Abstract
Purpose:
Provision of high-quality, evidence-based patient care that is sustainable for our universal health system is a core Canadian Medical Education Directions for Specialists (CanMEDs) expectation. The Transplant Manitoba Adult Kidney Program (TMAKP) embraced this responsibility by addressing inefficiencies in its practices through multipronged quality improvement (QI) strategies, including reducing unnecessary interventions, implementing innovative strategies, and aligning clinical practices with emerging evidence. Using seamlessly embedded continuous QI and clinical research with a learning health system, the program achieved substantial cost savings and increased opportunities for deceased donor kidney transplantation. The purpose of this analysis is to measure the cost savings associated with these QI initiatives.
Sources of information:
Transplant Manitoba Adult Kidney Program database and quality metrics, Manitoba Health Physician’s Manual (April 1, 2024), PubMed.
Methods:
To quantify the potential cost savings, we employed a 3-pronged approach. For reduced testing, a cost-counting exercise was conducted using historical transplant activity (831 prevalent and 83 incident patients) to project number of tests avoided and direct costs per test. Second, cost savings for generic mycophenolic acid was presented as ratios of generic to brand name drug costs, and projected cost savings for prevalent patients receiving average dosing. Third, for increased kidney utilization, cost savings per kidney transplant were derived from published studies and extrapolated using predicted additional transplants. Net health care system savings across payers were assessed at a 1-year time horizon.
Key Findings:
The TMAKP reduced unnecessary testing, adopted generic medications, and implemented innovative strategies, achieving $2,530,026 in projected annual 1-year cost savings. These QI initiative savings augment the overall cost-effectiveness of kidney transplantation compared with dialysis. Implementing evidence-based protocols using personalized risk-stratified approaches to viral monitoring and novel donor-specific antibody surveillance strategies aligned testing with clinical risk while minimizing patient burden, highlighting the benefits of seamlessly integrating research with learning health systems. Programs for hepatitis C-viremic donor kidneys and age-targeted allocation increased transplant opportunities and optimized deceased donor organ use. Manitoba’s initiatives demonstrate the importance of validation, stakeholder engagement, and iterative adaptation in driving sustainable improvements in transplantation care. Critically, this requires the foresight of health care administrative systems to invest in effective and ongoing QI and embed research with clinical practice, to improve patient and health system outcomes.
Limitations:
This analysis is limited by reliance on projected cost savings, which require validation through real-world audits to confirm impact. In addition, some valuable QI efforts, while improving patient outcomes, may increase costs, highlighting the need for balanced perspectives in assessing stewardship initiatives. Finally, this analysis is limited to projected cost savings and does not evaluate clinical outcomes, process adherence, or implementation effectiveness.
Implications:
This experience highlights the potential for QI initiatives to optimize care and resource utilization within Canada’s publicly funded health system. These efforts reduced unnecessary testing, minimized patient burden, and expanded transplant opportunities, illustrating how stewardship can balance fiscal responsibility with high-quality state-of-the-art patient care. By implementing evidence-based protocols, TMAKP achieved an additional $2,530,026 in projected cost savings at 1 year. Future annual cost savings will continue rising in a growing prevalent kidney transplant population in Manitoba. These savings can be reallocated to other critical health care services, expanding access and improving outcomes for patients beyond transplantation.
Introduction
A significant health care challenge is implementing proven clinical interventions cost-effectively. Scarce resources, including time and staff, often hinder adoption, despite evidence that health-savings from evidence-based practices can outweigh delivery costs. In kidney transplantation, these barriers are compounded by lack of high-quality evidence and standardized guidelines, resulting in variation across programs and missed opportunities for cost savings.1-7 Stewardship, a core Canadian physician competency under the CanMEDS framework, emphasizes the need for efficient resource-utilization while maintaining high-quality care. 8 This is particularly relevant in Canada’s publicly funded health care system, where cost savings in one area translates to improved access and resources in others. 9
The Transplant Manitoba Adult Kidney Program (TMAKP) embraced this responsibility by addressing inefficiencies in its practices through multipronged quality improvement (QI) strategies. By reducing unnecessary interventions, implementing innovative strategies, and aligning practices with emerging evidence, the program achieved substantial cost savings and increased opportunities for transplantation. By utilizing kidneys that would otherwise be discarded, these initiatives reduce wait-times in a province with prolonged dialysis dependency while better honoring the gift of life from donors. 10 In addition, reducing unnecessary testing decreases the already high medical burden of transplant recipients, improving overall care.
