Abstract
Background:
The illicit drug toxicity (overdose) crisis has worsened across Canada, between 2016 and 2021 more than 28 000 individuals have died of drug toxicity. Organ donation from persons who experience drug toxicity death has increased in recent years.
Objective:
This study examines whether graft loss after kidney transplantation differed by donor cause of death.
Design:
Retrospective cohort.
Setting:
Provincial transplant program of British Columbia, Canada.
Patients:
Transplant recipients who received kidney transplantation from deceased donors aged 12 to 70 years between 2013 and 2019 (N = 1012).
Measurements:
Transplant recipient all cause graft loss (graft loss due to any cause including death) was compared by donor cause of death from drug toxicity or other.
Methods:
Five-year Kaplan-Meier estimates of all-cause graft survival, and 3-year complete as well as stratified inverse probability of treatment weighted Cox proportional hazards models were conducted.
Results:
Drug toxicity death donors donated to 25% (252/1012) of kidney transplantations. Drug toxicity death donors were more likely to be young, white, males, with fewer comorbidities such as diabetes or hypertension but were more likely to have a terminal serum creatinine ≥1.5 mg/dL or be hepatitis C virus (HCV) positive. Unadjusted 5-year estimate of all-cause graft survival was 97% for recipients of drug toxicity donor kidneys and 83% for recipients of non-drug toxicity donor kidneys (P < .001). Recipients of drug toxicity death donor kidneys had decreased risk of all cause graft loss compared to recipients of non-drug toxicity death donor kidneys (hazard ratio [HR]: 0.30, 95% confidence interval [CI]: 0.12-0.77, P = .012). This is primarily due to the reduced risk of all-cause graft loss for recipients of younger (≤35 years) drug toxicity death donor kidneys (HR: 0.05, 95% CI: 0.00-0.55, P = .015).
Limitations:
Potential selection bias, potential unmeasured confounding.
Conclusions:
Donation after drug toxicity death is safe and should be considered more broadly to increase deceased donor kidney donation.
Introduction
Kidney transplantation is the preferred treatment option for individuals experiencing end-stage kidney disease (ESKD) as transplantation improves recipient survival and quality of life compared to dialysis. 1 However, there are not enough organs available to meet the need for transplantation. In Canada, between 2011 and 2020 the prevalence of ESKD increased 31% from 31 776 to 41 670 and annual incidence increased 30% from 4704 to 6124. During the same period, the number of kidney transplantations increased 28% from 947 to 1210. In this time period, 2% to 3% of patients died on the waitlist each year, and another 6% to 8% were withdrawn from the waitlist due to deteriorating medical conditions. 2
Canada is in the midst of an opioid epidemic, having recorded a total of 28 960 opioid toxicity deaths between 2016 and 2021. Since 2016, British Columbia (BC) has had the highest illicit drug toxicity death rate in Canada. In 2021, 43.9 deaths per 1000 population were recorded in BC, about 6 deaths per day. 3 Of note, in BC, fentanyl has been detected in over 80% of illicit drug toxicity deaths since 2017. 4 A public health emergency was declared on April 2016 by the BC Provincial Health Officer, after which there was an expansion of take home naloxone, opioid agonist treatment, overdose prevention, and substance use treatment services across the province. 5 Additionally, in response to the COVID-19 pandemic, new prescribed safer supply programs were introduced to reduce reliance on the illicit drug supply. Despite these efforts, illicit drug toxicity remains the leading unnatural cause of death in BC accounting for more deaths than homicides, suicides, and motor vehicle incidents combined.5,6 Individuals that die of drug toxicity are eligible to become organ donors if they meet criteria for organ donation such as self or surrogate consent, death in hospital on mechanical ventilation, and a lack of medical contraindications for donation. 7
