Abstract
Background:
Most patients with end-stage kidney disease (ESKD) appreciate the importance of exercise and would like to increase their physical activity; however, they report a few key barriers, including (1) lack of physician advice to do so, (2) lack of safe and convenient programs (ie, appropriate for home or neighborhood), and (3) cost. Importantly, patients indicated in a previous survey that they would prefer an exercise program that improves muscle strength and symptoms, and are less interested in cardiovascular disease prevention.
Objective:
To test the feasibility of a simple, prescribed exercise program using Nordic walking poles in patients with ESKD treated with dialysis.
Design:
Randomized multicenter pilot trial of an exercise intervention that includes Nordic walking poles, personalized physician exercise prescriptions, pedometers, and access to exercise videos, compared with standard of care, in patients being treated with maintenance dialysis.
Setting:
Multicenter tertiary care centers in Canada.
Patients:
Ambulatory adult patients with ESKD treated with peritoneal dialysis or hemodialysis (HD) for at least 6 months at participating sites are potentially eligible. Inclusion criteria include ability to use Nordic walking poles (either de novo or in place of mobility aid) and to provide informed consent in English or in French. Exclusion criteria include (1) any absolute contraindication to exercise, (2) baseline step count >8000 steps/day, (3) planned living donor kidney transplant, and (4) participation in another interventional trial that may affect the results of this study.
Methods:
This is a randomized multicenter pilot trial of an exercise intervention that consists of a prescription to exercise using Nordic walking poles, a pedometer to track activity, and access to exercise videos, with the comparator of standard of care (dialysis unit staff encouragement to exercise) in patients being treated with maintenance dialysis. Randomization is concealed and uses a 1:1 ratio for group assignment. Our specific aims are to determine the feasibility of patient recruitment, adherence to the exercise program (verified by step counts), and efficacy of the intervention on patient-important outcomes that were assessed as a priority by patients in a prior survey—specifically strength, fatigue, and sleep. We record days spent in hospital and loss of independent living to inform sample size calculations for a definitive trial of exercise in patient with ESKD treated with dialysis. Adverse events are closely monitored.
Outcomes:
Primary: Our recruitment goal is 90 to 150 patients over 27 months; adherence success will be defined if >75% of randomized patients, excluding those who are transplanted or deceased, achieve >80% of their prescribed steps at 6 and 12 months. Secondary Efficacy Outcomes: (1) strength—hand grip strength and 5 times sit to stand, (2) energy—Short Form (SF)-36 vitality subscale, and (3) sleep—Pittsburg Sleep Quality Index will be assessed at baseline, 6, and 12 months.
Results:
Trial recruitment started before the COVID-19 pandemic and the pandemic led to many interruptions and delays. Online exercise Web sites and a tailored video were added to the protocol to encourage activity when participants were unable or reluctant to walk in public places.
Limitations:
This trial was designed to include ambulatory patients with ESKD and does not address the burden of disease in patients with very restricted mobility.
Trial Registration:
NCT03787589.
Background
Why Is a Trial Needed Now?
Patients with end-stage kidney disease have high symptom burden and reduced quality of life
Most patients with end-stage kidney disease (ESKD) receive 3 times weekly in-center hemodialysis (HD) or home peritoneal dialysis (PD). Dialysis patients report their quality of life as significantly poorer than the general population,1,2 with symptom burden similar to hospitalized cancer patients. 1 ,3-5 While fatigue is most prevalent (weighted mean prevalence 71% in a systematic review), many patients had 6 to 10 additional disabling symptoms, including weakness, insomnia, pain, anorexia, difficulty concentrating, shortness of breath, decreased sexual function, cramps, dizziness, anxiety, and depression (weighted mean prevalence 35%-55%). 6 Symptom burden at baseline and changes over time are associated with all aspects of health-related quality of life (HRQOL) and explain 40% to 50% of the variation in the mental and physical health components of the Short Form (SF)-12. 5 In 2 scoping exercises used to define research priorities for ESKD patients, 2 of the top 10 priorities were symptom reduction and ways to enhance self-management.7,8
Loss of function after dialysis initiation predicts fracture, hospitalization, and mortality
After dialysis initiation, patients suffer significant declines in muscle strength and function. In nursing home residents with ESKD, declines in ability to complete activities of daily living occurred as early as 3 months after dialysis start. 9 After 12 months, 58% of patients had died and only 13% had maintained their pre-dialysis functional status. Among prevalent elderly dialysis patients, >50% require assistance with personal care with further functional declines during hospitalization. 10
Reduced strength and functional decline have serious consequences. Frailty in dialysis patients is associated with a 2-fold higher risk of first fall or fracture than non-frail dialysis patients, carrying with it a 2.5 times greater mortality risk than in the general population.11,12 While several risk factors for falls have been identified, loss of muscle strength is a major modifiable risk factor. Improvements in muscle strength and endurance are associated with significantly lower fracture risk in ESKD (odds ratio [OR] = 0.10-0.30). 13 Functional impairment is also associated with increased risk of hospitalization and early mortality in patients with ESKD.14-16 The observational nature of these studies prevents us from drawing firm conclusions about causality, but the impressive findings highlight the need to test interventions with the potential to reduce frailty in ESKD. Exercise may reduce the risk of becoming frail, thereby reducing hospital admissions and prolonged length of stay related to loss of functional independence.
