Abstract
Background:
Little was known about how chronic hyperkalemia (cHK) in patients with chronic kidney disease (CKD) is managed in British Columbia, Canada.
Objective:
To investigate the trend in sodium polystyrene sulfonate (SPS) and calcium polystyrene sulfonate (CPS) utilization and their efficacy in treating cHK in CKD patients from British Columbia, Canada.
Study Design:
Retrospective cohort.
Setting & Patients:
CKD patients aged ≥18 years, followed in Kidney Care Clinic (KCC), who had at least 2 potassium values ≥5.0 mmol/L separated by no more than 91 days during the period of June 1, 2015, to July 31, 2021, were included. Index date was the first date of the 2 potassium values ≥5.0 mmol/L. Patients who received SPS or CPS within 90 days before index date were excluded. Patients who were on dialysis or received kidney transplantation on or before index date were also excluded.
Exposure:
Continuous exposure to SPS and CPS.
Outcome:
SPS/CPS prescription utilization trend was described by the proportion of patients ever treated with SPS/CPS, median time in days between cHK diagnosis and initiating treatment with SPS/CPS, total and median number of SPS/CPS prescriptions dispensed. Change in mean serum potassium concentration before and after a 90-day continuous treatment with SPS/CPS was estimated.
Analytical Approach:
Descriptive.
Results:
This study included 10 495 patients with cHK (median age 74 years, 60% were male). Median follow-up time was 625 days. Only 2864 (27%) patients were dispensed at least 1 prescription of either SPS or CPS. A total 7300 prescriptions were dispensed; median prescriptions dispensed per patients were 2 (interquartile range [IQR]: 1-3). Median time from index date to the first prescription dispensing date was 154 days (IQR: 36-455). Continuous 90-day treatment with SPS/CPS decreased the mean serum potassium concentration by 0.60 mmol/L, from 5.58 to 4.98 mmol/L.
Limitations:
Descriptive observational study without control group.
Conclusions:
In British Columbia, only 1 in 4 CKD patients with cHK were dispensed with SPS/CPS, mostly with higher degrees of hyperkalemia. These medications appeared to be moderately effective in reducing the serum potassium concentration. Future research is necessary to evaluate the comparative effectiveness of newer generation medications.
Introduction
Hyperkalemia, increased serum potassium (k+) concentration, is usually rare in the general population but fairly common among people with reduced kidney function. 1 Recent research suggests that approximately 45% of the patients with chronic kidney diseases (CKD) may experience hyperkalemia. 2 Untreated hyperkalemia, especially when moderate or severe can cause cardiac arrhythmias and sudden death. 3
Sodium polystyrene sulfonate (SPS), a nonspecific sodium-cation exchange resin, is commonly used in managing hyperkalemia. Its use in treating chronic hyperkalemia in CKD patients was not evident due to small sample sizes in the Randomized Controlled Trials (RCTs), mild degree of hyperkalemia at baseline, and short study durations of 3 to 7 days. 4 Calcium polystyrene sulfonate (CPS) may have a few clinical advantages over SPS. As a mechanism of action, SPS exchanges sodium for potassium, whereas CPS exchanges calcium for potassium. 5 Nakayama et al 6 reported CPS as a safer option compared with SPS in treating hyperkalemia in patients with CKD because it does not induce hypervolemia or hyperparathyroidism. However, its efficacy in treating chronic hyperkalemia among CKD patients is also not well established. 7
In British Columbia, Canada, 1 in 10 individuals has kidney disease. 8 Little is known about the management of chronic hyperkalemia among these people with reduced kidney function. The objective of this study was to investigate the trend in SPS and CPS utilization, and their efficacy in treating chronic hyperkalemia in CKD patients from British Columbia, Canada.
Methods
Data Sources and Study Population
This retrospective cohort study was conducted using data from Patient Records and Outcome Management Information System (PROMIS), a population-based integrated registry database for CKD patients under the specialized nephrologist care in British Columbia, Canada. Individuals with abnormality in kidney structure or function that is present for at least 3 months with no specific estimated glomerular filtration rate (eGFR) cutoff are considered to be patients with CKD and are registered into PROMIS. CKD patients aged 18 years or more and followed in any of the 16 Kidney Care Clinics (KCC) under the provincial registry database (PROMIS), who had at least 2 potassium values ≥ 5.0 mmol/L separated by no more than 91 days during the period of June 1, 2015, to July 31, 2021, were included into the cohort of chronic hyperkalemia patients.1,9 -11 The first date of the 2 potassium values ≥5.0 mmol/L was recorded as the cohort entry date (index date). CKD patients with chronic hyperkalemia who received SPS or CPS within the last 90 days from the index date were excluded. Study individuals who were on dialysis or received kidney transplantation on or before index date were also excluded. This study was approved by the Clinical Research Ethics Board at the University of British Columbia, Canada (H21-02507).
