Abstract
Background:
Diabetes is the leading cause of kidney disease and contributes to 38% of kidney failure requiring dialysis. A gap in detection and management of type 2 diabetes (T2D) in chronic kidney disease (CKD) exists in primary care. Community pharmacists are positioned to support those not able to access kidney care through traditional pathways. Algorithms were developed and validated to assist community pharmacists in identifying individuals with T2D in CKD and prescribing kidney-protective medications.
Objective:
The objective was to develop and validate pharmacist algorithms to confirm T2D and CKD and to prescribe guideline-directed therapies for individuals with an estimated glomerular filtration rate (eGFR) of 30 to 60 mL/min/1.73 m² in community pharmacy primary care clinics in Nova Scotia.
Design:
Lynn’s method was utilized for algorithm development and content validation. Interview data were analyzed using qualitative descriptive analysis.
Setting:
Pharmacists working in primary care clinic settings completed content and face algorithm validation, and virtual interviews were conducted following each round of validation.
Patients:
The algorithms aim to support individuals with T2D and CKD in primary care by optimizing the resources and capacity of community pharmacists while ensuring safety and quality of care through a team-based approach. Patient partners were not part of algorithm development and validation.
Measurements:
Content validity was computed using an item-level content validity index (I-CVI) and scale-level content validity index (S-CVI/Ave) per round. To measure face validity, percentages of those that “agreed” or “strongly agreed” to five statements were calculated.
Methods:
Evidence- and expert-informed algorithms were developed and revised using Lynn’s 3-step method (domain identification, item generation per domain, and instrument formation). Best evidence was collated with literature searches, and experts in nephrology, endocrinology, family medicine, nursing, and pharmacy revised the algorithms until there was consensus agreement on 4 final algorithms (detection of T2D and CKD, initiation/titration of angiotensin-converting enzyme inhibitors or angiotensin receptor blockers, and initiation/management of sodium-glucose cotransporter-2 inhibitors and finerenone). Six community pharmacists per round for 3 rounds were needed to validate the algorithms. A 2-part questionnaire was utilized where pharmacists rated content and face validity using Likert scales. I-CVI and S-CVI/Ave per round and across 3 rounds were determined. Percentages were calculated for the rating level of agreement to 5 statements. Interviews were conducted and analyzed. Revisions were made to the algorithms between rounds.
Results:
Eighteen community pharmacists (6 per round) participated with a mean ± standard deviation of 18 ±11 years of experience. The I-CVI of each item of the algorithms per round ranged from 0.83 to 1, which met the content validity threshold of 0.83 (P < .05) for at least 6 participants. The overall S-CVI/Ave across 3 rounds was 0.97. The overall percentage of participants across 3 rounds who agreed or strongly agreed to 5 face validity statements ranged from 83% to 100%, which was above the prespecified threshold for face validity consensus.
Limitations:
The algorithms are intended for individuals with an eGFR of 30 to 60 mL/min/1.73m². While guideline medications are indicated below this threshold, this cut point was selected as these individuals should typically be referred to a nephrologist. There is a potential for delays in initiation of kidney-protective medications below this threshold while waiting to be seen by nephrology.
Conclusions:
This is the first study to develop and validate algorithms for a new model of care that utilizes community pharmacists to identify and manage T2D and CKD in primary care. The algorithms achieved high content and face validity. Future implementation and evaluation will determine the effectiveness and safety of the algorithms.
Trial Registration:
Not registered.
Introduction
Thirty percent of Canadians live with diabetes or prediabetes. 1 Diabetes is the leading cause of kidney disease and contributes to 38% of kidney failure requiring dialysis in Canada.2 -4 People with diabetes are also 12 times more likely to be hospitalized with kidney failure. 1 One out of 10 Canadians or nearly 4 million adults have chronic kidney disease (CKD).2,4 In Canadian primary care practices, the prevalence of CKD is 72 per 1000 individuals and is higher in rural settings than in urban settings. 5 As kidney function and estimated glomerular filtration rate (eGFR) decline in those with type 2 diabetes (T2D), there is an increased risk of cardiovascular events including heart failure, hospitalization, mortality, and higher utilization of health care resources.6 -8
Over the past several years, new drug therapies with cardiorenal benefits have emerged for the treatment of individuals with T2D and CKD.8 -13 These therapies include sodium-glucose cotransporter-2 inhibitors (SGLT2i), nonsteroidal mineralocorticoid receptor antagonists (MRA, finerenone), and glucagon-like peptide-1 receptor agonists (GLP1-RA). Clinical practice guidelines for diabetes management in CKD have been updated to include these therapies as standard of care. These therapies are recommended along with lifestyle, glycemic and blood pressure optimization, and treatment with angiotensin-converting enzyme inhibitors (ACEi) or angiotensin receptor blockers (ARBs) in those with hypertension or albuminuria.8 -13
Despite compelling evidence for these therapies in T2D and CKD, the use of guideline-directed prescribing, particularly in primary care, is sub-optimal.14 -19 Many barriers have been cited including complexity of prescribing and monitoring, concerns about side effects (hyperkalemia, eGFR dip, cough or reproductive concerns with ACEi or ARB, or genital mycotic infections with SGLT2i), medication costs, limited visit time for complex care, and lack of patient awareness.20 -23 In addition, testing for and recognizing CKD is another barrier to the use of kidney-protective medications.24 -27 Many individuals with T2D and early stages of kidney disease are mainly managed in primary care. They have the most to gain from early detection and persistent prescribing of the kidney-protective therapies known to slow progression and its complications.