Exercise improves mortality, hospitalizations, and fractures in other populations
Evidence suggests that exercise improves cardiovascular risk and mortality in non-dialysis patients. A recent meta-epidemiological study of 305 randomized controlled trials with 339 274 participants without ESKD found that exercise has similar effectiveness to drug interventions for the secondary prevention of coronary heart disease and mortality. 17 Exercise was more effective than any drug intervention at reducing the risk of death after a stroke. For patients with established cardiovascular disease, participation in exercise-based cardiac rehabilitation decreases total and cardiac mortality by 20% to 26% as well as hospitalizations.18,19
Exercise has also been shown to be of benefit in fall and fracture prevention. In a meta-analysis that included 19 478 patients, any type of exercise as a single intervention reduced the rate of falls in community-dwelling older people by 21%. 20 Programs that challenge balance and are of higher dose (>3 h/wk) had the largest effects. 20 Furthermore, in an epidemiologic observational study of 8600 postmenopausal women and 5049 men, the age-adjusted relative risk for hip fracture associated with exercise was 0.6 and 0.5 for females and males, respectively. 21
Physical function and symptoms are of higher priority to ESKD patients than other outcomes
Cardiovascular disease and mortality may not be the most important outcomes to patients. Using a questionnaire, developed with the assistance of the Champlain LHIN Regional Renal Patient and Family Council, we surveyed 424 dialysis patients. 22 They were asked to select the 3 most important benefits they would want for themselves if they were to begin an exercise program. Overwhelmingly, dialysis patients selected improvements in energy, strength, and sleep (Figure 1). From a policy perspective, the potential to reduce need for acute or long-term institutional care are clearly important objectives but were deemed less important by patients. An appropriately designed and implemented exercise program may mitigate some of the symptoms associated with ESKD and enhance participation. Low-intensity exercise has been shown to have beneficial effects on energy, difficulty with sleep, and physical functioning.23,24
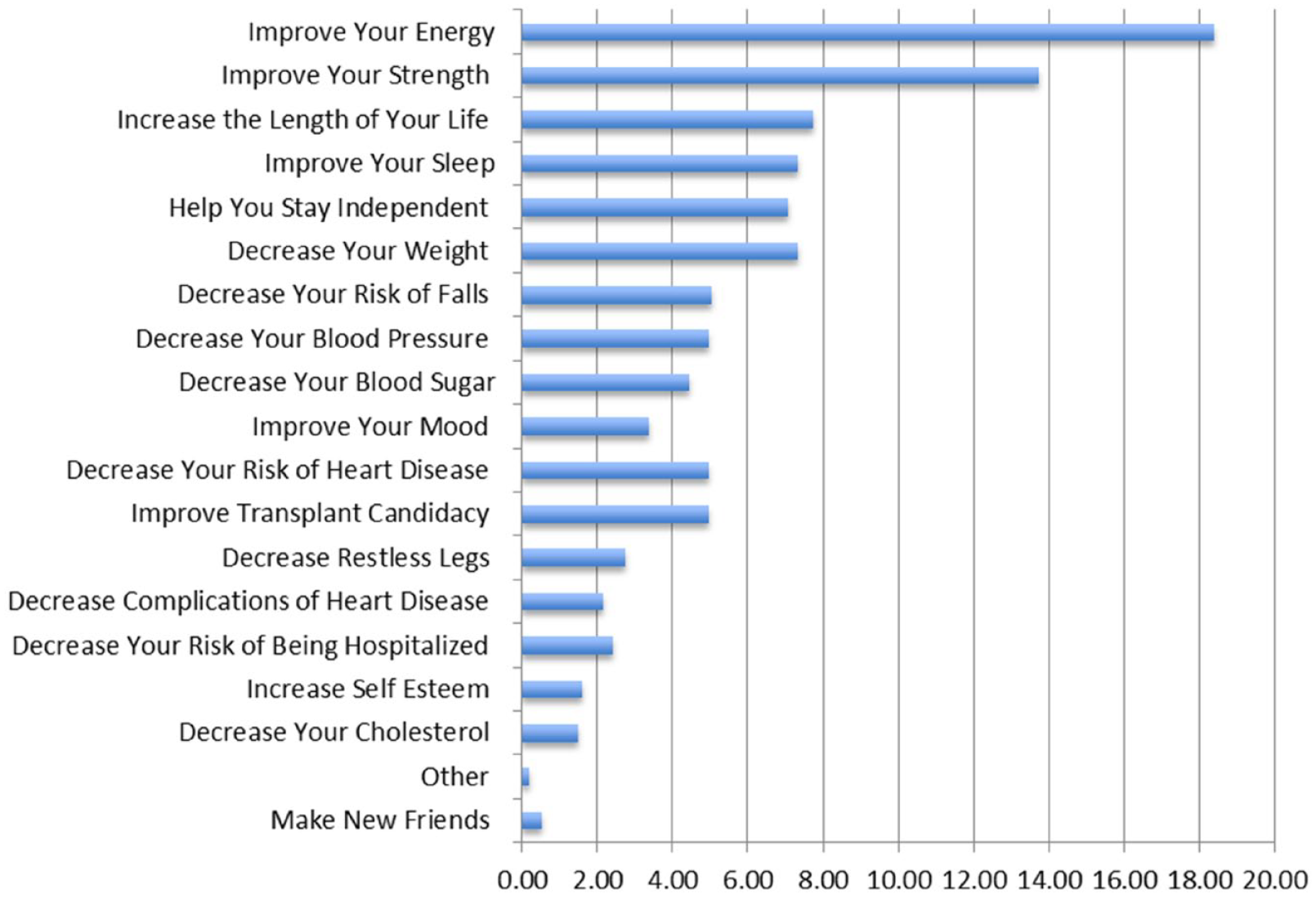
Desired exercise outcomes from our patient survey.
Patients with ESKD identify many barriers to exercising
In spite of the potential benefits of exercise, patients with ESKD have been found to be less active than sedentary individuals in the general population. 25 One study found ambulatory patients new to dialysis had physical activity scores that were below the 5th percentile of healthy individuals for all age and gender categories. 26 Only 44% of patients reported being able to climb a flight of 12 steps non-stop; just 56% could walk one block non-stop. In a study of 202 Japanese HD patients, the average number of steps on non-dialysis days was 3925 (interquartile range [IQR] = 2287, 6244); 99% of patients were only engaged in light-intensity physical activities. 