Abstract
Background:
Renin-angiotensin system inhibitors (RASi) are not re-initiated for almost a quarter of patients who suffered acute kidney injury 6 months after discharge. This discontinuation might be partly explained by the nephrotoxicity of these medications, yet they remain of benefit, especially for patients with heart failure.
Objective:
To determine the factors deemed by clinicians to influence RASi re-initiation and set threshold values for important safety parameters.
Design:
Three-round modified online Delphi survey.
Setting:
The study was conducted in Quebec, Canada.
Participants:
Twenty clinicians from nephrology, intensive care medicine, and internal medicine.
Measurements:
The factors’ importance was rated on 4-point Likert-type scale, ranging from “not important” to “very important” by the panelists.
Methods:
We conducted a brief literature review to uncover possible influencing factors followed by a 3-round modified Delphi survey to establish a consensus on the importance of these factors.
Results:
We recruited 20 clinicians (7 nephrologists, 3 internists, and 10 intensive care physicians). We created a list of 25 factors, 15 of which met consensus. Eleven of these factors, including serum creatinine, glomerular filtration rate, and acute kidney injury (AKI) stage, were deemed as important while 4, such as responsibility ambiguity and absence of feedback, were deemed as not important. The majority of the 10 factors which did not meet consensus were related to the clinical setting, such as a pharmacist follow-up and the required time to ensure optimal RASi re-initiation.
Limitations:
Quebec clinicians’ agreement might not reflect the opinion of the rest of Canada. The survey measures clinicians’ belief rather than their actual practice.
Conclusion:
Renin-angiotensin system inhibitors re-initiation is a rather complex concept which encompasses several factors. Our research uncovered some of these factors which may be used to develop guidelines on optimal RASi re-initiation.
Introduction
Despite their benefit, renin-angiotensin system inhibitors (RASi) tend to be discontinued after hospital discharge for patients who suffered acute kidney injury (AKI). A study based on the Alberta health database uncovered that almost a quarter of hospitalized patients taking chronically RASi and suffering from AKI did not reinitiate their medication 6 months after hospital discharge. 1 Moreover, a study examining frail patients’ misuse/underuse of medications 2 uncovered that the most prevalent case of underuse was the absence of Angiotensin Converting Enzyme inhibitors (ACEi) for a patient with systolic heart failure. The study found that misuse and underuse of medication decreased the 18-months survival rate from 97 to 96 and 88%, respectively. Additionally, a recent study 3 following patients with discontinued RASi after hospital discharge due to AKI episode uncovered that people with discontinued RASi had a 27% increase in mortality compared to those who did not experience a discontinuation. Thus, unwanted discontinuation might impact patients’ long-term outcomes. However, RASi re-initiation can be challenging due to its impact on renal function. Approximately 20% of patients taking RASi will suffer from at least one episode of hyperkalemia, 4 whilst 10% will see a more than 20% rise in their serum creatinine (sCr) levels. 5 Hence, the greater challenge to re-initiate these medications for intensive care unit (ICU) patients who already suffer multiple health complications, especially ICU patients with AKI.
To date, there is no consensus on specific safety parameters and optimal clinical settings for the re-initiation of RAS blockers, leaving the clinician to the best of their judgment. The aim of this study was to establish a consensus on RASi re-initiation practices for ICU patients who suffer from AKI.
Methods
We conducted a 3-round modified Delphi method to establish consensus on crucial factors influencing RASi re-initiation for ICU patients who suffered from AKI. The Delphi method is a well-established method to establish consensus and help develop guidelines. 6 We modified the classical Delphi by not conducting face-to-face meetings at the end of the survey with the participants due to COVID restrictions. In addition, we added multiple-choice questions and open-ended questions at the third round rather than merely asking participants to re-rate items that did not meet consensus.