According to a recent national survey, approximately one in five Canadians are without a family physician or nurse practitioner. 28 Community pharmacists commonly see individuals with diabetes and kidney disease. Pharmacists’ scope of practice in Canada has expanded over the past few years to allow for a more active role in managing chronic diseases in primary care. 29 In 2023, the Pharmacy Association of Nova Scotia (PANS) in partnership with the Government of Nova Scotia and Nova Scotia Health launched 26 community pharmacy primary care clinics (CPPCCs) in locations with the highest number of people without a family physician. 30 These pharmacist-led clinics provide an extended suite of pharmacy primary care services including assessment and prescribing for chronic disease management.30,31 These clinics are appointment-based, operate separately from the pharmacy dispensary, and are supported by a 24-hour consultation service staffed by a nurse practitioner and/or pharmacist.
Community pharmacists are in an ideal position to order blood work to support the collaborative diagnosis of CKD and to prescribe and monitor guideline-directed medications needed to preserve kidney function in this population. Community pharmacists’ perspectives from our previous research however identified that this is best accomplished through evidence- and expert-informed tools or algorithms. 32 Validated community pharmacist prescribing algorithms of kidney-protective medications in this population are absent in the literature. Our objective was to develop and validate pharmacist algorithms to confirm T2D and CKD diagnosis and to prescribe guideline-recommended therapies (ACEi or ARB, SGLT2i, and finerenone) for those with an eGFR of 30 to 60 mL/min/1.73 m² in CPPCCs in Nova Scotia. This research utilizes an innovative model of care to address the gap in CKD detection and management in primary care by optimizing the resources and capacity of community pharmacists while ensuring safety and quality of care through a team-based approach.
Methods
This study consisted of two phases. Phase 1 encompassed developing algorithms to assist pharmacists working in a CPPCC to confirm the diagnosis of T2D and CKD, and to manage pharmacotherapy to slow CKD progression in individuals with an eGFR of 30 to 60 mL/min/1.73 m². This eGFR cut point was selected as individuals with an eGFR below 30 mL/min/1.73 m2 are typically referred to and managed by a nephrologist. Phase 2 was comprised of validation of the algorithms by community pharmacists. The study was approved by Nova Scotia Health’s Research Ethics Board (File #1030369).
Phase 1: Development of the Algorithms
Our recent qualitative study of interviews of community pharmacists’ perspectives on assessing kidney function and prescribing in primary care guided the algorithm development phase. 32 Four algorithms were developed including detection of T2D and CKD, initiation and titration of ACEi or ARB to maximally tolerated doses, and initiation and management of SGLT2i and finerenone. Only publicly funded medications were included in the algorithms. To identify the best evidence for these algorithms, a medical librarian assisted with search strategies in Ovid MEDLINE, Embase, CINAHL, and Cochrane databases between January 1, 2020, and March 15, 2024. Evidence tables were created for the diagnostic criteria for T2D and CKD along with each algorithm medication to assist the research team in algorithm development (see Supplementary File 1). From here, evidence- and expert-informed algorithms were developed and revised using Lynn’s 3-step method (domain identification, item generation per domain, and instrument formation). 33 Team experts included nephrologists, pharmacists, an endocrinologist, a family physician, a nurse practitioner, an implementation scientist, policy-makers, and information system specialists. These experts identified three key domains for our T2D and CKD algorithms including screening/assessment, prescribing, and monitoring (laboratory values and physical assessment). Per domain, relevant evidence-based items were extracted from literature, guidelines, and medication product monographs. The research team met virtually to conceptualize the instrument or algorithm formation. A sequential algorithm format, which follows a linear set of steps, was preferred by the team. 34 Decision nodes were used to ensure each step was completed before moving onto the next and to enable navigation through different clinical scenarios. Sequential algorithms also supported the integration of loops where multiple steps need to be completed to achieve an outcome, such as with the titration of an ACEi or ARB to achieve a maximally tolerated dose. The first draft of the algorithms underwent three rounds of review and revisions with the research team until agreement was reached on the final algorithms using the methods outlined in the following sections for pharmacist content and face validation.
Phase 2: Pharmacist Validation of the Algorithms
Community pharmacists working in a CPPCC were recruited using an advertisement sent through email by PANS. Informed consent was obtained for those agreeing to participate. Six community pharmacist participants per round (18 community pharmacists in total across 3 rounds) were needed to minimize the risk of chance agreement based on previous work done by a team member (M.B.).35 -37 A minimum of three rounds were planned with revisions made by research team members between each round.
