Abstract
Background:
An emerging strategy to increase deceased organ donation is to use dedicated donation physicians to champion organ donation. We sought to conduct a systematic review of the effectiveness of donation physicians in improving organ donation outcomes.
Objective:
A systematic review was conducted following Cochrane principles. MEDLINE, Embase, and CINHAL databases were searched from inception to March 26, 2020.
Methods:
Quantitative studies examining the effects of donation physicians on all deceased organ donation outcomes were considered for inclusion. Review articles, editorials and opinion articles, and case studies were excluded. Study selection was completed independently by 2 team members; all discrepancies were resolved by consensus. Two team members independently extracted data from studies.
Results:
A total of 1017 studies were screened, and 12 met inclusion criteria. Included studies were published between 1994 and 2019. Half used an interrupted time series design (n = 6; 50%), 3 (25%) were cohort studies, and 3 (25%) used a before-and-after study design. Outcomes (reported in greater than 50% of included articles) included consent/refusal rate (n = 8; 67%), number of potential donors (n = 7; 58%), and number of actual donors (n = 7; 58%). Across studies and design types, there was an increase in potential organ donors ranging from 8% to 143% (Mdn = 33%), an increase in actual organ donors from 15% to 113% (Mdn = 27%), an increase in donor consent rate from −3% to 258% (Mdn = 12%), and an increase in deceased donor transplants from 13% to 24% (Mdn = 19%) following the introduction of donation physicians.
Conclusions:
Donation physicians have the potential to significantly improve deceased organ donation. Further implementation and evaluation of donation physician programs is warranted. However, implementation should be undertaken with a clear plan for a methodologically rigorous evaluation of outcomes.
Introduction
Despite improvements in organ donation procedures and practices in many countries across the world, a global shortage of organs remains. In the United States, for example, despite a nearly 5% annual increase in transplantation rates, the gap between organ supply and demand has widened due to an increase in patients awaiting transplants. 1 Internationally, specialists called “donation physicians,” “donor physicians,” or “transplant physicians” are increasingly recognized as playing a key role in improving organ donation rates. 2 All donation physicians have expertise on organ and tissue donation. 3 However, the role of donation physicians varies across countries. 3 In Spain, donation physicians take the center role in all components of the donation process and provide training to critical care staff. 3 These physicians evaluate and diagnose brain death, manage the donation, and communicate with the transplant team. 3 By contrast, in the United Kingdom and Australia, donation physicians promote donation but do not attend to every donor; instead, they work closely with nurse donor coordinators who provide direct care. 3 However, across most countries, besides acting as local champions for organ donation within intensive care units and the broader hospital, donation physicians have responsibility and accountability for organ deceased donation 4 and work alongside other health care professionals and administration to facilitate organ donation. 3
While donation physician programs exist, given the continued need to optimize organ donation internationally, it is essential that interventions being used to increase organ donation be evaluated for effectiveness so that resources can be allocated appropriately. 5 To this end, the purpose of this review was to examine the published literature that reported effectiveness of donation physicians on deceased organ donation outcomes, including the number of potential donors, the number of actual donors, and consent rates, across different interventions and study designs.
Methods
Search Strategy
The search strategy was developed and performed with a health sciences librarian who has expertise in systematic reviews. A total of 3 online databases were searched: EMBASE, MEDLINE, and CINAHL. There was no limit on the date of publication and the search accounted for studies published from the inception of each database up to March 26, 2020. Key terms used in the search included “donor physician,” “donation physician,” and “transplant physician.” The complete search strategy is provided (see Supplemental Additional File 1). Reference lists of included studies and excluded review papers were assessed for eligible studies.
Inclusion and Exclusion Criteria
All quantitative studies that examined the effects of donation physicians on deceased donation outcomes were included. The specific outcomes were not decided upon a priori to maximize inclusion of any articles that quantitatively evaluated one or more possible outcomes following implementation of donation physicians (eg, the number of potential donors, actual donors, consent rate). Review articles, editorials, opinion articles, and case studies were excluded. Articles focused on living donation and nonphysician donor coordinators were also excluded.
