Abstract
Background:
In 2013, the British Columbia (BC) Childhood Nephrotic Syndrome Clinical Pathway (CNSCP) was developed to standardize the care of children with nephrotic syndrome (NS). In BC, children access nephrology care at BC Children’s Hospital (BCCH) and multiple regional clinics.
Objective:
The primary objective was to compare induction therapy and clinical outcomes between BCCH and regional clinics since implementation of the CNSCP.
Design, setting, and patients:
This was a retrospective cohort study of children with NS in BC.
Measurements and methods:
We conducted a retrospective cohort study of children 1 to 17 years old with new-onset NS from 2013 to 2019 inclusive with minimum 12 months of follow-up. Children with non-minimal change disease, steroid resistance, incomplete induction therapy, or less than 6 months of pathway treatment within their first year post-diagnosis were excluded. Clinics were categorized as BCCH or regional (Surrey, Prince George, or Kelowna).
Results:
Sixty-nine patients were included, with 52 (75%) at BCCH and 17 (25%) at regional clinics. There were no significant between-group differences in age, sex, or clinical characteristics at time of diagnosis. Comparing BCCH and regional clinics, there was no difference in induction prednisone exposure (median 3400, interquartile range [IQR] 3331-3585 mg/m2 vs 3492, IQR 3397-3644 mg/m2, P = .167), annualized relapse rate (median 3.3, IQR 1.1-5.3 vs 2.3, IQR 0.5-4.2, P = .575), or development of frequently relapsing courses (50% vs 62%, P = .475). There was a similar number of first-year clinic visits (4.2 ± 1.2 vs 4.0 ± 1.8, P = .655) and dietitian-reviewed food records (67% vs 47%, P = .135, BCCH vs regional). More children at BCCH had a recommended ophthalmology surveillance visit (87% vs 59%, P = .01, BCCH vs regional).
Limitations:
Study limitations include small sample size and exclusion of children with complicated NS (ie, relapse during induction, steroid resistance).
Conclusion:
Since we implemented the CNSCP, children with NS received comparable care and had similar outcomes at BCCH and regional clinics without significant practice variation.
Introduction
Childhood idiopathic nephrotic syndrome (NS) is one of the most common pediatric kidney diseases, affecting 1 to 17 per 100 000 children. 1 Nephrotic syndrome is characterized by the triad of proteinuria, hypoalbuminemia, and edema. Appropriate recognition and management of NS are important to prevent morbidity from secondary complications, including severe edema, acute kidney injury, thromboembolic events, infection, and hyponatremia. 2 Standard first-line treatment for newly diagnosed NS is oral corticosteroids (prednisone). Up to 80% of patients will be steroid sensitive and achieve complete remission within the first 4 weeks of prednisone therapy. 3 The majority of these children will also experience at least one relapse of their disease, and almost half will go on to have multiple relapses requiring further exposure to corticosteroids. 4 While prednisone is effective for most children with NS, chronic corticosteroid use and increased cumulative exposure necessitate regular monitoring for weight gain, hypertension, impaired linear growth, and ocular complications. 5 Several guidelines exist for induction therapy dosing regimens and for treatment of subsequent relapses.6 -8 However, recent single- and multi-center data suggest that significant variability exists in corticosteroid dosing for children with newly diagnosed NS.9,10
In an effort to minimize local practice variation, the Pediatric Nephrology Clinical Pathway Development Team at BC Children’s Hospital (BCCH) created a Childhood Nephrotic Syndrome Clinical Pathway (CNSCP) to standardize the diagnosis, management, and surveillance of patients to improve the outcomes of children with idiopathic and uncomplicated NS in British Columbia (BC). 11 The CNSCP is a structured, multidisciplinary, evidence-based, and prescriptive care plan that adapts high-level guidelines to the local context.
The development process included weekly multidisciplinary discussions with experts from diverse disciplines (nephrologists, endocrinologists, ophthalmologists, pharmacists, registered dietitians, pediatricians, trainees), consensus building to formalize the CNSCP recommendations, and the publication and dissemination of said recommendations via a Physicians’ Handbook in 2013. 11 In addition to reviewing the most up-to-date Kidney Disease Improving Global Outcomes (KDIGO) Clinical Practice Guidelines for Glomerulonephritis at the time, 12 a historical practice audit of BCCH patients with NS diagnosed from 1990 to 2012 was completed in order to inform practices regarding prednisone dosing, 9 indications for kidney biopsy, 13 and dietary recommendations. 14 When new data were published in 2016 suggesting a reduction in prednisone dosing for induction therapy,15,16 a second edition of the CNSCP was published to reflect most recent data.