Validation of the algorithms was based on an approach outlined by Lynn’s method. 33 A two-part questionnaire was developed to validate the algorithm’s content (e.g., appropriateness, accuracy, completeness of content) and face validity (e.g., clarity, comprehensibility, and appropriateness to the user) based on Feinstein’s concept of clinical sensibility 38 (see Supplementary File 2). The algorithms were divided into items A to G to align with the two-part questionnaire (see Supplementary File 3). Pharmacist participants were emailed the two-part questionnaire along with the algorithms and were given 10 days to complete their questionnaire. After questionnaires were returned, a 1-on-1 short virtual interview was conducted (J.-A.W., J.M.) with each pharmacist to discuss ratings and comments.
Content Validation
Participants were asked to independently rate the algorithms on a four-point Likert scale (1: strongly disagree [unacceptable, remove], 2: disagree [unacceptable, major revisions needed], 3: agree [acceptable with minor revisions], 4: strongly agree [acceptable as is]). Participants were asked to provide comments about where revisions were needed. Any rating of 1 or 2 by one or more participants required revision before the subsequent round.
Face Validation
Participants were asked to rate statements regarding each of the algorithms on a five-point scale (1: strongly disagree, 2: disagree, 3: neither agree nor disagree, 4: agree, 5: strongly agree). Statements included questions whether the tool was clear and understandable, used appropriate language and wording, flowed in a logical manner, whether they would use it in their practice, and whether they would recommend its use. Participants were asked to provide comments to improve the algorithms. Any statement with a rating of 1 to 3 by one or more participant’s required revision.
Statistical Analysis
For content validity, the content validity index (CVI) was computed using an item-level CVI (I-CVI) and scale-level CVI (S-CVI/Ave) per round. The I-CVI is the proportion of pharmacist participants giving the item (A to G) a rating of 3 or 4. According to Lynn and others, for 6 participants in a round, at least 5 must agree (rating of 3 or 4) to establish content validity at P < .05 (I-CVI of ≥0.83). 33 ,39 -41 An item is considered invalid if 2 out of the 6 participants per round provide a rating of 1 or 2 (I-CVI ≤ 0.67). The S-CVI/Ave is the average of all I-CVI scores for all items (A to G) per round. We also calculated the overall I-CVI for each item and S-CVI/Ave across the three rounds. A S-CVI/Ave ≥ 0.9 indicates that the algorithms have excellent content validity.39 -41
For face validity, percentages were calculated for the rating of each statement. Ratings of 4 or 5 were deemed as face valid while ratings of 1 to 3 were not. A threshold for consensus was set at a minimum of 70% of the number of participants, with level of agreement set as agree or strongly agree (rating of 4 or 5). This threshold is commonly employed when using the Delphi technique.35,36,42 -44 Algorithm components that did not achieve content and face validation required revision before the next round.
Interview data were analyzed using a qualitative descriptive analysis. As described by Sandelowski, this approach is characterized by its focus on summarizing the informational contents of the data (what participants said) rather than on interpretation (what participants meant).45,46 Relevant to tool development, this low-inference approach is well suited to describe pharmacists’ perspectives on the tool in terms of content and face validity, ease of use, and identification of specific opportunities for improvement.
Results
Phase 1: Development of Algorithms
Between 10 and 14 research team content experts, based on availability, informed the algorithm development in 3 rounds of content and face validation between March 31, 2024, and May 31, 2024 (Supplementary File 4). Over 3 rounds, the I-CVI per algorithm item ranged from 0.80 to 1. The overall S-CVI/Ave across three rounds was 0.96. The overall percentage of participants (average across three rounds) who agreed or strongly agreed to five face validity statements ranged between 82% and 100%. Following content and face validation and when no further algorithm revision was required, we proceeded to the pharmacist algorithm validation in phase 2.
Phase 2: Pharmacist Validation of Algorithms
Study participants
Eighteen community pharmacists from different urban (61%) and rural (39%) community pharmacy primary care settings in all zones of Nova Scotia participated. The majority (94%) of participants were female. The mean ± standard deviation (SD) for participant age was 43 ± 10 years, with 18 ± 11 years of experience (Table 1). Most of the participants (89%) had a Bachelor of Science in Pharmacy as their highest academic qualification. The mean debrief interview duration was 30 minutes (range: 22-35 minutes).
Community Pharmacists’ Demographic and Practice Characteristics.

Note. CPPCC = community pharmacy primary care clinic; PharmD = doctor of pharmacy; SD = standard deviation; NSH = Nova Scotia Health.
Community pharmacist.
Community pharmacist/nurse practitioner.
Pharmacist/nurse practitioner or physician.
Content and face validation
Three rounds of content and face validation were undertaken between June 2, 2024, and July 17, 2024. Three rounds of interviews (18 total) were conducted between each round. Tool revisions were made after each round, and changes made are available in the Supplementary File 5. The first part of the questionnaire quantified content validity (Supplementary File 6). Over three rounds, the I-CVI of each algorithm item (A to G) ranged from 0.83 to 1. The I-CVI of each item of the algorithms per round of validation met the threshold of 0.83 (P < .05) for at least 6 participants (Table 2). The calculated S-CVI/Ave for rounds 1, 2, and 3 of the pharmacist content validation was 1, 0.93, and 0.98, respectively. The overall S-CVI/Ave across three rounds was 0.97 and exceeded the threshold for content validity. Therefore, community pharmacists found the algorithm content appropriate, accurate, and complete for the management of T2D in CKD.