This case study demonstrates how thoughtful investment in QI and seamlessly integrated research with learning health systems improves both patient care and resource-utilization, offering valuable lessons for Canadian transplant programs and health care administrators weighing the “costs” of funding QI.
Why Should We Change?
Kidney transplantation is highly cost-effective compared to dialysis. 11 Importantly, prior landmark economic analyses may underestimate the modern cost savings of transplant due to now routine use of low-cost generic immunosuppressants post-transplant and the high costs of delivering dialysis in rural and remote regions of Manitoba. 12
As a relatively young field, transplantation requires iterative fine-tuning to address inefficiencies and replace practices rooted in tradition versus evidence. Choosing Wisely Canada works to reduce unnecessary care, conserve resources, and minimize harm; however, transplantation currently lacks specific recommendations, underscoring the need for local programs to lead by example. 13
Examination of local TMAKP protocols revealed deviations from guidelines; unsurprising, given gaps and inconsistencies among guidelines themselves.14-21 For example, routine post-transplant annual chest x-rays (CXR) and electrocardiograms (ECG) persisted despite omission from KDIGO 2009 guidelines. 21 Post-transplant monitoring for BK virus (BKV), Epstein-Barr virus (EBV), and cytomegalovirus (CMV) diverged from KDIGO, AST-IDCOP, and COMMIT recommendations.14-16,21 Donor-specific antibody (DSA) monitoring, supported by The European Society of Organ Transplantation (ESOT) and The Transplantation Society (TTS), lacked clear guidance on testing frequency. 22 Emerging research demonstrated safety of hepatitis C virus (HCV)-viremic donors, such that, KDIGO 2022 guidelines recommend their use in both HCV-positive and HCV-negative recipients with access to direct-acting antivirals 23 (Table 1).
Summary of Kidney Transplantation Guideline Recommendations.
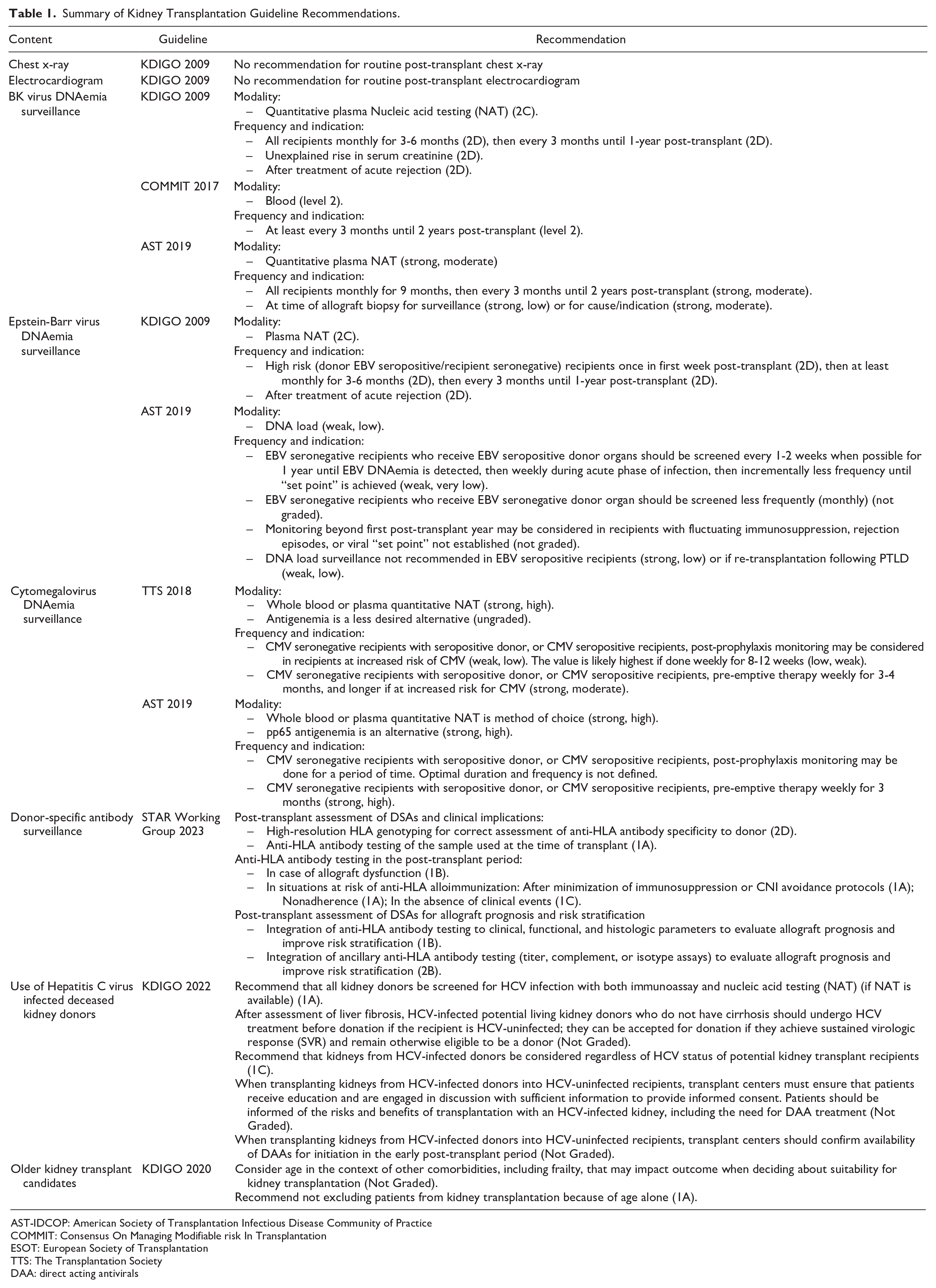
AST-IDCOP: American Society of Transplantation Infectious Disease Community of Practice
COMMIT: Consensus On Managing Modifiable risk In Transplantation
ESOT: European Society of Transplantation
TTS: The Transplantation Society