In the last decade, kidney donation from deceased donors has increased in BC, partly attributed to increased donation from individuals that have died of illicit drug toxicity. 8 The United States is also undergoing an opioid epidemic which began earlier than Canada and has recorded similar increases in organ donation from individuals that died of illicit drug toxicity.9 -11 Studies in the United States have found that recipient survival after kidney transplantation from individuals who died from drug toxicity was similar for recipients of kidneys from donors that died of any other cause of death.12,13 Individuals that exhibit a history of behaviors with a theoretical increased risk of transmitting a communicable disease (eg, hepatitis B virus [HBV], hepatitis C virus [HCV], human immunodeficiency virus [HIV]) are more likely to be labeled as increased risk donors (IRDs). Many donors who die of drug toxicity are labeled as IRDs due to a history of high-risk behaviors, 14 despite the low risk of undetectable infection. National and regional organ registry studies conducted in the United States found that organs from illicit drug toxicity death donors are disproportionately discarded prior to transplantation, despite similar post-transplant outcomes compared to transplantation from donors with other causes of death.10,12,15 -17
With increased organ donation from individuals that died of drug toxicity in BC, it is timely that a review of transplant recipient outcomes is conducted as literature on kidney transplant outcomes for recipients of drug toxicity death (DTD) donor kidneys is absent in Canada. The objectives of this study are to (1) describe utilization of DTD donor kidneys in BC and (2) to compare recipient all-cause graft loss after kidney transplantation by cause of donor death (DTD or other).
Methods
Data Source and Study Population
We studied recipients of kidney transplantation from within province deceased donors aged 12 to 70 years (to represent the age of persons who died from drug toxicity) between January 2013 and December 2019 captured in the Patient Records and Outcome Management Information System (PROMIS) database. PROMIS is an administrative and clinical database that records data from transplant programs in BC to facilitate patient management, organ donation, transplantation, and transplant research. 18 Patients waitlisted for a kidney transplantation meet general listing criteria such as having progressive and irreversible kidney disease, not having major contraindications to transplantation, having projected life-expectancy greater than 5 years given successful transplantation, and having a good support system as well as being willing to undergo post-transplant care. Recipient and donor matching of kidneys takes into consideration a number of high-priority groups (highly sensitized patients, medically urgent, combined kidney pancreas transplants, pediatric patients, and previous living donors) as well as donor and recipient age matching. 19 Donor death from drug toxicity was identified by a medical professional, as the cause of donor death through a combination of toxicology, ambulance services, emergency room, intensive care unit (ICU) admission, and consult history notes. Transplantations where donor cause of death were not able to be ascertained were excluded. This study was approved by the University of British Columbia Human Research Ethics Board (H18-01784).
Descriptive Analyses
We examined the number and proportion of kidney transplantations by donor cause of death (DTD or non-DTD) over time. Recipient, donor, and transplant characteristics were compared by donor cause of death using frequencies and proportions for categorical variables and medians and quartiles for continuous variables. Categorical variable comparisons were performed using the chi-square test or Fisher’s exact test as appropriate, continuous variables were compared using the Wilcoxon rank-sum test.
