Abstract
Background:
Malnutrition and protein-energy wasting (PEW) are nutritional complications of advanced chronic kidney disease (CKD) that contribute to morbidity, mortality, and decreased quality of life. No previous studies have assessed the effect of oral nutritional supplements (ONSs) on patient-reported symptom burden among patients with non-dialysis CKD (CKD-ND) who have or are at risk of malnutrition/PEW.
Objective:
The objective of this study was (1) to quantify the associations between baseline nutritional parameters and patient-reported symptom scores for wellbeing, tiredness, nausea, and appetite and (2) to compare the change in symptom scores among patients prescribed ONS with patients who did not receive ONS in a propensity-score–matched analysis.
Design:
This study conducted observational cohort analysis using provincial registry data.
Setting:
This study was done in multidisciplinary CKD clinics in British Columbia.
Patients:
Adult patients >18 years of age with CKD-ND entering multidisciplinary CKD clinics between January 1, 2010-July 31, 2019 who had at least 2 Edmonton Symptom Assessment System Revised: Renal (ESASr:Renal) assessments.
Measurements:
The measurements include nutrition-related parameters such as body mass index (BMI), serum albumin, serum phosphate, serum bicarbonate, neutrophil-to-lymphocyte ratio (NLR), and ESASr:Renal scores (overall and subscores for wellbeing, tiredness, nausea, and appetite).
Methods:
Multivariable linear regression was applied to assess associations between nutritional parameters and ESASr:Renal scores. Propensity-score matching using the greedy method was used to match patients prescribed ONS with those not prescribed ONS using multiple demographic, comorbidity, health care utilization, and temporal factors. Linear regression was used to assess the association between first ONS prescription and change in ESASr:Renal overall score and subscores for wellbeing, tiredness, nausea, and appetite.
Results:
Of total, 2076 patients were included. Higher baseline serum albumin was associated with lower overall ESASr:Renal score (−0.20, 95% confidence interval [CI] = −0.40 to −0.01 per 1 g/L increase in albumin), lower subscores for tiredness (−0.04, 95% CI = −0.07 to −0.01), nausea (−0.03, 95% CI = −0.04 to −0.01), and appetite (−0.03, 95% CI = −0.06 to −0.01). Higher BMI was associated with higher overall ESASr:Renal score (0.32, 95% CI = 0.16 to 0.48 per 1 kg/m2 increase in BMI), higher symptom subscores for wellbeing (0.02, 95% CI = 0.00 to 0.04) and tiredness (0.05, 95% CI = 0.02 to 0.07). Higher baseline NLR was associated with higher overall score (0.21, 95% CI = 0.03 to 0.39 per 1 unit increase in NLR), higher symptom subscores for wellbeing (0.03, 95% CI = 0.01 to 0.05) and nausea (0.03, 95% CI = 0.02 to 0.05). In the propensity-score–matched analysis, there were no statistically significant associations between ONS prescription and change in overall ESASr:Renal (beta coefficient for change in ESASr:Renal = 0.17, 95% CI = −2.64 to 2.99) or for subscores for appetite, tiredness, nausea, and wellbeing.
Limitations:
Possible residual confounding. The ESASr:Renal assessments were obtained routinely only in patients with G5 CKD-ND and/or experiencing significant CKD-related symptoms.
Conclusions:
This exploratory observational analysis of patients with advanced non-dialysis CKD demonstrated BMI, serum albumin, and NLR were modestly associated with patient-reported symptoms, but we did not observe an association between ONS use and change in ESASr:Renal scores.
Introduction
Malnutrition and protein-energy wasting (PEW) are nutritional complications of advanced chronic kidney disease (CKD) that contribute to morbidity, mortality, and decreased quality of life.1,2 Among patients with non-dialysis CKD (CKD-ND), the prevalence of PEW is 11% to 54%. 3 Following nutritional counseling, treatment with oral nutritional supplements (ONSs) is recommended by renal nutrition guidelines for management of malnutrition/PEW. 4
We previously demonstrated in an observational study that ONS prescription was associated with improvements in nutritional parameter slopes among patients with CKD-ND who were at risk of malnutrition/PEW in British Columbia,5,6 which has a province-wide, government-funded ONS policy stewarded by dietitians. 7 Although a few previous studies of ONS therapy have assessed quality-of-life domains among patients receiving hemodialysis (HD),8-10 there are no studies assessing the effect of ONS on patient-reported symptom burden among patients with CKD-ND who have or are at risk of malnutrition/PEW.
