Abstract
Background:
There is limited evidence on the association of serum phosphate with mortality in patients receiving continuous renal replacement therapy (CRRT).
Objective:
To assess the association of serum phosphate with mortality in critically ill patients requiring CRRT for acute kidney injury (AKI).
Design:
A cohort study.
Setting:
A tertiary referral hospital in the United States.
Patients:
Acute kidney injury patients receiving CRRT from 2006 through 2015 in intensive care units.
Measurements:
(1) Serum phosphate before CRRT and (2) mean serum phosphate during CRRT were categorized into 3 groups; ≤2.4 (hypophosphatemia), 2.5 to 4.5 (normal serum phosphate group), and ≥4.6 (hyperphosphatemia) mg/dL.
Methods:
Multivariable logistic regression was used to assess the association between serum phosphate and 90-day mortality.
Results:
A total of 1108 patients were included in this study. Of these, 55% died within 90 days after CRRT initiation. Before CRRT, 3%, 30%, and 66% had hypophosphatemia, normophosphatemia, and hyperphosphatemia, respectively. Before CRRT, both hypophosphatemia and hyperphosphatemia were significantly associated with higher 90-day mortality with the adjusted odds ratio (OR) of 2.22 (95% confidence interval [CI]: [1.03, 4.78]) and 1.62 (95% CI: [1.21, 2.18]), respectively. During CRRT, 3%, 85%, and 12% had mean serum phosphate in hypophosphatemia, normophosphatemia, and hyperphosphatemia range. During CRRT, hyperphosphatemia was significantly associated with higher 90-day mortality with adjusted OR of 2.22 (95% CI: [1.45, 3.38]).
Limitations:
Single center, observational design, lack of information regarding causes of serum phosphate derangement.
Conclusion:
Most CRRT patients had hyperphosphatemia before CRRT initiation but maintain normal serum phosphate during CRRT. Before CRRT, hypo- and hyperphosphatemia, and during CRRT, hyperphosphatemia predicted higher mortality.
Trial registration:
Not registered.
Introduction
Phosphorus is the most abundant intracellular anion that is responsible for the integrity and structure of cell membranes through phospholipids, cellular signaling, and nucleotide metabolism through adenosine monophosphates and triphosphates and bone mineralization.1 -3 Phosphate is present in the serum as an inorganic form, and normal levels range between 2.5 to 4.5 mg/dL. 4 Serum phosphate derangements, ie, hypophosphatemia and hyperphosphatemia, in critically ill patients are due to redistribution from transcellular shifts, malnutrition, impaired excretion due to kidney injury, acid-base disturbances, and tissue injury. 5 In critically ill patients, the prevalence of hypophosphatemia ranges between 15% and 45%, while hyperphosphatemia has been reported to be around 26%, depending on the study population.6,7 Serum phosphate derangements are associated with acute kidney injury (AKI), respiratory failure, myocardial dysfunction, ventricular arrhythmias, hemolysis, and impaired neuromuscular activity resulting in catastrophic outcomes in critically ill patients.8 -10
Continuous renal replacement therapy (CRRT) is a dialysis modality that is used in critically ill patients with AKI or end-stage kidney disease (ESKD) in which there is a slower solute and fluid removal when compared with intermittent hemodialysis, leading to a lower propensity to hemodynamic instability. 11 The literature highlights that patients who need CRRT have a substantially high in-hospital mortality ranging from 30% to 60%.12,13 Several clinical characteristics predicted mortality in CRRT patients.12 -14 Among these factors is the derangement of various electrolytes reported to be associated with higher mortality in CRRT patients.15 -18 Although serum phosphate derangement has been associated with increased mortality risk in various study populations such as hospitalized individuals, particularly critically ill and septic patients,5,7,19 -24 there is limited evidence on whether serum phosphate is associated with mortality or has any predictive value in critically ill AKI patients receiving CRRT. Therefore, this study aimed to determine the mortality risk based on serum phosphate levels before CRRT and during CRRT among AKI patients requiring CRRT.
