Abstract
Background:
Individuals with kidney disease are at a high risk of bleeding and as such tools that identify those at highest risk may aid mitigation strategies.
Objective:
We set out to develop and validate a prediction equation (BLEED-HD) to identify patients on maintenance hemodialysis at high risk of bleeding.
Design:
International prospective cohort study (development); retrospective cohort study (validation).
Settings:
Development: 15 countries (Dialysis Outcomes and Practice Patterns Study [DOPPS] phase 2-6 from 2002 to 2018); Validation: Ontario, Canada.
Patients:
Development: 53 147 patients; Validation: 19 318 patients.
Measurements:
Hospitalization for a bleeding event.
Methods:
Cox proportional hazards models.
Results:
Among the DOPPS cohort (mean age, 63.7 years; female, 39.7%), a bleeding event occurred in 2773 patients (5.2%, event rate 32 per 1000 person-years), with a median follow-up of 1.6 (interquartile range [IQR], 0.9-2.1) years. BLEED-HD included 6 variables: age, sex, country, previous gastrointestinal bleeding, prosthetic heart valve, and vitamin K antagonist use. The observed 3-year probability of bleeding by deciles of risk ranged from 2.2% to 10.8%. Model discrimination was low to moderate (c-statistic = 0.65) with excellent calibration (Brier score range = 0.036-0.095). Discrimination and calibration of BLEED-HD were similar in an external validation of 19 318 patients from Ontario, Canada. Compared to existing bleeding scores, BLEED-HD demonstrated better discrimination and calibration (c-statistic: HEMORRHAGE = 0.59, HAS-BLED = 0.59, and ATRIA = 0.57, c-stat difference, net reclassification index [NRI], and integrated discrimination index [IDI] all
Limitations:
Dialysis procedure anticoagulation was not available; validation cohort was considerably older than the development cohort.
Conclusion:
In patients on maintenance hemodialysis, BLEED-HD is a simple risk equation that may be more applicable than existing risk tools in predicting the risk of bleeding in this high-risk population.
Introduction
Bleeding is a common diagnosis requiring hospitalization in patients with end-stage kidney disease often requiring invasive testing, interventions, and/or blood product transfusion.1 -4 In older patients with end-stage kidney disease, 1 in 7 patients will experience a major bleeding event within 3 years of dialysis initiation, with an incidence rate of 52.6 per 1000 patient years from the international Dialysis Outcomes and Practice Patterns Study (DOPPS). 5 Multiple factors contribute to the high bleeding risk in dialysis patients, including (but not limited to) disorders of primary hemostasis (uremic platelet dysfunction), secondary hemostasis (reduced or abnormal coagulation factor activity), vessel wall disruption (vascular access needling or placement), and antithrombotic medication use (during the hemodialysis procedure as stroke prophylaxis with atrial fibrillation [AF]).6 -8
Several bleeding scores, developed and validated in the non-dialysis population, are used to identify patients at high risk of bleeding with varying degrees of accuracy and applicability.9 -12 However, a number of commonly used prediction models, such as the Hypertension, Abnormal kidney and liver function, Stroke, Bleeding, Labile INR, Elderly and Drugs or Alcohol (HASBLED), the Anticoagulation and Risk factors in Atrial Fibrillation (ATRIA), and the Hepatic or kidney disease, Ethanol abuse, Malignancy, Older age, Reduced platelet count or reduced platelet function, Hypertension, Anemia, Genetic factors, Excessive fall risk and Stroke (HEMORR2HAGES), did not include individuals receiving chronic dialysis and as such demonstrate questionable prediction in patients with advanced chronic kidney disease (CKD) and on dialysis. 4
As the risk of bleeding is high in long-term patients on dialysis with multiple factors unique to this population, it follows that a dialysis-specific risk equation may improve on existing risk scores. Furthermore, a risk algorithm may aid in clinical decision making such as initiation of anticoagulation in AF. As such, we set out to develop, validate, and compare a novel risk equation to predict bleeding events in patients on chronic hemodialysis (BLEED-HD) using the international DOPPS prospective cohort study.
