Abstract
Rationale:
Infection-related glomerulonephritis (IRGN) is an immune-mediated glomerulonephritis caused by extra-renal infectious diseases. There has been an important shift in epidemiology in recent years, with a significant proportion of adults affected. The incidence of IRGN is higher amongst Indigenous populations and especially in those with multiple comorbidities. Beginning in 2019, we observed several IRGN cases amongst adult Indigenous peoples referred to the McGill University Health Center (MUHC). The aim of this article is to describe the demographic, clinical, and outcome data of these individuals and highlight the heterogeneity of IRGN in this population through 2 illustrative cases.
Presenting concerns of the patient:
In total, 8 cases of IRGN were identified between 2019 and 2022. All patients presented with features of acute glomerulonephritis.
Diagnoses:
All patients had documented evidence of an infection that preceded their diagnosis of IRGN. IRGN was not the initial clinical diagnosis in all cases.
Interventions:
Half the patients received immunosuppression while the others received supportive care only.
Outcomes:
Four patients required initiation of hemodialysis at time of presentation and at 2 years of follow-up, 3 of the 4 remained hemodialysis-dependent.
Teaching points:
Our case series emphasizes the heterogenous clinical, laboratory, and pathological presentations that make the diagnosis of IRGN quite challenging. A high index of suspicion should be present when a patient presents with acute kidney injury, features of a glomerulonephritis, and an infection, especially those with multiple comorbidities and a preceding history of chronic kidney disease.
Introduction
Infection-related glomerulonephritis (IRGN) is an immune-mediated glomerulonephritis caused by extra-renal infectious diseases. While IRGN (specifically poststreptococcal GN) is traditionally thought to be a childhood disease, the last few decades have seen an important shift in epidemiology with a significant proportion of cases occurring in adults. 1 The sites of index infection, spectrum of causative pathogens, severity of illness, and duration of infection differ significantly in this group. 2 Adult patients with multiple comorbidities are a particularly susceptible group; one study reported that nearly 60% of patients with IRGN had diabetes mellitus, 14% malignancy, and 4% had liver disease. 3 In addition, in contrast to children in whom IRGN can manifest without long-term consequences, studies in adults note that between 30% and 50% experience residual hypertension, persistently abnormal urinalysis or elevated creatinine, and glomerulosclerosis on kidney biopsy.4,5
Incidence of IRGN is notably high amongst Indigenous populations. 3 A longitudinal case-series among the First Nations of Sioux Lookout in Northwestern Ontario found over a 6-year period an incidence of 4.0 per 100 000 person-years as compared with 0.3 per 100 000 person-years in similar economies to Canada. 6
Beginning in 2019, we observed several IRGN cases amongst adult Indigenous peoples referred to the McGill University Health Center (MUHC). In this article, we describe the demographic, clinical, and outcome data of these individuals and highlight the heterogeneity of IRGN in this population through 2 illustrative cases. Consent for this study was obtained from the Cree Board of Health and Social Services of James Bay and the MUHC Research Ethics Board.
Presenting Concerns
We report 8 cases of IRGN in adult Indigenous patients referred to our Nephrology service between 2019 and 2022 (Table 1). Age at presentation ranged from 19 to 59 years, and 4 patients (50%) already had established chronic kidney disease. All patients had documented clinical or microbiological evidence of an infection that preceded their diagnosis of IRGN including pharyngitis, tonsillitis, urinary tract infection, skin and soft tissue infection, and COVID-19 (Table 2). All presented with hypertension, edema, proteinuria, and acute kidney injury. Kidney biopsies were performed in 7 of 8 cases and showed diffuse endocapillary proliferation with numerous neutrophils, a pattern referred to as exudative glomerulonephritis, which is consistent with IRGN (Table 3).
Baseline Characteristics.

DM2: Diabetes Mellitus Type 2, defined as an HbA1c ≥ 6.5%; GDM: Gestational Diabetes Mellitus; CVD: Cardiovascular disease, defined as previous history of myocardial infarction or cardiac catheterization confirming coronary artery disease; HTN: Defined as blood pressure above 140/90 mm Hg before clinical presentation; UACr: Urine albumin to creatinine ratio; Y: Yes N: No N/A: Not available.