Therefore, we performed an observational study utilizing registry data for patients with CKD-ND in BC, with the following aims: (1) to quantify the associations between baseline nutritional parameters and symptom scores for wellbeing, tiredness, nausea, appetite, in an exploratory analysis and (2) to compare the change in symptom scores among patients prescribed ONS with patients who did not receive any ONS prescription, using a propensity-score–matched analysis.
Materials and Methods
This cohort analysis included adult patients >18 years of age with CKD-ND who entered multidisciplinary CKD clinics in BC between January 1, 2010 and July 31, 2019, and who had at least 2 Edmonton Symptom Assessment System Revised: Renal (ESASr:Renal) assessments done. For patients prescribed ONS, at least 1 ESASr:Renal assessment before ONS initiation and at least 1 ESASr:Renal assessment after ONS initiation were required. For non-ONS users, they were required to have at least 2 ESASr:Renal assessments done 0.5 to 24 months apart. Data were extracted from the Patient Records and Outcome Management Information System, an electronic registry of kidney patients in BC. Patients with previous renal transplant were excluded from the analysis. Ethics approval was obtained from the University of British Columbia-Providence Health Care Institute (study number: H19-01154).
British Columbia has a provincial Nutritional Supplement Policy 7 that provides ONS free of charge for patients in whom any of the following conditions occur and cannot be addressed through nutritional counseling alone: unintentional weight loss >10% of usual body weight in the past 6 months, current weight <90% of desirable body weight, nutrient intake <80% recommended, and/or current hypercatabolic state. Oral nutritional supplement available under this policy (Supplemental Table 1) include general ONS, renal-specific ONS, diabetes-specific ONS, and protein-only ONS. Renal dietitians determine the ONS prescription based on the patient’s nutritional status, dietary intake, caloric/protein needs, comorbidities, medical status, and patient preference. Duration of ONS treatment is variable and based on regular evaluation by a renal dietitian.
British Columbia has a province-wide Systematic Symptom Assessment and Management policy that utilizes ESASr:Renal to assess symptom burden of patients followed at multidisciplinary CKD clinics who meet the following criteria: eGFR<15 mL/min/1.73 m2, and/or reporting significant CKD-related symptoms. 11 ESASr:Renal encompasses 12 symptoms: pain, tiredness, nausea, depression, anxiety, drowsiness, appetite, wellbeing, shortness of breath, pruritus, sleeping problems, and restless legs. Patients can also add and rate a 13th symptom that is not already listed. For each symptom, patients are asked to rate their experience over the past week using a scale of 0 to 10 (0 = no symptom, 10 = severe). Patients complete their own ESAS, but if they cannot independently perform this task, it is completed with the assistance of a caregiver. The ESAS can also be administered by a Kidney Care Clinic team member by telemedicine if the patient is not attending clinic in person.
Any ESASr:Renal subscore of symptom that was left unanswered by the patients was assumed to be no symptom with a score of “0.” Sensitivity analyses were performed by excluding patients with unanswered subcomponent values to validate the assumption. Missing data were observed in some nutritional parameters of interest; for example, 34.2% missingness in body mass index (BMI) and 18.3% in urine albumin-to-creatinine ratio (ACR). We assumed data were missing at random and generated 20 imputed data sets based on multivariate imputation by chained equations (MICE). All 13 baseline and post-ONS ESASr:Renal subscores, nutritional parameters of interest, and patients’ baseline characteristics were included in the imputation model.