27 A 10-minute/day increase in activity was associated with a 22% decrease in all-cause mortality. In a study of US HD patients, 56% reported exercising less than once per week. 28 Similar results were reported using international data. 29 Regular exercisers had higher HRQOL, physical functioning, and sleep quality scores (P ≤ .0001 for all), and a 24% lower hospitalization risk from fractures. 30
One reason patients with ESKD do not exercise may be due to severe limitations to vigorous or moderate physical activity reported by 75% and 42% of patients, respectively. 31 Also, the majority of nephrologists do not assess or recommend physical activity to their patients; citing the belief that patients would not increase their activity level if advised to do so. 32 However, a survey of US dialysis patients suggested 93% were interested in and would exercise if counseled to do so by their physician. 33 They also identified barriers including a lack of places to exercise and cost associated with the use of fitness facilities. 33 In our 424 patient survey, we found similar results. Our patients identified the key barriers to exercising were: (1) symptom burden (mostly fatigue, 55%), (2) concern about the risks of exercise (53%), (3) lack of ability to design an appropriate and safe exercise program (42%), and (4) cost (27%). More than 75% of patients stated they would exercise if a physician suggested it. In contrast with the hospital-based exercise interventions most studied to date with HD patients, the majority of our patients stated that they would prefer to exercise at home or in their neighborhood; less than 10% of respondents were interested in exercising in the HD unit. 22
Previous studies of exercise in dialysis patients have had limitations
Studies examining the benefits of exercise in dialysis patients are few. A Cochrane Review concluded that exercise was beneficial for most patients with chronic kidney disease, but most studies did not include dialysis patients. 34 Two more recent systematic reviews of exercise studies in patients with ESKD demonstrated that most studies were single center with a small number of non-representative participants, included only HD and not PD patients, were of short duration (6 months or less), and often did not study patient identified important outcomes.35,36 Recruitment rates and participant adherence to the prescribed exercise protocol tended to be low. The most common intervention was stationary cycling during HD. These in-center programs of supervised exercise training are resource intensive, associated with unit work-flow challenges in an already busy dialysis unit, are not superior for improving strength or daily physical activity compared to pedometers, and are not well accepted by patients (consistent with our survey).37-39 Furthermore, they necessarily exclude patients receiving HD or PD at home.