Clinical Presentation and Laboratory Findings.
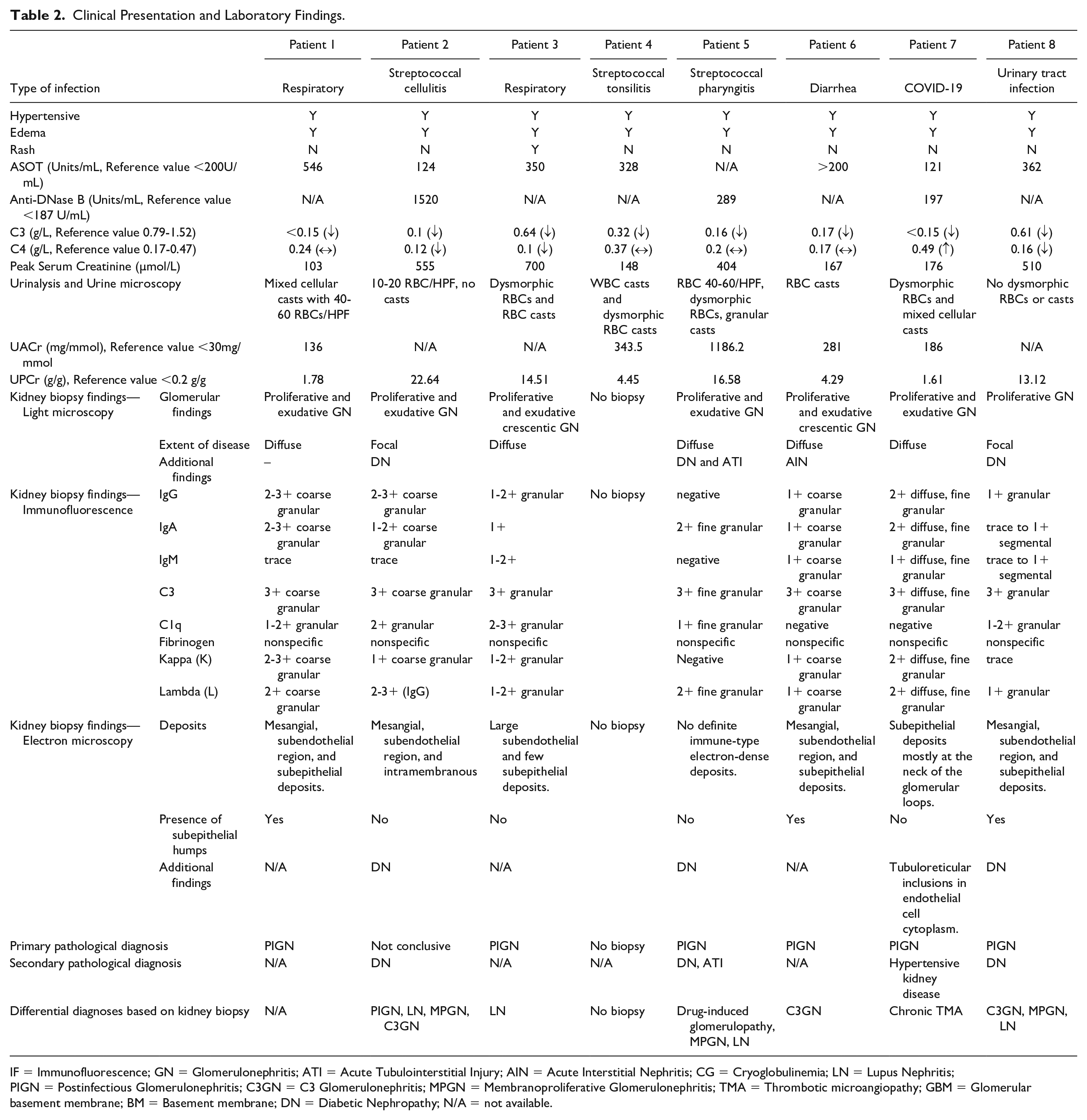
IF = Immunofluorescence; GN = Glomerulonephritis; ATI = Acute Tubulointerstitial Injury; AIN = Acute Interstitial Nephritis; CG = Cryoglobulinemia; LN = Lupus Nephritis; PIGN = Postinfectious Glomerulonephritis; C3GN = C3 Glomerulonephritis; MPGN = Membranoproliferative Glomerulonephritis; TMA = Thrombotic microangiopathy; GBM = Glomerular basement membrane; BM = Basement membrane; DN = Diabetic Nephropathy; N/A = not available.