Panel Selection
Our expert panel was composed of clinical experts in AKI, medical reconciliation, and/or intensive care. We focused on clinicians because they are the key decision makers for medication re-initiation in most centers. The selection was based on clinicians’ involvement in treatment and care of ICU patients with AKI, especially in determining medication re-initiation throughout the full episode of care (in ICU, in hospital and out of hospital); hence our focus on the following specialties: nephrology, intensive care medicine, and internal medicine. We sent a preformatted email invitation distributed by the different professional associations: the Association des spécialistes en Médecine Interne du Québec (ASMIQ) for internists (600 members), Association des néphrologues du Québec (ANQ) for nephrologist (201 members), and Société des intensivistes du Québec (SIQ) for intensive care physicians (100 members). We also recruited panelists through internal hospital services (McGill University Health Center [MUHC], Hôpital Maisonneuve-Rosemont [HMR], Center hospitalier de l’Université de Montréal [CHUM], and Center hospitalier de l’Université de Sherbrooke [CHUS]). To ensure equal representation, we aimed to recruit 5 clinicians from each specialty for a total of 15 clinicians. 7
Study Variables
Our concept of interest was RASi re-initiation. Our influencing factors were divided into 3 categories: demographic, clinical setting, and patient’s safety parameters (see supplemental S3). We inferred the clinical setting variables from existing concepts surrounding guideline compliance and medication reconciliation. We conducted a brief literature review of papers on these topics and based our concepts mostly on those mentioned by Cabana et al, 8 a pioneer in guidelines compliance. Moreover, these concepts were reviewed by our research team members (2 nephrologists, 1 internist-intensivist, 1 family physician, and 1 researcher with experience conducting Delphi studies). We inferred the safety parameters from RASi initiation guidelines and risk factors of AKI mentioned in the literature.9-11 The survey was then reviewed for clarity and completion time by 5 senior residents in internal medicine. Following their comments, we reformulated some questions to enhance their clarity.
Overview of the Modified Delphi Process
The Delphi started in May 2021 and ended in August 2021. The survey was distributed via a user-unique link sent to each panelist using the online survey platform LimeSurvey. The survey was in French only (see Supplemental S2 for an English version). We sent weekly reminders to panelists who did not answer. All panelists provided consent, via a written form sent by email, to participate and complete all 3 rounds.
Round 1
Round 1 started on May 17 and ended on June 10. The purpose of the first round was to collect demographic data and establish a primary consensus. Panelists were asked during this round to first answer questions about their specialty, number of years of practice, experience in treating ICU patients and other demographic questions. Then they were asked to rate, on a 4-point Likert-type scale (“not important,” “a little important,” “important,” and “very important”) the list of influencing factors we extracted from literature review. Finally, they evaluate the relevance (“not relevant,” “a little relevant,” “relevant,” and “very relevant”) of a series of questions to be used in the final round. A comment box was also added in this section so panelists could provide comments and suggestions about the questions (see Supplemental Questionnaire 1).
Round 2
Round 2 started on June 22 and ended on July 19. The purpose of this round was to establish consensus on the remaining items and to further improve questions quality and clarity used in the third round. After reviewing comments from the first round, we added 2 multiple-choice questions enquiring about the panelists preferred measure to estimate renal function in the second round (see Supplemental Questionnaire 1). Answers collected from these 2 questions were used to refine the third round questions.
Round 3
Round 3 started on August 3 and ended on August 30. The purpose of this round was to establish consensus on the remaining items and to set threshold values for safety parameters. Panelists evaluated the importance of the remaining influencing factors and answered a series of multiple-choice and open-ended questions to establish values for certain influencing factors such as sCr levels, glomerular filtration rate (GFR), and arterial pressure (see Supplemental Questionnaire 3). These questions focused on patient’s clinical picture and safety parameters. Only questions deemed as relevant by the panelists during the 2 previous rounds were kept for this round.
Statistical Analysis
Data were tabulated after each round using IBM SPSS version 26. We entered the percentage of ratings of each element. We defined consensus as a 75% combined rating as either “not important” and “little important,” or “important” and “very important.” The same consensus definition was used for questions’ relevance. We used 75% as a cut-off value based on previous Delphi studies. 12 Data collected in the third round (e.g., optimal GFR and arterial pressure values) is presented as the number of panelists per value. During the third round, some panelists provided brief comments which we analyzed manually.
Results
We successfully recruited 20 panelists: 7 nephrologists, 3 internists, and 10 intensive care physicians (see Table 1). The latter came from different base specialties: 4 internal medicine, 1 respirology, 1 respirology and internal medicine, 1 anesthesiology, 1 emergency medicine, while the remaining 2 stated intensive care medicine as their sole specialty. Furthermore, 12 out of 20 (60%) of our panelists were females. All our panelists stated that their perceived knowledge and competencies in treating ICU patients with AKI were either “good” or “very good.” Eighteen of the panelists treated ICU patients, and 2 did not take care of ICU patients in their usual practice.