The CNSCP not only outlines recommendations for prednisone dosing and clinic follow-up, but also clearly delineates the importance of surveillance monitoring for adverse effects of steroid exposure, like ocular assessments (for cataracts or glaucoma) and food intake records (to ensure appropriate sodium, calcium, vitamin D, and caloric intake; see Figure 1 for summary of all recommendations). Given that the pathway’s target patient population are those with typical NS, those that relapse during induction, develop steroid resistance, or have non-minimal change disease subsequently exit the pathway and are treated per physician preference with steroid-sparing agents (SSAs) including tacrolimus, mycophenolate mofetil, or cyclophosphamide.

Recommended scheduled visits during the first year of follow-up post-diagnosis, including history, physical exam, laboratory, and other surveillance monitoring.
Since its initial publication, the CNSCP has been disseminated through multiple avenues, including BC’s Pediatrics Grand Rounds presentations, continuing medical education sessions through the BC Pediatric Society, didactic teaching sessions with BCCH pediatrics residents, ad hoc information sessions at regional sites, and robust consensus-building discussions at our BCCH divisional rounds, which include all the practicing pediatric nephrologists in the province. We also leveraged the relationships built between our regional clinics and local pediatricians for word-of-mouth distribution.
Located in Vancouver, BCCH is the only tertiary pediatric referral center for the province of BC. Children across the province access kidney care at the BCCH home campus or one of its regional clinics. Regional clinics in Surrey and Prince George are serviced by BCCH staff (pediatric nephrologist, nurse, and dietitian) who travel on a regular basis to provide outreach care. The regional clinic in Kelowna is a private practice, with one BCCH-affiliated nephrologist who works independently in the community. Given that one of the goals of the CNSCP was to standardize care for children with NS, we sought to determine whether children cared for at regional clinics had clinical characteristics and outcomes similar to those seen at BCCH. We completed an audit of the care provided at BCCH versus the regional clinics to assess fidelity to the CNSCP recommendations, that is whether care provided was consistent with the standards set by the CNSCP. Our objectives were to compare (1) demographic and clinical characteristics, (2) induction prednisone exposure, (3) relapse rates, and (4) fidelity to care recommendations of the CNSCP. We hypothesized that clinical characteristics and outcomes would be similar between the BCCH and regional clinics because of the standardized recommendations of the CNSCP.
Methods
This retrospective cohort study was conducted as part of a quality improvement audit of the CNSCP. We included patients from the BCCH Division of Nephrology clinical database, using the term “nephrotic syndrome,” who were followed in pediatric nephrology clinics in Vancouver, Surrey, and Prince George. This search included patients with a new diagnosis of NS from 2013 to 2019 inclusive. In Kelowna, patients were followed in a private clinic by a BCCH-affiliated nephrologist. Patients followed in Kelowna were identified from the nephrologist’s clinic list, which included all their patients with NS. Patients in Surrey and Prince George were seen by our regional clinic staff, including a BCCH-affiliated nephrologist. Clinical data were extracted from hospital and clinic charts and entered into a secure Excel datasheet.
An Alberta Research Ethics Community Consensus Initiative Ethics Screening Tool was completed, 17 classifying this study as a Quality Improvement and Evaluation Project with minimal risk. Ethics approval was not sought as quality improvement initiatives are exempt from Research Ethics Board review, in accordance with article 2.5 of the Tri-Council Policy Statement (TCPS 2). 18
Case Ascertainment
Children were eligible for inclusion in the study if they (1) were 1 to 17 years old; (2) presented with new-onset idiopathic NS during the study period of 2013 to 2019; (3) met the diagnostic criteria for NS (edema, proteinuria defined as ≥3+ on dipstick, >3 g/L on urinalysis, or urine protein-to-creatinine ratio ≥200 mg/mmol, and serum albumin <25 g/L); and (4) had at least 1 year of follow-up data available post-diagnosis. Children were ineligible if they (1) had induction treatment completed outside of BC; (2) were treated per the CNSCP for less than 6 months within their first year of follow-up; (3) had a known kidney anomaly or previous history of kidney disease; (4) had evidence suggestive of secondary NS; or (5) lacked sufficient clinical information, including prednisone dosing or relapse course. Children were excluded if they (1) were resistant to steroids, defined as persistence of proteinuria after 6 weeks of daily prednisone therapy from time of diagnosis; (2) had biopsy-proven evidence of non-minimal change disease; or (3) relapsed early (ie, prior to completing the initial induction prednisone course prescribed). These patients were excluded because they would require a deviation from the CNSCP’s recommended induction therapy.