Materials and Methods
Setting and Study Population
A single-center cohort study was conducted at Mayo Clinic Hospital, Minnesota, USA. All adult patients (age ≥ 18 years) who received CRRT in intensive care unit (ICU) from December 2006 to November 2015 were screened. We included patients who had available serum phosphate measurement within 24 hours before CRRT initiation and those who received CRRT for at least ≥24 hours. We excluded patients with ESKD or those who received any dialysis modalities within 7 days before CRRT initiation, were moribund, and died within 24 hours of CRRT initiation. If a single patient received multiple CRRT sessions, we only included the first session in the analysis. However, we considered CRRT sessions with the interruption between sessions of ≤24 hours as a single CRRT session to account for CRRT interruption due to tests or procedures. The Mayo Clinic institutional review board approved this study and waived the need for informed consent for patients who provided research authorization due to the minimal risk of this retrospective medical review study (IRB number 10-000657). We excluded patients without research authorization.
Continuous Renal Replacement Therapy
The detail of the CRRT setting in our hospital was previously described.15-17 In brief, continuous venovenous hemofiltration was the only CRRT modality performed in our hospital. Continuous renal replacement therapy was prescribed and managed by the nephrology-intensive care service. The standard CRRT order set included the blood flow rate of 200 mL/min, and the replacement fluid rate of 30 mL/kilogram of body weight/hour, in which 50% were delivered as prefilter replacement fluid. Standard replacement fluids used during the study period were phosphate-free fluids, and we used continuous intravenous sodium phosphate infusion to maintain normal serum phosphate levels. In addition, we used regional citrate anticoagulation with a dextrose citrate solution formula A unless there was an absolute contraindication. Owing to regional citration anticoagulation, all patients received continuous intravenous calcium chloride infusion to maintain normal serum ionized calcium. The comprehensive electrolyte panel, including serum phosphate, was monitored at least every 8 to 12 hours throughout the CRRT session. While dialysis nurses set up and periodically assessed the CRRT circuit, intensive care nurses handled the beside CRRT management.
Data Collection and Definitions
Clinical characteristics, treatments, and laboratory data were abstracted from the hospital and ICU databases. The primary predictors were (1) serum phosphate before CRRT, defined as the most recent serum phosphate measured within 24 hours before CRRT initiation and (2) the mean serum phosphate during CRRT. The primary outcome was 90-day mortality after CRRT initiation, obtained from our institutional registry and the Social Security Death Index database.
Statistical Analysis
Clinical characteristics were compared between serum phosphate groups using the analysis of variance (ANOVA) test for continuous variables and the chi-square test for categorical variables. Serum phosphate was categorized into 3 groups according to the reference range in our hospital, ie, ≤2.4 (hypophosphatemia), 2.5 to 4.5 (normal serum phosphate group), and ≥4.6 (hyperphosphatemia) mg/dL. Logistic regression was performed to estimate the odds ratio for 90-day mortality based on (1) serum phosphate before CRRT and (2) the mean serum phosphate during CRRT, using the normal range of serum phosphate, ie, 2.5 to 4.5 mg/dL, as the reference group. Multivariable analysis was performed to adjust the associations for priori-defined clinically pertinent variables, including age, sex, race, body mass index, Charlson comorbidity index, SOFA score, sepsis, mechanical ventilation, vasopressor use, fluid balance before CRRT, ICU type, and baseline serum creatinine at hospital admission. The association of the mean serum phosphate with mortality was additionally adjusted for serum phosphate before CRRT. A 2-tailed P value <.05 was considered statistically significant. All analyses and model buildings were performed using RStudio, version 1.3.1093, RStudio PBC, 2009-2020.
Results
Clinical Characteristics
One thousand seven-hundred fifty-eight patients underwent CRRT in ICU. Of these, we excluded 157 patients who with ESKD or need for dialysis before CRRT initiation, 216 patients who received CRRT for <24 hours, and 277 patients who did not have serum phosphate measured within 24 hours before CRRT initiation. Therefore, there were 1,108 eligible patients in the final analysis. Table 1 shows the clinical characteristics of all included patients. The mean age was 61 ± 15 years, 58% (n = 645) were male, and 87% (n = 968) were white. Seventy-two percent (n = 794) and 64% (n = 714) were on mechanical ventilation and vasopressors, respectively, at CRRT initiation. Twenty-eight percent (n = 306) were admitted in medical ICU, 20% (n = 225) in surgical ICU, 16% (n = 176) in cardiac surgery ICU, 7% (n = 84) in cardiac ICU, and 29% (n = 317) in mixed ICU. The median duration of CRRT was 6 (interquartile range [IQR]: 4–9) days. Fifty-five percent (n = 607) of patients died within 90 days following the CRRT initiation.