Panel’s Sociodemographic Characteristics (n = 20).

Note. Sociodemographic characteristics. SD = standard deviation; ICU = intensive care unit.
Rounds 1 and 2
All panelists answered the first 2 rounds. Fifteen items met consensus during this round. Nine items, mainly items related to patient’s health such as GFR, arterial pressure, and vital prognosis, were deemed important (see Table 2). Four items, 3 of which were all related to clinical setting, were deemed not important (see Table 2). We also enquired about clinicians preferred parameter for renal function estimation and the preferred method to present it. Fourteen (70%) panelists chose eGFR as an absolute value to estimate renal function and thus we kept these choices to design our questions for the third round.
Factors’ Importance.

Note. Factors’ importance according to clinician’s rating. In green: factors deemed important or very important. In orange: factors which did not meet consensus. In red: factors deemed little or not important. AKI = acute kidney injury; GFR = glomerular filtration rate; RASi = renin-angiotensin system inhibitors; sCr = serum creatinine; absence of feedback = absence of feedback on the clinician’s decision impact.
Round 3
Eighteen out of 20 (90%) panelists answered this round. Ten elements were evaluated for their importance, and none met consensus (see Table 2).
Aside from item rating, this round included multiple-choice and open-ended questions (see Supplemental Questionnaire 3). In the first section, we enquired about eGFR. We suggested a range of values inspired by the CKD scale. There was no consensus on the general eGFR value for RASi re-initiation. Seven (39%) panelists stated that an acceptable value would be between 30 and 59 mL/min/1.73 m2 (see Figure 1), while 7 others indicated a value other than the ones we suggested (patients’ baseline value or values between 15 and 90 mL/min/1.73 m2). These values changed slightly when asked about optimal eGFR values for RASi re-initiation for ICU patients with AKI. Six (33%) stated that a value between 30 and 59 mL/min/1.73 m2 was optimal for RASi re-initiation, 5 (28%) chose a value between 60 and 89 mL/min/1.73 m2 (see Figure 1), and 7 panelists stated that no value was optimal, or they would wait until ICU discharge for re-initiation (see Figure 1). Finally, we enquired about an optimal eGFR value for RASi re-initiation for ICU patients who suffered from AKI with 1 of the 4 following conditions: hypertensive, suffering from recent episodes of hyperkalemia, with heart failure, and diabetes. No consensus value was established for any of the pathologies (see Figure 2).

Optimal eGFR values for RAS re-initiation.

Optimal eGFR values for RASi re-initiation in different conditions.
In the second part, we enquired about optimal arterial pressure for RASi re-initiation in ICU patients who suffered from AKI and whose primary indication of RASi was hypertension. We suggested values from 90 to 140 mm Hg for the systolic pressure and from 40 to 80 mm Hg for diastolic pressure. No consensus value was established. An equal number of panelists opted for 140 mm Hg or 120 mm Hg (see Figure 3). On the other hand, 5 (28%) panelists suggested other values than those we proposed for the systolic pressure (range 140-180 mm Hg or wait until ICU discharge). As per diastolic pressure, no consensus was established. The numbers were distributed equally between 70 mm Hg, 80 mm Hg, and other values (suggestions were 90-95 mm Hg; see Figure 3).
