few participants).
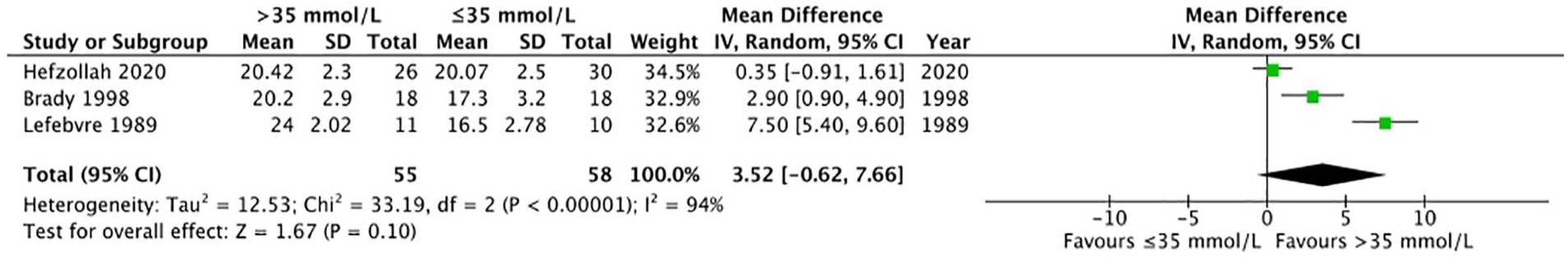
Forest plot of comparison: >35 mmol/L versus ≤35 mmol/L dialysate bicarbonate, outcome: pre-dialysis serum bicarbonate at end of study.

Forest plot of comparison: >35 mmol/L versus ≤35 mmol/L dialysate bicarbonate, outcome: pre-dialysis total calcium at end of study.a

Forest plot of comparison: >35 mmol/L versus ≤35 mmol/L dialysate bicarbonate, outcome: pre-dialysis potassium at end of study.
Meta-analysis was also performed on three crossover RCTs22 -24 with 74 total participants treated with HD that measured pre-dialysis and post-dialysis ionized calcium and potassium comparing dialysate bicarbonate concentrations ≥32 mmol/L and ≤29 mmol/L. There was an uncertain effect of higher vs lower dialysate bicarbonate concentration on pre-dialysis ionized calcium (MD 0.0 mmol/L, 95% CI −0.03 to 0.03), post-dialysis ionized calcium (MD −0.05 mmol/L, 95% CI −0.08 to −0.02), pre-dialysis serum potassium (MD −0.04 mmol/L, 95% CI −0.2 to 0.3), and post-dialysis serum potassium (MD −0.2 mmol/L, 95% CI −0.3 to −0.1) (Supplemental Figures 2-5, Supplemental Table 10). The overall certainty of the evidence is very low due to very serious concerns with risk of bias (selection bias and randomization concerns) and imprecision (few participants).
There were 14 HD/HDF studies that reported PTH as an outcome.16,18,19,25 -28,32,33,37,45,47,50,52 Of these, the RCTs which compared higher to lower dialysate bicarbonate concentrations (n = 4) and oral sodium bicarbonate supplementation to no supplementation (n = 2), did not find significant differences in PTH,16,18,19,25,52 except for one RCT that found a decrease in iPTH in the oral bicarbonate group and an increase in iPTH in the control group. 33 The largest PD RCT found higher serum bicarbonate and pH in the high dialysate alkali plus oral sodium bicarbonate group (27.2 ± 0.3 mmol/L, 7.4 ± 0.004) compared to the low dialysate alkali group (23.0 ± 0.3 mmol/L, 7.4 ± 0.004, both P < .001 between-groups) but no within-group differences in potassium, calcium, or PTH after one year (Supplemental Table 8). 48
Discussion
This systematic review included 41 studies, most of which included patients on maintenance HD or HDF. There were 16 RCTs, with the largest RCTs in HD and PD including 93 and 200 patients, respectively.17,48 Studies in HD/HDF examining mortality and hospitalizations were all non-randomized, with most evidence coming from one large, international cohort study demonstrating harm with higher dialysate bicarbonate concentrations. 3 Studies in PD were small and lacked power to meaningfully determine effects on mortality and hard cardiovascular outcomes, such as stroke. The largest PD RCT reported fewer hospitalizations per patient per year in the high alkali group compared to the low alkali group. 48
With respect to cardiovascular outcomes in HD/HDF RCTs, stroke and myocardial infarction were not reported, and none of the trials were powered to examine these effects. The parallel-group RCTs that we identified were few (n = 4) and small;15,17,33,52 most data were derived from small crossover RCTs and non-randomized interventional studies. Some studies suggest that higher dialysate bicarbonate concentrations may increase the risk of intradialytic hypotension and arrhythmia, but meaningful conclusions cannot be made due to small sample sizes and inconsistent results across studies.