Clinical Course and Outcomes.

GC = Glucocorticoids; MMF = Mycophenolate mofetil; HCQ = Hydroxychloroquine; HD = Hemodialysis; N/A = not available.
Clinical Findings, Timeline, Diagnostic Focus, and Assessment
Patient 1—Typical Presentation of IRGN
A 19-year-old previously healthy woman with normal baseline kidney function (Cr 50µmol/L) presented with a 1-week history of rhinorrhea, sore throat, and cough. She subsequently developed pleuritic chest pain and shortness of breath which led her to present to the emergency room. She was found to be hypertensive with a blood pressure of 159/100 mm Hg and had evidence of volume overload manifest as peripheral edema. She had acute kidney injury (Cr 103 µmol/L) with urine sediment showing dysmorphic red blood cells, red blood cell casts, and proteinuria (urine protein to creatinine ratio 10.5 g/g).
Her laboratory evaluation revealed a decreased C3 level, normal C4 level, and a positive anti-streptolysin titer (ASOT). The rest of her auto-immune panel including ANA, anti-double-stranded DNA, and ANCA serologies were all negative. In addition, viral serologies of Hepatitis B, C and HIV were negative. A diagnosis of streptococcal pharyngitis was made, and she underwent kidney biopsy to confirm etiology of the acute kidney injury. The biopsy demonstrated diffuse proliferative and exudative glomerulonephritis with immunofluorescence positive for IgG (2-3+), IgA (3+), C3 (3+), and C1q (1-2+) that was most consistent with a poststreptococcal glomerulonephritis. Electron microscopy demonstrated hypercellularity and immune complex electron-dense deposits in mesangium, subendothelial region, and characteristic large subepithelial deposits (“humps”).
She received supportive therapy and did not require any immunosuppression. At discharge from hospital 7 days later, her infection had resolved, and creatinine levels returned to her previous normal baseline. Her C3 remained low.
On follow-up 2 years later, her Cr was 50 µmol/L and urine albumin to creatinine ratio normal. She was no longer on any antihypertensives, nor diuretics. Her repeat complement levels normalized on yearly follow-up visits.
Patient 2—Atypical Presentation of IRGN
A 45-year-old female with previously known stage 3b A3 chronic kidney disease thought to be secondary to diabetic nephropathy (baseline creatinine 163µmol/L, eGFR of 33 mL/min and urine ACr of 330mg/mmol) presented to the emergency department with a 1-week history of fever and a painful left leg that was erythematous and swollen. A clinical diagnosis of cellulitis was made, and she was started on antibiotics. During her hospitalization, she developed shortness of breath and chest X-ray revealed an atypical pneumonia, thus her antibiotic coverage was broadened. She subsequently developed worsening kidney function over the following days with hypertension and peripheral edema.
Laboratory evaluation revealed an acute kidney injury with peak serum creatinine 550 µmol/L and urine microscopy demonstrated 10 to 20 red blood cells per high power field but no cellular casts. C3 and C4 levels were low as well as positive ASOT. She was also noted to have a positive ANA titer of 1 in 160, anti-DNA antibody 288IU/mL (normal 0-99 IU/mL), negative anti-glomerular basement membrane antibody, normal serum protein electrophoresis, and a normal free light chain ratio of 1.46. A presumed diagnosis of lupus nephritis was made on a background of diabetic nephropathy despite the absence of extra-renal manifestations of systemic lupus erythematosus (SLE).