In the largest randomized trial of exercise in ESKD to date (N = 296), patients were encouraged to walk at home for 6 months in synch with a metronome beat. The number of steps per minute exercise prescription was based on their baseline 6MW test. 40 There was a statistically significant increase in the 6MW distance and 5 times sit-to-stand test. Hospital-free survival was greater but only for patients who completed the study (P = .04). 40 This study had notable limitations. Most importantly, it is difficult to know how to adopt a home-based walking prescription that is based on baseline 6-MW distance, a test that is not normally performed in clinical practice. Furthermore, patients were required to use a metronome, which is not practical outside of the home. Finally, the study was underpowered to show a statistically significant improvement in hospitalizations in an intent-to-treat analysis, making cost-effectiveness of the intervention unclear.
Use of Nordic walking poles is a safe, simple, and acceptable way of increasing daily activity
The tremendous burden of co-morbid disease and frailty in patients with ESKD mandates a different type of exercise program. Patients with ESKD in our survey indicated they were interested in both aerobic and resistance exercise, provided it was safe, improved their symptoms, and could be done anywhere at their own convenience. An exercise program using Nordic walking poles would meet these aims. Nordic walking poles simultaneously condition the upper and lower body similar to rehabilitative programs that incorporate both aerobic and resistance training, can be used by patients experiencing instability (including those requiring mobility aids) or muscular weakness, and have been found to be superior to brisk walking alone on resting heart rate, blood pressure, exercise capacity, maximal oxygen consumption, and quality of life.41,42 Furthermore, they can be used anywhere and are inexpensive. They have been found to be safe and easy to use with excellent (98%) long-term compliance (12 months) in a home walking program. 43
Although their rehabilitative potential has not been explored in patients with ESKD, members of our team have demonstrated positive results in a pilot study of patients with congestive heart failure and in a randomized controlled trial of patients with coronary artery disease.42,44 They showed that functional capacity, self-reported physical activity, and grip strength improved more with Nordic pole walking compared to standard cardiac rehab. 42 They also showed improvements in quality of life. 44 Nordic pole walking has also been shown by others to be beneficial in Parkinson’s disease and peripheral arterial disease.43,45
Simple ways to increase exercise behavior
Despite the overwhelming evidence of benefit to increasing activity, changing behavior is difficult. In a randomized trial of 491 sedentary individuals comparing an exercise prescription and verbal advice to exercise to verbal advice alone, the proportion of participants who increased their activity was significantly higher in the exercise prescription group as was the amount of time each individual engaged in activity compared with baseline. 46 The use of step counters such as pedometers and accelerometers has also been associated with increases in physical activity including in patients with ESKD.47,48 For these reasons, we believe that a successful ESKD exercise program should incorporate a specific, individualized exercise prescription, a method to measure step counts, and verbal advice to exercise.
Methods
What Is the Proposed Trial Design?
After receiving REB approval (20180789-01H), we will conduct a 1:1 randomized multicenter pilot trial of an exercise intervention versus standard of care in patients receiving chronic dialysis. Our specific aims will be to determine feasibility of patient recruitment, adherence to the exercise program, and efficacy of the intervention on patient-important outcomes. The intervention will consist of a prescription to exercise using Nordic walking poles and a pedometer to track activity compared with standard of care.
What Are the Planned Trial Interventions?
After obtaining signed informed consent, demographic information, comorbidities, laboratory tests, and current living situation (living independently, living with assistance, nursing home) will be collected from the patient’s medical record and supported by patient interview.