Using baseline data within 6 months of entry to the CKD clinic, multivariable linear regression was applied to assess associations between nutritional parameters (BMI, serum albumin, serum phosphate, serum bicarbonate, NLR) and ESASr:Renal scores (overall and subscores for wellbeing, tiredness, nausea, and appetite) within each imputed data set. We selected these 4 ESASr:Renal subscores because we hypothesized they may be linked with nutritional status. The models were adjusted for age, sex, health region, estimated glomerular filtration rate (eGFR), urine ACR, cause of CKD, hypertension, diabetes, any cardiovascular disease, parathyroid hormone (PTH), ferritin, iron saturation, and hemoglobin. Estimated effects of the covariates and confidence intervals were pooled via Rubin’s rules from each imputed data set. 12
Propensity-score matching using the greedy method13-15 was used to match patients prescribed ONS with those not prescribed ONS using covariate assessment period of 12 months prior to first ONS prescription within each imputed data set. The propensity score included age, sex, eGFR, urine ACR, hypertension, diabetes, other cardiovascular disease comorbidities, BMI, serum albumin, serum phosphate, serum bicarbonate, serum PTH, iron saturation, serum ferritin, hemoglobin, NLR, health authority, date of entry to CKD clinic, time between ESASr:Renal measurements, and number of medications prescribed during the covariate assessment period. Laboratory data from the closest date to first ONS prescription were used for patients prescribed ONS. For non-ONS users, data from the date closest to the first ESASr:Renal assessment were used. The primary outcome was change in overall ESASr:Renal score, and secondary outcomes included change in ESASr:Renal subscores for wellbeing, tiredness, nausea, and appetite. For ONS users, change in ESASr:Renal was defined as the difference between the first available ESASr:Renal scores post-ONS vs baseline ESASr:Renal during covariate assessment period, and for non-ONS users, the change in ESASr:Renal was the difference between 2 consecutive ESASr:Renal scores assessed 0.5 to 24 months apart. Linear regression