Clinical Variables
We sought to characterize the clinical characteristics, practice variation, and outcomes of children with NS followed at BCCH compared to regional clinics. The clinic at which the patient received the majority of their care in the first year after diagnosis was designated their “primary clinic.” Patients who had an equal number of visits in two clinics were classified based on the location of their first visit, since the majority of CNSCP teaching is done within the first 3 months post-diagnosis. Elevated blood pressure was defined as systolic or diastolic blood pressure ≥95th percentile for age, sex, and height. 19 Clinically significant hematuria was defined as ≥20 red blood cells per high powered field; this cut-off was based on the threshold from our center’s historical practice audit. 9 Estimated glomerular filtration rate (eGFR) was calculated using the revised Schwartz formula. 20
Induction therapy was defined as the first course of prednisone treatment following diagnosis, including daily and alternate day dosing. Cumulative induction prednisone dosing was defined as the total dose of prednisone received during induction therapy in milligrams per meter-squared body surface area (BSA, mg/m2). If the patient’s BSA was greater than 1 m2, BSA was adjusted to 1 m2 for dosing and exposure calculations, as our maximum daily prednisone dose was 60 mg. In light of the demonstrated non-inferiority of shorter courses of induction prednisone (ie, 2-3 months) compared to long courses (ie, 6 months) for relapse outcomes,15,16 a second edition of the CNSCP was published in 2016; the prednisone induction was decreased from 4095 mg/m2 over 24 weeks to 3360 mg/m2 over 12 weeks. 11 In this report, we compared cumulative induction prednisone for patients who received the short induction course of prednisone, as this is the current version of the pathway that is being actively used in practice.
Clinical outcomes of interest occurring within 2 years of follow-up from diagnosis were examined for the subset of patients who received the short induction course, including: days until first relapse from end of induction therapy; proportion of patients that developed frequently relapsing NS (FRNS), defined as 2 or more relapses in the first 6 months following induction therapy or 4 or more relapses in any 12-month period; proportion of patients that developed steroid-dependent NS, defined as meeting criteria for FRNS with 2 or more relapses occurring during or within 2 weeks of stopping prednisone; and proportion of patients that required treatment with SSAs, including tacrolimus, cyclophosphamide, or mycophenolate mofetil. We also examined the annualized relapse rate, defined as the number of relapses per year over the 2 years after start of induction prednisone or until initiation of a SSA (whichever occurred earlier).
We assessed and compared fidelity to the CNSCP between BCCH and regional clinics (ie, whether care provided was consistent with the standards set by the CNSCP). The pathway care components included scheduled outpatient clinic visits, review of immunization status, relapse history, handbook worksheet review (where parents document the patient’s proteinuria, weight, and symptoms), physical examination looking for evidence of relapse or steroid side effects, formal urine monitoring for protein, review of diet and fluid recommendations, and eye exams (Figure 1). However, we restricted our analysis to the number of nephrology clinic visits in the first year, completion of a food record reviewed by a registered dietitian, and completion of an ophthalmology appointment for review of ocular complications due to steroid use. If there was no formal documentation of the activity, it was assumed as not completed.
Statistical Methods
All statistical analyses were performed using SPSS Software (IBM SPSS Statistics for Macintosh, Version 27, Armonk, New York). Descriptive statistics were expressed as mean and standard deviation for normally distributed data, and median and interquartile range (IQR) for non-normally distributed data. Categorical variables were expressed as proportions. Groups were compared using two-sided Student’s t tests, comparison of medians, and Chi-square tests. Levene’s test was used to compare differences in variance. Statistical significance was defined as P < .05.