Study Identification
Screening was undertaken using Distiller Systematic Review (Distiller SR) Software, 6 a web-based software that facilitates collaboration among reviewers during the study selection and data extraction processes. Screening occurred in 2 phases. In both phases, all records were assessed using 2 screening questions: (1) Does the study report on the role of the donation physician? and (2) Does the study report on the effect of a donation physicians on an organ donation outcome such as referral, consent, and/or donation rates? In level 1 screening, 2 team members (L.D.A., W.J.S.) independently assessed the titles and abstracts of all records identified in the search strategy. All potentially relevant articles, as well as those where there was insufficient information to decide eligibility, progressed to level 2 screening. In level 2 screening, full-text articles were assessed for eligibility based on the inclusion criteria by 2 team members (L.D.A., W.J.S.). All discrepancies in study selection were resolved through consensus between team members with consultation of a third senior team member (J.E.S.) when necessary. Reasons for exclusion were documented for all full-text articles.
Data Extraction
A structured data extraction form was developed and piloted on a sample of 5 studies. Once the form was finalized, data from each article were extracted independently by 2 of 3 possible team members (L.D.A., W.J.S., N.G.). Data extracted included (1) study identification—authors, publication year, country/province/territory, language, publication status, funding, study design, data collection dates; (2) participant characteristics—sample description including sample size and gender, age, education, health care professionals’ role, health care professionals’ experience; health care setting; and (3) outcomes—any information pertaining to organ donation outcomes. Any discrepancies in data extraction were resolved through a consensus process between the data extractors. A third senior team member (J.E.S.) was consulted when necessary.
Quality of Included Studies
Two team members assessed methodological quality of the studies independently (L.D.A., W.J.S., D.C.-Y.). Discrepancies in methodological quality assessments between reviewers were resolved through consensus. Two validated assessment tools were used to conduct the methodological quality assessments. The Checklist for Quasi-Experimental Studies (nonrandomized experimental studies) from the Joanna Briggs Institute was used for interrupted time series studies. 7 This checklist contains 9 items and assesses studies in the areas of sampling, measurement, outcomes, and statistical analysis. The Quality Assessment and Validity Tool for Pre/Post-test Studies tool was used for cohort and before-and-after study designs.8-12 The Pre/Post-test Studies tool assesses studies in 6 core areas: sampling, design, control of confounders, data collection and outcome measurement, statistical analysis, and dropout. A quality score for each article was obtained by dividing the sum of the scores by the total amount possible. All included studies were classified using the same rating scale: weak (≤0.50), low-moderate (0.51-0.65), high-moderate (0.66-0.79), or strong (≥0.80). This rating system is based on a previously developed system 13 and has been used in previous systematic reviews.8-12
Data Synthesis
The data were categorized according to the organ donation outcome addressed in the study. Data synthesis differed by study design. For interrupted time series studies, the relative year-to-year percent changes were calculated (eg, [(2001-2000)/2000] and [(2002-2001)/2001]) and presented as dot plots. The average relative percent change was calculated and presented as bar graphs. For cohort and before-and-after study designs, the relative percent change between the cohorts or before-and-after data points was synthesized and presented as bar graphs.
Results
Eligible Studies
A total of 1621 records were identified in the search. After the removal of duplicates (n = 487), 1134 articles remained and underwent screening based on title and abstract. This first level of screening led to the exclusion of 1014 articles. A total of 120 articles were assessed for eligibility through full-text screening and 12 studies met the inclusion criteria for the review (see Figure 1 for PRISMA [Preferred Reporting Items for Systematic Reviews and Meta-Analyses] flow diagram). A list of studies that were excluded upon full-text review, including the reason(s) for their exclusion, are provided in Supplemental Additional File 2.

Preferred Reporting Items for Systematic Reviews and Meta-Analyses chart.
Study Characteristics
Characteristics of the included studies are provided in Table 1. All studies were published between 1994 and 2019. Nearly half of the studies were conducted in Spain (n = 5; 42%). Other countries represented were Netherlands (n = 2; 17%), Poland (n = 2; 17%), Brazil (n = 1; 8%), Greece (n = 1; 8%), Uruguay (n = 1; 8%). None of the included studies were conducted in North America.
Information for Included Studies.