was used to assess the association between first ONS prescription and change in ESASr:Renal scores within each imputed data set. A pooled estimated average marginal treatment effect of ONS prescription was calculated using Rubin’s rule. All analyses were performed using SAS version 9.4 (SAS Institute, Inc, Cary, North Carolina).
Results
A total of 2076 patients with CKD-ND were included in the analysis (Supplemental Figure 1). Patients prescribed ONS were older, had lower eGFR, hemoglobin and serum albumin, and greater serum PTH compared with those not prescribed ONS (Table 1). Oral nutritional supplement users had higher baseline overall ESASr:Renal scores (median = 32.5, interquartile range [IQR] = 18-51), compared with non-users (23, IQR = 10-39), as well as higher subscores for tiredness, appetite, and wellbeing (Supplemental Table 2).
Baseline Patient Characteristics After Multiple Imputation.
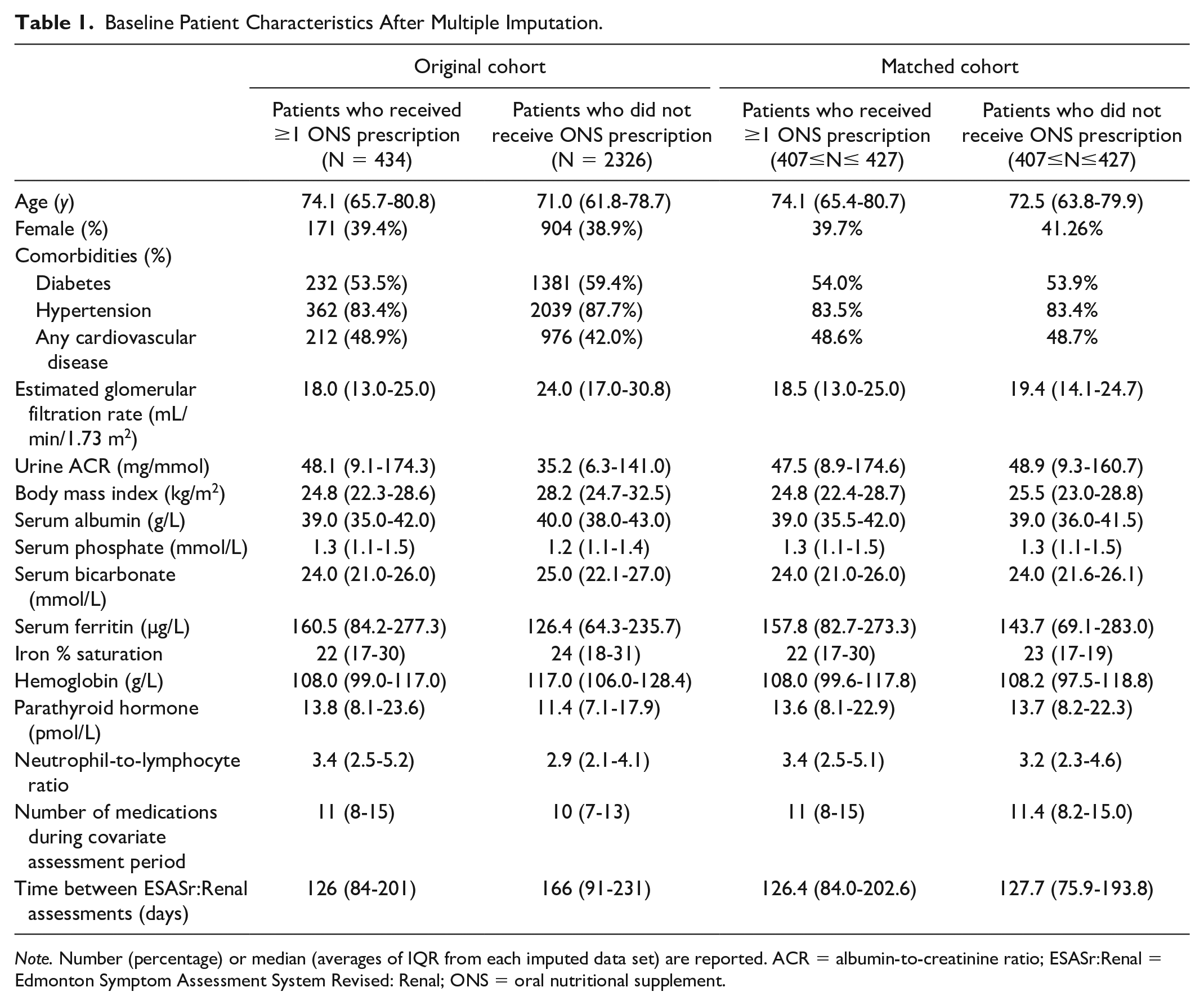
Note. Number (percentage) or median (averages of IQR from each imputed data set) are reported. ACR = albumin-to-creatinine ratio; ESASr:Renal = Edmonton Symptom Assessment System Revised: Renal; ONS = oral nutritional supplement.
Higher baseline serum albumin was associated with lower overall ESASr:Renal score (−0.20, 95% CI = −0.40 to −0.01 per 1 g/L increase in albumin), lower tiredness subscore (−0.04, 95% CI = −0.07 to −0.01), lower nausea subscore (−0.03, 95% CI = −0.04 to −0.01), and lower appetite subscore (−0.03, 95% CI = −0.06 to −0.01) (Figure 1). Higher BMI was associated with higher overall score (0.32, 95% CI = 0.16 to 0.48 per 1 kg/m2 increase in BMI) and higher symptom subscores for wellbeing (0.02, 95% CI = 0.00 to 0.04) and tiredness (0.05, 95% CI = 0.02 to 0.07). Higher baseline NLR was associated with higher overall score (0.21, 95% CI = 0.03 to 0.39 per 1 unit increase in NLR) and higher symptom subscores wellbeing (0.03, 95% CI = 0.01 to 0.05) and nausea (0.03, 95% CI = 0.02 to 0.05). Baseline serum bicarbonate and phosphate did not demonstrate statistically significant associations with the overall ESASr:Renal score or for wellbeing, nausea, tiredness, or appetite subscores.