Exposure to SPS and CPS
Study individuals were followed from the index date to a PROMIS registration end or deactivation date, death, kidney dialysis start date, kidney transplantation date, or the study end date (July 31, 2021), whichever occurred first.
All outpatient prescriptions of SPS (or SPS + Sorbitol) and CPS were identified from the PROMIS dispensing records. For each prescription, the duration of treatment was from the prescription start date to the earliest of the prescription discontinuation date (cancel prescription), prescription end date (for medications that were prescribed with an end date), or follow-up end date. If a new prescription was dispensed within 7 days from the previous prescription duration end date, then we assumed that the patient was continuously exposed to the medication from the first prescription start date to the consecutive prescription stop date. This continuous medication exposure was ended when there was a more than 7-day gap between 2 prescriptions. Switching between SPS and CPS was allowed to capture the real-world trend in medication utilization.
Outcomes
To describe the trend in SPS/CPS prescriptions, we first identified the CKD patients with chronic hyperkalemia who were dispensed at least 1 prescription. This would tell us the proportions of chronic hyperkalemia (HK) patients ever treated with SPS/CPS during the entire study period. In addition, we estimated the median time in days before initiating treatment with SPS/CPS. We also looked into the total and median number of SPS/CPS prescriptions dispensed.
For the clinical outcome, we used the outpatient potassium measurements. We categorized hyperkalemia based on serum potassium concentration (mmol/L): normal (<5.0), mild (5.0 to <5.5), and moderate (≥5.5). We looked into the distribution of potassium level at index date, that is, proportions of chronic HK patients with mild and moderate HK. Finally, the change in mean serum potassium concentration after a 90-day continuous treatment with SPS/CPS was estimated. Serum potassium concentrations were recorded as the closest one measured within 15 days before dispensing the first prescription and within 15 days from the 90th day of continuous treatment.
Co-variables to Describe Patient Characteristics
Although this was an observational descriptive study, we recorded a number of co-variables to describe the patient characteristics for overall study sample as well as by treatment status. Biological variables included age at index, sex, and self-reported race. Treatment strategy may vary based on the patients’ follow-up location. Therefore, we assessed whether the patient was followed at the multidisciplinary kidney care clinics (KCC) or both in the physician’s office and KCC. Certain medication may contribute to hyperkalemia. We looked into the baseline history of treatment with renin-angiotensin-aldosterone system inhibitors (RAASi), ie, if the patient were dispensed any RAASi within 90 days before index date. Baseline eGFR was recorded as the closest eGFR measured within 90 days before the index date. We also recorded a series of comorbidities at baseline, including diabetes, cardiovascular disorders, and malignancy.
Results
The study included 10 495 CKD patients with chronic hyperkalemia during the study period of June 1, 2015, to July 31, 2021. The median age was 74 years (IQR: 65-81), and approximately 60% were male (Table 1). The proportion of male patients was higher in the treated group compared with untreated group (66% vs. 58%). The self-reported race was approximately evenly distributed except for a lower proportion of Caucasians in the treated group. As expected, the median serum potassium concentration at cohort entry was higher among those who were treated with SPS/CPS, 5.30 versus 5.20. In terms of severity, the prevalence of moderate hyperkalemia was more than double in the treated group compared with untreated group, 40% and 19%, respectively. Presence of other baseline comorbidities was similar across the groups except for slightly higher proportion of diabetes among the treated group. For overall study sample, the baseline eGFR was 24 ml/min/1.73 m2, which was similar across the treatment groups. Approximately 12% of the patients were dispensed RAASi within 90 days before the index date.
Patient Characteristics by Treatment With Sodium Polystyrene Sulfonate or Calcium Polystyrene Sulfonate Status.