Outcome Analyses
We analyzed the first instance of kidney-only transplantation in the study period for each recipient. Median transplant recipient follow-up time was calculated. All-cause graft loss (ACGL), defined as graft loss due to any cause (re-transplant, return to dialysis, or death), censored at end of follow-up (August 23, 2020) was the primary outcome. Secondary outcomes included death with graft function (experiencing death with a functioning graft, censored at re-transplant, return to dialysis, or end of follow-up), death censored graft loss (DCGL) (defined as graft loss due to graft failure characterized by re-transplantation or return to dialysis, censored at death or end of follow-up), and first episode of acute rejection. The Kaplan-Meier method was used to estimate all outcomes at 1-, 3-, and 5-years post-transplant. Estimated Kaplan-Meier curves by donor cause of death were compared using the log-rank test. The association between drug toxicity as the cause of donor death and ACGL was modeled using unadjusted and weighted multivariable Cox proportional hazards models. As DTD donors are on average younger, we also examined 2 additional models dividing the dataset by median DTD donor age to determine the association of drug toxicity as the cause of donor death on ACGL in younger and older donor age groups. Weights were calculated using the inverse probability of treatment weighting (IPTW) method. Propensity scores used to derive inverse probability of treatment (IPT) weights were estimated using multivariable logistic regression adjusting for a vector of donor, recipient, and transplant characteristics. In this study, the propensity score was defined as the probability of receiving a DTD donor kidney. Inverse probability of treatment weights were calculated as the inverse of the probability of receiving the type of kidney given the donor cause of death group the recipient received the kidney from. Therefore, the IPT weight for the recipient of a DTD donor kidney is one divided by the propensity score, whereas the IPT weight for a recipient of a non-DTD donor kidney is one divided by one minus the propensity score. Based on donor, recipient, and transplant characteristics this method estimates a lower weight for a recipient that shares more similarities with other recipients within their donor death group and a higher weight for a recipient that is more dissimilar compared with other recipients from their donor death group. Detailed IPTW methodology can be found elsewhere.20,21 IPT weights for each covariate of interest were assessed using standardized mean differences (SMDs). IPT weights were included in the multivariable Cox proportional hazards model. Variables were adjusted for in the multivariable Cox proportional hazards model if the variable had a SMD value greater than 0.1 or if the variable was not initially included in the logistic regression model. 20 The proportional hazards assumption was examined using log-log plots. Variables considered for inclusion in the propensity score model were donor factors (ie, sex, age, race, body mass index [BMI], HCV, terminal serum creatinine, history of hypertension, history of diabetes, and deceased donor type), recipient factors (ie, sex, age, race, BMI, HCV), and transplant factors (ie, time on dialysis prior to transplantation, human leukocyte antigen [HLA] mismatch, and transplant number). All analyses were conducted in R v.3.6.2.
Results
Figure 1 depicts steps taken to arrive at the analytical sample starting with 1082 kidney-only transplantations as a result of 591 deceased donors donating kidneys to 1079 transplant recipients. For these donors, cause of death could not be ascertained for 35 donors resulting in 67 transplants being excluded. Three transplants were excluded as they were not the first transplant for the patient in the study period. The final analytical sample comprised of 1012 kidney transplantations from 555 donors, of these 252 (25%) transplants were from DTD donors.

Flowchart of kidney transplant inclusion criteria.
Transplant Trends
Figure 2 shows the number of kidney transplantations in BC over time by donor cause of death. The number of kidney transplantations increased 89.2% from 93 in 2013 to 176 in 2019 due to increases in both DTD (350% increase) and non-DTD (57.8% increase) donor transplantation. The average number of DTD donor transplantations was 6-fold higher after 2016 (63 transplants per year) compared to before 2016 (10 transplants per year). Proportionally, transplantations from DTD donors peaked at 38% of the analytic sample in 2017, then dropping to 25.6% of the analytic sample in 2019.

Count of kidney-only transplantations between 2013-2019 categorized by DTD and other cause of donor death (non-DTD).
Kidney Transplantation Characteristics
Recipients of DTD donor transplantations were more likely to be young (median age [Q1; Q3]): 51 [42, 59] years vs. 60 [48, 67] years, P < .001). Other recipient traits showed similar distributions between donor cause of death groups. DTD donors were more likely to be young (35.0 [27.0, 42.2] years vs. 49.5 [34.0, 59.0] years, P < .001), white (84.1% vs 79.3%, P < .001), males (75.8% vs 58.9%, P < .001), with fewer comorbidities such as diabetes (1.6% vs 8.6%, P < .001) and hypertension (6.3% vs 24.9%, P < .001), and were less likely to be expanded criteria donors (4.8% vs 35.5%, P < .001). However, DTD donors were more likely to be flagged as IRDs (82.1% vs 37.0%, P < .001), have a terminal serum creatinine ≥1.5 mg/dL (22.6% vs 11.6%, P < .001), and were more likely to be HCV positive (6.7% vs 1.7%, P < .001). Transplant recipients of DTD donor kidneys were less likely to have been on dialysis for more than 3 years prior to transplantation (46.0% vs 58.4%, P = .003). Delayed graft function defined by requirement for dialysis in the first week after transplantation occurred in similar frequencies between recipients of DTD and non-DTD donor kidneys (28.6% vs 29.3%, P = .878) (Table 1).
