Abstract
Background:
Iron deficiency is common in chronic kidney disease (CKD) patients not on dialysis (ND). Restoring depleted iron stores through intravenous (IV) route is faster and associated with less side effects. There is conflicting data regarding intravenous iron use and its impact on clinical outcomes in this population.
Objective:
This study aims at evaluating any negative clinical impact associated with IV iron use in CKD patients at stages (3-5) before dialysis.
Design:
Retrospective chart review.
Setting and Population:
Chart analysis of ND CKD 3-5 (estimated glomerular filtration rate [eGFR] <60 mL/min/1.73 m2) patients who received IV iron between January 2013 and January 2018 in 3 major hospitals in Kuwait.
Methods:
Outcomes analyzed were rates of all-cause hospitalizations, infection-related hospitalizations, mortality rates, and eGFR decline over 12 months after IV iron infusion in this population divided into 3 groups according to CKD stage.
Results:
A total of 738 patients were included in our analysis. Mean initial hemoglobin concentration was 111.5 ± 15.0 g/L in group 1 (CKD 3: eGFR 30-59 mL/min/1.73 m2), 103.6 ± 17.7 g/L in group 2 (CKD 4: eGFR 15-29 mL/min/1.73 m2), and 99.4 ± 14.5 g/L in group 3 (CKD 5: eGFR < 15 mL/min/1.73 m2 but not on dialysis). All-cause hospitalization and infection-related hospitalization were more common among group 3 subjects (adjusted odds ratio =2.12 [95% confidence interval, CI: 1.32-3.41] and 2.02 [95% CI: 1.15-3.55]), respectively. No deaths occurred during 12 months of follow-up.
Limitations:
Lack of control group, retrospective study.
Conclusion:
Intravenous iron use in CKD 3-5 ND is generally safe. Higher hospitalization rates in patients with eGFR <30 mL/min are possibly associated with lower baseline hemoglobin, lower baseline eGFR, and higher comorbidity burden, and not related to iron infusion.
Introduction
Anemia is a common manifestation in chronic kidney disease (CKD). 1 It results mainly from lack of erythropoietin production from diminished kidney function. Iron plays a major role in hemoglobin production, and lack of iron can lead to anemia in patients with CKD-ND (not on dialysis). 2 Low iron stores in patients with CKD is multifactorial in nature and can result from chronic bleeding due to uremia-associated platelet dysfunction, frequent phlebotomy, and impaired dietary iron absorption. 3 Chronic inflammation can play a significant role in iron metabolism by increasing hepcidin levels, resulting in reduced gastrointestinal iron absorption and iron delivery from body storage sites and can impede the use of iron for erythropoiesis, leading to an increase in ferritin levels. 4 Treatment involves the use of erythropoiesis-stimulating agents (ESAs) and iron, which can be administered orally or through intravenous (IV) infusion. According to the Kidney Disease: Improving Global Outcomes (KDIGO) and the Kidney Disease Outcomes Quality Initiative (KDOQI) guidelines, both routes are acceptable for CKD-ND patients with transferrin saturation (TSAT) ≤30% and ferritin levels ≤500 ng/mL.5,6
Intravenous iron is more effective than oral iron for increasing hemoglobin and TSAT levels, lowering ESA dosage requirements, reducing the pill burden associated with oral iron, and preventing gastrointestinal upset. 7 However, excessive IV iron may increase free iron, which can lower immunity by helper T-cell depletion and poor phagocytosis, increase bacterial growth and virulence (iron is essential micronutrient for microorganisms), endothelial injury, and monocyte adhesion due to oxidative stress which hastens atherosclerosis. Free iron may get deposited in sensitized microvasculature increasing the risk of calcific uremic arteriolopathy (CUA). 8
Moreover, there are conflicting reports on the impact of IV iron on hospitalization and infection rates, mortality rates, and decline in estimated glomerular filtration rates (eGFR).
In this study, we aim to evaluate whether there is any negative impact of IV iron use on clinical outcomes in CKD 3-5 ND patients.