Methods
Study Population
The DOPPS is an international prospective cohort study that collects information on adults (≥18 years) on maintenance, in-center hemodialysis in over 20 countries. 13 The current study included data from 53 147 patients participating in DOPPS phase 2-6 (2002-2018, Supplementary Table 1). The DOPPS uses a 2-stage stratified random cluster sample where facilities are randomly sampled within country-specific strata followed by a random selection of 20 to 40 patients within each facility. 13 The DOPPS collects detailed information on individual patients’ demographics, comorbid illnesses, medications, laboratory values, dialysis characteristics, hospitalizations, and mortality. Details on the DOPPS study design and methodology can be found elsewhere. 13 Regional ethics board approval was obtained for this de-identified retrospective study by the Ottawa Hospital Regional Ethic Board.
Study Design
Development of the risk equation required selection and identification of factors that were associated with bleeding events requiring hospitalization, creating a parsimonious model, and evaluating the model operating characteristics. Model validation was performed externally, in an independent dataset (the Canadian Organ Replacement Registry, CORR) linked and housed at ICES. 14 Finally, BLEED-HD was compared to existing bleeding risk models (HAS-BLED, ATRIA, HEMORR2GHAGE) in terms of discrimination and calibration.
Predictors
We created a large list of candidate list predictor variables a priori based on previous literature, clinical plausibility, and use in other bleeding risk scores.1 -3,7,15,16 Variables included demographics (sex, age, country), era (DOPPS phase), comorbid illness (previous gastrointestinal bleeding, cardiovascular diseases, prosthetic heart valve, arrhythmias, diabetes, cancer, neurological disease, lung disease, HIV, psychiatric illness, recurrent cellulitis), dialysis characteristics (vintage, vascular access, pre-dialysis blood pressure, average ultrafiltration, average treatment time, adequacy), laboratory values (hemoglobin, albumin), and medications (anticoagulants, anti-platelet agents, gastric acid inhibitors). Characteristics are derived from patient charts and/or medical records by a trained study coordinator. Comorbid illnesses are based on prior medical history while vintage, vascular access, laboratory values, and medication use are based on information obtained at DOPPS enrollment. The average ultrafiltration (UF), treatment time, and pre-dialysis systolic blood pressure were based on 1 week of data at DOPPS enrollment (averaging of 3 values across 3 HD sessions in a week).
Study Outcome
The primary outcome definition was hospitalization for a bleeding event. A DOPPS-specific set of hospitalization diagnosis and procedure codes was used by study coordinators to classify hospitalizations based on chart abstraction; codes relevant to the outcome of interest were identified by the current study investigators. Bleeding events could include gastrointestinal bleeding, subdural hematoma, evacuation of a hematoma, abnormal bleeding, hemoptysis, hematuria, cerebral hemorrhage, epistaxis, or vascular access bleeding.
Statistical Analysis
Model development
We investigated the association between the predictor variables of interest and the study outcome of bleeding using time to event models. Follow-up time was censored upon the first of death, kidney transplantation, loss to follow-up, the study outcome, or end of the follow-up period. Predictor variables with >15% missing data were excluded. 17 Variables with <15% missingness were imputed using the predictive mean matching method for continuous variables and discriminant function method for categorical variables. 18 We prespecified all predictor variables in our initial model. Continuous variables were standardized and centered on their respective mean. 19 We used Cox proportional hazards models to examine the relationship between predictor variables and the outcome of interest. To develop a parsimonious model, we used the step-down procedure described by Ambler et al. 20 First, an initial model including all predictors was created followed by n-1 variable removal based on the beta coefficient. The final model contained the lowest number of predictors with a stable Akaike information criteria (AIC) value.20,21 The AIC is an estimate of the quality of a prediction model that optimizes the trade-off between model goodness of fit and simplicity. A plot of AICs versus n-1 predictive models is presented in Supplementary Figure 1 and Supplementary Table 2. Predictive performance was assessed and reported using measures of model discrimination (area under the receiver operator curve) and calibration (plots of the observed versus predicted risk of hemorrhage, Brier score) for the total cohort and stratified predictor variables included in the final model (ie, age [< or >= 65 years], sex, country, previous gastrointestinal bleeding, prosthetic heart valve, or vitamin K antagonist use). 22 Perfect calibration would yield a low Brier score with complete miscalibration yielding a Brier score of 1. 19 The time-averaged c-statistic at 3 years of follow-up was reported. We conducted all analyses with SAS software, SAS Enterprise guide version 7.1 (SAS Institute Inc., Cary, NC, USA).