DAA: direct acting antivirals
Transplant Manitoba Adult Kidney Program addressed these inefficiencies by aligning protocols with guidelines, applying personalized risk stratification, and implementing innovative approaches to reduce kidney discard and expand donor organ use. These efforts included eliminating routine annual CXR and ECG, tailoring viral load surveillance to patient risk, 24 developing a novel risk score for DSA monitoring, 25 switching to generic mycophenolic acid (MPA) immunosuppression, introducing kidneys from HCV-viremic donors, 26 and creating an age-targeted program matching kidneys with shorter expected graft survival to older recipients. 27 By reducing unnecessary testing, expanding transplant opportunities, and optimizing resource-utilization, TMAKP demonstrated how stewardship can transform kidney transplantation practices.
What is the Scope of the Problem?
To quantify the scope of inefficiencies and potential cost savings, this high-level analysis employed a 3-pronged approach. First, for reduced testing, a cost-counting exercise was conducted using historical transplant activity (831 prevalent and 83 incident patients) to project number of tests avoided and direct costs per test. Second, cost savings for generic MPA was presented as ratios of generic to brand name drug costs, and projected cost savings for prevalent patients receiving average dosing. Third, for increased kidney utilization, cost savings per kidney were derived from published studies and extrapolated using predicted additional transplants.28,29 Net health care system savings across payers were assessed at a 1-year time horizon, with cost inputs summarized in Table 2.
Cost Input Parameters Used in Analysis.

This analysis focuses exclusively on estimating the cost savings of QI initiatives already implemented in clinical practice, and previously reported (Table 3). These initiatives were developed and introduced based on internal audits, available evidence, and clinical consensus. Although they were designed with quality and safety in mind, the present analysis is limited to quantifying economic impact, rather than assessing patient outcomes or conducting a formal QI process evaluation.
Transplant Manitoba Quality Improvement Initiatives.