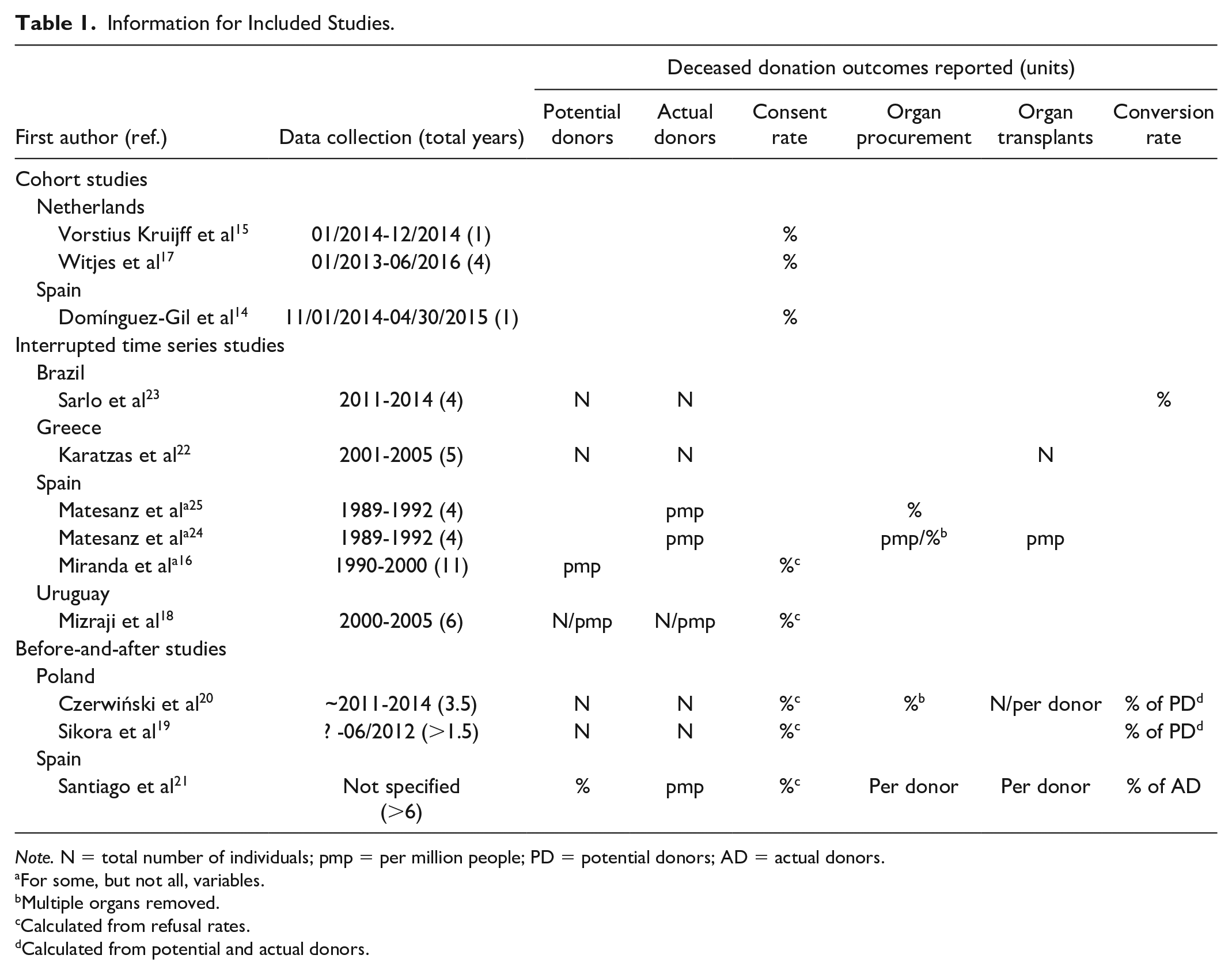
Note. N = total number of individuals; pmp = per million people; PD = potential donors; AD = actual donors.
For some, but not all, variables.
Multiple organs removed.
Calculated from refusal rates.
Calculated from potential and actual donors.
Half of the studies (n = 6; 50%) used an interrupted time series design, whereas 3 (25%) studies used cohort designs, and 3 (25%) used a before-and-after study design. The period of data collection in the studies ranged from 1 year14,15 to 11 years. 16 Several different organ donation outcomes were reported in included studies: consent or refusal rate (n = 8; 67%),14,16-21 number of potential organ donors (n = 7; 58%),14,15,17-20,22 number of actual organ donors (n = 7; 58%),14,17-20,22-24 number of deceased donor transplantations (n = 4; 33%),20-22,24 conversation rate (ie, how many potential donors become actual donors) (n = 4; 33%),19-21,23 number of organs removed (n = 3; 33%),21,24,25 and number of multiple organs removed (n = 2; 17%)20,25 (Table 1).