All participants will wear a pedometer for 2 weeks to determine their baseline steps per day (average of the highest 6 days) and divided into levels of activity (level 1: <3000, level 2: 3000-5000, level 3: >5000 steps per day). For participants randomized to the intervention group, the research coordinator (certified in the use of poles) will instruct the participant on the proper use of the Nordic walking poles. All participants in the intervention arm will be encouraged to wear the pedometer throughout the trial to monitor adherence and follow the step-count prescription (Figure 2). The baseline steps will be increased by 1200 steps, 1600 steps, and 2000 steps at least 3 days per week in levels 1, 2, and 3, respectively, over the first 3 months. If tolerated and accepted, they will increase their step count by an additional 600, 800, and 1000 steps per day at least 3 days per week in each of the 3 levels for the next 3 months followed by a 6-month maintenance phase. Prescription refills with the step goals will be written every 3 months. To increase adherence and applicability of the exercise program, participants will be encouraged to use their poles anywhere they wish, including in the home, in the neighborhood (patients will be provided with a list of indoor facilities that they can use during poor weather), or on the way into the dialysis unit.

Exercise prescription.
What Are the Proposed Practical Arrangements for Allocating Participants to Trial Groups?
Baseline measures will be entered by the local coordinator into an electronic database (housed at Ottawa Hospital Research Institute) accessible by the world-wide web. A concealed computer-generated randomization scheme using random numbers will allocate participants to the intervention or control group in a 1:1 ratio, stratified for center and baseline step count (levels 1-3).
What are the proposed methods for protecting against sources of bias?
Blinding of participants is not possible. However, to minimize bias, assessors (those administering the physical function tests and HRQOL questionnaires) will ideally be different than the main study coordinator and blinded to group. In the case that multiple coordinators are not available, all coordinators will be trained to remain as objective as possible and not lead or encourage subjects during assessments. Single assessors versus multiple assessors will be tracked for information purposes on the case report forms.
Over time, participants in the intervention group may continue to walk (and thereby have the prescribed number of steps per day) but decide not to use the Nordic walking poles. Control participants may choose to buy their own Nordic walking poles. To detect these “crossovers,” we will question participants from both groups each month about pole use and report this information with the study results.
There is a small possibility that this trial may cause increased interest and possibly participation in exercise in both groups. These effects are unlikely but if present, are likely to be short term. To reduce the risk of augmenting Hawthorne effects, we have chosen to use pedometers in the control group only for the 2-week periods at baseline, 6, and 12 months to measure their step counts. The 2-week period was chosen as artificial increases in step counts by control participants when they are given the pedometers are unlikely to be maintained for a full 2 weeks.
Finally, given that participants on HD may have variable responses to physical function tests and HRQOL questionnaires depending on timing in relation to their dialysis session, all tests for HD patients will be administered pre-dialysis during a mid-week treatment.
What are the planned inclusion and exclusion criteria?
All ambulatory adult patients with ESKD treated with PD or HD for >6 months at participating sites will be approached. Inclusion criteria are able to (1) understand English or French, (2) use Nordic walking poles, and (3) provide informed consent. Exclusion criteria are (1) any absolute contraindication to exercise, (2) baseline step count >8000 steps/day (average of 6 highest days over 2 weeks), (3) planned living donor kidney transplant, (4) potential for recovery of renal function, (5) patients who feel unsafe using Nordic walking poles in place of their mobility aid, and (6) participation in another interventional trial that may affect the results of this study. Numbers of patients screened and reasons for exclusion will be recorded in aggregate. Our eligibility criteria are broad to improve generalizability and maximize recruitment.
What is the proposed frequency and duration of follow-up?
Participants will be followed from randomization until they are transplanted, die, or study completion. This will allow tracking of event rates and inclusion of up to 150 participants in the larger definitive trial. All participants will have a detailed baseline assessment. Tests of physical function and the quality-of-life questionnaires will be administered at baseline, 6, and 12 months. The 6-month assessment is to minimize missing data due to death in this high-risk population. End point outcomes will be assessed monthly.
What Are the Proposed Outcome Measures?
The 5 times sit to stand test requires lower extremity strength and is an important predictor of falls in the elderly. 50 It is also responsive to change: in the 6-month walking trial in patients with ESKD, mean speed improved as did the number of patients able to complete any repetitions in the intervention group. 40 Patients are instructed to move from a sitting to standing position from a 42- to 45-cm high chair as quickly as possible; time is recorded in seconds.