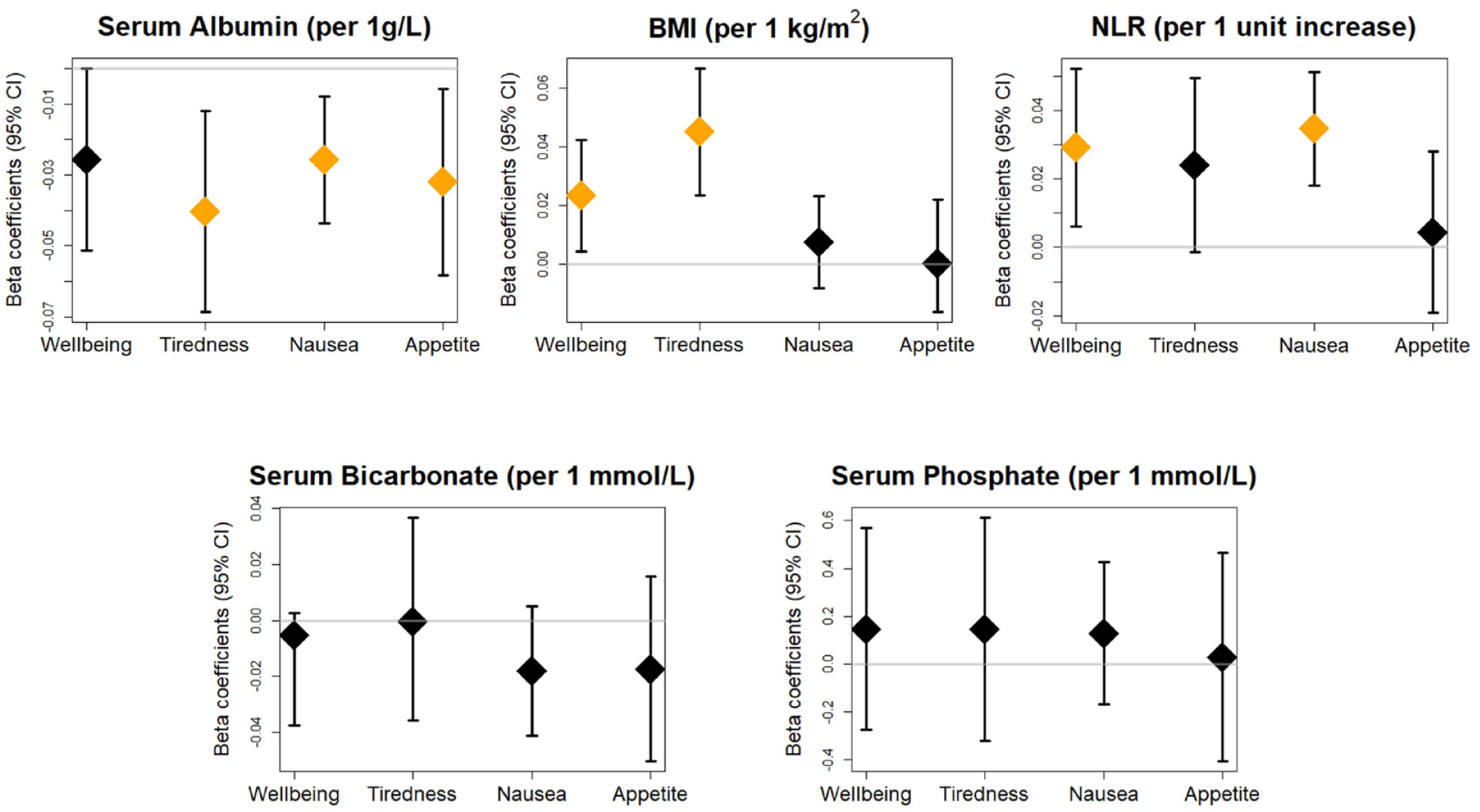
Associations between baseline nutritional parameters and patient-reported symptom burden among patients with non-dialysis CKD.