Recipient, Donor, and Transplant Characteristics Compared by Donor Cause of Death (Drug Toxicity (DTD) or Other (Non-DTD) for Kidney Transplant Recipients.

Note. Variable level frequencies between DTD and non-DTD donor organ recipients were compared using the chi-square test, Fisher’s exact test, or the Wilcoxon rank sum test as appropriate. Each donor may contribute more than one row of data for donor variable frequencies as each donor may donate up to 2 kidneys. DTD = drug toxicity death; BMI = body mass index; HCV = hepatitis C virus; PRA = panel reactive antibody; CVA = cerebrovascular accident; NDD = neurologic determination of death; DCD = donation after circulatory death; HLA = human leukocyte antigen; CIT = cold ischemic time.
Missing: recipient race (0.2%); recipient BMI (0.69%); recipient peak PRA % (13.04%); donor BMI (0.2%); donor terminal serum creatinine, mg/dL (0.2%); dialysis duration prior to transplant, years (0.79%); number of HLA mismatches (0.3%); and CIT, hours (1.98%).
Outcomes
Median follow-up time was 3 years. The cumulative probability of all-cause graft survival after DTD donor transplantation was 98% at 1-year and 97% at 3- and 5-years post-transplant, compared to 94%, 88%, and 83% at 1-, 3-, and 5-years post-transplant among non-DTD donor transplantations. Compared to recipients of non-DTD donor kidneys, recipients of DTD donor kidneys were less likely to experience ACGL at 5-years post-transplant (log-rank, P < .001) (Figure 3). The probability of acute rejection after DTD donor kidney transplantation was 8%, 9%, and 11% at 1-, 3-, and 5-years post-transplant, the probability of acute rejection for non-DTD donor kidney transplantation was similar at 8%, 10%, and 11% at 1-, 3-, and 5-years post-transplant (log-rank, P = .7) (Figure 4). Estimates of recipient death with function and DCGL are shown in the appendices.

Five-year Kaplan-Meier curves and corresponding risk tables for the probability of all-cause graft survival among kidney transplant recipients by DTD and other cause of donor death (non-DTD).
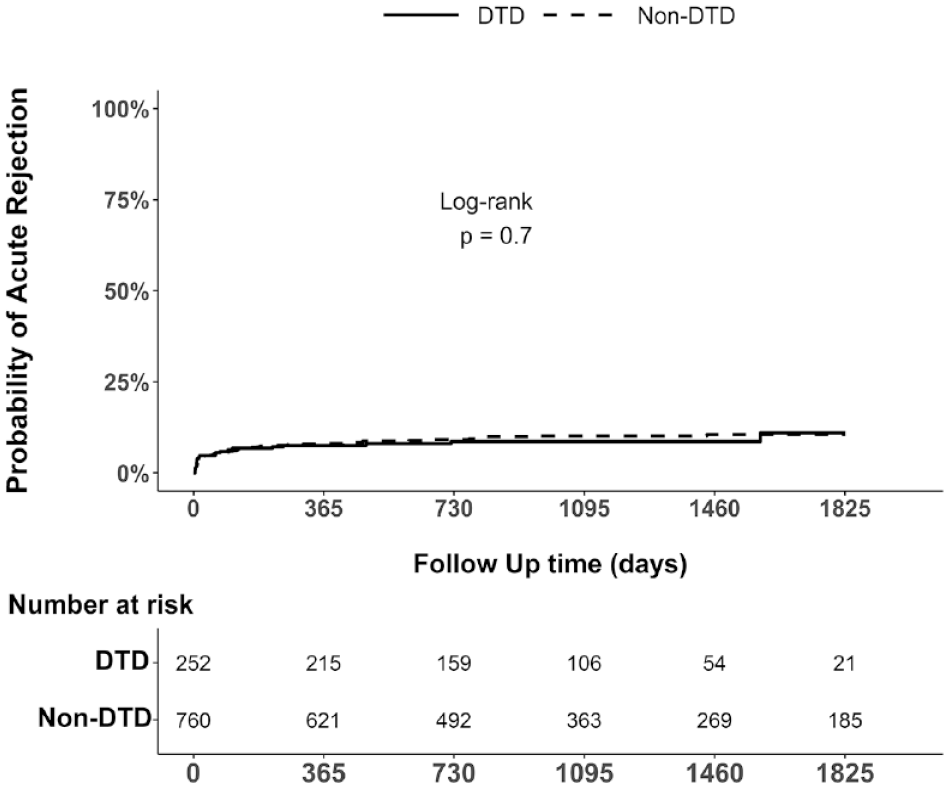