Methods
Data Curation
We retrospectively reviewed the files of patients who had received IV iron therapy in outpatient setting from 3 major public hospitals in Kuwait (Adan, Mubarak, and Jahra) between January 1, 2013, and January 1, 2018. Included patients were adults >18 years of age with established CKD as defined in KDIGO. 5 Exclusion criteria: patients on dialysis, kidney transplant patients, glomerulonephritis patients on immunosuppressive therapy, and patients with active malignancy. Indications for IV iron were a hemoglobin level <100 g/L and/or TSAT <30%. Patients were then divided into 3 groups: group 1 included patients with an eGFR between 30 and 59 mL/min, group 2 included patients with an eGFR between 15 and 29 mL/min, and group 3 included patients with eGFR <15 mL/min but not yet on dialysis. Estimated glomerular filtration rate values were calculated using the Chronic Kidney Disease Epidemiology Collaboration formula (CKD-EPI). We collected patients’ demographic data, including age, sex, comorbidities, as well as use of rennin-angiotensin-aldosterone system (RAAS) blockers and ESAs. Laboratory parameters collected included hemoglobin, eGFR, TSAT, and parathyroid hormone (PTH). Laboratory values and eGFR levels of each patient were collected at the time of infusion and 12 months later. Infection rates were calculated based on patients’ charts within a period of 12 months from the first IV iron infusion date. Only infections that required hospitalization were included: chest infections, urinary tract infections, and diabetic foot infections. We also collected data on mortality and any adverse reactions occurring within the 12-month follow-up.
The study aimed at evaluating any negative clinical outcomes such as all-cause hospitalization rates, infection-related hospitalization rates, and the rate of decline in eGFR at 12 months after administering IV iron. Data were analyzed between the 3 groups. Subjects with multiple hospitalizations were counted only once in the analysis and a percentage was calculated for each group. Study was approved by the Joint Ministry of Health and the Kuwait University Committee on Medical Research.
Statistical Analysis
The data analysis was performed using the statistical software Stata (version 16, Stats Corp, College Station, TX, USA). Continuous variables were reported using the mean ± standard deviation, and categorical variables were reported using numbers and percentages. An unpaired Student t-test was applied to compare the mean values of continuous variables between eGFR groups, and a chi-square test was applied for categorical variables. Binary univariable logistic regression was first performed for each of the outcome: all-cause hospitalization and infection-related hospitalization. Variables with P <.10 were included in the multivariable logistic regression in addition to the 2-way interaction between eGFR and age, and between eGFR and comorbidity to assess the pattern of eGFR on outcome according to age and comorbidity. A linear mixed model was applied to assess the estimated marginal changes in hemoglobin and TSAT levels between the baseline and at 12 months within and between the eGFR groups after controlling for plausible confounders (age, gender, total iron dose, and comorbidities) within and between groups. The change in mean hemoglobin and TSAT levels was also calculated between the groups using the interaction between time and baseline and final values. A P-value of less than .05 was considered significant. The chi-square goodness-of-fit test was performed to reject the null hypothesis.
Results
The study included 738 patients (Table 1 shows their characteristics and baseline laboratory data): 289 (39%) were assigned to group 1 (eGFR between 30 and 59 mL/min), 270 (37%) were assigned to group 2 (eGFR 15-29 mL/min), and 179 (24%) were assigned to group 3. As shown in Table 1, the 3 groups were heterogeneous, with a significant female predominance (66%). Only 32% of patients were on RAAS blockers, with a higher usage rate in group 1. Erythropoiesis-stimulating agents were administered to 52%, highest in group 3. Iron saccharate was used in 76.6% of the patients and was administered in doses of 900 to 1000 mg divided in multiple sessions, and ferric carboxymaltose was used in 23.4% and was administered in doses of 1000 mg in 1 or 2 sessions.
Patients’ Baseline Characteristics and Initial Laboratory Data.