Clinical Characteristics of Included Patients According to Serum Phosphate Before CRRT Initiation.

Note. Continuous data are presented as mean ± SD. Categorical data are presented as count (%). BMI = body mass index; COPD = chronic obstructive pulmonary disease; CRRT = continuous renal replacement therapy; ICU = intensive care unit; eGFR = estimated glomerular filtration rate; SOFA score = Sequential Organ Failure Assessment score.
Serum Phosphate Before CRRT and Mortality
The mean serum phosphate before CRRT was 5.7 ± 2.2 mg/dL. Before CRRT, 3% (n = 36) had hypophosphatemia, 30% (n = 336) had normal serum phosphate, and 66% (n = 736) had hyperphosphatemia. Table 1 shows clinical characteristics according to serum phosphate levels before CRRT. Compared to normal serum phosphate group, both hypophosphatemia and hyperphosphatemia groups were younger and had less comorbidity burden. However, hypophosphatemia group was more female and likely to be admitted to cardiac surgery ICU. In contrast, hyperphosphatemia group was more male, and likely to be admitted to medical and mixed ICU but less likely to be admitted to cardiac surgery ICU. Hyperphosphatemia group had higher serum creatinine but lower estimated glomerular filtration rate.
The 90-day mortality was 61% in hypophosphatemia, 49% in the normal serum phosphate group, and 57% in hyperphosphatemia. In adjusted analysis, hypophosphatemia and hyperphosphatemia before CRRT were significantly associated with increased 90-day mortality with odds ratio (OR) of 2.22 (95% confidence interval [CI]: [1.03, 4.78]) and 1.62 (95% CI: [1.21, 2.18]), respectively (Table 2).
The Association Between Serum Phosphate Levels Before CRRT Initiation and 90-Day Mortality.

Note. CI = confident interval; OR = odds ratio; CRRT = continuous renal replacement therapy; ICU = intensive care unit; SOFA score = Sequential Organ Failure Assessment score.
Adjusted for age, sex, race, body mass index, Charlson comorbidity index, SOFA score, sepsis, mechanical ventilation, vasopressor, fluid balance, ICU type, and baseline serum creatinine.
Serum Phosphate During CRRT and Mortality
During CRRT, the mean serum phosphate was 3.7 ± 1.0 mg/dL. While on CRRT, 3% (n = 31) had a mean serum phosphate in hypophosphatemic range, 85% (n = 940) in normal serum phosphate range, and 12% (n = 137) in hyperphosphatemic range. Table 3 shows clinical characteristics according to the mean serum phosphate during CRRT. The hypophosphatemia group was more likely to be female, have a higher Charlson Comorbidity Index and be admitted to the medical ICU. In contrast, the hyperphosphatemia group was more likely to be male, with a higher SOFA score, and admitted in a mixed ICU.
Clinical Characteristics of Included Patients According to the Mean Serum Phosphate During CRRT.