Five-year Kaplan-Meier cumulative incidence curves and corresponding risk tables for the first episode of acute rejection among kidney transplant recipients by DTD and other cause of donor death (non-DTD).
The univariate Cox proportional hazards model showed that recipients of DTD donor kidneys had significantly decreased risk of ACGL (hazard ratio (HR): 0.23, 95% confidence interval (CI): 0.10-0.53, P < .001) compared to recipients of non-DTD donor kidneys. After IPT weighting and multivariable adjustment, donor DTD status was still significantly protective (HR: 0.30, 95% CI: 0.12-0.77, P = .012) (Table 2). All recipients when stratified by median DTD donor age were split into 2 subgroups (≤35 years, >35 years) and a IPTW multivariable Cox proportional hazards model was fit for each subgroup. In the donor age ≤35 years group, recipients of DTD donor kidneys had significantly decreased risk of ACGL (HR: 0.05, 95% CI: 0.00-0.55, P = .015) compared to recipients of non-DTD donor kidneys. In the donor age >35 years group, risk of ACGL was similar for recipients that received a DTD donor kidney compared to recipients of non-DTD donor kidneys (HR: 0.46, 95% CI: 0.17-1.25, P = .129) (Table 3). The proportional hazards assumption was visually adequate in all log-log survival plots.
Modeling the Association Between Donor Drug Toxicity as a Cause of Donor Death and Recipient All Cause Graft Loss for Single Kidney Transplantations.
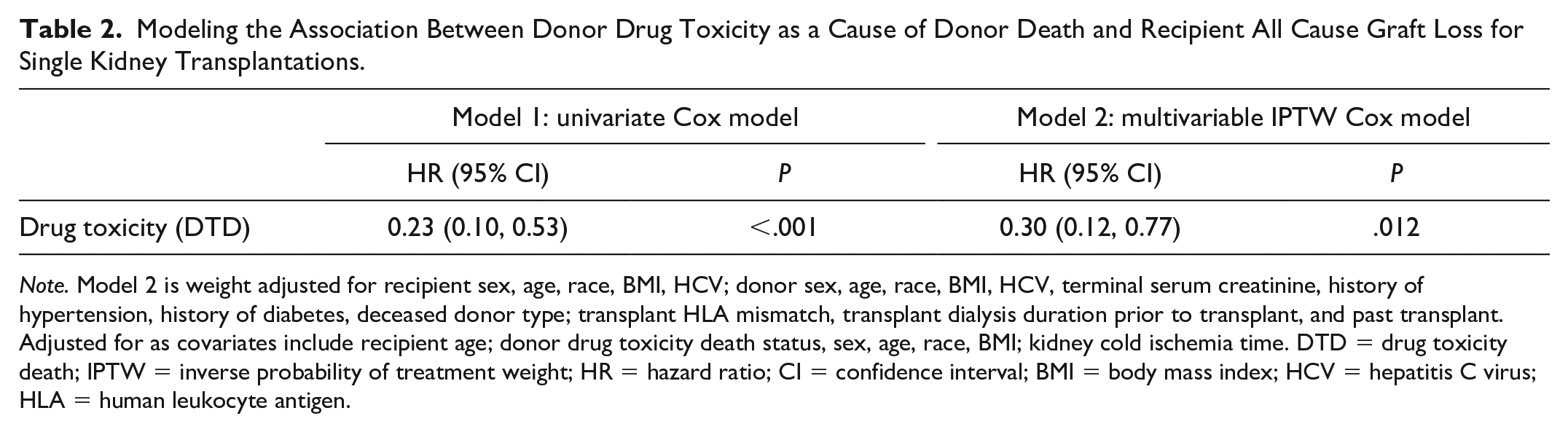
Note. Model 2 is weight adjusted for recipient sex, age, race, BMI, HCV; donor sex, age, race, BMI, HCV, terminal serum creatinine, history of hypertension, history of diabetes, deceased donor type; transplant HLA mismatch, transplant dialysis duration prior to transplant, and past transplant. Adjusted for as covariates include recipient age; donor drug toxicity death status, sex, age, race, BMI; kidney cold ischemia time. DTD = drug toxicity death; IPTW = inverse probability of treatment weight; HR = hazard ratio; CI = confidence interval; BMI = body mass index; HCV = hepatitis C virus; HLA = human leukocyte antigen.