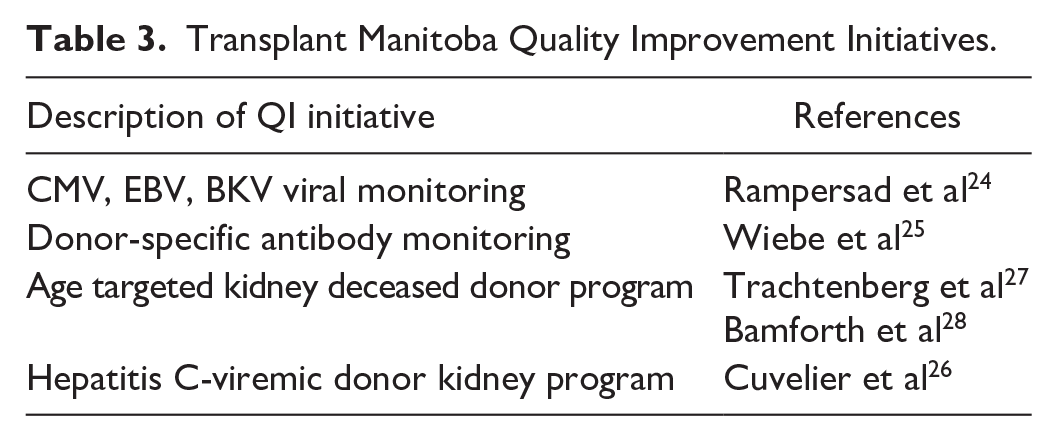
Chest X-Ray and Electrocardiogram
Routine annual CXRs and ECGs in prevalent patients were long-standing practices without evidence or guideline support. Eliminating these produced immediate annual cost savings of $20,983 for CXRs and $20,933 for ECGs.
Viral Load Surveillance
Risk-stratified viral monitoring reduced unnecessary tests for incident patients. For CMV, high-risk recipients were projected to undergo 18 to 30 fewer tests annually, while moderate-risk groups would avoid 9 to 18 tests, yielding cost savings of $17,014 to $28,358. Epstein-Barr virus monitoring, targeted to high-risk mismatched recipients, projected 1 to 3 fewer tests annually, saving $432 to $1296. BK virus testing was projected to decrease by 9 to 21 tests annually, saving $74,999 to $174,997. 24 Combined, these estimates totaled $92,446 to $204,651 annually. Real-world audits revealed poor adherence to overly frequent prior protocols, suggesting revised protocols may be more practical and tolerable for patients while also cost-saving.
DSA Surveillance
Donor-specific antibody monitoring was refined using a novel risk-based approach incorporating age and HLA molecular mismatch. 25 Testing would be reduced by 50% over 10 years (from 453 to 1200 tests per 100 patients), with projected annual savings of $248,303 for prevalent patients while maintaining detection of clinically significant cases.
Generic MPA
Mycophenolic acid is a maintenance immunosuppressive agent used in all transplant recipients. In 2012, TMAKP adopted generic versions of MPA for all prevalent patients, representing 17% and 50% of previous costs for CellCept and Myfortic, respectively. 30 Projected annual savings are $2,021,775 (93% patients, average dose 500 mg twice daily).
HCV-Viremic Donor Program
The HCV-viremic donor program enables use of previously discarded HCV-viremic kidneys. 26 This approach is highly cost-effective, with incremental cost-effectiveness ratio of $56,018 per quality-adjusted life year (QALY) from the payer perspective and $4647 per QALY from the societal perspective. Over 5 years, the program reduces costs by $191,000 (USD) per patient.29,31 Projecting 2 additional transplants annually, this program is expected to save $76,400 (USD) or $109,720 (CAD) per year.
Age-Targeted Program
The age-targeted program optimizes use of previously discarded high Kidney Donor Profile Index (KDPI 86-100) kidneys, by allocating them to older recipients (≥65 years) who pre-consented for this strategy to improve quality of life and survival. 27 Cost-utility analysis demonstrated this approach reduces costs by $23,452 per transplant over 10 years, compared to standard allocation. 28 Projecting 10 transplants annually, the program achieves $23,452 in annual cost savings.
How Does Manitoba’s Experience Reflect the Global Landscape of Transplant Stewardship?
Examining global practices highlights common challenges and opportunities for improvement. Manitoba’s initiatives to optimize transplant stewardship align with broader international efforts to improve resource-utilization while maintaining high-quality care.