A kidney biopsy confirmed the presence of mesangial nodular expansion and thickening of the glomerular basement membrane that was consistent with diabetic glomerulopathy. In addition, the biopsy showed that 30% of the glomeruli had active proliferation with neutrophil rich exudation and immunofluorescence was strongly suggestive of immune complex deposition with predominant C3 in a coarse granular pattern, although still a full-house pattern. Electron microscopy demonstrated mesangial, subendothelial, and intramembranous deposits. At this time, there was uncertainty between a diagnosis of lupus nephritis, membranoproliferative GN, C3 GN, and IRGN as the main differential diagnoses given the C3 dominant staining. After discussion between nephrology and rheumatology services, it was decided to treat the patient for lupus nephritis with a high dose of glucocorticoids and subsequently start on a steroid sparing regimen of mycophenolate mofetil. During her initial admission, she was also started on hemodialysis for persistent hyperkalemia in the context of acute kidney injury. At 2 years of follow-up, she continued to have no response to immunosuppressants, and she remained on chronic hemodialysis.
Since her initial presentation, multiple repeat lupus serologies including ANA, anti-DNA antibody, and complement levels have all been normal. In combination with the complete lack of other lupus symptoms, the diagnosis of SLE was put into question, and in retrospect, a diagnosis of IRGN rather than lupus nephritis was made. Over time, the patient had episodes of profuse diarrhea secondary to mycophenolate. Thus, considering the lack of response to therapy and low likelihood of SLE, mycophenolate was discontinued after 2 years of therapy.
Therapeutic Focus, Assessment, Follow-up, and Outcomes
Management of IRGN beyond treatment of the underlying infection is unproven. 7 The value of high-dose steroids is uncertain, and there are no trials to help guide therapy. In our cohort, 4 of the patients received immunosuppression which initially consisted of high-dose glucocorticoids (Table 3). Two of these patients required dialysis and remain dialysis-dependent at 2 years of follow-up. One patient (patient 6) was given a course of steroids and demonstrated kidney recovery with a residual GFR of 60 mL/min, and the outcome of 1 patient (patient 7) who received glucocorticoids is unknown. The remainder of the patients received only supportive care with 1 patient (patient 1) returning to her baseline kidney function and the 3 others being left with residual chronic kidney disease ranging from stage 3a to 3b CKD.
Discussion
We have highlighted the wide spectrum of clinical, laboratory, and pathological presentations of IRGN in an adult Indigenous population referred to our center. There appears to be no unifying reason why a high number of cases, approximately 4% of biopsies at our center from 2019 to 2022, was observed. In comparison, a large registry of 14 138 kidney biopsies performed at a New York referral center involving mostly Caucasian adults showed an incidence of IRGN of only 0.6% between 1995 and 2005. 8 Observational data have shown that Indigenous communities have a higher incidence of IRGN. 6 Possible hypotheses for this are the higher rate of comorbidities, specifically the higher incidence of chronic kidney disease and diabetes mellitus, and under recognition of this disease in adults. 3
Our cohort had a broad spectrum of infections ranging from classic streptococcal pharyngitis to skin and soft tissue and urinary tract infections. However, 5 of the 8 patients had an upper respiratory tract infection. This is like other large cohort studies where the most common site of infection was upper respiratory tract followed by skin, lung and endocarditis. 8 On review of the electronic record for these patients, the diagnosis of glomerulonephritis was not always on the treating team’s differential of acute kidney injury and therefore complete microbiology studies were not systematically ordered for all patients during their presentation. This again highlights the importance of being aware of the association of all infections with IRGN and to obtain complete microbiologic specimens and appropriate serology, specifically an ASOT and anti-Deoxyribonuclease B (anti-DNase B) titer, as part of the standard glomerulonephritis workup. The combination of ASOT and anti-DNase B has been shown to have a sensitivity of 95.5% and specificity of 88.6% in the diagnosis of poststreptococcal disease. 9
Infection-related glomerulonephritis is often caused by the accumulation of complement and immunoglobulins in the kidneys, leading to an immune and inflammatory response. The typical histo-morphological features include glomerular enlargement and hypercellularity due to proliferation of capillary endothelial cells and influx of inflammatory cells. 10 Acute tubulitis and tubular injury may also be seen along with hyaline and red cell casts. 11 On electron microscopy, the characteristic alteration is the occurrence of prominent subepithelial deposits (“humps”) and less commonly subendothelial deposits. 10 The precise immune-mediated mechanism underlying IRGN is not completely understood, despite recent research providing greater insights into the condition. It is known that streptococcal infections can release nephritogenic proteins, such as nephritis-associated plasmin receptor (NAPlr) 12 and streptococcal pyrogenic exotoxin B (SPEB) 13 into the bloodstream. These proteins accumulate in the endothelium of glomerular capillaries, where they trap and activate plasmin. 14 Plasmin degrades glomerular connective tissue and activates inflammatory cells, thereby resulting in glomerular damage and accumulation of immune complexes formed either in the kidneys or the circulation. Besides NAPIr, enterotoxins associated with staphylococcal infections 15 and cryoglobulins 16 play a role in the pathogenesis of IRGN.