Pharmacist Algorithm Content Validity Indices Scores.

Note. A = Screen/Confirm T2D; B = Screen/Confirm CKD; C = ACEi or ARB Titration to Maximally Tolerated Dose; D = ACEi or ARB Initiation and Titration to Maximally Tolerated Dose; E = ACEi or ARB Dose Table; F = SGLT2i; G = Finerenone.
The second part of the questionnaire quantified face validity (Supplementary File 7). The overall percentage of participants (average across 3 rounds) who agreed or strongly agreed to 5 face validity statements for algorithm 1 (confirming T2D and CKD diagnosis), algorithm 2 (ACEi or ARB), algorithm 3 (SGLT2i), and algorithm 4 (finerenone) ranged from 89% to 94%, 83% to 100%, 89% to 100%, and 83% to 100%, respectively (Table 3). All algorithms were above the prespecified threshold for face validity consensus of 70% by round 3. Therefore, community pharmacists considered the algorithms clear, comprehensive, and appropriate for users. Algorithm agreement (across 3 rounds) for statement 1 (algorithm is clear and understandable), 2 (algorithm uses appropriate language and wording), 3 (algorithm flows in a logical manner), and 4 (confident in using algorithm in their practice) ranged from 67% to 100%. Agreement for those who would be confident recommending the use of the algorithms (statement 5) ranged from 83% to 100%. The algorithms with the highest overall agreement scores (5 statements combined across 3 rounds) were SGLT2i (95.4%), followed by ACEi or ARB (93.2%), confirming T2D in CKD (91%), and finerenone (89.8%). The finerenone algorithm required the most revision for face validation.
Pharmacist Algorithm Face Validity: Level of Agreement per Statement.

Note. ACEi = angiotensin-converting enzyme inhibitor; ARB = angiotensin receptor blocker; CKD = chronic kidney disease; CPPCC = community pharmacy primary care clinic; T2D = type 2 diabetes.
Interview feedback
Recurring feedback was identified throughout the 3 rounds of algorithm validation. Most comments were related to the formatting and wording of certain aspects of the algorithms to improve clarity and usability. For example, sentences were reworded, some words were bolded to add emphasis, and details were added to certain steps to minimize ambiguity. Several participants from round 1 of validation commented that the ACEi or ARB algorithms were too complex. In response to these comments, items C and D were split into two separate algorithms (for round 2 and 3), where item C provides guidance on titration to maximally tolerated dose, and item D provides guidance on initiation and titration to maximally tolerated dose. In addition, it was suggested to integrate links to provide resources for lifestyle and relevant guidelines for T2D in CKD. In response to comments regarding the algorithm eGFR cut points of 30-60 mL/min/1.73 m2, a disclaimer was added to include SGLT2i and finerenone drug product monograph labeling around initiation of these medications at lower eGFRs (<20 mL/min/1.73 m2 [SGLT2i] and ≤25 mL/min/1.73 m2 [finerenone]). This disclaimer also added clarity to ensure that those with an eGFR below 30 mL/min/1.73 m2 are referred to nephrology. A few participants commented that they had a lack of clinical experience with the finerenone algorithm to provide content validation and would require education and training. Furthermore, there were concerns with the finerenone algorithm provincial funding criteria as it required consultation with a nephrologist for coverage. To overcome this barrier, we sought provincial funding approval that the use of the finerenone algorithm would meet the nephrologist consultation criteria as it was extensively developed by these stakeholders. Figure 1 shows the final version of the validated algorithms.
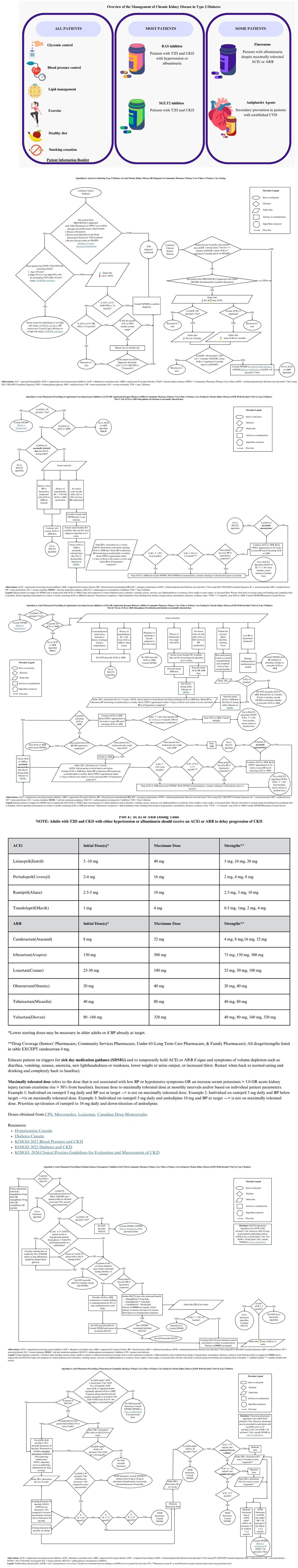
Final validated algorithms.