Results
Case Ascertainment
A total of 151 patients were identified from the patient databases, of whom 33 were deemed ineligible (Figure 2). Of the remaining 118 patients with idiopathic NS, 49 (42%) were excluded due to steroid resistance, early relapse during induction, or evidence of non-minimal change disease on kidney biopsy. We included the remaining 69 patients for data analysis.
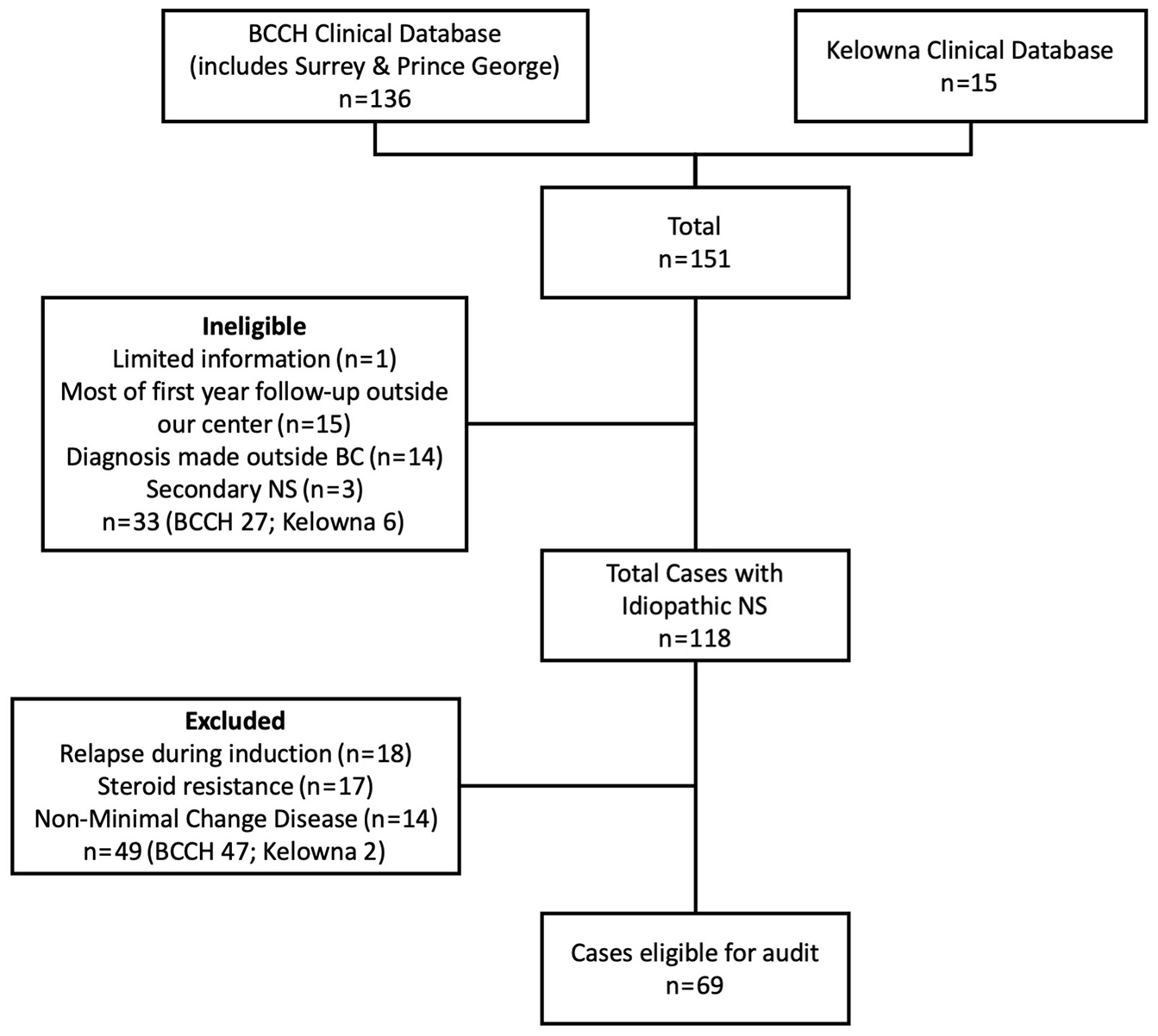
Identification of cases. Of the 151 children diagnosed with new onset NS, 118 children were managed according to the pathway and had non-primary NS.