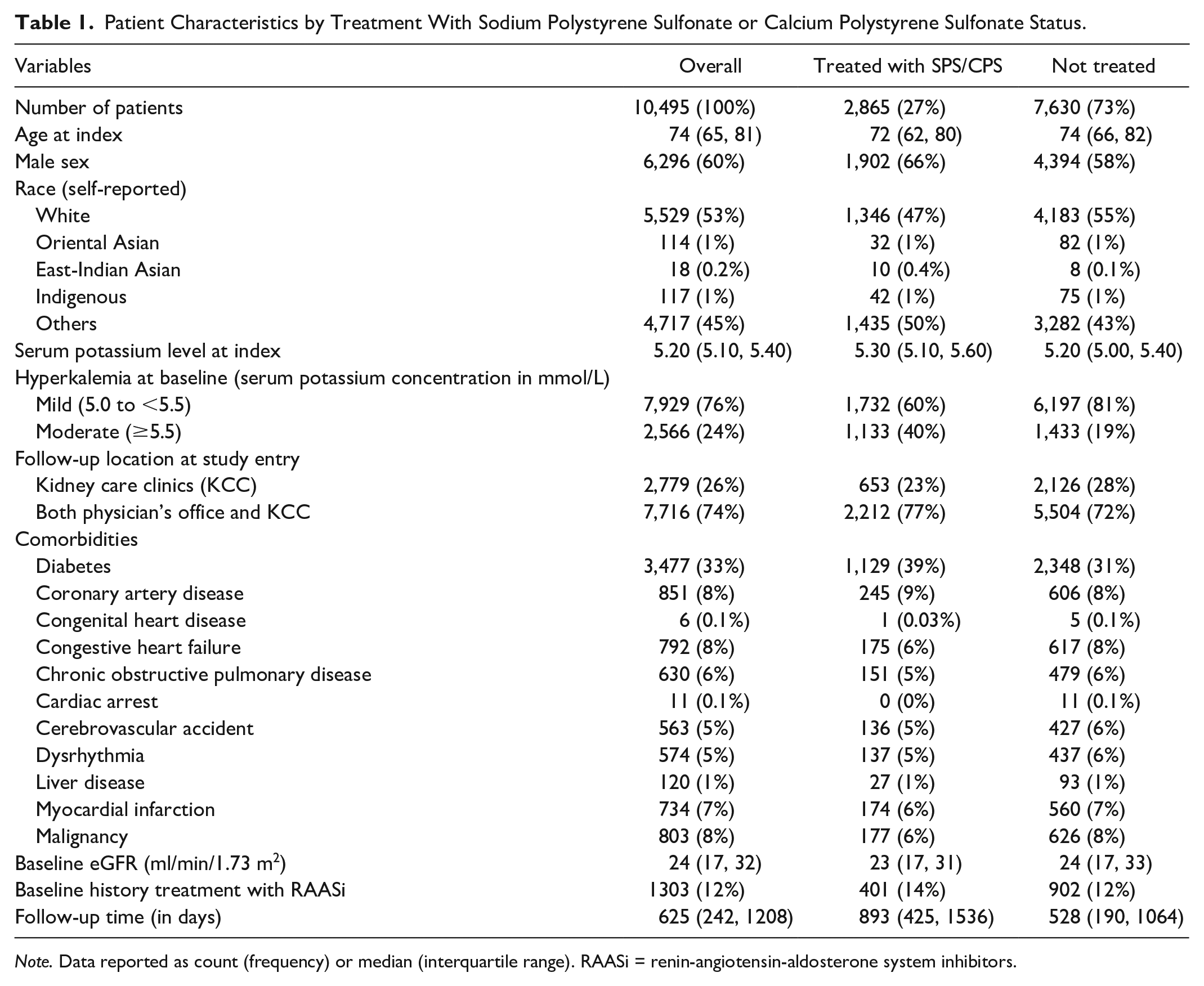
Note. Data reported as count (frequency) or median (interquartile range). RAASi = renin-angiotensin-aldosterone system inhibitors.
Median follow-up time was 625 days (IQR: 242-1208). Only 2864 (27%) CKD patients with chronic hyperkalemia were dispensed at least 1 prescription of either SPS or CPS. A total 7300 prescriptions were dispensed, of which 6428 (88%) were for SPS and remaining 872 (12%) were for CPS. Median number of prescriptions dispensed per patients was 2 (IQR: 1-3). It appeared that these patients were often not treated with SPS/CPS immediately after diagnosis with chronic hyperkalemia. The median time from the index date to the first prescription dispensing date was 154 days (IQR: 36-455). This latency period appeared to be a period of active monitoring by the nephrologists. The median number of serum potassium measurements was 5 (IQR: 2-13) during this period. Overall, 7.5, 15, and 30 gm were the most frequently prescribed doses, comprising 81% and 85% of SPS and CPS prescriptions, respectively (Figure 1). Once daily or 3 times weekly were the most common frequency for both SPS and CPS. However, the proportion of once daily dosage was much higher for CPS prescriptions compared with SPS prescriptions for all 3 doses. We also looked into the doses and frequencies that were prescribed to a subset of 649 patients for whom the mean serum potassium concentration was 5.58 mmol/L before the SPS/CPS treatment start date (see clinical outcome analysis below). The distribution was similar to the overall cohort.