Methodological Quality of Included Studies
All studies were rated as “weak” in methodological quality. Reasons for reduced quality scores were largely around sampling and statistical analyses. The quality ratings for each included study are presented in the supplementary file (see Supplemental Additional File 3 for studies with an interrupted time series study design; see Supplemental Additional File 4 for cohort and before-and-after study design).
Organ Donation Outcomes
Potential organ donors
Of the studies reporting on potential organ donors (n = 7), 4 reported data as absolute values,19,20,22,23 2 as donors per million,16,18 and 1 as percent change. 21 All 7 studies found a relative increase, between 8% 16 and 143%, 21 in potential organ donors following implementation of donation physicians (median increase, 33%). Among the interrupted time series studies (Figure 2A and 2B), the average relative year-to-year increase in potential organ donors across all studies ranged from 8% 16 to 67%. 22 Among the before-and-after studies (Figure 2C), the relative percent change in potential organ donors ranged from 27% 20 to 143%. 21

Relative percent change in potential organ donors for interrupted time series (A-B) and before-and-after (C) studies.
Actual organ donors
Of the studies reporting on actual organ donors (n = 7), 3 reported data as part per million18,21,24 and 4 as the absolute value.19,20,22,23 All 7 studies found an increase between 15% 25 and 113% 19 in actual organ donors following implementation of donation physicians (median increase, 27%). Among interrupted time series studies (Figure 3A and 3B), the average relative year-to-year increase in actual organ donors ranged from 15% 25 to 73%. 23 Among the before-and-after studies (Figure 3C), the relative percent change in actual donors ranged from 24% 20 to 113%. 19

Relative percent change in actual organ donors for interrupted time series (A-B) and before-and-after (C) studies.
Donor consent rate
Of the studies reporting donor consent or refusal rates (n = 8), the average relative change in donor consent rates ranged from −3% to 258% (median increase, 12%). Among interrupted time series studies (Figure 4A and 4B), the average relative increase in the year-to-year percent change in donor consent rates ranged from 1% 16 to 17%. 18 In before-and-after studies (Figure 4C), the average relative increase in donor consent rate ranged from −1% 20 to 11%. 19 In cohort studies (Figure 4D), the relative percent change in donor consent rate varied from 5% 17 to 259%. 15

Percent change in donor consent rates for interrupted time series (A-B), before-and-after (C), and cohort (D) studies.
Organ procurement
Of the 3 studies21,24,25 reporting on single organs procured, 2 reported data as absolute numbers24,25 and 1 as per donor. 21 The change in single organs procured (Supplemental Additional File 5) ranged from 16% 21 to 81%.24,25 Both studies20,25 reporting on the procurement of multiple organs provided data as a percent change (Supplemental Additional File 6). For the interrupted time series study, the average relative year-to-year increase was 38%, 25 and for the before-and-after study, the average relative change in multiple organs procured was 4%. 20
Transplants
Four studies20,21,22,24 reported on the number of transplants from deceased donors (Supplemental Additional File 7). Of these, 2 reported data as absolute numbers,20,22 2 as per donor,20,21 and 1 as part per million. 24 Among all studies, there was an increase in deceased donor transplants from 13% to 24% (median increase, 19%). Among interrupted time series studies, the average relative year-to-year increase in deceased donor transplants ranged from 13% 24 to 24%. 22 Among before-and-after studies, the average relative year-to-year increase in deceased donor transplants ranged from 13% 21 to 20%. 20
Organ donor conversion rate
Of the 4 studies19-21,23 reporting on organ donor conversion rates (Supplemental Additional File 8), 3 reported data as percent of potential donors19,20,23 and 1 as percent of actual brain deaths. 21 Among interrupted time series data, the average relative year-to-year increase in the organ donor conversion rate was 30%. 23 Among before-and-after studies, the relative percent change in organ donor conversion rates ranged from 6% 19 to 23% 21 among studies reporting a positive change and a −3% percent change in one study reporting a decrease. 20
Discussion
Summary of Findings
Of the studies that reported actual and potential numbers of deceased organ donors in this review, all found an increase in these outcomes following the implementation of a donation physician program. Organ donor consent rates and actual deceased donor transplantations also experienced positive increases in most studies. Overall, the implementation of donation physicians was associated with a positive effect on deceased organ donation and transplantation. However, due to a lack of direct comparisons between different programs and in the absence of well-controlled studies, it is difficult to draw conclusions regarding the role of donation physicians in promoting organ donation rates. Consequently, many other factors may have affected the results reported in the reviewed studies.