Note. DM = diabetes mellitus; HTN = hypertension; BMI = body mass index; DLP = dyslipidemia; CAD = coronary artery disease; eGFR = estimated glomerular filtration rate; Hb = hemoglobin; TSAT = transferrin saturation; PTH = parathyroid hormone; ESA = erythropoiesis-stimulating agent; RAAS = renin-angiotensin-aldosterone system; ANOVA = analysis of variance.
One-way ANOVA.
Patients in group 1 were younger (mean age of 55.3 years compared to 57 years in group 3). Hypertension and diabetes were highly prevalent in all patients, 94% and 65%, respectively. Mean initial hemoglobin was lowest in group 3, 99.4 g/L compared to 111.5 g/L in group 1. Mean initial TSAT was 16.2% in all patients (Table 1).
Table 2 shows the estimated marginal mean values for hemoglobin, TSAT, and eGFR at baseline and at 12 months after adjusting for age, gender, total iron dose, and comorbidities. Mean hemoglobin and TSAT were significantly higher at 12 months compared to baseline in all groups.
Change in Mean for Hemoglobin and TSAT at 12 Months Compared With Baseline in Stage III, IV, and V Adjusting for Age, Gender, Total Iron Dose, and Comorbidity.

Note. TSAT = transferrin saturation; CI = confidence interval.
Hospitalization rates were highest in group 3 (45.8%) compared to 33% and 19.7% in groups 2 and 1, respectively. Infection-related hospitalization was also higher in group 3 (27.4%) compared to 12% in group 1 (Table 3).
Rates of Hospitalization and Hospitalization for Infections in All Groups.

Note. UTI = urinary tract infection.
The association of hospitalization in patients who received IV iron was significant in advanced CKD (group 3) with an adjusted odds ratio (OR) of 2.12 [1.32-3.41], P = .002 (Table 4). Infection risk was higher in group 3 patients who received IV iron with adjusted OR of 2.02 [1.15-3.55], P = .014 (Table 5).
The Association of Hospitalization With Other Risk Factors in Patients Who Received Intravenous Iron.
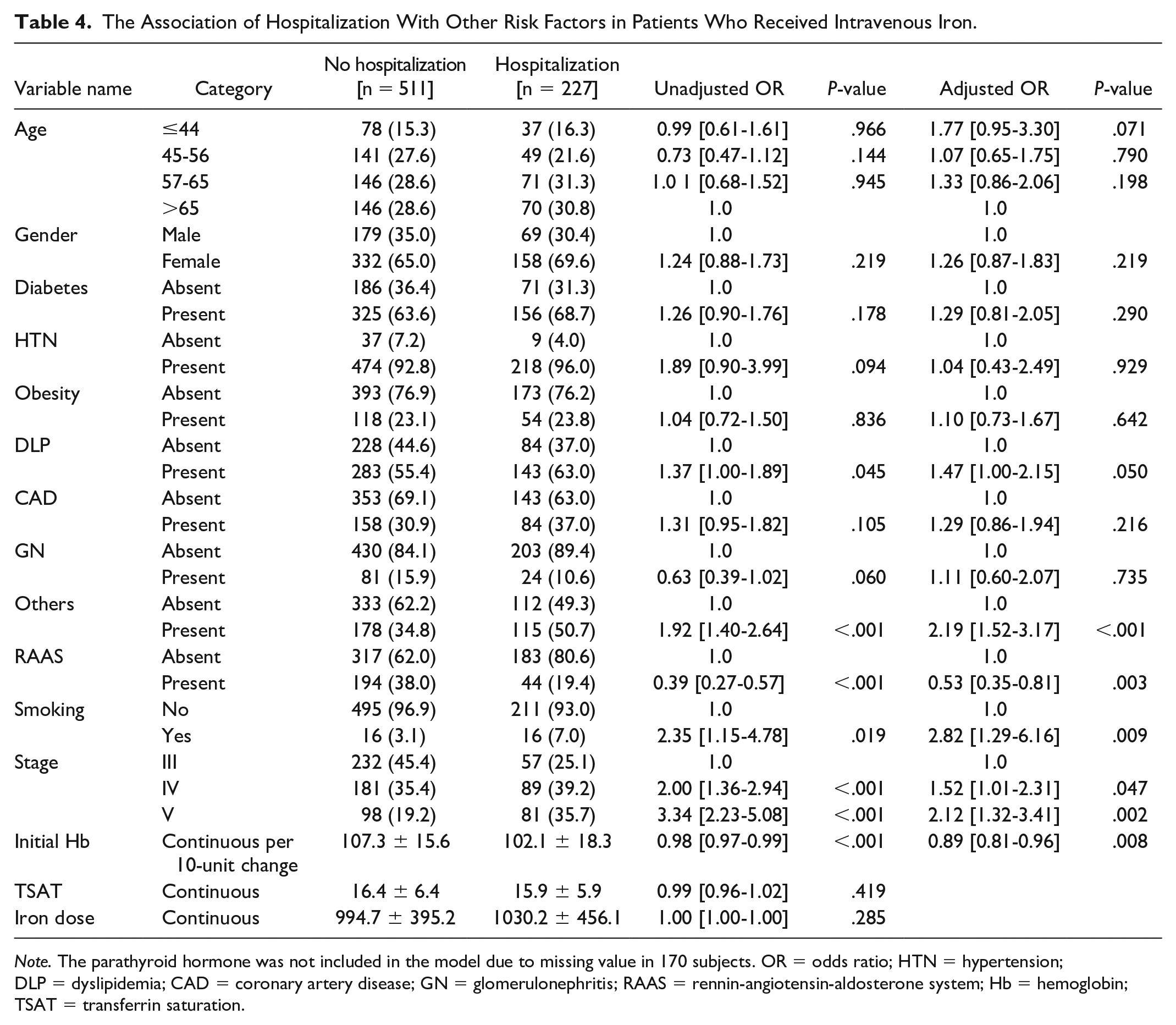
Note. The parathyroid hormone was not included in the model due to missing value in 170 subjects. OR = odds ratio; HTN = hypertension; DLP = dyslipidemia; CAD = coronary artery disease; GN = glomerulonephritis; RAAS = rennin-angiotensin-aldosterone system; Hb = hemoglobin; TSAT = transferrin saturation.
The Association of Infection With Other Risk Factors in Patients Who Received Intravenous Iron.

Note. The parathyroid hormone was not included in the model due to missing value in 170 subjects. OR = odds ratio; HTN = hypertension; DLP = dyslipidemia; CAD = coronary artery disease; GN = glomerulonephritis; RAAS = renin-angiotensin-aldosterone system; Hb = hemoglobin; TSAT = transferrin saturation.