Patient Characteristics
Of the 69 patients reviewed, 36% (25/69) were female, with a median age of 5 years (IQR 3.4-8.3; Table 1). Fifty-two patients were followed primarily at BCCH (75%), and 17 (25%) received care at one of the regional clinics. At initial presentation, 88% of patients (61/69) were initially assessed by emergency room physicians and 51% (35/69) were admitted to hospital. At the time of diagnosis, 44% (23/52) had elevated blood pressure (systolic or diastolic blood pressure ≥95th percentile), 11% (8/69) had hematuria (≥20 red blood cells per high-power field), and the mean eGFR was 128 ± 35 mL/min/1.73 m2 (calculated by Schwartz equation). Frequently relapsing NS developed in 44% of patients by 2 years after the start of induction therapy (30/69). One child had a biopsy, which demonstrated minimal change disease.
Characteristics of Children With Steroid-Responsive Nephrotic Syndrome.
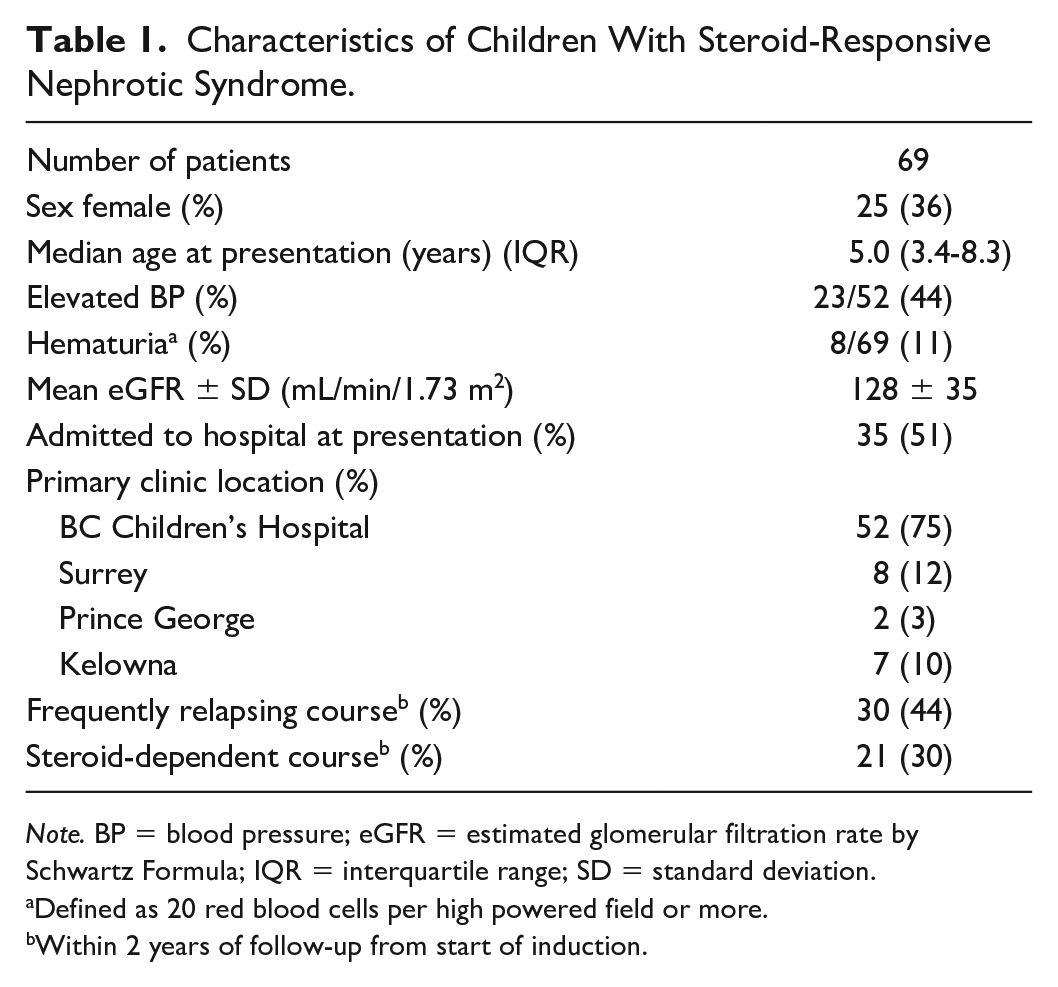
Note. BP = blood pressure; eGFR = estimated glomerular filtration rate by Schwartz Formula; IQR = interquartile range; SD = standard deviation.