Stratified Models By Median DTD Donor Age Modeling the Association Between Donor Drug Toxicity as a Cause of Donor Death and Recipient All Cause Graft Loss for Single Kidney Transplantations.

Note. Model 1 is weight adjusted for recipient sex, age, race, BMI, HCV; donor sex, age, race, BMI, HCV, terminal serum creatinine, history of hypertension, deceased donor type; transplant HLA mismatch, transplant dialysis duration prior to transplant, and past transplant. Adjusted for as covariates include recipient race and donor drug toxicity death status. Model 2 is weight adjusted for recipient sex, age, race, BMI, HCV; donor sex, age, race, BMI, HCV, terminal serum creatinine, history of hypertension, history of diabetes, deceased donor type; transplant HLA mismatch, transplant dialysis duration prior to transplant, and past transplant. Adjusted for as covariates include recipient sex, age, race; donor drug toxicity death status, sex, age, race, BMI, history of hypertension, deceased donor type, transplant HLA mismatch, and cold ischemia time. DTD, drug toxicity death; HR, hazard ratio; CI, confidence interval; BMI = body mass index; HCV = hepatitis C virus; HLA = human leukocyte antigen.
Discussion
Kidney transplantation from DTD donors increased more than 7-fold over the study period, reaching a high of 73 in 2017. DTD donor characteristics were consistent with characteristics of individuals who died of drug toxicity in the province (i.e., DTD donors were oftf transmission to the recipient but is likely en young, white, and male). In addition, compared to non-DTD donors, DTD donors had fewer comorbidities but higher terminal serum creatinine and a higher prevalence of HCV. Compared to recipients of non-DTD donor kidneys, recipients of DTD donor kidneys were significantly less likely to experience ACGL. In stratified analysis, we saw that these trends were largely driven by recipients of kidneys from younger DTD donors. There was no difference in the probability of experiencing the first episode of acute rejection by donor cause of death.