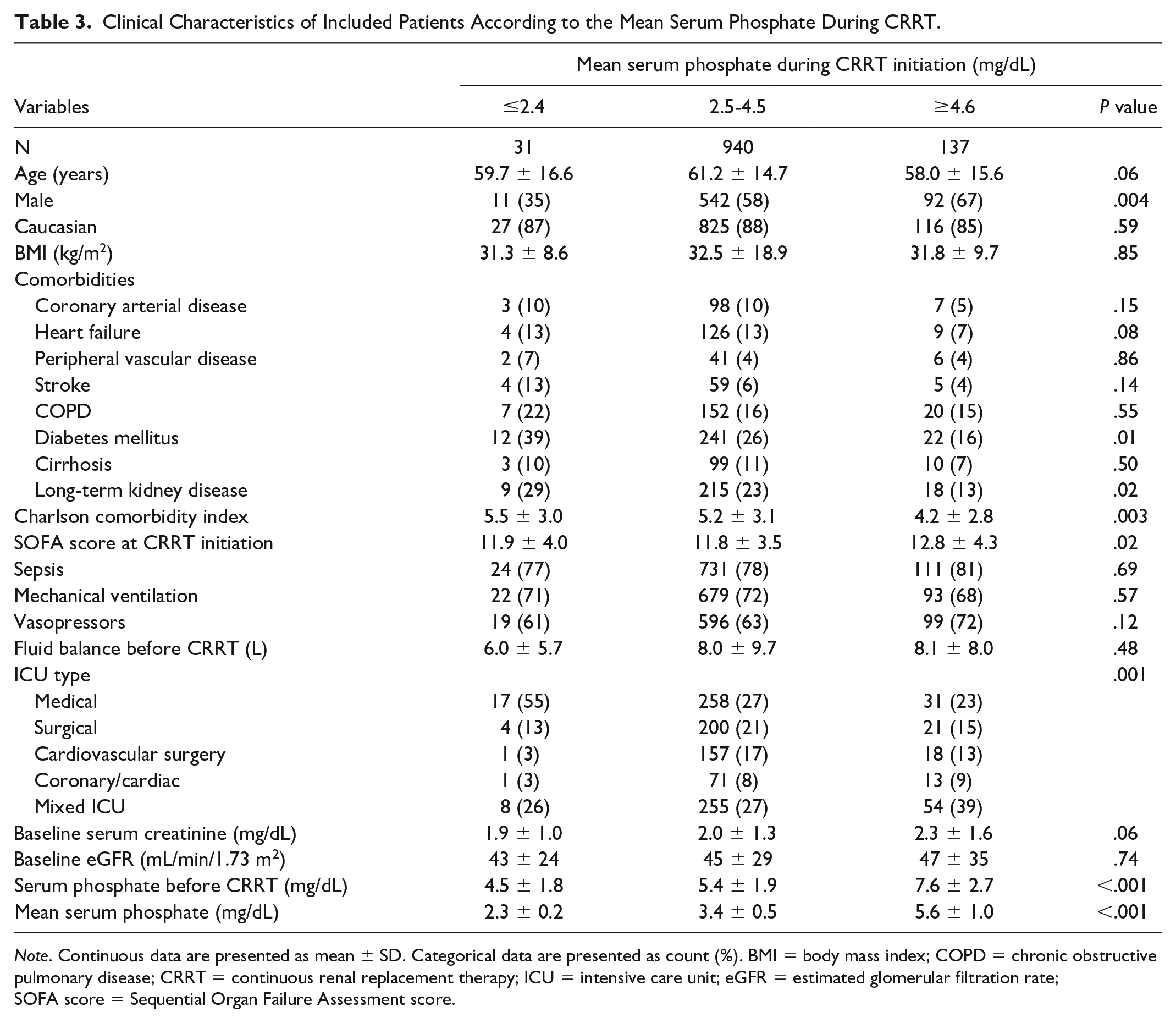
Note. Continuous data are presented as mean ± SD. Categorical data are presented as count (%). BMI = body mass index; COPD = chronic obstructive pulmonary disease; CRRT = continuous renal replacement therapy; ICU = intensive care unit; eGFR = estimated glomerular filtration rate; SOFA score = Sequential Organ Failure Assessment score.
The 90-day mortality was 42%, 53%, and 69% in patients with mean serum phosphate during CRRT in hypophosphatemia, normal serum phosphate, and hyperphosphatemia group, respectively. In adjusted analysis, hyperphosphatemia during CRRT was significantly associated with increased 90-day mortality with OR of 2.22 (95% CI: [1.45, 3.38]). However, hypophosphatemia during CRRT was not significantly associated with mortality (Table 4).
The Association Between the Mean Serum Phosphate During CRRT and 90-Day Mortality.