Defined as 20 red blood cells per high powered field or more.
Within 2 years of follow-up from start of induction.
When the clinical characteristics at the time of initial presentation were compared between the BCCH and regional clinics (Table 2), there were no statistically significant differences in the proportion of females (37% vs 35%; P = .926), median age at presentation (5.1 [IQR 3.6-9.3] vs 4.7 [IQR 3.2-7.1] years; P = .624), proportion of patients admitted to hospital (46% vs 65%; P = .184), mean eGFR (130 ± 38 vs 125 ± 26 mL/min/1.73 m2; P = .692), proportion of patients with elevated blood pressure (41% vs 53%; P = .708) or hematuria (17% vs 8%; P = .474), or need for an SSA (44% vs 35%, P = .157). A similar proportion of patients received the 12-week induction course at BCCH (36/52; 69%) and at regional clinics (13/17; 75%; P = .568), that is were treated per the second edition of the CNSCP published in 2016. The remainder of the patients (BCCH 16/52, regional 4/17, overall 20/69; 29%) received the 24-week induction course that was initially recommended in the first edition of the CNSCP (ie, 2013-2016).
Characteristics of Children With Steroid-Responsive Nephrotic Syndrome by Clinic.

Note. BP = blood pressure; eGFR = estimated glomerular filtration rate by Schwartz Formula; SSA = steroid-sparing agent.
Defined as 20 red blood cells per high powered field or more.
Within 2 years of follow-up from start of induction.
Induction Prednisone Exposure
In the subset of patients that received the 12-week induction course (n = 49), median prednisone exposure was 3400 mg/m2 (IQR 3331-3585) for the BCCH cohort and 3492 mg/m2 (IQR 3397-3644) for the regional cohorts (P = .167; Table 3). Levene’s test indicated equal variances between the BCCH and regional cohorts (F[1,47] = 0.705; P = .405).
Outcomes of Children With Steroid-Responsive NS on 12-Week Induction Course by Clinic.

Note. NS = nephrotic syndrome; BCCH = British Columbia Children’s Hospital; IQR = interquartile range; ARR = annualized relapse rate.
From end of induction.
From start of induction to 2 years after start of induction (or start of SSA).
Relapse Metrics
Relapse metrics were calculated for patients who received the 12-week induction course. There was no significant difference in the proportion that developed FRNS comparing the BCCH (18/36, 50%) and the regional clinic cohorts (8/13, 62%; P = .475). The median annualized relapse rate over the first 2 years post-diagnosis was 3.3 (IQR 1.1-5.3) relapses per year at BCCH and 2.3 (IQR 0.5-4.2) relapses per year at regional clinics (P = .575). There were a similar number of days to first relapse from end of induction (BCCH 72, IQR 23.5-117 vs regional clinic 44, IQR 4.5-174, P = .560; Table 3).
Pathway Fidelity Metrics
In assessing the adherence to pathway recommendations, all 69 patients were included. Pathway recommendations for the first year of follow-up are outlined in Figure 1; these were determined based on best available evidence and multidisciplinary input based on the local context. While the CNSCP has many recommendations for the first year post-diagnosis, analysis was limited to clinic visits, food record review, and ophthalmology assessment due to limitations of the chart review. Patients followed at BCCH and the regional clinics had a similar number of nephrology clinic visits in the first year (mean 4.2 ± 1.2 vs 4.0 ± 1.8 visits; P = .655). Median days to first clinic visit were similar between the BCCH group and the regional group (9.5, IQR 4-29 vs 9.0, IQR 3-26; P = .945). There was no statistically significant difference in the proportion that had food records reviewed by a dietitian in the first year of follow-up (BCCH 35/52, 67%, regional clinics 8/17, 47%, P = .135) or in the median days to first food record review post-diagnosis (BCCH 30, IQR 10-61, regional clinics 46, IQR 20-78, P = .750). There was, however, a significant difference in the number of patients who were reviewed by ophthalmology in the first year post-diagnosis: at BCCH, 87% (45/52) of patients had documented ophthalmology visits, whereas 59% (10/17) of those followed at regional clinics had a documented ophthalmology visit (P = .014). For patients seen by ophthalmology, the median number of days after diagnosis to first ophthalmology appointment was 189, IQR 109 to 319; there was no difference between groups (BCCH 189, IQR 125-304, regional 143, IQR 70-351; P = .874). Of those who had ophthalmology assessments, one patient (2%) had documented steroid-related ocular pathology (bilateral subcapsular cataracts). Eighty-seven percent (48/55) had normal ocular exams, and 11% (6/55) did not have ophthalmology documentation available to review.