External validation
We externally validated BLEED-HD in a cohort of adults (>66 years old) on chronic hemodialysis from Ontario, Canada, captured in the Canadian Organ Replacement Registry (CORR, a federally mandated, validated data registry for chronic dialysis patients) and housed at ICES.14,23 -25 Individuals under 66 were excluded as medications are only captured in those 65 or older and an additional 1 year was added to allow for medication refills. We included patients from April 2008 to March 2019 with follow-up to March 2020 (Supplementary Table 3). Outcomes were defined using a validated algorithm for bleeding requiring an emergency department visit or hospitalization (94% sensitivity, positive predictive value 87%, Supplementary Table 4 for definitions used). 26 We calculated the event rate, area under the curve (AUC), and plots of deciles of risk for observed versus predicted events using the BLEED HD algorithm.
Comparison to HAS-BLED, ATRIA, and HEMORR2HAGE
We visually examined outcome events by raw score risk and AUC for our comparator bleeding scores.9,10,12 Definitions were modified to the dialysis population and/or based on data availability as required (Supplementary Table 5). To examine calibration, we plotted the observed and predicted probability of a bleeding event by risk score points for our comparator bleeding scores. Risk scores were compared to BLEED-HD by calculating the difference in Uno’s c-statistic with 95% confidence intervals, the net reclassification index, and integrated discrimination index.27,28
Results
A total of 53 147 hemodialysis patients with a median follow-up time of 1.6 (interquartile range [IQR], 0.9-2.1) years were examined (Table 1). Bleeding events occurred in 2770 (5.2%, event rate 32.0 per 1000 person-years) individuals. Bleeding events were more common with an older age, in those with cardiovascular disease (coronary artery disease, other cardiac disease, stroke, peripheral vascular disease, prosthetic heart valves), AF and other arrhythmias, neurological or lung disorders, previous gastrointestinal bleeding, and cancer. Bleeding events differed by geographic region (higher in Europe, North America, and Australia/NZ). In terms of dialysis characteristics, bleeding was higher in those with catheters, with longer dialysis vintage and lower mean ultrafiltration per session. No difference was noted by mean dialysis session time or pre-dialysis blood pressure. Anticoagulant or acetylsalicylic acid, proton pump inhibitors (PPIs), or H2-receptor blockers use was more commonly in those with bleeding events.
Baseline Characteristics of Hemodialysis Patients With and Without a Bleeding Event in the DOPPS.

Belgium, France, Germany, Italy, Spain, and the United Kingdom.
Japan, Russia, Turkey, China, Gulf Cooperation Council (Saudi Arabia, Kuwait, the United Arab Emirates, Qatar, Bahrain, and Oman).
Ultrafiltration: average post weight minus pre-weight for first 3 dialysis sessions.