Note. CI = confident interval; OR = odds ratio; CRRT = continuous renal replacement therapy; ICU = intensive care unit; BMI, body mass index; SOFA score = Sequential Organ Failure Assessment score.
Adjusted for age, sex, race, BMI, Charlson Comorbidity Index, SOFA score, sepsis, mechanical ventilation use, vasopressor, ICU type, baseline serum creatinine, and serum phosphorus before CRRT.
Discussion
In a large cohort of ICU patients requiring CRRT, while hypophosphatemia was not common, two-third of patients were hyperphosphatemic before CRRT initiation. Higher 90-day mortality was observed when serum phosphorus before CRRT initiation was ≤2.4 mg/dL and ≥4.6 mg/dL compared to normophosphatemia. In contrast, higher mortality was noted in only patients who had mean serum phosphate ≥4.6 mg/dL during CRRT. These findings demonstrate the prognostic importance of serum phosphorus before and during CRRT on mortality in CRRT patients.
Our study results are consistent with most of the studies that have described the mortality association with serum phosphate derangements. In a study done by Shor et al, 22 severe hypophosphatemia (serum phosphate <1 mg/dL) was associated with poor outcomes and increased risk of mortality in patients with sepsis. Wang et al 23 reported that hypophosphatemia at the time of admission was associated with longer ICU stay, prolonged mechanical ventilation, and independent risk factor of 28-day mortality in critically ill patients. Suzuki et al 25 and Haider et al 7 contradicted the above findings by showing that hypophosphatemia was not an independent predictor of mortality in critically ill patients after adjusting for illness severity. We suspect that the contradicting results may be due to sample size, study population, and varying laboratory ranges affecting the generalizability of the results. In contrast, hyperphosphatemia has been consistently associated with increased mortality in various study populations such as hospitalized patients, critically ill patients, severe burns, and septic shock patients.5,7,26,27 When compared to other studies described above, our study included a critically ill AKI patients needing CRRT and showed that hypophosphatemia and hyperphosphatemia before CRRT were associated with increased 90-day mortality after adjusting for severity of illness. In contrast, prior studies demonstrated higher serum phosphate at CRRT initiation was associated with increased mortality in AKI patients requiring CRRT.21,28
Serum phosphate derangements are common in critically ill patients due to various reasons. Hypophosphatemia can occur due to transcellular shifts due to hormones such as insulin, glucagon, medications such as vasopressors, refeeding syndrome, respiratory alkalosis from mechanical ventilation, and sepsis.1,29 In addition, alcoholism, kidney losses due to tubulopathy, or the use of diuretics and continuous renal replacement therapies are associated with hypophosphatemia.1,29,30 Hyperphosphatemia in critically ill patients can occur due to the release of intracellular phosphate in the setting of cellular lysis, eg, tumor lysis syndrome, rhabdomyolysis, shock liver, decreased renal clearance due to AKI or long-term kidney disease, and transcellular shifts due to metabolic acidosis. 1
The mortality association can be explained by the interference of hypophosphatemia on critical functions including glycolysis, generation of adenosine triphosphate (ATP) leading to derangements in cellular metabolism, structure, and function as well as tissue hypoxia resulting from the increased affinity of hemoglobin to oxygen due to changes in concentration of 2,3 diphosphoglycerate on erythrocytes. 30 This ultimately leads to multiorgan dysfunction, such as cardiac arrhythmias, respiratory failure, encephalopathy, seizures, hemolysis, and impaired leukocyte function. 8 On the contrary, hyperphosphatemia can lead to hypocalcemia which can interfere with neuromuscular conduction, cardiac function, and AKI development due to calcium phosphate crystallization.1,5 In addition, hyperphosphatemia can also cause inflammation and calcification of vascular smooth muscle leading to increased cardiovascular mortality. 26
Our study demonstrated that mean serum phosphate ≥4.6 mg/dL was associated with increased mortality in contrast to mean phosphate ≤2.4 mg/dL during CRRT. Similar to our study, Jung et al 21 and Wang et al 28 showed higher serum phosphate at 24 hours after CRRT was associated with increased mortality in AKI patients requiring CRRT. In addition, higher serum phosphate after CRRT initiation than the baseline levels before CRRT initiation was associated with increased mortality. Bai et al 31 found that serum phosphate at 24 hours after CRRT initiation was 1 of 4 variables in their final prediction model for mortality in AKI patients requiring CRRT. While hyperphosphatemia resolved in most patients after starting CRRT, our study did not investigate the causes of persistent hyperphosphatemia during CRRT. Accordingly, we could not delineate whether the mortality association was due to hyperphosphatemia or a result of unmeasured confounders related to underlying causes of persistent hyperphosphatemia. Factors such as inadequate CRRT dose, clotting of filters, CRRT interruption for procedures, ongoing cellular breakdown, increased catabolism, rhabdomyolysis, and worsening metabolic acidosis leading to transcellular shifts may lead to hyperphosphatemia during CRRT. These factors are associated with increased mortality risk in critically ill patients and may explain why hyperphosphatemia during CRRT was associated with higher mortality.