Discussion
These algorithms were developed to enhance detection and use of guideline-directed kidney-protective medications for those with T2D and CKD in primary care using a collaborative community pharmacist model of care. While content validity was achieved in round 1, we continued with three rounds as face validity did not occur until round 3. The algorithms achieved high content validation with an overall S-CVI/Ave across three rounds of 0.97. The overall percentage of participants (average across three rounds) who agreed or strongly agreed to 5 face validity statements ranged from 83% to 100%. Overall, 83% to 94% of participants (average across 3 rounds) agreed they would use the algorithms in their own practice.
Clinical pathways for diagnosis, management, and referral of adults with CKD have been developed for health care providers including the potential clinical use for pharmacists. 47 However, this is the first study to develop and validate algorithms for a model of care that utilizes community pharmacists to identify and manage T2D and CKD in primary care.
The algorithms provide a systematic, comprehensive, evidence- and expert-informed approach to assist community pharmacists screen, order blood work to identify T2D and CKD, and prescribe and monitor ACEi or ARB, SGLT2i, and finerenone.
The algorithms target those with an eGFR of 30 to 60 mL/min/1.73 m2 as these individuals are typically managed in primary care and not by a nephrologist. A study in a community pharmacy conducting point-of-care eGFR testing in high-risk individuals led to the identification of nearly 40% of participants with CKD stages 3 to 5. 48 Our algorithms also include triggers to refer to a nephrologist when the eGFR falls below 30 mL/min/1.73 m2. They were developed to support individuals with or without a family physician; however, there are several algorithm branch points where collaborative care is required. One example is where pharmacists must consult with a nurse practitioner or the individual’s family physician to confirm a diagnosis of CKD or to refer to nephrology. Coordination of care has been shown to increase the use of guideline-directed medication therapies particularly in higher risk populations.49,50 The algorithms also build in regular contact points for the pharmacist to assess response, provide education, and support ongoing treatment. Ongoing use of kidney-protective medications is required to derive their benefits. Higher risk of death and kidney failure have been associated with increasing duration of ACEi or ARB discontinuation. 51 Significant improvements in adherence and medication knowledge have been reported with pharmacist-managed or cross-team collaboration.52,53
The addition of a pharmacist in ambulatory or primary care settings has been shown to support greater use of guideline-directed therapies in individuals with CKD. 19 ,54 -59 The DRIVE trial, Diabetes Remote Intervention to Improve Use of Evidence-Based Medications, was a remote virtual team-based approach which included a clinical pharmacist who prescribed an SGLT2i or a GLP1 receptor agonist under a collaborative drug therapy management agreement, along with a nurse practitioner and physicians. 60 The trial led to greater detection and prescribing of guideline therapeutics.
Strengths of this study include the utilization of evidence and content experts in nephrology, endocrinology, family medicine, nursing, pharmacy, informatics, professional associations, and regulators to inform the algorithm development. The algorithms foster collaborative team-based care with the individual’s health care team and facilitate referrals where needed. The algorithms support screening individuals at risk of T2D and CKD, which may improve detection. To remove the barrier of medication access, we only included medications with provincial funding. Individuals lacking medication insurance have been associated with lower prescribing rates of guideline-directed kidney-protective medications. 14 ,20 -23 Following team expert consensus, content and face validation with community pharmacists was undertaken with a balanced number of urban and rural pharmacy clinics, which may support adoption and scalability in other jurisdictions. Furthermore, we utilized the Lynn method for establishing content validity, which is commonly used for algorithm validation.35 -37 This method allows for multiple rounds of feedback until validation of the final algorithms. 33 There are several study limitations. First, although Lynn’s method is widely used to validate algorithms, there is no universally accepted threshold for content validation. Second, research team content experts validating the algorithm initially were also those who developed the algorithms and therefore may have been biased in their validation scores. However, we achieved high I-CVI and S-CVI across three rounds with the community pharmacist algorithm validators, which adds confidence that validation was achieved. Third, not all comments could be integrated in the algorithms. These will be included in future education and training for algorithm implementation. Fourth, GLP-1 receptor agonists were not included as an algorithm due to the lack of public funding but will be a part of future algorithm development given the reported benefits in reducing kidney and cardiovascular clinical outcomes in individuals with CKD with T2D. 61 It is important to note that the generalizability of these algorithms in other jurisdictions may be impacted by medication coverage. Furthermore, this validation study did not include patient voice. Patient partners have joined the research team to support the implementation and evaluation of the algorithms. Lastly, these algorithms did not focus specifically on lifestyle considerations, but we plan to incorporate resources and education and strengthen linkages to provincial diabetes centers with future algorithm implementation.
Conclusion
We developed and validated algorithms for confirming the diagnosis of T2D and CKD and for prescribing kidney-protective medications (ACEi or ARB, SGLT2i, and finerenone) by community pharmacists working in CPPCCs in Nova Scotia. The algorithms underwent rigorous content and face validation, first by the research team and then by the community pharmacists end users. Community pharmacists are positioned to support individuals who are not able to access kidney care through traditional pathways. This research has the potential to improve access to CKD care as well as create greater responsiveness and coordination of team-based care through CPPCCs. Future research seeks to implement and evaluate the algorithm’s effectiveness and safety in CPPCCs.