In a national registry study in the United States, Durand et al showed that recipients of kidney transplantation from DTD donors (defined as overdosed deceased donors in the study) had similar survival outcomes compared to recipients of non-DTD donor (categorized as either trauma death donors [TDD] or medical death donors [MDD]) kidneys. Compared to the BC sample of kidney transplant recipients, unadjusted 5-year recipient survival was lower for DTD donor (86.3%), TDD (86.2%), and MDD kidney transplant recipients (80.7%). Estimated standardized hazard ratio of death stratified by transplant center also indicated that risk of death was not significantly different between DTD donor and TDD transplant recipients (HR: 1.08, 95% CI: 0.96-1.23) and was significantly protective for DTD donor compared to MDD transplant recipients (HR: 0.83, 95% CI: 0.72-0.96). Similar to the BC DTD donor population, DTD donors studied by Durand et al 12 were more likely to be younger, white, have higher terminal serum creatinine, and were less likely to have a history of diabetes or hypertension. Wanis et al also studied the national Scientific Registry of Transplant Recipients database but only looked at transplantation from donation after circulatory death donors between 2006 and 2016 by estimating graft failure cumulative incidence curves standardized by baseline recipient, donor, and transplant characteristics finding no difference in graft failure between DTD and non-DTD donor kidney transplant recipients. Wanis et al reported a 5-year risk of graft failure of 21.7% among DTD donor kidney transplant recipients. DTD kidney donors studied by Wanis et al 13 were more likely to have a history of cocaine or other drug use, but similar to the BC DTD donor population were more likely to be younger with fewer comorbidities such as diabetes or hypertension. Similar to the comparator group of MDDs in Durand et al, recipients of DTD donor kidneys in BC were significantly less likely to experience all cause graft loss compared to recipients of non-DTD donor kidneys. This is likely attributable to the younger demographic of DTD donors.
In this study, a greater proportion of DTD donors are flagged as IRDs, a categorization that refers to a very small but real risk of communicable disease transmission to the recipient but is likely not reflective of lower organ quality. When patients are listed for transplantation, they are informed of the potential risks and benefits of transplantation with an IRD organ. When an IRD organ becomes available for transplantation, patients are further counseled by transplant physicians on the definition of an IRD, the process of donor communicable disease screening, and the risk trade-offs of accepting or declining an IRD organ. The final decision to accept an IRD organ relies on informed consent from the patient. 19 There has been underutilization of IRD kidneys in the United States despite the low risk of communicable disease transmission.10,16,17 Survival outcomes are similar when comparing recipients of an IRD kidney compared to recipients of non-IRD kidneys, cumulative mortality is lower for those that accept an IRD kidney compared to patients that choose to remain on the waitlist.10,15 In our study, a greater proportion of DTD donors were labeled as IRDs and a greater proportion of DTD donors were also HCV positive. HCV transmission was not of great concern as most HCV positive donors were HCV antibody positive but nucleic acid tested (NAT) negative. However, in 2019, 2 NAT positive non-DTD donors donated kidneys to 4 recipients with no history of HCV as a pilot study for the use HCV NAT positive donors. Appropriate direct acting antiviral prophylactic treatment was conducted, and no HCV transmission occurred for the 4 recipients. Direct-acting antiviral regimens have been shown to successfully suppress and eradicate HCV viral load in HCV negative kidney transplant recipients.22 -24
Strengths and Limitations
Limitations of our study include the fact that we cannot differentiate whether drug toxicity deaths were due to opioids or other illicit drugs, however, the majority of drug toxicity deaths since 2015 involved fentanyl, in 2019 a staggering 91% of all drug toxicity deaths involved fentanyl. 25 Also due to the observational aspect of this study we are susceptible to selection bias as well as potential unmeasured confounding. The decision to offer and accept DTD donor kidneys may have differed by transplant team and patients waiting for transplantation resulting in subgroups of donors or recipients being disproportionately represented. That is, some potential DTD donor demographic groups (eg, donors 60 years and older) may be under-represented in this study. As the proportion of individuals dying of drug toxicity from older age groups has increased in recent years, 6 it is not clear from our results whether recipients of organs from older deceased individuals would yield similar outcomes.