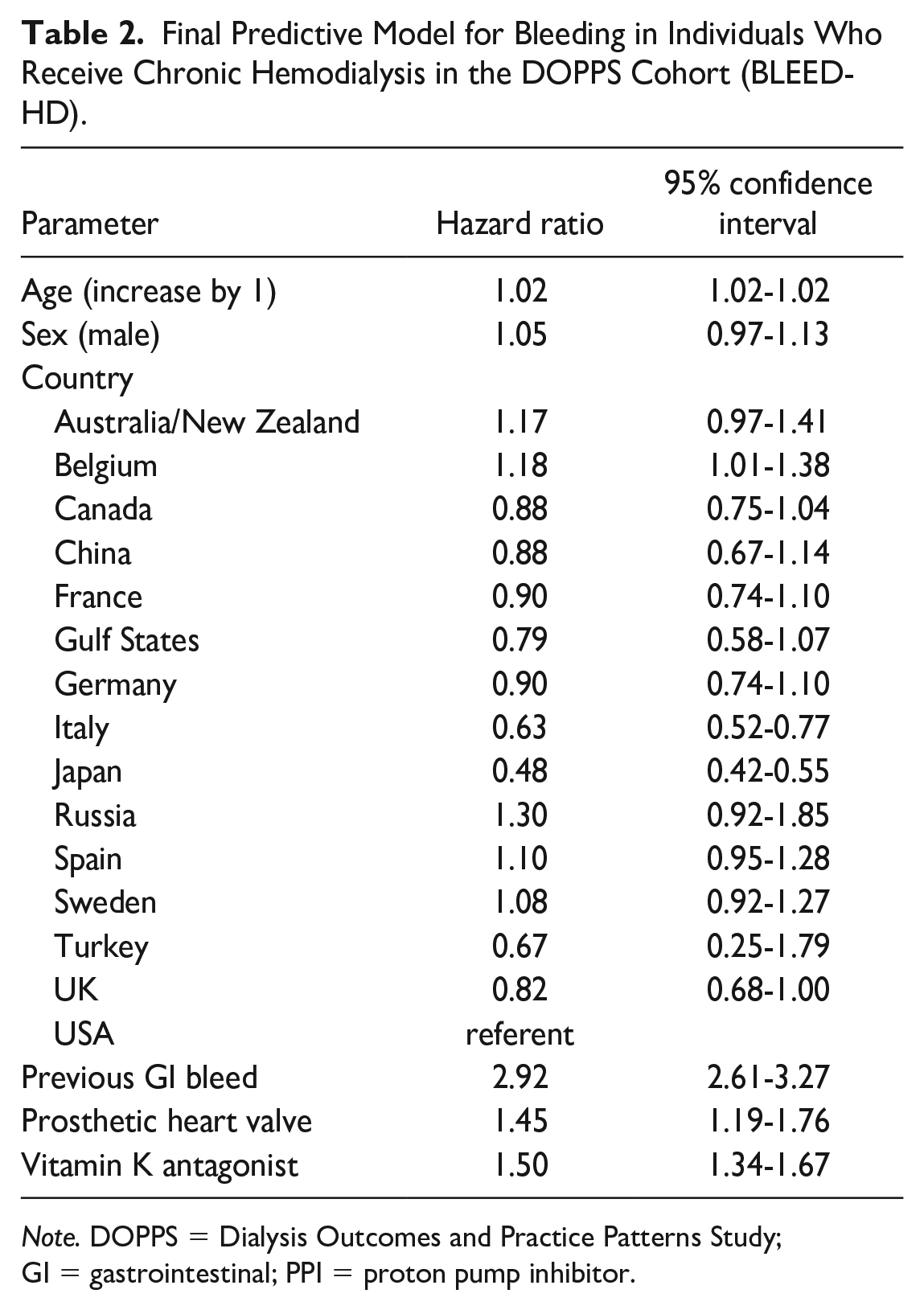
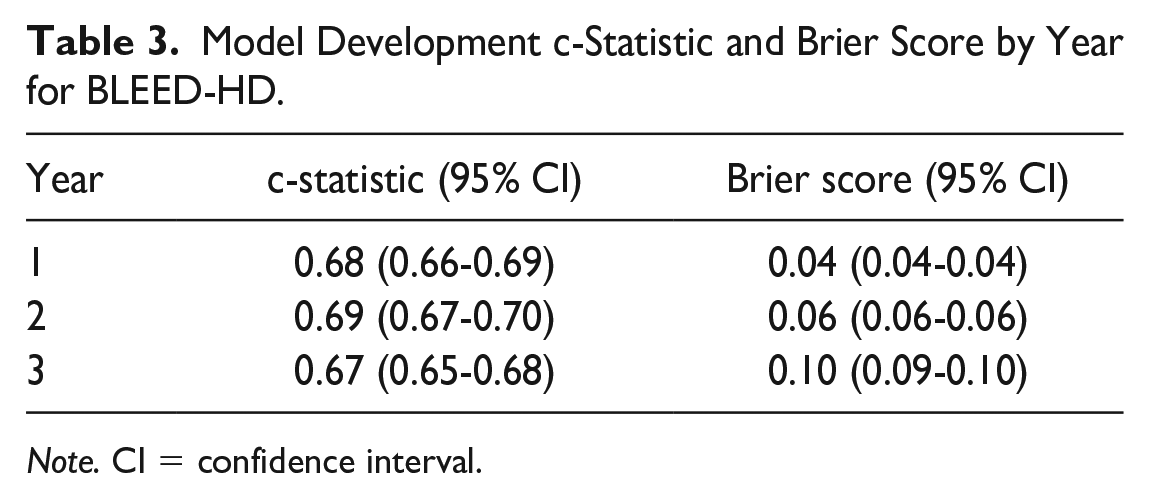
In time to event models, significant predictors of bleeding included in our final BLEED-HD model were age, sex, country, previous gastrointestinal bleed, prosthetic heart valve, and vitamin K antagonist use (Table 2, model equation is presented in Supplementary Box 1). No interactions were retained in the final model (based on AIC). The c-stat for BLEED-HD ranged from 0.66 to 0.69 over years 1 to 3, with a Brier score ranging from 0.035 to 0.095 (Table 3). The observed versus predicted risk difference did not exceed 2% across any decile risk category and the probability of predicted risk ranged from 3% to 10% (Figure 1). Calibration was consistent across individual predictors with no absolute difference between observed and predicted risk >2% (Figure 2).
Final Predictive Model for Bleeding in Individuals Who Receive Chronic Hemodialysis in the DOPPS Cohort (BLEED-HD).
Model Development c-Statistic and Brier Score by Year for BLEED-HD.

Plot of observed versus predicted probabilities across deciles of 3-year risk for bleeding for BLEED-HD among individuals on hemodialysis in the Dialysis Outcomes and Practice Patterns Study (DOPPS).

Observed versus predicted probability of bleeding (y-axis) for BLEED-HD among individuals on hemodialysis in the Dialysis Outcomes and Practice Patterns Study (DOPPS) by model predictors: (1) age, (2) sex, (3) previous gastrointestinal (GI) bleeding, (4) vitamin K antagonist, (5) country, and (6) prosthetic cardiac valve.