Supplemental Material
sj-docx-1-cjk-10.1177_20543581241309974 – Supplemental material for Optimizing Prescribing for Individuals With Type 2 Diabetes and Chronic Kidney Disease Through the Development and Validation of Algorithms for Community Pharmacists
Supplemental material, sj-docx-1-cjk-10.1177_20543581241309974 for Optimizing Prescribing for Individuals With Type 2 Diabetes and Chronic Kidney Disease Through the Development and Validation of Algorithms for Community Pharmacists by Jennifer Morris, Marisa Battistella, Karthik Tennankore, Steven Soroka, Cynthia Kendell, Penelope Poyah, Keigan More, Mathew Grandy, Thomas Ransom, Natalie Kennie-Kaulbach, Daniel Rainkie, Jaclyn Tran, Syed Sibte Raza Abidi, Samina Abidi, Nicole Fulford, Heather Neville, Heather Naylor, Lisa Woodill, Andrea Bishop, Glenn Rodrigues, Diane Harpell, Michelle Stewart and Jo-Anne Wilson in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-docx-10-cjk-10.1177_20543581241309974 – Supplemental material for Optimizing Prescribing for Individuals With Type 2 Diabetes and Chronic Kidney Disease Through the Development and Validation of Algorithms for Community Pharmacists
Supplemental material, sj-docx-10-cjk-10.1177_20543581241309974 for Optimizing Prescribing for Individuals With Type 2 Diabetes and Chronic Kidney Disease Through the Development and Validation of Algorithms for Community Pharmacists by Jennifer Morris, Marisa Battistella, Karthik Tennankore, Steven Soroka, Cynthia Kendell, Penelope Poyah, Keigan More, Mathew Grandy, Thomas Ransom, Natalie Kennie-Kaulbach, Daniel Rainkie, Jaclyn Tran, Syed Sibte Raza Abidi, Samina Abidi, Nicole Fulford, Heather Neville, Heather Naylor, Lisa Woodill, Andrea Bishop, Glenn Rodrigues, Diane Harpell, Michelle Stewart and Jo-Anne Wilson in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-docx-11-cjk-10.1177_20543581241309974 – Supplemental material for Optimizing Prescribing for Individuals With Type 2 Diabetes and Chronic Kidney Disease Through the Development and Validation of Algorithms for Community Pharmacists
Supplemental material, sj-docx-11-cjk-10.1177_20543581241309974 for Optimizing Prescribing for Individuals With Type 2 Diabetes and Chronic Kidney Disease Through the Development and Validation of Algorithms for Community Pharmacists by Jennifer Morris, Marisa Battistella, Karthik Tennankore, Steven Soroka, Cynthia Kendell, Penelope Poyah, Keigan More, Mathew Grandy, Thomas Ransom, Natalie Kennie-Kaulbach, Daniel Rainkie, Jaclyn Tran, Syed Sibte Raza Abidi, Samina Abidi, Nicole Fulford, Heather Neville, Heather Naylor, Lisa Woodill, Andrea Bishop, Glenn Rodrigues, Diane Harpell, Michelle Stewart and Jo-Anne Wilson in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-docx-12-cjk-10.1177_20543581241309974 – Supplemental material for Optimizing Prescribing for Individuals With Type 2 Diabetes and Chronic Kidney Disease Through the Development and Validation of Algorithms for Community Pharmacists
Supplemental material, sj-docx-12-cjk-10.1177_20543581241309974 for Optimizing Prescribing for Individuals With Type 2 Diabetes and Chronic Kidney Disease Through the Development and Validation of Algorithms for Community Pharmacists by Jennifer Morris, Marisa Battistella, Karthik Tennankore, Steven Soroka, Cynthia Kendell, Penelope Poyah, Keigan More, Mathew Grandy, Thomas Ransom, Natalie Kennie-Kaulbach, Daniel Rainkie, Jaclyn Tran, Syed Sibte Raza Abidi, Samina Abidi, Nicole Fulford, Heather Neville, Heather Naylor, Lisa Woodill, Andrea Bishop, Glenn Rodrigues, Diane Harpell, Michelle Stewart and Jo-Anne Wilson in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-docx-13-cjk-10.1177_20543581241309974 – Supplemental material for Optimizing Prescribing for Individuals With Type 2 Diabetes and Chronic Kidney Disease Through the Development and Validation of Algorithms for Community Pharmacists
Supplemental material, sj-docx-13-cjk-10.1177_20543581241309974 for Optimizing Prescribing for Individuals With Type 2 Diabetes and Chronic Kidney Disease Through the Development and Validation of Algorithms for Community Pharmacists by Jennifer Morris, Marisa Battistella, Karthik Tennankore, Steven Soroka, Cynthia Kendell, Penelope Poyah, Keigan More, Mathew Grandy, Thomas Ransom, Natalie Kennie-Kaulbach, Daniel Rainkie, Jaclyn Tran, Syed Sibte Raza Abidi, Samina Abidi, Nicole Fulford, Heather Neville, Heather Naylor, Lisa Woodill, Andrea Bishop, Glenn Rodrigues, Diane Harpell, Michelle Stewart and Jo-Anne Wilson in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-docx-2-cjk-10.1177_20543581241309974 – Supplemental material for Optimizing Prescribing for Individuals With Type 2 Diabetes and Chronic Kidney Disease Through the Development and Validation of Algorithms for Community Pharmacists