The chi-square goodness-of-fit test was performed to reject the null hypothesis that there are statistically significant, χ2(2) = 28.1, P < .001, differences in the 3 stages (Stage III n = 289; Stage IV n = 270; Stage V n = 179) (Supplement Table 1).
Discussion
Our study confirmed the utility of IV iron infusion in CKD 3-5ND that resulted in an average increase of 6 to 11 g/L of hemoglobin and 9% to 12% in TSAT levels during 12-month follow-up. In the literature, there is conflicting evidence with regard to the direct impact of IV iron on mortality rates, hospitalization rates, cardiovascular events, infection rates, and rates of GFR decline in CKD-ND patients. Observational data, meta-analyses, and data from large hemodialysis registries on the safety of IV iron report conflicting results regarding mortality rates.9,10 Several studies have shown that IV iron has no impact on hemodialysis patients11 -14 or on hemodialysis and patients with CKD. 15 Two studies, using data from the Dialysis Outcomes and Practice Patterns Study and from a large dialysis provider in the United States,16,17 have shown an increased mortality rate in hemodialysis patients with higher IV doses. However, a national database analysis of an Asian population showed lower mortality in patients with CKD-ND. 18
Evidence is again conflicting regarding hospitalization rates, cardiovascular events, and infections. Intravenous iron could theoretically increase the risk of infection through the impairment of neutrophil and T-cell functions and the promotion of microbial growth. 19 However, evidence associating IV iron with increased infection risk in patients with CKD is scarce, mainly because of the heterogeneity of the studies and biases in treatment indications. 10 A meta-analysis 20 and the Ferinject Assessment in Patients with Iron Deficiency Anemia and Non-Dialysis-Dependent Chronic Kidney Disease (FIND-CKD) study 21 showed no increase in the risk of infections, cardiovascular events, or hospitalization rates among patients who received IV iron compared with those taking oral iron. However, the opposite result was reported by the single-center Randomized Trial to Evaluate Intravenous and Oral Iron in Chronic Kidney Disease (REVOKE) trial, 22 which was terminated early due to an increased risk of infections and cardiovascular events in patients who received IV iron compared with those taking oral iron.