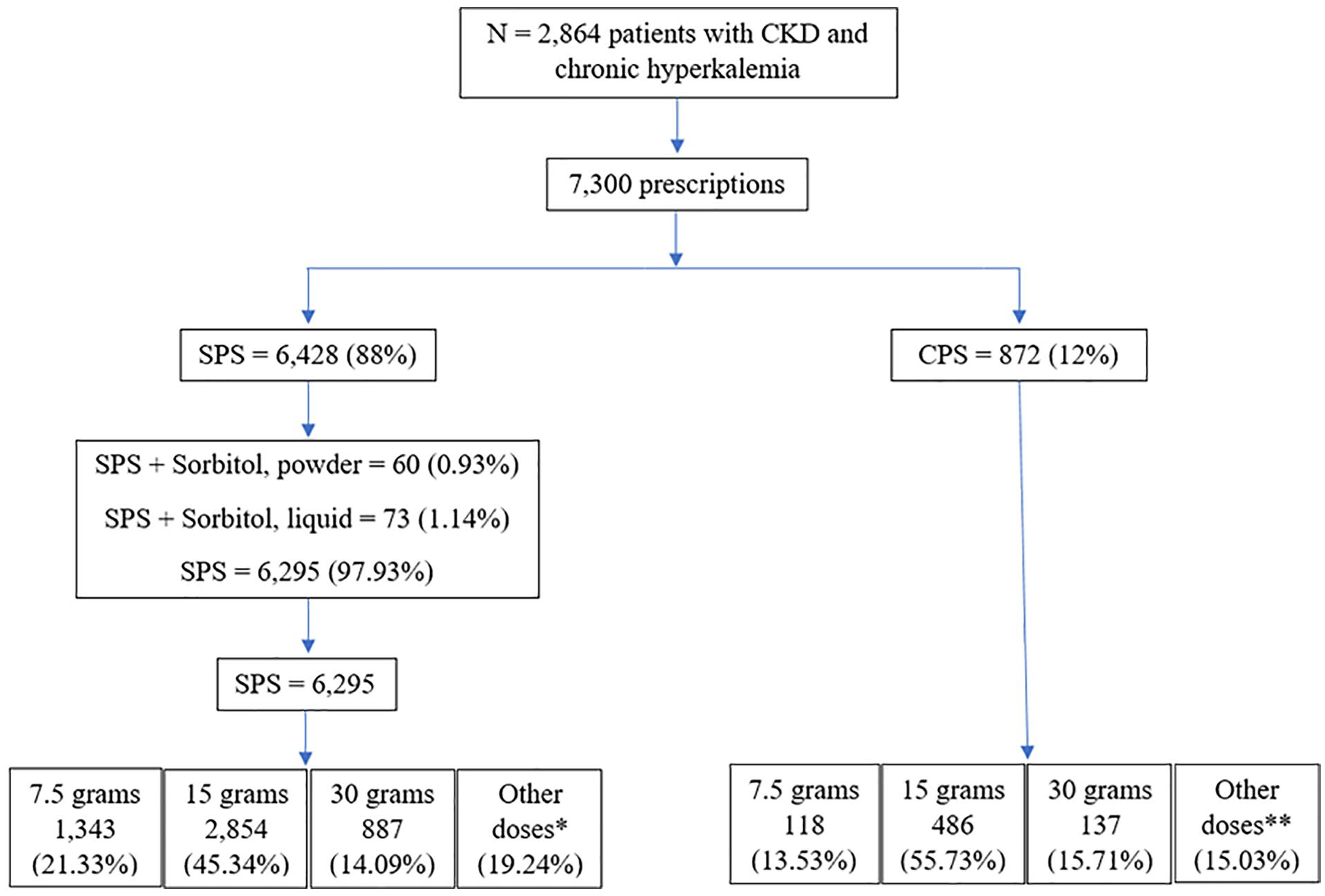
Distribution of doses for sodium polystyrene sulfonate (SPS) and calcium polystyrene sulfonate (CPS).