Supplemental material, sj-docx-2-cjk-10.1177_20543581241309974 for Optimizing Prescribing for Individuals With Type 2 Diabetes and Chronic Kidney Disease Through the Development and Validation of Algorithms for Community Pharmacists by Jennifer Morris, Marisa Battistella, Karthik Tennankore, Steven Soroka, Cynthia Kendell, Penelope Poyah, Keigan More, Mathew Grandy, Thomas Ransom, Natalie Kennie-Kaulbach, Daniel Rainkie, Jaclyn Tran, Syed Sibte Raza Abidi, Samina Abidi, Nicole Fulford, Heather Neville, Heather Naylor, Lisa Woodill, Andrea Bishop, Glenn Rodrigues, Diane Harpell, Michelle Stewart and Jo-Anne Wilson in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-jpg-3-cjk-10.1177_20543581241309974 – Supplemental material for Optimizing Prescribing for Individuals With Type 2 Diabetes and Chronic Kidney Disease Through the Development and Validation of Algorithms for Community Pharmacists
Supplemental material, sj-jpg-3-cjk-10.1177_20543581241309974 for Optimizing Prescribing for Individuals With Type 2 Diabetes and Chronic Kidney Disease Through the Development and Validation of Algorithms for Community Pharmacists by Jennifer Morris, Marisa Battistella, Karthik Tennankore, Steven Soroka, Cynthia Kendell, Penelope Poyah, Keigan More, Mathew Grandy, Thomas Ransom, Natalie Kennie-Kaulbach, Daniel Rainkie, Jaclyn Tran, Syed Sibte Raza Abidi, Samina Abidi, Nicole Fulford, Heather Neville, Heather Naylor, Lisa Woodill, Andrea Bishop, Glenn Rodrigues, Diane Harpell, Michelle Stewart and Jo-Anne Wilson in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-jpg-4-cjk-10.1177_20543581241309974 – Supplemental material for Optimizing Prescribing for Individuals With Type 2 Diabetes and Chronic Kidney Disease Through the Development and Validation of Algorithms for Community Pharmacists
Supplemental material, sj-jpg-4-cjk-10.1177_20543581241309974 for Optimizing Prescribing for Individuals With Type 2 Diabetes and Chronic Kidney Disease Through the Development and Validation of Algorithms for Community Pharmacists by Jennifer Morris, Marisa Battistella, Karthik Tennankore, Steven Soroka, Cynthia Kendell, Penelope Poyah, Keigan More, Mathew Grandy, Thomas Ransom, Natalie Kennie-Kaulbach, Daniel Rainkie, Jaclyn Tran, Syed Sibte Raza Abidi, Samina Abidi, Nicole Fulford, Heather Neville, Heather Naylor, Lisa Woodill, Andrea Bishop, Glenn Rodrigues, Diane Harpell, Michelle Stewart and Jo-Anne Wilson in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-jpg-5-cjk-10.1177_20543581241309974 – Supplemental material for Optimizing Prescribing for Individuals With Type 2 Diabetes and Chronic Kidney Disease Through the Development and Validation of Algorithms for Community Pharmacists
Supplemental material, sj-jpg-5-cjk-10.1177_20543581241309974 for Optimizing Prescribing for Individuals With Type 2 Diabetes and Chronic Kidney Disease Through the Development and Validation of Algorithms for Community Pharmacists by Jennifer Morris, Marisa Battistella, Karthik Tennankore, Steven Soroka, Cynthia Kendell, Penelope Poyah, Keigan More, Mathew Grandy, Thomas Ransom, Natalie Kennie-Kaulbach, Daniel Rainkie, Jaclyn Tran, Syed Sibte Raza Abidi, Samina Abidi, Nicole Fulford, Heather Neville, Heather Naylor, Lisa Woodill, Andrea Bishop, Glenn Rodrigues, Diane Harpell, Michelle Stewart and Jo-Anne Wilson in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-jpg-6-cjk-10.1177_20543581241309974 – Supplemental material for Optimizing Prescribing for Individuals With Type 2 Diabetes and Chronic Kidney Disease Through the Development and Validation of Algorithms for Community Pharmacists
Supplemental material, sj-jpg-6-cjk-10.1177_20543581241309974 for Optimizing Prescribing for Individuals With Type 2 Diabetes and Chronic Kidney Disease Through the Development and Validation of Algorithms for Community Pharmacists by Jennifer Morris, Marisa Battistella, Karthik Tennankore, Steven Soroka, Cynthia Kendell, Penelope Poyah, Keigan More, Mathew Grandy, Thomas Ransom, Natalie Kennie-Kaulbach, Daniel Rainkie, Jaclyn Tran, Syed Sibte Raza Abidi, Samina Abidi, Nicole Fulford, Heather Neville, Heather Naylor, Lisa Woodill, Andrea Bishop, Glenn Rodrigues, Diane Harpell, Michelle Stewart and Jo-Anne Wilson in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-jpg-7-cjk-10.1177_20543581241309974 – Supplemental material for Optimizing Prescribing for Individuals With Type 2 Diabetes and Chronic Kidney Disease Through the Development and Validation of Algorithms for Community Pharmacists