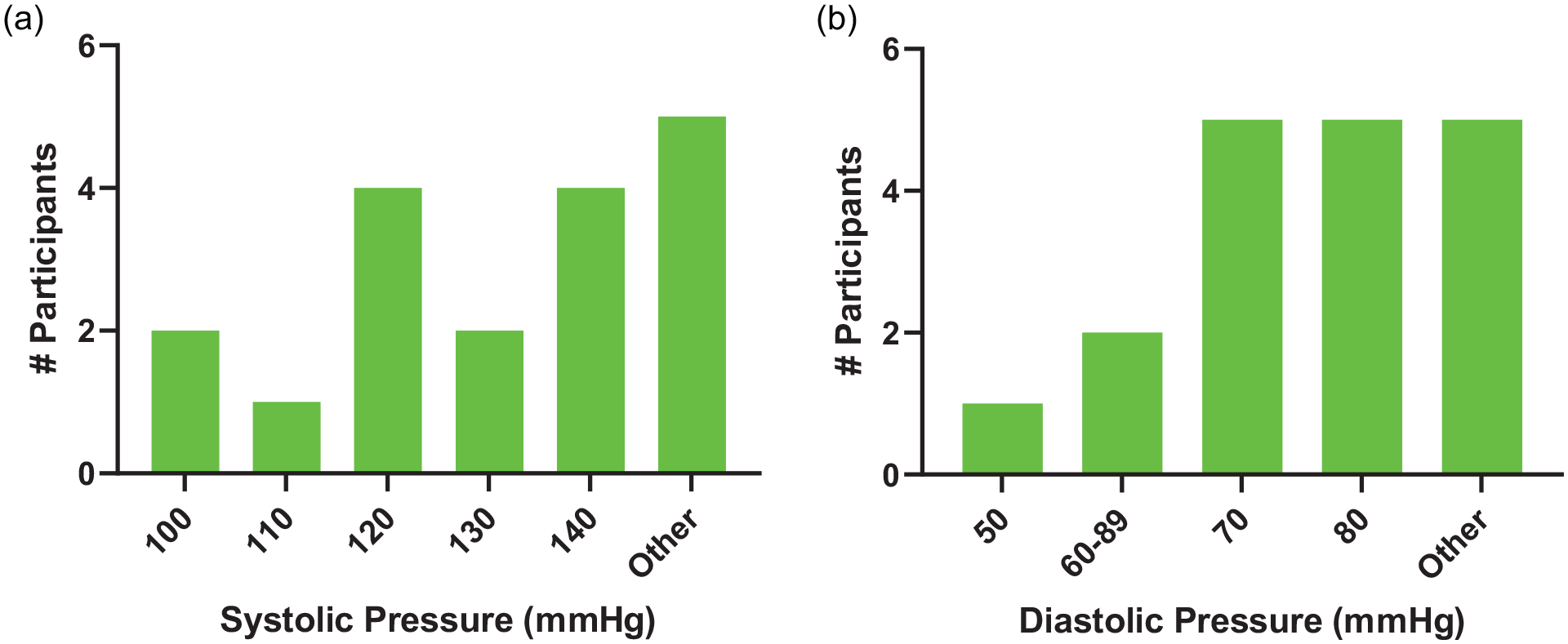
Optimal arterial pressure values.
In the third part, we enquired about the order of importance in re-initiating several long-term medications for ICU patients who suffered from AKI whose primary indication of RASi was either heart failure or diabetic nephropathy. We proposed the following medication: diuretics, antihypertensives, beta-blockers, and RASi. For patients with heart failure, 4 (22%) panelists chose RASi as the first medication to re-initiate while 8 (44%) panelists chose them as the second, and 6 (33%) put them as either third or fourth (see Figure 4). For patients with diabetic nephropathy, 15 (83%) panelists deemed RASi as the first medication to re-initiate, while 3 (17%) chose them as either second or third (see Figure 4). No participant put RASi as the last medication to re-initiate for these patients.

Re-initiation order of long-term medication.
Finally, for this part, we enquired about re-initiating RASi for ICU patients who suffered from AKI and taking either diuretics, beta-blockers or both. For patients whose principal RASi indication is heart failure, there was a consensus (89%) that both beta-blockers and diuretics were safe to re-initiate with RASi together or alone (see Figure 5). By contrast, there was no consensus (72%) on RASi re-initiation concomitant to diuretics for patients with diabetic nephropathy (see Figure 6).

RASi re-initiation concomitant to other medications for patients with heart failure.

RASi re-initiation for patients with diabetic nephropathy.
The last part of this questionnaire was about time and resources. When asked about whether their workload enables them to have enough time to ensure optimal RASi re-initiation, 11 (61%) panelists stated yes, and 7 (39%) stated no (see Figure 7). Moreover, there was a consensus on the timing of RASi re-initiation with 15 panelists (83%) choosing a timing after ICU discharge. Indeed, only 1 (5.56%) participant opted for ICU stay, while 11 (61%) opted for hospital ward, 3 (17%) for external clinic and 3 (17%) for another moment (after ICU discharge; see Figure 8). Finally, when asked about the best resources for optimal RASi re-initiation, 14 (78%) panelists stated that a pharmacist’s follow-up was the best resource.