The 7300 SPS/CPS prescriptions dispensed resulted in 3302 continuous treatment episodes, ie, no gap more than 7 days between consecutive prescriptions. The median length of the first continuous treatment episode was 289 days (IQR: 82-693).
A total of 2101 (73%) of 2,864 chronic hyperkalemia patients who were treated with SPS/CPS had first continuous treatment episode of at least 90 days. Measurement of serum potassium concentration following 90 days of treatment was inconsistent. For example, 84 patients did not have serum potassium concentration recorded after 90 days of treatment. Median time of post-treatment serum potassium measurement was 23 days (IQR: 9-44), with a range of 0 to 637 days.
We restricted our clinical outcome analysis to 649 patients with chronic hyperkalemia who were continuously treated with SPS/CPS for at least 90 days and had a serum potassium concentration recorded within 15 days before SPS/CPS treatment start and had another serum potassium concentration recorded within 15 days from the 90th day of treatment. The mean serum potassium concentration increased by 0.16 mmol/L from index date to SPS/CPS treatment start date (5.42 at baseline vs 5.58 within 15 days before treatment started). A continuous 90-day treatment with SPS/CPS resulted in a mean decrease of 0.60 mmol/L, from 5.58 to 4.98 mmol/L (Table 2).
Effect of 90-Day Continuous Treatment With SPS/CPS on the Serum Potassium Concentration Among Chronic Kidney Patients With Chronic Hyperkalemia.

Note. CPS = calcium polystyrene sulfonate; IQR = interquartile range; SPS = sodium polystyrene sulfonate.
When looked into the severity of hyperkalemia, at baseline 61% experienced mild hyperkalemia versus 39% with moderate hyperkalemia. With a median of 154 days before the SPS/CPS treatment started, the proportion of moderate hyperkalemia was increased by 31%, ie, from 39% to 70%. Interestingly, 64 of 649 patients (10%) reached the normal serum potassium concentration level even before the start of SPS/CPS treatment. After a continuous 90-day treatment with SPS/CPS, a substantial proportion (45%) of the chronic hyperkalemia patients achieved the normal level. The proportion of moderate hyperkalemia decreased by 52%, ie, from 70% before treatment start to 18% after 90-day treatment with SPS/CPS (Table 3).
Effect of 90-Day Continuous Treatment With SPS/CPS on the Severity of Hyperkalemia.

Note. CPS = calcium polystyrene sulfonate; SPS = sodium polystyrene sulfonate.
Discussion
Multiple approaches exist to manage hyperkalemia, including conservative treatment with preventive approaches or pharmacological treatment with potassium binders such as SPS, CPS, and other newer agents. Recent research suggests that the treatment strategy in treating chronic hyperkalemia is not standardized. 10 The frequency and management practices of chronic hyperkalemia in CKD patients in British Columbia, Canada, had not previously been described. Thus, we undertook this observational study describing the SPS and CPS utilization pattern as well as the real-world effectiveness in treating chronic hyperkalemia among CKD patients in British Columbia, Canada. We found that a quarter of the CKD patients with chronic hyperkalemia were treated with potassium binders, primarily with SPS. In term of clinical effect, the average serum potassium concentration was decreased by 0.60 mmol/L after a 90-day continuous treatment with SPS/CPS.
In randomized control trials (RCTs), SPS appeared to reduce the mean serum potassium concentration by 1.25 to 1.5 mmol/L after only 3 to 7 days of treatment.12,13 The decrease in serum potassium concentration observed in the study sample was much less (−0.60 mmol/L change). This could be explained by several factors. First, the RCTs included CKD patients with only mild hyperkalemia, whereas approximately 24% of the British Columbia cohort had moderate hyperkalemia at baseline. The sample size was much less in RCTs, for example, 47 and 16 compared with more than 10 000 in the current study. Also, we allowed switching from SPS to CPS to capture the real-life trend in cation exchange resin use. Our findings were closer to the estimate from a previous retrospective observational study investigating the effectiveness of SPS in treating hyperkalemia. Batterink et al 14 reported a mean decrease of 0.58 in serum potassium after short-term treatment with SPS.