Most studies reported no significant differences in nutritional outcomes, including outcomes of known prognostic value: serum albumin, nPCR, and SGA. These data suggest that dialysate bicarbonate concentrations within typically prescribed ranges, whether higher or lower, are sufficient to prevent protein-energy wasting related to metabolic acidosis. Evidence for the effect of higher vs lower dialysate bicarbonate concentrations on pre-HD serum bicarbonate, total calcium, and potassium was graded as very low certainty, as was the evidence for the effect on post-HD ionized calcium and potassium. As expected, most HD RCTs examining different dialysate bicarbonate concentrations reported a significantly higher post-dialysis serum bicarbonate with the use of higher dialysate bicarbonate concentrations,17,19,21,24 but the trial examining oral bicarbonate supplementation did not find a significant difference. 15
A previous systematic review and meta-analysis 53 on this topic included 3 RCTs18,19,43 and pooled both HD and PD studies.18,43 This approach may be problematic because we do not know whether the effect of dialysate bicarbonate or supplementation differs between PD and HD. This review is larger, including 41 studies and examined more diverse outcomes. The results of our meta-analysis of 3 HD RCTs18,19,25 yielded an uncertain effect of higher vs lower dialysate bicarbonate concentration on pre-dialysis serum bicarbonate (MD 3.5 mmol/L, 95% CI −0.6 to 7.7) and substantial heterogeneity (I1 = 94%). A previous systematic review and meta-analysis that pooled one PD and 2 HD RCTs with dialysate bicarbonate or oral bicarbonate intervention groups found a greater magnitude of effect (MD 4.6, 95% CI 1.1 to 8.2) but also substantial heterogeneity (I1 = 87.5%). 53
Some of the heterogeneity between included studies could be explained by changes in characteristics of patients receiving maintenance dialysis through secular effects. For example, the mean age of participants in studies published in 1989 and 1998 was about 50 years,18,19 while more recent studies had a higher mean age of approximately 60 years. 16 In addition, a study included in our meta-analysis published in 1989 excluded patients with diabetes, 19 while contemporary study populations included up to 70% of participants with diabetes. 16 The evidence is also limited by small sample sizes of published RCTs; the largest HD and PD trial sample sizes were 93 and 200 patients, respectively and had a high risk of bias 17 or some concerns. Most HD/HDF studies had a moderate or high risk of bias, while 2 of 4 PD studies had a low risk of bias. Sufficient data were not available to meta-analyze patient-important outcomes, such as myocardial infarction, stroke, mortality and hospitalizations. HD studies examining nutritional outcomes and surrogate cardiovascular outcomes were too heterogeneous in terms of methods and outcome definitions for meta-analysis.
A strength of this systematic review is the use of broad inclusion criteria accepting any type of adult population on maintenance HD/HDF or PD that reported any comparisons of dialysate bicarbonate concentration or oral bicarbonate supplementation with no supplementation with no limitations on study design. This review also focused on a wide range of biochemical and clinically relevant outcomes that would be plausibly affected by alterations in bicarbonate status and commonly used by nephrologists to guide management decisions. Limitations of the review include the exclusion of non-English literature and gray literature, which might introduce publication bias. However, the magnitude of this bias may not be large, and the quantity and quality of the data found were such that it is unlikely that we missed work that would change our conclusions. 54 Another limitation of this review is the lack of a registered protocol, which may affect transparency. However, there were no deviations from the initial review plan. Additionally, this review relied primarily on small or observational studies, as there was a lack of large RCTs.