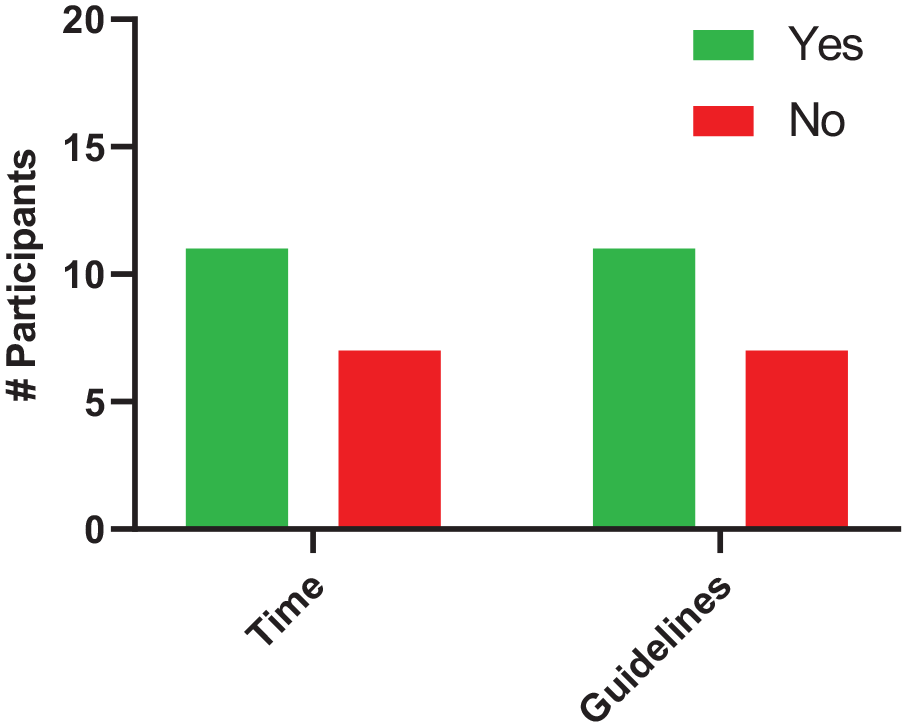
Available time and guidelines.
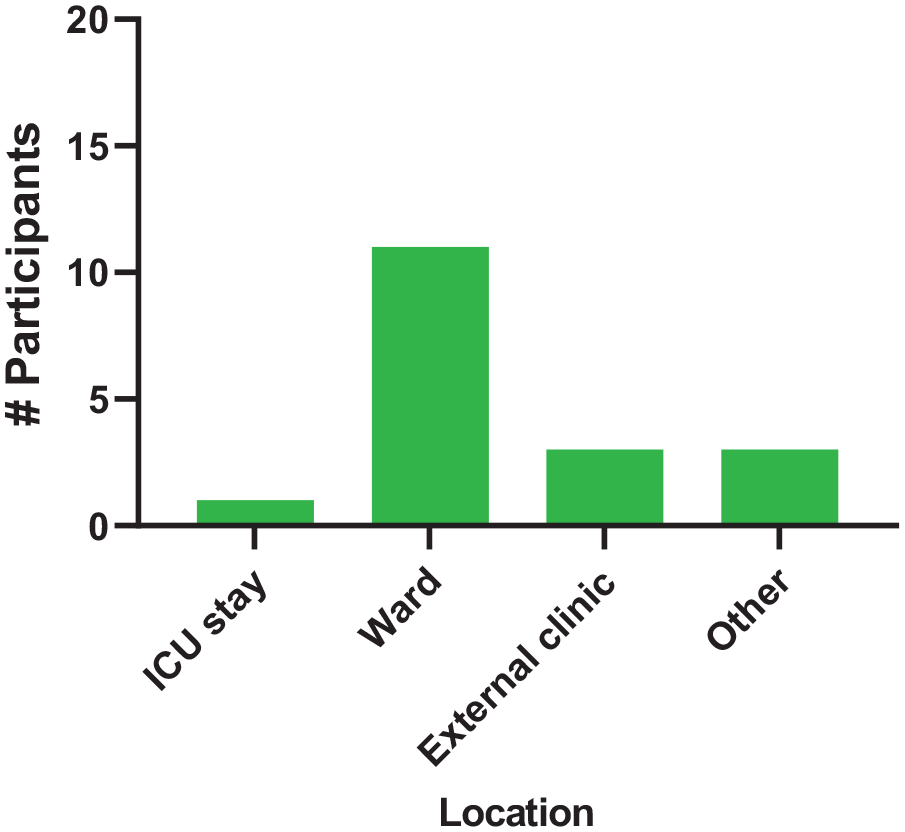
Optimal location for RASi re-initiation.