In our cohort, we observed no deaths over the 12 months, although 31% of the patients required hospitalization, mainly for non-infection reasons (82%). Subjects on RAAS blockers had a reduced likelihood of hospitalization (Table 4). The all-cause and infection-related hospitalization rates were higher in CKD 5-ND patients. However, the need for hospitalization in our patients was likely due to lower baseline eGFR, lower hemoglobin, and comorbidities.
Finally, regarding the impact of IV iron on eGFR decline in CKD-ND patients, the rate of eGFR decline was similar between IV and oral iron groups in randomized trials.21,22 However, IV iron was reported to be associated with a higher risk of faster progression to end-stage kidney disease in a national database analysis of an Asian population. 18 The rate of eGFR decline in our groups was statistically significant, albeit small.
The limitations of our study include its observational and retrospective nature and as well as being subject to biases and confounders. As we lacked control group, we can only state the effect of IV iron on certain outcomes in patients with eGFR less than 60 mL/min. Another limitation was the lack of knowledge on the length of time between IV iron administration and hospitalization. We also did not collect data on other diseases that may increase the risk of infections, such as malignancy, or immunosuppressive medications for immune diseases like glomerulopathies. Nevertheless, this study was one of the largest from the Middle East (738 patients) to evaluate IV iron safety in CKD-ND patients followed for an adequate period. Another strength of our study was that it demonstrated that IV iron does not increase mortality, and hospitalization was more likely to be due to the patients’ comorbidities and lower eGFR.
Conclusion
Intravenous iron is well tolerated in CKD 3-5ND patients and effective at improving hemoglobin and TSAT levels, with a small decline in eGFR within 12 months. Hospitalization after the use of intravenous iron (after adjusting for confounders) appears to be related to the patients’ low eGFR and medical conditions (smoking, dyslipidemia, and low hemoglobin level at baseline) and not to the iron infusion. In addition, subjects on RAAS blockers had a lower hospitalization rate than subjects not on RAAS blockers. There was no apparent association between IV iron use and increased mortality.
Supplemental Material
sj-docx-1-cjk-10.1177_20543581221130685 – Supplemental material for Safety of Intravenous Iron in CKD Stage 3-5 Patients Not on Dialysis: A Retrospective Cohort Study
Supplemental material, sj-docx-1-cjk-10.1177_20543581221130685 for Safety of Intravenous Iron in CKD Stage 3-5 Patients Not on Dialysis: A Retrospective Cohort Study by Ali AlSahow, Anas AlYousef, Hani Nawar, Bassam AlHelal, Ahmed AlQallaf, Abdullah AlMuhaiteeb, Yasser ElSharkawi, Heba AlRajab, Hamad Ali and Rajeev Kumar in Canadian Journal of Kidney Health and Disease
Footnotes
Ethics Approval and Consent to Participate
Study was approved by the Joint Committee on Medical and Scientific Research.
Consent for Publication
All authors provided consent for publication.
Availability of Data and Materials
Data can be provided by the corresponding author in case of a reasonable request.
Author Contributions
All authors contributed to the design of this study, the acquisition, analysis, and interpretation of data, the drafting of the study, and the final approval of this manuscript. All authors agree to be accountable for all aspects of this study in ensuring that questions related to the accuracy and integrity of any part of the study are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