External validation was conducted on 19 318 hemodialysis patients from Ontario, Canada (Supplementary Table 6). In general, the external validation cohort was older (mean age 76.7), 40% female, 64% with diabetes mellitus, 58.7% coronary artery disease, 14% previous gastrointestinal bleed, and 3.6% prosthetic heart valve. Atrial fibrillation was present in 24.6% at baseline, 17.1% were on a vitamin K antagonist, and 46.8% were on a PPI. Overall, there were 2406 (12.5%, event rate 49.4 per 1000 person-years) bleeding events, with a median follow-up time of 1.95 years per patient. The BLEED-HD model discrimination yielded a time-averaged c-stat of 0.60. Examining observed versus predicted deciles of risk, BLEED HD underestimated most deciles of risk by 3% or less except in the highest risk decile (Figure 3).

External validation: Plot of observed versus predicted probabilities across deciles of 3-year risk for bleeding for BLEED-HD among individuals on hemodialysis on external validation.
In comparison to existing bleeding risk scores, BLEED-HD demonstrated improved discrimination and calibration in the DOPPS-HD cohort (Supplementary Table 5). The c-statistic for the HAS-BLED, ATRIA, and HEMORR2HAGE scores were 0.55, 0.55, and 0.57, respectively, compared to 0.65 for BLEED-HD (
Comparison of BLEED-HD Model Discrimination to Existing Bleeding Risk Prediction Models (1) HAS-BLED, (2) ATRIA, and (3) HEMORRHAGE in DOPPS Hemodialysis Patients.


Observed versus predicted probability of bleeding (y-axis) for (A) HEMORR2HAGES, (B) HAS-BLED, and (C) ATRIA among individuals on hemodialysis in the Dialysis Outcomes and Practice Patterns Study (DOPPS).