Discussion
We uncovered 11 elements, such as sCr and GFR, that were deemed important factors and 4 others, such as absence of feedback and absence of guidelines, that were deemed not important. Also, more than the third of proposed elements, such as patient’s ejection fraction, did not meet consensus. Finally, while we did not establish consensus on optimal GFR nor arterial pressure values for RASi re-initiation for ICU patients who suffered AKI, we did establish a consensus on the optimal timing for RASi re-initiation.
One objective of this study was to determine thresholds of safety parameters surrounding RASi re-initiation, hence the third questionnaire with open-ended questions. Unfortunately, the answers were scattered, and no value could be used as a reference or threshold. Indeed, when we enquired about eGFR values optimal for RASi re-initiation, no value had more than 50% of the panelists’ agreement. However, 3 values stood out: values between 30 and 59 mL/min/1.73 m2, 15 mL/min/1.73 m2 or higher, and patient’s baseline value. These values might be explained by the fact that some patients in ICU suffer from CKD; therefore, their normal GFR range might fall as low as 15 mL/min/1.73 m2. In addition, elderly people, which constitute the majority of the ICU population, tend to have a lower GFR. 13 Finally, using patient’s baseline value as an optimal value account for GFR variations between patients. Thus, what might be optimal for one patient is not necessarily optimal for another. Similarly, when specifying a condition, no suggested value had more than 50% agreement. Nevertheless, 2 values stood out: 60 to 89 mL/min/1.73 m2 for hypertensive and hyperkalemic patients and 30 to 59 mL/min/1.73 m2 for patients with heart failure and patients with diabetes. These values are comparable to those found in the inclusion criteria of clinical trials on patients with heart failure, where patients with a GFR > 30 mL/min/1.73 m2 are excluded.14,15 Also, the shift toward lower values for patients with heart failure and diabetes reveal that clinicians see an urgency in re-initiating RASi when there is a perceived benefit. This is further corroborated by several comments from our panelists indicating that RASi re-initiation for patients with heart failure is necessary and should be re-initiated as soon as possible. In addition, the panelists deemed RASi indication as an important factor for their re-initiation. In addition, most panelists indicated that RASi should be re-initiated first for patients with diabetic nephropathy. This may be due to the beneficial effects of RASi on this population, namely the prevention of kidney nephropathy progression through proteinuria reduction. 16
Akin to optimal eGFR values, no arterial pressure value had more than 50% panelists agreement, while 2 values stood out for systolic (120 and 140 mm Hg) and 2 for diastolic pressure (70 and 80 mm Hg). It is worth noting that both systolic values were target values for antihypertensive treatment in the Systolic Blood Pressure Intervention Trial (SPRINT), 17 which is the reference study for hypertensive patients. The target value for an intensive antihypertensive treatment was 120 mm Hg, while the target for standard treatment was 140 mm Hg. In addition, a diastolic blood pressure of 80 mm Hg or more corresponds to the definition of hypertension. 18 Thus, it is normal for clinicians to follow this threshold value given that RASi are antihypertensive medications and that we set, in our question, the principal RASi indication as hypertension. Interestingly, some panelists opted for lower systolic and diastolic pressure values, such as 100 mm Hg for systolic and 50 mm Hg for diastolic. This might be explained by the panelists’ potential consideration that ICU patients suffering from severe heart failure have lower blood pressure values. 19
Finally, when trying to establish an order of importance of long-term medication re-initiation, there was no explicit agreement aside from antihypertensive medication being re-initiated last for ICU patients with heart failure and RASi first for patients with diabetic nephropathy. In sum, setting optimal thresholds of patient’s safety parameters for RASi re-initiation is complex due to interindividual variations of these parameters and the numerous comorbidities affecting ICU patients’ health.