What is the planned recruitment rate? How many centers are involved? Timelines?
There are more than 1500 dialysis patients currently treated at the 4 participating centers for this feasibility study. We anticipate that 60% of patients with ESKD will be eligible to participate, 40 and 30% of those eligible would consent to be randomized. Initial recruitment will start at the Ottawa Hospital and then be rolled out to the other sites (Montreal, London, and Toronto) once we have addressed any unique issues in the ESKD population compared with the congestive heart failure population. 42
Are there likely to be any problems with compliance?
We have attempted to improve upon previous studies of exercise in ESKD that have had long-term adherence problems by designing a trial consistent with patients’ wishes. In our survey, most patients indicated they were not interested in exercising in the dialysis unit but wanted a program that they could follow in their home or neighborhood. Furthermore, unlike previous studies of vigorous exercise, our program entails mild and tolerable increases in activity with individualized, attainable goals. Ease of use of the Nordic walking poles, ability to incorporate the program into usual daily activities, and unique inclusion of step counters should enhance participation rates. All participants will keep their pedometers after completion of the 12-month follow-up visit.
What is the likely rate of loss to follow-up?
Loss to follow-up is <1% per year in dialysis patients as they require life-sustaining therapy. Approximately 10% of patients are expected to die before completion of one 6-month evaluation, but 76% of these patients will be admitted in the last month of life and counted in the hospitalization totals for the definitive trial. 66 Each year, about 4% of patients will receive a renal transplant; we will censor at the time of transplant as this outcome and prolonged hospitalization post-transplant are likely to be independent of the intervention. 67
What is the proposed type of analyses?
The numbers of patients receiving dialysis, screened, and eligible, and reasons for nonparticipation and exclusion will be assessed at each center in aggregate. Recruitment rates will be calculated per center per month. Adherence will be assessed in real time every 3 months during the trial by evaluating the proportion of patients in the treatment group who are achieving an average of at least 80% of their prescribed steps per day over 2 weeks. The final adherence assessment will be based on the proportion of patients who achieve >80% of their prescribed steps per day over 2 weeks at 12 months. At study end, baseline variables will be analyzed with chi-square and Student t test, as appropriate to describe the population. We will remain blinded to the outcomes so that we can roll our participant results into the larger trial. We will calculate aggregate rates to facilitate adjusting our sample size target, but we will not look at these outcomes by group. If the study is deemed not to be feasible, we will publish summaries of our outcomes but since we will not be powered to detect differences, we will not conduct formal hypothesis testing.
Trial Progress
More than 100 patients have been recruited across 3 sites with an additional site starting recruitment in May 2023; the initial protocol included 3 sites and a 15-month recruitment window. However, the COVID-19 pandemic had a significant impact on the ability to recruit and perform some of the follow-up visits. The initial participants in the intervention group were invited to join anonymous social media groups by level of activity and facilitated group sessions to address barriers to exercise; this was ultimately abandoned due to a lack of interest partially related to stalls in recruitment, small participant numbers at any point in time, and anonymity that negates the benefit of social support. 68
Additional challenges created by the COVID-19 pandemic including restricted use of indoor spaces such that participants in the intervention group are also being provided with access to additional self-help exercise videos (including one specifically designed to be used in the home with Nordic walking poles, https://www.barbgormley.com/free-on-demand-fitness-classes-with-walking-poles-1). An activity log detailing use of the exercise videos or other forms of exercise was added to the protocol.
Conclusions
Despite its comprehensiveness, we believe this program will be simple to implement. Our program is safe and can be used by patients with functional impairment or instability; is individually tailored to patient ability; incorporates both aerobic and resistance training; can be done anywhere; requires minimal training to prescribe; and it is not costly. Our major objective is to test whether this simple, prescribed exercise program can be easily adopted by dialysis programs and patients, is sustainable, and cost-effective.
Footnotes
Ethics Approval and Consent to Participate
This study was approved by the Ottawa Health Science Network Research Ethics Board (20180789-01H).
Consent for Publication
All authors reviewed and approved the final version of the manuscript and provided consent for publication.
Availability of Data and Materials
As this is a protocol for a research study, no data is currently available.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Canadian Institutes of Health Research (Funding Reference Number 159737).