Discussion
Long-term hemodialysis patients represent one of the highest risk populations for major bleeding events and the ability to accurately identify those at high or low risk would aid in mitigation strategies and safe prescribing practices. Using data from an international, prospective cohort study of 53 147 individuals on long-term hemodialysis, we developed a clinical prediction equation (BLEED-HD) to help identify individuals at risk of clinically significant bleeding events requiring hospitalization. BLEED-HD included age, sex, country, previous diagnosis of a gastrointestinal (GI) bleeding, prosthetic heart valve, or vitamin K antagonist use in the final model. The overall model discrimination was low to moderate (c-stat = 0.65) with excellent calibration, was externally validated, and demonstrated some improvement to existing non-dialysis specific risk scores (HAS-BLED, ATRIA, HEMORR2GHAGE). Additional risk factors or novel biomarkers may further improve hemorrhage prediction in individuals on long-term dialysis.
Bleeding events are exceedingly common in the chronic hemodialysis population with estimates ranging from 40 to 80 per 1000 person-years.8,29 -33 In our DOPPS cohort, we found a rate consistent and slightly lower than previous studies (32 per 1000 person-years).34,35 We previously reported international variation in bleeding rates ranging from 40 (Japan) to 160 (Sweden) per 1000 person-years in the DOPPS phase 1-4 cohort. 36 These events are not without consequence, as they are estimated to occur in 14.4% of patients within the first 3 years of initiating HD and associated with considerable procedure-related morbidity and health care costs. 37
The BLEED-HD risk equation included several established risk factors for bleeding. Comorbidity, age, and use of vitamin K antagonist are well-known risk factors for major bleeding. 38 Previous GI bleeding was the single strongest risk factor for predicting future bleeding risk consistent with a previous DOPPS study. 5 Surprisingly, anti-platelet agents and dialysis-related characteristics, such as treatment time or vascular access, were not retained in the final model, illustrating the importance of traditional bleeding-related risk factors in predicting risk. Whether additional risk factors not included in the present study such as anticoagulation use with the dialysis procedure itself or as a catheter locking solution are associated with bleeding events remains unknown.
Our findings add to the current body of evidence demonstrating the challenges of exiting bleeding risk scores developed in the general population when applied to those on hemodialysis.4,39 -41 We demonstrated c-statistics <0.6 for HASBLED, ATRIA, and HEMORR2HAGES. A previous study of the NECOSAD dialysis cohort reported similar poor discrimination (c-statistics of 0.58 or lower) with currently available bleeding risk tools. 42 Calibration, a measure of more importance in prediction, was variable with existing risk scores with little ability to differentiate across risk groups and an inability to identify highest risk individuals. In contrast, BLEED-HD demonstrated improved calibration with a steady increase across risk deciles and an ability to predict from low (3%) to high (10%) risk. In some respects, these discrepancies are not surprising as many of the predictor definitions are difficult to apply directly to an HD population. For example, there is no clear definition of hypertension or anemia. Furthermore, existing scores were originally developed to identify bleeding risk in anticoagulated individuals with AF and not bleeding risk in general per se. Thus, BLEED-HD is targeted to estimate bleeding risk in the general hemodialysis population and demonstrates improvements in model operating characteristics (discrimination, calibration) to existing AF-based, general population tools.
Despite our large international cohort and data-driven model development approach, the BLEED-HD equation demonstrated low to moderate model discrimination and is of questionable clinical application. This suggests bleeding events may be relatively difficult to predict in the hemodialysis population and/or further studies or approaches may lead to model improvement. Key variables expanding on types, therapeutic effectiveness, routes of administration, and dosing of anticoagulation use may aid in that regard. Alternative approaches such as the accounting for the competing risk of death or model reduction techniques should be examined.
The clinical need for an accurate bleeding risk score in the hemodialysis population remains as it could aid in identifying and counseling individuals at risk in general or before/after the addition of an anticoagulant. Furthermore, a determination of high risk may prompt considerations of bleeding reduction strategies such as discontinuation of anti-platelet agents or prophylactic PPI use in individuals with AF. Routine PPI co-prescription has been estimated to have a number needed to treat of 25 to prevent one disabling or fatal upper gastrointestinal bleed in those aged 85 years or older with vascular events in the general population.