Limitations of Included Studies
While we saw a trend for a positive effect on donation outcomes with donation physicians, the included studies had several limitations. First, as most studies included in this review found a positive result, it is possible that the presence of publication bias may have distorted the results of the current study. It is therefore possible that the true effects of donor physicians are not as favorable as the current findings suggest. As well, there were several methodological weaknesses in the studies. Notably, all included studies were rated as weak, the lowest grading in methodological quality, illustrating a need for well-designed studies in this area. For before-and-after and cohort studies, it was not clear if the interventions (eg, training sessions for physicians) would need to be regularly implemented and, if so, how regularly they would need to be implemented to ensure the positive effects are maintained. Thus, future longitudinal studies would be an asset in this area of study. The amount and type of training physicians received also varied between studies. When defined, training appeared to range from online modules to in-person training courses; however, in many studies, the training of physicians was not explicitly defined. This may have affected the study outcomes. Studies also varied in statistical rigor, and few studies performed inferential statistical tests. As most studies did not report statistical significance, we could not consider effect sizes or precision of estimates in evaluating the findings. As well, none of the interrupted time series studies reported baseline data, making it difficult to evaluate the extent to which the intervention improved organ donation outcomes over and above secular trends. As with all uncontrolled studies, it is possible that other interventions were implemented during the period that resulted in improved outcomes.
Limitations of the Present Review
The present review has some limitations. First, as we did not search gray literature databases, this review may not include all relevant work. Second, we did not contact the study authors to clarify data that could have made pooling easier. Third, the wide diversity in how the data were reported complicated our ability to compare results across studies. For instance, some studies reported results as absolute values, others as percent change, and others as parts per million or per donor, leading to difficulties in data synthesis. To address this in interrupted time series data, we calculated the average relative percent change year-over-year for each variable. This allowed us to better compare intervention outcomes across studies, irrespective of how the data were reported. Fourth, a protocol was not in place before the searchers were conducted. This limits the rigor of the approach taken to conduct the search, data review, extraction, and appraisal. Fifth, studies conducted in Spain are overrepresented in this review, with 5 out of the 12 using Spanish data. While Europe is well represented, with 9 out of 12 studies from European countries, and South America also has representation, with 3 out of 12 studies, none of the included studies were from North America or Asia. Thus, it is not clear to what extent the findings would be applicable globally. Finally, we also excluded studies that examined the role of nonphysician coordinators in increasing organ donation. While we were specifically interested in the role of physician coordinators, future reviews should include studies evaluating the role of nonphysician coordinators, including conducting comparative analyses regarding their efficacy as compared with their physician colleagues. It is important to identify if nonphysician coordinators, who would be less expensive, are equally effective in increasing donation rates.
Conclusions and Future Research
In summary, this review found important improvements in deceased organ donation outcomes following the introduction of organ donation physicians. Although the quality of the included data was weak, the identified positive trends suggest that further implementation and evaluation of donation physician programs is warranted. However, implementation should only be undertaken with a clear plan for a methodologically rigorous evaluation of outcomes, including economic indicators. To this end, future studies must consider other factors that may influence increasing donation and transplantation rates. For instance, concurrent changes in policies and procedures may positively affect organ donations rates, making it difficult to ascertain whether the effects are truly due to donor physician programs. Thus, subsequent research must take into a careful account any additional or parallel interventions that may be taking place. As well, the presence or implementation of organ donation committees or tissue donation coordinators may enhance the work of donation physicians and should also be accounted for in subsequent research. Finally, more details should be provided regarding the structural aspects of the locations where the interventions are implemented, as different locations will have access to different resources, which may shape the efficacy of the interventions.