All biopsied patients showed active glomerulonephritis with an exudative pattern. Interestingly, 1 patient (patient 7) had documented COVID-19 infection, and the renal biopsy showed minimal interstitial fibrosis and tubular atrophy in addition to exudative glomerulonephritis. Electron microscopy findings showed tubuloreticular inclusions and subepithelial deposits, both of which have been reported in cases of COVID-19-related glomerulopathy. 17 An unusual full-house immunostaining pattern (IgG, IgM, IgA, C1q, and C3), which is typical of lupus nephritis was observed in 2 of the 7 patients in this case report series. Although not typical of IRGN, full-house immunostaining pattern has been reported in other cases of IRGN. 18 The reasons for full-house pattern in non-lupus nephropathy are still being elucidated, however, non-lupus etiologies presenting with a full-house pattern are associated with poor kidney survival. 19
The prognosis remains poor for patients with this condition. In our cohort, 50% of the patients required hemodialysis at presentation and the majority remained dialysis-dependent which emphasizes the poor long-term outcomes of this disease. 3 Compared with a larger cohort involving mostly Caucasian adults from New York, only 17.1% developed end stage kidney disease and 26.8% developed chronic kidney disease. 8 This observation has major implications for Indigenous patients we care for including possible relocation for dialysis treatments and leaving their family and personal responsibilities behind.
A high index of suspicion for IRGN is needed when evaluating adult patients with acute kidney injury in the context of an acute glomerulonephritis. The second illustrative case demonstrates just how difficult it can be to diagnose this condition in patients with multiple comorbidities and atypical presentations. A large proportion of Indigenous patients have advanced kidney disease due to comorbidities such as diabetes and hypertension. 20 In addition, they can have atypical presentations of auto-immune conditions that make it difficult to establish a definite diagnosis and can lead to delays in implementing appropriate therapy.
Conclusion
Our case series emphasizes the heterogenous clinical, laboratory, and pathological presentations which can make the diagnosis of IRGN quite challenging. The clinician needs to have a high index of suspicion when patients present with an acute kidney injury, features of a glomerulonephritis and an infection, especially those with multiple comorbidities including chronic kidney disease.
Footnotes
Acknowledgements
McGill University is on land which has long served as a site of meeting and exchange amongst Indigenous peoples, including the Haudenosaunee and Anishinaabeg nations. We acknowledge and thank these Indigenous peoples on whose lands this study was performed. The authors thank all First Nation participants and the Cree Board of Health and Social Services of James Bay for their involvement. The results of this case series will be shared with members of the Cree Board of Health and Social Services of James Bay to help promote improved awareness of this disease.
Ethics Approval and Consent to Participate
Consent for this study was obtained from the Cree Board of Health and Social Services of James Bay and the MUHC Research Ethics Board. Each patient provided informed consent to participate in the study.
Consent for Publication
All authors have provided their consent for publication of this article.
Availability of Data and Materials
Data will be stored as per institional guidelines outlined by the Cree Board of Health and Social Services of James Bay and the MUHC research ethics board. The manuscript has a Creative Commons lisence via SAGE Open Access Portal.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