43
However, their use in the dialysis population can be more problematic due to the risk of hypomagnesemia, fracture, and/or
The current study developed and validated a novel bleeding score using data from a large international and well-established hemodialysis cohort that substantively improves on existing prediction tools. Nevertheless, there are noteworthy limitations. Important factors that may be associated with bleeding were not readily available such as duration and dosage of anticoagulation, dialysis procedure-related anticoagulation use, INR lability, applicability in peritoneal dialysis, and there was a limited number of individuals using direct oral anticoagulants, an emerging anticoagulant in the dialysis population.45,46 Our external validation differed from the development cohort as they were in Canada only and older relative to the DOPPS. This may explain the mildly lower model discrimination observed in the validation cohort. Despite the use of a large international cohort, we were limited by a small number of events in examining subgroups of high clinical interest such as the bleeding risk in individuals with or without an anticoagulant. For the existing risk scores (HEMORR2HAGES, HAS-BLED, ATRIA), all risk factors were not available for inclusion and modifications were required.
In conclusion, BLEED-HD predicts the risk of bleeding in individuals on hemodialysis with potential clinical use in determining individual risk and trial enrollment. Further studies examining its utility in high-risk populations, such as those on peritoneal or home hemodialysis, are required.
Supplemental Material
sj-docx-1-cjk-10.1177_20543581231169610 – Supplemental material for Development and Validation of a Predictive Risk Algorithm for Bleeding in Individuals on Long-term Hemodialysis: An International Prospective Cohort Study (BLEED-HD)
Supplemental material, sj-docx-1-cjk-10.1177_20543581231169610 for Development and Validation of a Predictive Risk Algorithm for Bleeding in Individuals on Long-term Hemodialysis: An International Prospective Cohort Study (BLEED-HD) by Mohit Madken, Ranjeeta Mallick, Emily Rhodes, Roshanak Mahdavi, Anan Bader Eddeen, Gregory L. Hundemer, Dearbhla M. Kelly, Angelo Karaboyas, Bruce Robinson, Brian Bieber, Amber O. Molnar, Sunil V. Badve, Peter Tanuseputro, Gregory Knoll and Manish M. Sood in Canadian Journal of Kidney Health and Disease
Footnotes
Acknowledgements
This study was supported by ICES, which is funded by an annual grant from the Ontario Ministry of Health (MOH) and the Ministry of Long-Term Care (MLTC). This study also received funding from: [list additional funders of this work, e.g., CIHR]. This document used data adapted from the Statistics Canada Postal Code OM Conversion File, which is based on data licensed from Canada Post Corporation, and/or data adapted from the Ontario Ministry of Health Postal Code Conversion File, which contains data copied under license from ©Canada Post Corporation and Statistics Canada. Parts of this material are based on data and/or information compiled and provided by: [list relevant data sources, e.g., MOH, CIHI]. The analyses, conclusions, opinions and statements expressed herein are solely those of the authors and do not reflect those of the funding or data sources; no endorsement is intended or should be inferred.
Ethics Approval and Consent to Participate
Regional ethics board approval was obtained for this deidentified retrospective study by the Ottawa Hospital Regional Ethic Board.
Consent for Publication
Not applicable.
Availability of Data and Materials
The dataset from this study is held securely in coded form at ICES. While legal data sharing agreements between ICES and data providers (e.g., healthcare organizations and government) prohibit ICES from making the dataset publicly available, access may be granted to those who meet pre-specified criteria for confidential access, available at ![]() (email:
(email:
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.M.S. has received a CME speaker fee from AstraZeneca. A.K., B.R., and B.B. are employees of Arbor Research Collaborative for Health, which administers the DOPPS.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Global support for the ongoing DOPPS Programs is provided without restriction on publications. See ![]() for more information. All funding is provided to Arbor Research Collaborative for Health and not to the authors directly.
for more information. All funding is provided to Arbor Research Collaborative for Health and not to the authors directly.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