Discussion
This study demonstrates similar treatment and outcomes for children with newly diagnosed NS who were seen at our tertiary and regional clinics, suggesting standardized NS care throughout the province regardless of primary clinic geographic location. To our knowledge, this is the first study to describe outcomes for children with NS based on regional clinic location. Similar findings have been described in other pediatric subspecialties, for example, in type 1 diabetes care in Western Australia. 21 In general, families who live in rural regions experience barriers to care that impact health outcomes. 22 In the United States, despite an increasing number of pediatric nephrologists, 23% of children have to drive a distance of >40 miles to the nearest pediatric nephrology center. 23 By providing services that are geographically distributed, we can begin to address issues of equity and access to specialist services, reducing the difficulty and cost associated with patient travel to tertiary centers. 24 Our study reinforces the value and efficacy of the regional clinics and a standardized CNSCP in caring for children with NS.
Importantly, we have demonstrated that there was no difference in the prednisone induction treatments, relapse rates, or fidelity to our clinical pathway recommendations in the tertiary and regional clinic settings. Interestingly, of the outcomes examined, the only significant difference was access to ophthalmology follow-up, with a smaller proportion of patients in regional clinics having documentation of ophthalmology review. Studies of ocular complications in children with NS report ranges in incidence of cataract from 1% to 33% and glaucoma from 4.5% to 20%, with an association between total dose of corticosteroid and ocular complications.25,26 While review by an ophthalmologist for a baseline examination is recommended by our clinical pathway, we recognize that access to pediatric ophthalmologists is variable across the province. Continuing to engage with community eye care providers, including optometrists, pediatric ophthalmologists, and community adult ophthalmologists, will be a key component to improving this pathway-fidelity metric and ensuring children have ocular screening completed in a timely manner.
In this study, we also demonstrated that there was similar variance in prednisone dosing between the BCCH and regional clinics, which we attribute to the implementation of the CNSCP. While clinical practice guidelines have been published by multiple groups, several studies demonstrate ongoing practice variation in the management of children with NS. In a longitudinal, prospective pan-Canadian study, Rodriguez-Lopez et al 10 identified site of treatment, as well as age and race of the child to be associated with differences in induction prednisone dosing. This phenomenon has been described in many countries: surveys completed in the United States and the Netherlands demonstrate similar practice variation.27 -29 A survey of pediatric nephrologists in Canada identified several factors that contribute to practice variation: years in practice, uncertainties in evidence, anecdotal or center experience, and center-specific standardized protocols. 30 In an international survey of pediatric nephrologists, perceived complexity and physician attitudes contributed to non-adherence to clinical practice guidelines. 31 Even within our center, we identified significant variance in prednisone dosing among BCCH-affiliated nephrologists prior to the CNSCP 9 ; this is what initially prompted the development of our pathway. Historically, BCCH patients diagnosed with NS between 1990 and 2012 revealed a median prednisone induction dose of 2546 mg/m2 with a large range of dosing (IQR 2040-3091). 9 However, our current data demonstrate a median induction prednisone dosing of 3400 mg/m2 (IQR 3331-3585) at BCCH and 3492 mg/m2 (IQR 3397-3644) at regional sites, both of which are quite similar to our CNSCP recommendation of 3360 mg/m2. While we do not have site-specific data available prior to implementation of the CNSCP, our results demonstrate that physicians are adhering to the CNSCP recommendations, and that care has been standardized among patients followed at BCCH and at regional clinics. To our knowledge, our study is the first to highlight the role of a clinical pathway as a knowledge translation tool to implement international NS guideline recommendations in a local context.