In the analysis assessing the association between ONS prescription and change in ESASr:Renal score, a range of 407 to 427 matched pairs were identified using propensity score matching over the 20 imputed data sets (Table 1). The mean change in overall ESASr:Renal score was 0.02 (95% CI = −1.71 to 1.75) among patients prescribed ONS, and −0.16 (95% CI = −2.38 to 2.06) among patients not prescribed ONS, over a median period of 127 days. There were no statistically significant associations between ONS prescription and change in ESASr:Renal for the overall score (beta coefficient for change in ESASr:Renal = 0.17, 95% CI = −2.64 to 2.99) and for subscores for appetite (−0.08, 95% CI = −0.56 to 0.39), tiredness (−0.28, 95% CI = −0.74 to 0.18), nausea (−0.18, 95% CI = −0.53 to 0.17), and wellbeing (0.18, 95% CI: −0.28 to 0.64).
Discussion
To our knowledge, this is the first study assessing the association between ONS and patient-reported symptom burden among patients with advanced CKD-ND. There were weak associations between some nutritional lab parameters and ESASr:Renal subscores, and there was no statistically significant association between ONS prescription and change in ESASr:Renal subscores.
Symptom assessment and management have been identified by patients with advanced CKD as top research priorities. 16 Among the multiple physical and psychologic symptoms experienced by patients with CKD, fatigue is one of the most common and has been established as a core outcome measure among patients receiving HD.17,18 The ESASr:Renal is a symptom assessment tool that has been validated for longitudinal assessment in patients with end-stage kidney disease. 19 Implementing ESASr:Renal in routine care has been shown to improve patient and provider symptom awareness and to empower patients to raise issues with providers. 20 Nutritional interventions, including ONS, are also an important part of symptom management among patients opting for conservative kidney management. 21
In our study, the observed associations between nutrition-related variables and ESAS scores were of small magnitude and not clinically significant. Previous studies have established that the minimal clinically important difference for each ESAS subscore is 1 point. 22 The lack of clinically meaningful associations between biochemical parameters and symptom burden has been observed in previous studies of patients receiving dialysis, with negligible correlations between changes in biochemical markers and changes in ESASr:Renal. 19 However, composite nutrition scores, such as the Malnutrition Inflammation Score and Subjective Global Assessment, have demonstrated associations with quality-of-life domains in HD23-25 and non-dialysis CKD. 26
Despite the small magnitude of the associations between nutritional parameters and ESAS scores observed in our study, the directions of the effects are biologically plausible. Higher serum albumin was associated with lower overall symptom burden and lower tiredness and nausea subscores. Hypoalbuminemia often has multifactorial causes in patients with CKD, including malnutrition, inflammation, volume overload, and comorbidities. 1 In the Chronic Renal Insufficiency Cohort, patients with more severe gastrointestinal symptoms were more likely to have hypoalbuminemia. 27 Our study also found an association between higher BMI and worse overall symptom burden, tiredness, and wellbeing. Fatigue frequently occurs in people with obesity, and this association may be due to inflammation, sleep apnea, comorbidities, and psychiatric disorders. 28 A study that assessed different dimensions of fatigue found that obesity explained a significant portion of physical fatigue symptoms, even after adjustment for depression and inflammation. 28 Higher NLR was associated with worse overall ESAS score, wellbeing, and nausea. As a marker of inflammation, NLR has been associated with mortality, cardiovascular events, uremic toxin levels, and other inflammation markers.29,30
Although no studies have assessed the effect of ONS on symptom burden in patients with kidney disease, quality-of-life outcomes have been assessed in a few previous studies. One such study was conducted among patients with CKD-ND prescribed a low-protein diet; however, patients with malnutrition/PEW were excluded, and there was no control group. 31 In the HD population, there have been inconsistent results, with some studies demonstrating improved Short Form (SF)-36 scores for physical role functioning8,10 with ONS treatment, and 1 study that did not observe changes in any SF-36 domain. 9
There are a few possible explanations for the lack of association between ONS and ESASr:Renal scores in our study. A previous analysis of prescription patterns in our population demonstrated that a majority of patients with CKD-ND prescribed ONS have infrequent use (1-2 ONS prescriptions per year). 5 The frequency of ESASr:Renal assessment in routine care (every 6 months or more frequently as indicated) may not capture effectively the short-term effects of ONS. We included time between ESAS measurements in the propensity score to account for the variability in this parameter.