Conclusions
In conclusion, current guidelines for treatment of metabolic acidosis in kidney failure recommend a serum bicarbonate ≥22 mmol/L. 2 This recommendation has resulted in the common use of high (≥35 mmol/L) dialysate bicarbonate concentrations or, less commonly, the use of oral bicarbonate supplementation, with the assumption that these approaches will normalize serum bicarbonate (measured pre-dialysis in HD patients), and therefore improve patient outcomes. 3 The results of this review support that this recommendation and the interventions used to treat the metabolic acidosis of kidney failure are based on limited, weak evidence. This review highlights the need for large, high-quality RCTs that examine the impact of higher vs lower dialysate bicarbonate concentrations or oral bicarbonate supplementation on clinically relevant outcomes in patients receiving maintenance dialysis.
Supplemental Material
sj-docx-1-cjk-10.1177_20543581251356182 – Supplemental material for The Effect of Dialysate Bicarbonate Concentration or Oral Bicarbonate Supplementation on Outcomes in Patients on Maintenance Dialysis: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-cjk-10.1177_20543581251356182 for The Effect of Dialysate Bicarbonate Concentration or Oral Bicarbonate Supplementation on Outcomes in Patients on Maintenance Dialysis: A Systematic Review and Meta-Analysis by Ashlee M. Azizudin, Samuel A. Silver, Amit X. Garg, Zoe K. Friedman, Andrea C. Cowan, Catherine M. Clase and Amber O. Molnar in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-docx-2-cjk-10.1177_20543581251356182 – Supplemental material for The Effect of Dialysate Bicarbonate Concentration or Oral Bicarbonate Supplementation on Outcomes in Patients on Maintenance Dialysis: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-2-cjk-10.1177_20543581251356182 for The Effect of Dialysate Bicarbonate Concentration or Oral Bicarbonate Supplementation on Outcomes in Patients on Maintenance Dialysis: A Systematic Review and Meta-Analysis by Ashlee M. Azizudin, Samuel A. Silver, Amit X. Garg, Zoe K. Friedman, Andrea C. Cowan, Catherine M. Clase and Amber O. Molnar in Canadian Journal of Kidney Health and Disease
Footnotes
List of Abbreviations
DBP, diastolic blood pressure; HD, hemodialysis; HDF, hemodiafiltration; HR, hazard ratio; iPTH, Intact parathyroid hormone; MD, mean difference; nPCR, normalized protein catabolic rate; PD, peritoneal dialysis; PNA, protein nitrogen appearance; PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses; PTH, parathyroid hormone; RCT, randomized controlled trial; SBP, systolic blood pressure; SGA, subjective global assessment.
Ethical Approval and Consent to Participate
Not applicable.
Authors’ Contributions
Study conception and design: AMA, AOM. Data analysis and interpretation: AMA, AOM. Data acquisition and study implementation: AMA, AOM, ZKF. Manuscript drafting: AMA, AOM. Manuscript revision: AMA, SAS, AXG, ACC, CMC, AOM.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Catherine Clase has received consultation, advisory board membership, or research funding from the Ontario Ministry of Health, Sanofi, Pfizer, Leo Pharma, Astellas, Janssen, Amgen, Boehringer-Ingelheim, and Baxter. In 2018, she co-chaired a KDIGO potassium controversies conference sponsored at arm’s length by Fresenius Medical Care, AstraZeneca, Vifor Fresenius Medical Care, Relypsa, Bayer HealthCare, and Boehringer-Ingelheim. The other authors declare that they have no competing interests.
Data Availability Statement
All relevant data are included in this manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