Supplemental material, sj-jpg-7-cjk-10.1177_20543581241309974 for Optimizing Prescribing for Individuals With Type 2 Diabetes and Chronic Kidney Disease Through the Development and Validation of Algorithms for Community Pharmacists by Jennifer Morris, Marisa Battistella, Karthik Tennankore, Steven Soroka, Cynthia Kendell, Penelope Poyah, Keigan More, Mathew Grandy, Thomas Ransom, Natalie Kennie-Kaulbach, Daniel Rainkie, Jaclyn Tran, Syed Sibte Raza Abidi, Samina Abidi, Nicole Fulford, Heather Neville, Heather Naylor, Lisa Woodill, Andrea Bishop, Glenn Rodrigues, Diane Harpell, Michelle Stewart and Jo-Anne Wilson in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-jpg-8-cjk-10.1177_20543581241309974 – Supplemental material for Optimizing Prescribing for Individuals With Type 2 Diabetes and Chronic Kidney Disease Through the Development and Validation of Algorithms for Community Pharmacists
Supplemental material, sj-jpg-8-cjk-10.1177_20543581241309974 for Optimizing Prescribing for Individuals With Type 2 Diabetes and Chronic Kidney Disease Through the Development and Validation of Algorithms for Community Pharmacists by Jennifer Morris, Marisa Battistella, Karthik Tennankore, Steven Soroka, Cynthia Kendell, Penelope Poyah, Keigan More, Mathew Grandy, Thomas Ransom, Natalie Kennie-Kaulbach, Daniel Rainkie, Jaclyn Tran, Syed Sibte Raza Abidi, Samina Abidi, Nicole Fulford, Heather Neville, Heather Naylor, Lisa Woodill, Andrea Bishop, Glenn Rodrigues, Diane Harpell, Michelle Stewart and Jo-Anne Wilson in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-jpg-9-cjk-10.1177_20543581241309974 – Supplemental material for Optimizing Prescribing for Individuals With Type 2 Diabetes and Chronic Kidney Disease Through the Development and Validation of Algorithms for Community Pharmacists
Supplemental material, sj-jpg-9-cjk-10.1177_20543581241309974 for Optimizing Prescribing for Individuals With Type 2 Diabetes and Chronic Kidney Disease Through the Development and Validation of Algorithms for Community Pharmacists by Jennifer Morris, Marisa Battistella, Karthik Tennankore, Steven Soroka, Cynthia Kendell, Penelope Poyah, Keigan More, Mathew Grandy, Thomas Ransom, Natalie Kennie-Kaulbach, Daniel Rainkie, Jaclyn Tran, Syed Sibte Raza Abidi, Samina Abidi, Nicole Fulford, Heather Neville, Heather Naylor, Lisa Woodill, Andrea Bishop, Glenn Rodrigues, Diane Harpell, Michelle Stewart and Jo-Anne Wilson in Canadian Journal of Kidney Health and Disease
Footnotes
Ethical Approval
This study was approved by the Nova Scotia Health Research Ethics Board (REB file number 1030369).
Consent to Participate
All authors consented to publication. Written informed consent was obtained from participants prior to the commencement of the study.
Availability of Data and Materials
The data collected and anlyzed in this study are not made publicly available to protect the identity of particpants.
Authors’ Note
We are committed to improving patient care for individuals with type 2 diabetes and chronic kidney disease. Our team developed and validated algorithms to empower community pharmacists with optimized prescribing strategies. Together, we can make a significant impact! #Kidney #Pharmacy #DiabetesCare.
Author Contributions
J.-A.W. initiated and lead this project, supported database searches and collated best evidence to support algorithm development, developed and revised the algorithms per round, created a two-part questionnaire per round, conducted data collection, performed analysis, interviewed participants, drafted the initial draft and final manuscript. J.M. conducted database searches, prepared evidence tables to assist in algorithm development, completed revisions between rounds for the algorithms, supported data collection and analysis, and supported initial manuscript draft, tables, and supplemental materials. All other authors supported J.W. in the project initiation through concept development and methodology, provided content expertise and completed 3 rounds of team content and face validation, and reviewed and revised initial and final manuscripts.
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: There are no conflicts of interest related to this work except for the following authors: J.-A.W. has received moderator honorarium from Otsuka; K.T. has received honorarium for advisory board with Otsuka, Bayer, and Vifor Pharmaceuticals; has received unrestricted investigator-initiated grant funding from Otsuka; and is a deputy editor for the Canadian Journal of Kidney Health and Disease; K.M. has received speaker honorarium from Otsuka; M.S. has received honorarium from GSK; S.S. has received honorarium for advisory boards for Amgen, GSK, Bayer, and Otsuka; and L.W. is an employee of the non-profit Pharmacy Association of Nova Scotia where she advocates on behalf of pharmacy professionals in the province and is the project lead for the Community Pharmacy Primary Care Clinic Demonstration Project.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded through grants from the Dalhousie Pharmacy Endowment Fund and the Dalhousie University Faculty of Health Summer Research Internship.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