In clinical practice, the level of hyperkalemia to initiate treatment is not standardized. 10 This has been reflected in our study sample. We found that the patients with chronic hyperkalemia were not dispensed the SPS/CPS immediately after diagnosis. Rather, there appeared to be approximately 5 months period of active monitoring before initiating treatment. This latency in dispensing SPS/CPS appeared to be due to 2 reasons. First, due to engagement of dietitians, and review of impact of conservative interventions. Second, we used a broader definition of chronic hyperkalemia (serum potassium concentration ≥5.0 mmol/L) to capture the entire cohort of CKD patients with chronic hyperkalemia. It is possible that nephrologists may not initiate treatment for a serum potassium concentration of 5.0 mmol/L in patients with CKD but rather may have engaged in active monitoring and started SPS/CPS only when even higher serum potassium values were resulted. The median number of serum potassium measurements during this period was 5 (IQR: 2-13) that supports active monitoring. This would also be in keeping with our findings that indicate that compared with the baseline value, a much greater proportion of the serum potassium values immediately preceding SPS/CPS initiation tended to be in the moderately severe range as opposed to the mild range.
It was interesting to notice that approximately 10% of the study sample achieved normal concentration level before starting treatment with potassium binder. Perhaps this was due to dietary modifications, changes in existing medications, or other preventative approaches, which is out of scope of this analysis. In a retrospective cohort study investigating the SPS and CPS utilization pattern in patients with moderate hyperkalemia (serum potassium concentration ≥5.5 mmol/L) in CKD patients receiving RAASi, Ren et al 15 reported that only ~4% of the study population were dispensed a SPS or CPS prescription within 30 days from the hyperkalemia diagnosis date. Approximately 76% of our study population had mild hyperkalemia (<5.5 mmol/L) at baseline. As such, an active monitoring period of 154 days before treatment initiation appeared to be plausible.
The frequency at which the serum potassium concentration should be checked in chronic hyperkalemia patients is also not standardized. 10 In the study sample, we observed CKD patients with chronic hyperkalemia without a post-treatment record of serum potassium measurement. There was approximately 3 weeks gap between the 90th day of continuous treatment and serum potassium measurement with a median of 23 days (IQR: 9-44). Ren et al 15 reported a median time of 7 days (IQR: 3-29) between SPS or CPS prescription and post-treatment serum potassium measurement among patients with moderate hyperkalemia. Hence, a 3-week gap among British Columbia patients appeared to be plausible as majority (76%) of the British Columbia cohort were diagnosed with mild hyperkalemia. It is unlikely that the long-delayed measurements could be attributable to the treatment effect. This was an observation descriptive study. As such, establishing standard frequency based on health administrative data was out of scope of the project. Future research is required to standardize the frequency of testing that might inform practice.
Currently, only SPS and CPS are funded for treating chronic hyperkalemia in patients with CKD, and the newer medications for example patiromer and sodium zirconium cyclosilicate are not publicly funded in British Columbia, Canada. Long-term use of SPS and CPS in treating chronic hyperkalemia may be limited due to lack of evidence and increasing reports of serious gastrointestinal side effects.7,16 In a number of RCTs, patiromer has shown excellent efficacy and safety in treating hyperkalemia in CKD patients.4,7,10,16 Future research is necessary to prospectively evaluate the comparative efficacy and safety of current treatment options in British Columbia, including SPS/CPS with newer treatment choices, for example, patiromer and sodium zirconium cyclosilicate.
Our study has several strengths. First, to the best of our knowledge, this is the first observational study using population-level data to describe the utilization trend as well as clinical effectiveness of SPS/CPS in treating chronic hyperkalemia among CKD patients in British Columbia. The large sample size allowed us to understand the current real-life approach in treating hyperkalemia in CKD patients. The major limitation was that there was no control group in the study. However, in this descriptive observational study, our objective was to elucidate the current treatment approach. Thus, this is a relative limitation.
In conclusion, effective treatment is important to prevent the risk of morbidity and mortality associated with chronic hyperkalemia. In British Columbia, Canada only 1 in 4 CKD patients with chronic hyperkalemia received pharmacological therapy with SPS/CPS, mostly with higher degrees of hyperkalemia, and when it was used, the vast majority were used for a relatively long period. These medications appeared to be moderately effective in reducing the serum potassium concentration. However, cautions need to be exercised in interpreting this result as hyperkalemia can improve due to a number of other reasons, including changes in diet, de-prescribing RAASi or non-steroidal anti-inflammatory drugs, and addition of diuretics. In this descriptive observational study, establishing causality was not possible due to lack of a control group. Future research is necessary to evaluate the comparative effectiveness of these cation exchange resins (SPS/CPS) with an appropriate control group or other newer generation medications, including patiromer and sodium zirconium cyclosilicate.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