Despite the lack of consensus on safety parameters, 3 quarters of our panelists agreed that RASi re-initiation should be after ICU. This aligns with what Silver et al 20 stipulated in their study about post-AKI ambulatory clinics. This agreement is further explained because not all ICU patients recover from AKI during their stay. 21 Similarly, some ICU patients do not recover their normal organ function even after ICU discharge and are re-admitted 48 h after. 22 Yet, when RASi are not re-initiated upon ICU discharge, the burden falls on the ward clinician, who does not always know that the medication has been discontinued nor why it has been discontinued. 23 This might partly explain some panelists’ choice of re-initiating RASi during ICU or upon discharge. Moreover, 17 panelists mentioned pharmacist’s follow-up as the best resource to ensure optimal RASi re-initiation. Taken together, these findings might suggest that the optimal clinical setting is a rather complex concept to agree on, which requires more attention. Indeed, most guidelines implementation and medication reconciliation challenges are related to the clinical setting rather than the patient’s health. Truly, Cabana et al 8 mentions lack of time and resources and organizational constraints as barriers hindering clinicians’ adherence to guidelines, while Sanchez et al 24 stipulates that medication reconciliation requires an increased cognitive focus. Our data further reinforce the complexity in implementing guidelines for proper medication reconciliation, especially for patients with complex diseases such as AKI.
Strengths
This study is the first to provide an insight on the complexity of RASi re-initiation for ICU patients who suffered from AKI and on Quebec’s clinicians needs for an optimal re-initiation. Several efforts were made to optimize our Delphi study. We chose 3 rounds to minimize attrition and respondents’ fatigue. Moreover, our selection criteria allowed for a diverse panel from different specialties leading to a wider point of view. We also made efforts to minimize questionnaire-related biases. Indeed, we chose a 4-points scale to avoid discrepancies between each point’s interpretation and reduce the response burden. Moreover, we used an even number of points to avoid a neutral midpoint. In addition, to avoid acquiescence bias, we used a bipolar scale. Finally, the questions used for round 3 have been acknowledged as pertinent by the panelists, which minimized non-response bias.
Limitations
Despite our efforts to optimize our method, some limitations remained. Indeed, this project reflects clinicians’ beliefs rather than the actual practice. Moreover, we acknowledge that a consensus among experts is not necessarily synonymous with best practices. In addition, we limited our selection to Quebec clinicians, thus excluding clinicians from other provinces, where safe and optimal RAS re-initiation could be defined differently. It would be better to conduct a future study examining the opinion of clinicians from several provinces. Finally, we failed to recruit family doctors whom we believe are important in long-term medication follow-up, especially since several clinicians in our study deemed that the optimal timing for RASi re-initiation was during external follow-up.
Conclusion
In sum, our study further corroborates the need to address RASi re-initiation for ICU patients post-AKI. In addition, it shed light on the complexity of this task and clinicians’ needs to ensure an optimal re-initiation. With a better understanding of clinicians’ need, it would be interesting to further explore safety parameters by adding more detailed questions and factors.
Supplemental Material
sj-docx-1-cjk-10.1177_20543581221112266 – Supplemental material for Determining Factors Influencing RAS Inhibitors Re-Initiation in ICU: A Modified Delphi Method
Supplemental material, sj-docx-1-cjk-10.1177_20543581221112266 for Determining Factors Influencing RAS Inhibitors Re-Initiation in ICU: A Modified Delphi Method by Hadjer Dahel, Jean-Philippe Lafrance, Mathilde Patenaude, Kelley Kilpatrick, William Beaubien-Souligny, Mathieu Moreau and Han Ting Wang in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-docx-2-cjk-10.1177_20543581221112266 – Supplemental material for Determining Factors Influencing RAS Inhibitors Re-Initiation in ICU: A Modified Delphi Method
Supplemental material, sj-docx-2-cjk-10.1177_20543581221112266 for Determining Factors Influencing RAS Inhibitors Re-Initiation in ICU: A Modified Delphi Method by Hadjer Dahel, Jean-Philippe Lafrance, Mathilde Patenaude, Kelley Kilpatrick, William Beaubien-Souligny, Mathieu Moreau and Han Ting Wang in Canadian Journal of Kidney Health and Disease
Footnotes
Ethics Approval and Consent to Participate
This modified Delphi followed a pre-established protocol and was approved by the research ethics committee of the CIUSSS de l’Est-de-l’Île-de-Montréal (2021-2592).
Consent for Publication
Research data collected from this study are not published in a public data base.
Availability of Data and Materials
Data sharing is possible upon request.
Authors’ Note
This manuscript has not been previously published or submitted elsewhere.
Author Contributions
All authors contributed significantly to meet the authorship criteria. HD and HTW were involved in every step of the process. JPL was involved in study design, survey design and data analysis and interpretation. MP contributed to manuscript writing and survey design. KK was involved in study design and survey design. WBS was involved in study design and data analysis and interpretation. MM was involved in survey design and manuscript review. All authors have seen and approved the final version of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