The next step for fully integrating the CNSCP at the provincial level is to empower and encourage general pediatricians to utilize the CNSCP to provide evidence-based care, since pediatricians are usually the primary longitudinal care providers for NS patients. We recently conducted a survey of 47 general pediatricians in BC where 100% of respondents thought that general pediatricians should be involved in the care of children with uncomplicated NS, though many lacked confidence in some aspects of management (unpublished data). While the current study did not assess general pediatricians’ role in the care of patients with NS, we are hopeful that the CNSCP will equip pediatricians with the tools to further improve access to pathway-recommended care for children across the province. Future goals include working to further disseminate this resource to pediatricians in BC through Continuing Medical Education events.
This study adds to the relative paucity of data on the effects of clinical pathways in assessing and improving care in pediatric nephrology, 32 and highlights the complexity of pathway implementation locally and at regional sites. While there was no significant difference in the number of clinic visits patients attended in the first year between BCCH and regional sites, patients attended on average only 4 of the 6 recommended in our CNSCP. Similarly, overall food record completion rate was only 62% (BCCH 67%, regional 47%). That said, our food record completion rate was consistent to that described in the literature, with one study suggesting that living further from study centers was associated with lower rates of food record completion. 33 Despite a similar time to ophthalmology assessment between BCCH and regional clinics, both were delayed relative to CNSCP targets. Results suggest that barriers to pathway fidelity may persist in spite of providing equitable care, which signals the need for continuous improvement across all sites. Unfortunately, the retrospective nature of our study makes it challenging to assess the root cause of these issues. As the role of telemedicine has rapidly increased over the last few years, this may be one avenue for improving adherence to pathway recommendations. 34
One of the weaknesses of our study is that as a retrospective cohort study, it allows opportunity for error in the data collection process. We may have underestimated whether certain activities (clinic visits, ophthalmology appointments, food record reviews by a dietitian) were completed due to incomplete documentation. Furthermore, fidelity to other components of the pathway was not examined. Given that the CNSCP was originally intended for the management of uncomplicated NS, several patients were excluded (ie, relapse during induction, steroid resistance). Future work to broaden the scope of the CNSCP to more complicated disease phenotypes is needed. Our small sample size may have limited our ability to identify differences between the two groups. Due to the retrospective nature of our study, we were limited to the data that were already available, and our sample size was fixed by the total number of cases that met our inclusion and exclusion criteria for the study time frame and were referred to BCCH Nephrology for care. With a larger sample size, we may have been powered to detect differences in components like time to ophthalmology review and food record review, which did not meet statistical significance in our study. Another limitation of our study is that we were only able to capture patients followed by pediatric nephrology and did not include patients followed by general pediatricians without nephrology involvement. This likely overestimates the compliance to the pathway recommendations, as care was provided by nephrologists in all instances.
Our study highlights the need for an extension of the CNSCP to include more patients with atypical or complicated clinical courses of NS. Because our pathway focuses on patients with typical NS and excludes those who relapsed during induction therapy, were steroid resistant, or had non-minimal change disease identified on biopsy, we excluded 42% of the patients identified with NS from our study. This is because our CNSCP focuses only on typical NS management; those who require a deviation from the pathway’s recommended induction therapy are treated by physician preference with SSAs. More recently published KDIGO Clinical Practice Guidelines on management of glomerular diseases give recommendations for SSAs in the context of steroid resistant or frequently relapsing disease courses, including clinical tips for when to choose each SSA. 35 The next iteration of the CNSCP will include more of these complex cases and comment on available SSA options for further management.
Conclusion
In summary, in our cohort of pediatric patients with NS in BC, similar care and clinical outcomes were observed in children who were followed at our tertiary care site and our regional clinics. This highlights the role of a clinical pathway to decrease variability in patient therapy and outcomes. Discrepancies remain for children’s access to multidisciplinary care, specifically ophthalmology, in regional clinics. We hope this study serves to inform future initiatives for improving regional access to tertiary care and highlights the utility of clinical pathways in providing consistent care to children with NS.
Footnotes
Acknowledgements
The authors would like to acknowledge the patients and their families and the Clinical Pathway Development Team at the British Columbia Children’s Hospital. They would also like to thank Dr Scott Wenderfer for reviewing their manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by BC Renal.