While hypophosphatemia before initiating CRRT was associated with mortality, hypophosphatemia during CRRT was not associated with mortality in our study. The difference in mortality was likely due to the different causes of hypophosphatemia between the 2 groups. While hypophosphatemia at the time of CRRT initiation is related to transcellular shifts, respiratory alkalosis, malnutrition as described above, these patients will likely become normophosphatemic during CRRT because continuous intravenous sodium phosphate infusion was given to maintain normal serum phosphate levels during CRRT. However, hypophosphatemia during CRRT is likely due to kidney losses signifying renal recovery, transcellular shifts due to cellular regeneration, signifying recovery from the critical illness. Hypophosphatemia in high-dose CRRT prescriptions is more common compared to standard-dose CRRT prescriptions due to increased phosphate losses in the effluent. 32 However, the 2 dosing groups did not have any difference in outcomes and mortality.
There are some inherent limitations in our study. This was an observational study; therefore, we could not conclude a causal relationship between serum phosphate derangement and mortality. In addition, there might be unknown confounders we did not account for in the adjusted analysis. Furthermore, we conducted this study using a large electronic database and could not investigate the cause and circumstance of serum phosphate derangement before and during CRRT. Therefore, we could not conclude whether higher mortality was mediated by serum phosphate derangement or unmeasured factors related to underlying causes of serum phosphate derangement. We did not have the data regarding the cause of death to more explicitly define their mechanistic linkage. In addition, the information on factors or treatments that can alter serum phosphate, eg, intravenous/enteral phosphate supplement, insulin, nutrition, acid/base status, was not available in our data set. Furthermore, the findings from our single hospital with most of the white patient population might have limited the generalizability of our results. Finally, our investigation only showed the association of serum phosphate with mortality based on a single value before CRRT and the average value during CRRT. Future studies assessing serum phosphate change, variability, and trajectory during CRRT might be informative.
Conclusion
The serum phosphate level among patients with AKI who need CRRT could be determined by different factors before and after CRRT initiation. Before CRRT initiation, as kidney clearance due to kidney function is very limited, its levels are often determined by its absorption or release from cells. However, after CRRT initiation, serum phosphate level would also be impacted by CRRT characteristics, including dialysis dose and phosphorus replacement rates. We reported that hyperphosphatemia and hypophosphatemia before CRRT initiation are associated with worse outcomes. This is while after CRRT initiation, only hyperphosphatemia is related to a higher rate of death. Refractory hyperphosphatemia after CRRT initiation is often due to the uncontrolled release of phosphorus from the damaged cells, which makes this relationship physiologically plausible. Therefore, serum phosphate levels before and during CRRT could be due to different mechanisms, and clinicians should remain cautious in their interpretation for prognostications.
Footnotes
Ethical Approval and Consent to Participate
The Mayo Clinic Institutional Review Board approved this study (IRB number 10-000657). The informed consent was waived by institutional review board due to minimal risk nature of the study. However, all patients provided authorization for their data use for research purpose.
Consent for Publication
All authors consent for publication of the manuscript in its current form.
Availability of Data and Material
No additional data and material are available.
Authors’ Note
Institution: This work was performed at Mayo Clinic in Rochester, MN.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