Viral Monitoring: International Variability
Adoption of streamlined risk-based viral monitoring aligns with international guideline recommendations but contrasts with variability observed globally. A survey of European transplant centers showed that 38% of programs performed routine EBV testing on all recipients. 1 In South Korea, duration of CMV and BKV monitoring durations ranged below and beyond guideline recommendations. 2 A US survey found that only 40% of centers routinely screened for BKV despite recommended universal testing, while intra-center protocol discrepancies were common. 3 These studies highlight real-world divergence from guidelines. Our experience demonstrates how aligning protocols with risk-based recommendations can reduce unnecessary testing and promote cost savings without compromising care. 24
DSA Monitoring: Emerging Approaches
While ESOT and TTS guidelines support DSA monitoring, they lack specificity on frequency, resulting in varied practices. 22 Manitoba’s novel DSA surveillance protocol focuses testing in high-risk patients, reflecting broader trends toward precision medicine in transplantation, and highlighting benefits of integrating research with clinical systems. 25
Expanding Donor Utilization: Lessons From HCV and Age-Targeted Programs
Innovations like HCV-viremic donor and age-targeted programs reflect successful strategies seen in other jurisdictions. The US has embraced HCV-viremic donor transplantation as a cost-effective solution to expand the donor pool, with 46% of recipient candidates willing to accept these kidneys, and with 6% of deceased donors being HCV-viremic. 32 Similarly, high-KDPI kidneys are utilized in many jurisdictions with satisfactory outcomes.32,33 Manitoba’s application of these strategies underscores the adaptability of international innovations to local challenges, addressing long dialysis wait-times for transplant candidates.
A Broader Perspective
Manitoba’s efforts to address inefficiencies resonate with global challenges in transplantation. By aligning with international best practices and tailoring to local contexts, Manitoba provides insights into how programs can balance resource optimization while achieving improved outcomes, such as increased rates of transplantation. These experiences underscore the need for continued evaluation and adaptation in transplantation stewardship worldwide.
What Are the Next Steps?
This experience demonstrates how QI initiatives can optimize resource-utilization, enhance patient outcomes, and expand access to kidney transplantation. A critical component of advancing QI initiatives is validation of changes through robust quality assurance studies. To sustain these successes, 4 key actions are required: funding QI efforts, research expanding evidence-based protocols, fostering collaboration, and embedding iterative adaptation.
Invest in Foundational Support for QI
Complex, multiyear QI initiatives demand dedicated time and resources. Protected physician time and administrative investment are critical to driving innovation and achieving better patient outcomes. Although QI initiatives generate long-term savings through improved care and reduced inefficiencies, upfront and ongoing investment is essential.
Expand Evidence-Based Protocols
Manitoba’s viral monitoring and DSA surveillance protocols highlight the value of aligning testing with clinical risk. For viral monitoring, follow-up studies demonstrated safety of streamlined protocols, showing no increase in viremia or tissue-invasive disease. Similarly, the novel DSA surveillance protocol developed by our group demonstrated stronger associations with alloimmune outcomes than conventional whole-antigen HLA matching. While these protocols minimized patient burden and optimized resource use, ongoing real-world evaluations are critical to refine approaches and address practice-variations.
Foster National Collaboration
Heterogeneous transplant protocols underscore the need for collaboration to standardize and disseminate best practices across Canada. Establishing national platforms for sharing QI initiatives, clinical outcomes, and cost savings would promote consistency while accelerating innovation. Partnerships between professional societies like the Canadian Society of Transplantation and Choosing Wisely Canada could formalize evidence-based protocols, enhancing efficiency and reducing unnecessary testing. Academic publications documenting QI planning and implementation are essential for transparency and rigor. Manitoba’s approach, adapted to address local challenges like long pre-transplant wait-times, highlights how global innovations can be tailored to regional contexts.
Embed Iterative Adaptation and Stakeholder Engagement
Iterative adaptation ensures QI initiatives remain effective, responsive, and aligned with emerging evidence and patient needs. Manitoba’s age-targeted program, which allocates high-KDPI kidneys to older recipients, exemplifies this approach. Developed through multidisciplinary collaboration and patient engagement, the program addresses long dialysis wait-times while maximizing donor organ use. Embedding audits and long-term monitoring will refine protocols and validate outcomes. Beyond cost-analyses, incorporating quality-of-life metrics ensures that stewardship efforts focus on patient well-being. Iterative adaptation fosters dynamic feedback loops between stakeholders, clinicians, and researchers, supporting patient-centered care.