Conclusion
Transplant recipients of DTD donor kidneys in BC show high all-cause graft survival rates, similar to recipients of kidneys from donors that die of other causes. This suggests that DTD donor transplantation following current practices is safe. At present, the number of individuals dying of drug toxicity is increasing in BC. Allowing these individuals their right to become organ donors, if eligible, is important. Monitoring short- and long-term outcomes for recipients of DTD donor kidneys should continue as characteristics of individuals who die from drug toxicity change.
Supplemental Material
sj-docx-1-cjk-10.1177_20543581231156853 – Supplemental material for Observational Study Examining Kidney Transplantation Outcomes Following Donation From Individuals That Died of Drug Toxicity in British Columbia, Canada
Supplemental material, sj-docx-1-cjk-10.1177_20543581231156853 for Observational Study Examining Kidney Transplantation Outcomes Following Donation From Individuals That Died of Drug Toxicity in British Columbia, Canada by Max Wenheng Xie, Sean Patrick Kennan, Amanda Slaunwhite and Caren Rose in Canadian Journal of Kidney Health and Disease
Footnotes
Acknowledgements
The authors acknowledge staff at BC Transplant: Yvonne Sun, Parvind Grewal, and Heidi Butler for their assistance with data extraction and clarification of data variables.
The authors acknowledge Younathan Abdia, Biostatistician at the BC Center for Disease Control for statistical consultation
The authors wish to acknowledge the devastating impact of the ongoing illicit drug toxicity crisis on people who use substances and their families, friends, and communities throughout British Columbia. Thousands of loved ones have passed far too soon. Deaths from illicit drug toxicity are entirely preventable and efforts must be made to expand access to safer supply, harm reduction, and related services. This work is dedicated to the persons, and families and friends of persons, who have died from illicit drug toxicity who made the selfless gift of organ donation. Your donations have saved the lives of hundreds of British Columbians. The authors also acknowledge with gratitude that the institutions of BC Transplant, BC Center for Disease Control, and University of British Columbia are located on the ancestral, unceded, and traditional homelands of the Musqueam, Squamish and Tsleil-Waututh Nations.
List of Abbreviations
ACGL, all cause graft loss; BC, British Columbia; BMI, body mass index; DCGL, death censored graft loss; DTD, drug toxicity death; ESKD, end-stage kidney disease; HBV, hepatitis B virus; HCV, hepatitis C virus; HIV, human immunodeficiency virus; HLA, human leukocyte antigen; IPTW, inverse probability of treatment weight; IRD, increased risk donor; MDD, medical death donor; NAT, nucleic acid test; PROMIS, patient records and outcome management information system; SMD, standardized mean difference; TDD, trauma death donor; US, United States.
Ethics Approval and Consent to Participate
This study was approved by the Human Research Ethics Board at the University of British.
Consent for Publication
All authors consent to the publication of this study.
Availability of Data and Materials
Data for this study is not available due to Provincial Health Services Authority and BC Transplant Health Information Management policies. Data can be requested from BC Transplant through a data request form and a Research Information Sharing Agreement.
Author Contributions
Max Xie was responsible for literature review, analyses, interpretation of data, writing, and finalization of the manuscript.
Dr. Sean Patrick Keenan assisted in drafting of manuscript and provided organ donation content area knowledge. Dr. Keenan conducted chart review to identify whether donors died of drug toxicity poisoning or had other causes of death. Dr. Keenan also took part in interpretation of data and work, revised manuscript drafts, and approved the final version of the manuscript.
Dr. Amanda Slaunwhite provided content area knowledge regarding the opioid crisis.
Dr. Caren Rose was responsible for formulation of concept, grant application, review of literature, review of manuscript, interpretation of data, and finalization of the manuscript.
All authors also took part in interpretation of data and work, revised manuscript drafts, and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Grant funding through BC Transplant. Dr. Sean Patrick Kennan is the Medical Director, Donation Services at BC Transplant. Staff at BC Transplant assisted with the extraction of data.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge the funding received from the Transplant Research Foundation of British Columbia, Venture Grant Program.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