Supplemental Material
sj-docx-1-cjk-10.1177_20543581221107751 – Supplemental material for Impact of Donation Physicians on Deceased Organ Donation: A Systematic Review
Supplemental material, sj-docx-1-cjk-10.1177_20543581221107751 for Impact of Donation Physicians on Deceased Organ Donation: A Systematic Review by Janet E. Squires, Laura D. Aloisio, Wilmer John Santos, Danielle Cho-Young, Monica Taljaard, Nills Gritters, Sonny Dhanani and Gregory Knoll in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-docx-2-cjk-10.1177_20543581221107751 – Supplemental material for Impact of Donation Physicians on Deceased Organ Donation: A Systematic Review
Supplemental material, sj-docx-2-cjk-10.1177_20543581221107751 for Impact of Donation Physicians on Deceased Organ Donation: A Systematic Review by Janet E. Squires, Laura D. Aloisio, Wilmer John Santos, Danielle Cho-Young, Monica Taljaard, Nills Gritters, Sonny Dhanani and Gregory Knoll in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-docx-3-cjk-10.1177_20543581221107751 – Supplemental material for Impact of Donation Physicians on Deceased Organ Donation: A Systematic Review
Supplemental material, sj-docx-3-cjk-10.1177_20543581221107751 for Impact of Donation Physicians on Deceased Organ Donation: A Systematic Review by Janet E. Squires, Laura D. Aloisio, Wilmer John Santos, Danielle Cho-Young, Monica Taljaard, Nills Gritters, Sonny Dhanani and Gregory Knoll in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-docx-4-cjk-10.1177_20543581221107751 – Supplemental material for Impact of Donation Physicians on Deceased Organ Donation: A Systematic Review
Supplemental material, sj-docx-4-cjk-10.1177_20543581221107751 for Impact of Donation Physicians on Deceased Organ Donation: A Systematic Review by Janet E. Squires, Laura D. Aloisio, Wilmer John Santos, Danielle Cho-Young, Monica Taljaard, Nills Gritters, Sonny Dhanani and Gregory Knoll in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-docx-5-cjk-10.1177_20543581221107751 – Supplemental material for Impact of Donation Physicians on Deceased Organ Donation: A Systematic Review
Supplemental material, sj-docx-5-cjk-10.1177_20543581221107751 for Impact of Donation Physicians on Deceased Organ Donation: A Systematic Review by Janet E. Squires, Laura D. Aloisio, Wilmer John Santos, Danielle Cho-Young, Monica Taljaard, Nills Gritters, Sonny Dhanani and Gregory Knoll in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-docx-6-cjk-10.1177_20543581221107751 – Supplemental material for Impact of Donation Physicians on Deceased Organ Donation: A Systematic Review
Supplemental material, sj-docx-6-cjk-10.1177_20543581221107751 for Impact of Donation Physicians on Deceased Organ Donation: A Systematic Review by Janet E. Squires, Laura D. Aloisio, Wilmer John Santos, Danielle Cho-Young, Monica Taljaard, Nills Gritters, Sonny Dhanani and Gregory Knoll in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-docx-7-cjk-10.1177_20543581221107751 – Supplemental material for Impact of Donation Physicians on Deceased Organ Donation: A Systematic Review
Supplemental material, sj-docx-7-cjk-10.1177_20543581221107751 for Impact of Donation Physicians on Deceased Organ Donation: A Systematic Review by Janet E. Squires, Laura D. Aloisio, Wilmer John Santos, Danielle Cho-Young, Monica Taljaard, Nills Gritters, Sonny Dhanani and Gregory Knoll in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-docx-8-cjk-10.1177_20543581221107751 – Supplemental material for Impact of Donation Physicians on Deceased Organ Donation: A Systematic Review
Supplemental material, sj-docx-8-cjk-10.1177_20543581221107751 for Impact of Donation Physicians on Deceased Organ Donation: A Systematic Review by Janet E. Squires, Laura D. Aloisio, Wilmer John Santos, Danielle Cho-Young, Monica Taljaard, Nills Gritters, Sonny Dhanani and Gregory Knoll in Canadian Journal of Kidney Health and Disease
Footnotes
Ethics Approval and Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Author Contributions
All authors contributed to the conceptualization of the project. Screening, data extraction, and quality assessment was completed by L.D.A., W.J.S., and D.C.-Y. Tables and figures were approved by all authors. L.D.A. and J.E.S. were major contributors to the writing of the manuscript. All authors read and approved the final manuscript.
Availability of Data and Materials
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is part of the Canadian National Transplant Research Program (CNTRP) and is supported by the Canadian Institutes of Health Research (CIHR) (grant number TFU 127880).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