Conclusion
This experience highlights the potential for QI initiatives to optimize care and resource-utilization within Canada’s publicly funded health system. By implementing evidence-based protocols, TMAKP achieved $2,530,026 in projected cost savings at 1 year. Future annual cost savings will continue rising in a growing prevalent kidney transplant population in Manitoba. These savings can be reallocated to other critical health care services, expanding access and improving outcomes for patients beyond transplantation.
A critical lesson is the necessity of validating changes through rigorous quality assurance studies, ensuring clinical outcomes are preserved or improved while optimizing resources. These efforts reduced unnecessary testing, minimized patient burden, and expanded transplant opportunities, illustrating how stewardship can balance fiscal responsibility with high-quality state-of-the-art patient care. They may also yield indirect cost savings by reducing environmental impact and patient burden—lowering lab waste, energy use, and dialysis-related resource demands, while streamlining care to improve patient experience and system efficiency. However, this analysis is limited to projected cost savings and requires validation through real-world audits. Future studies should also evaluate clinical outcomes, process adherence, and implementation effectiveness, as some QI efforts may improve care while increasing real-world costs, highlighting the need for balanced perspectives in assessing stewardship initiatives.
Further opportunities include standardizing evidence-based testing protocols across Canada, fostering collaboration through national platforms to share QI practices, and incorporating quality-of-life metrics into ongoing evaluations. Manitoba’s initiatives demonstrate that embedding iterative adaptation and stakeholder engagement into the planning and implementation process is key to sustaining improvements over time.
The TMAKP’s success underscores the broader potential of stewardship in transplantation: not only improving care for transplant recipients but also supporting a sustainable health system that benefits all patients. By embracing innovation, accountability, and collaboration, Canada’s transplantation community can ensure every health care dollar and donor organ is used to its fullest potential, setting a benchmark for programs worldwide.
Key Points
Cost Savings Achieved: The TMAKP reduced unnecessary testing, adopted generic medications, and implemented innovative strategies, achieving over $2,500,000 in projected annual 1-year cost savings.
Evidence-Based Protocols: Risk-stratified approaches to viral monitoring and novel DSA surveillance strategies aligned testing with clinical risk while minimizing patient burden, highlighting the benefits of seamlessly integrating research with learning health systems.
Expanding Transplant Opportunities: Programs for HCV-viremic donor kidneys and age-targeted allocation increased transplant opportunities and optimized deceased donor organ use.
Sustainable Stewardship: Manitoba’s initiatives demonstrate the importance of validation, stakeholder engagement, and iterative adaptation in driving sustainable improvements in transplantation care. Critically, this requires the foresight of health care administrative systems to invest in effective and ongoing QI.
Footnotes
Acknowledgements
The authors would like to gratefully acknowledge the Transplant Manitoba multidisciplinary pre- and post-kidney transplant team members for all their hard work in implementing these clinical quality improvement initiatives. Transplantation is a team sport and we could not accomplish any of this without everyone pulling together in the same direction. We would also like to acknowledge the Doctors Manitoba Group CME program for supporting a learning and growth environment in our transplant program. Our multiorgan transplant rounds series has invited expert speakers, funded by Doctors Manitoba, which directly helped guide the development of our quality improvement initiatives.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: CR is supported by a Kidney Research Scientist Core Education and National Training Program (KRESCENT) salary award co-funded by the Kidney Foundation of Canada, the Canadian Society of Nephrology, and the Canadian Institutes of Health Research (CIHR). PN is supported by the Flynn Family Chair in Renal Transplantation. JH’s research program is supported by the Canadian Institutes of Health Research (CIHR). The funders had no role in defining the content of this article.
Ethics Approval and Consent to Participate
Formal research ethics board review was not required by the University of Manitoba based on the Tri-Council Policy Statement.
Consent for Publication
All authors approved the final version of this manuscript
Availability of Data and Materials
The R code for this analysis is available upon reasonable request.
Open Access Statement
The R code for this analysis is available upon reasonable request.