Second, the effects of ONS on surrogate outcomes, such as nutritional lab parameters, may not translate directly to effects on patient-reported outcomes. Confounding pathways between the nutritional lab parameters and symptom burden may exist, and alternative pathways between the ONS intervention and symptom burden are also possible. 32 In addition, the multifactorial nature of CKD-related symptoms suggests that ONS alone may not be sufficient to alleviate symptoms. For example, fatigue may be caused by several factors, including depression, decreased oxygen delivery, decreased muscle mass/function due to metabolic derangements (acidosis, hyperphosphatemia, PEW), and sarcopenia. 18
Finally, the ESASr:Renal outcome may not be a sensitive indicator of patient symptoms related to nutritional status. It is possible that other outcomes related to quality of life, such as SF-36 which has been studied in nutritional intervention trials8-10 and the nutrition-specific quality-of-life instrument (NSQOL) which focuses on appetite and food enjoyment,26,33 may better capture these patient-reported outcomes. This highlights the importance of the Standardized Outcomes in Nephrology (SONG) initiative in developing core outcomes of importance to patients with respect to CKD complications such as malnutrition and PEW.
One of the strengths of the study is the inclusion of a diverse patient population using a large provincial registry and standard protocols for management of malnutrition/PEW and assessment of symptom burden for patients with advanced CKD-ND. One limitation of our study is possible residual confounding. Owing to lack of registry data regarding nutritional scoring systems, such as subjective global assessment and malnutrition-inflammation-score, our analysis of nutritional status was limited to BMI and laboratory data. However, BMI is often difficult to interpret in patients with CKD due to edema or muscle wasting. Other laboratory parameters such as prealbumin, and other tools measuring body composition, such as bioimpedance or handgrip strength, may allow a more precise portrait of the nutritional state. However, these tools are not used by BC Renal dietitians in routine practice. Future prospective research is necessary to measure these parameters. The registry does not collect data on patient adherence to taking ONS. Our study is limited to the subset of patients who completed ESASr:Renal assessments as per provincial guidelines (G5 CKD-ND and/or experiencing significant CKD-related symptoms) 11 and therefore cannot be extrapolated to all patients with CKD prescribed ONS.
In conclusion, this exploratory observational analysis of patients with advanced non-dialysis CKD demonstrated that nutrition-related parameters are associated with patient-reported symptoms, but we did not observe an association between ONS use and ESASr:Renal scores. Consensus surveys and workshops involving patients and other stakeholders can aid in determining optimal outcome measures to assess patient-reported symptom burden and functional status among patients with CKD and malnutrition/PEW. This process will then facilitate assessment of the effectiveness of ONS in prospective studies.
Supplemental Material
sj-docx-1-cjk-10.1177_20543581241228731 – Supplemental material for Oral Nutritional Supplement Prescription and Patient-Reported Symptom Burden Among Patients With Late-Stage Non-Dialysis Chronic Kidney Disease
Supplemental material, sj-docx-1-cjk-10.1177_20543581241228731 for Oral Nutritional Supplement Prescription and Patient-Reported Symptom Burden Among Patients With Late-Stage Non-Dialysis Chronic Kidney Disease by Michelle M. Y. Wong, Yuyan Zheng, Bingyue Zhu, Lee Er, Mohammad Atiquzzaman, Alexandra Romann, Dani Renouf, Zainab Sheriff and Adeera Levin in Canadian Journal of Kidney Health and Disease
Footnotes
Acknowledgements
The authors acknowledge Ognjenka Djurdjev, BC Renal, the Patient Records and Outcome Management Information System team, the BC Renal Dietitians Group for their assistance and input with this project.
Ethics Approval and Consent to Participate
This study was approved by the research ethics board at the University of British Columbia-Providence Health Care Institute (H19-01154).
Consent for Publication
All authors provided their consent for publication.
Availability of Data and Materials
The de-identified data underlying this article will be shared on reasonable request to the corresponding author.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.M.Y.W. has a consultancy agreement with George Clinical.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Michael Smith Health Research BC (M.M.Y.W., grant number 18465).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
